Biomarkers of Broccoli Consumption: Implications for Glutathione Metabolism and Liver Health
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
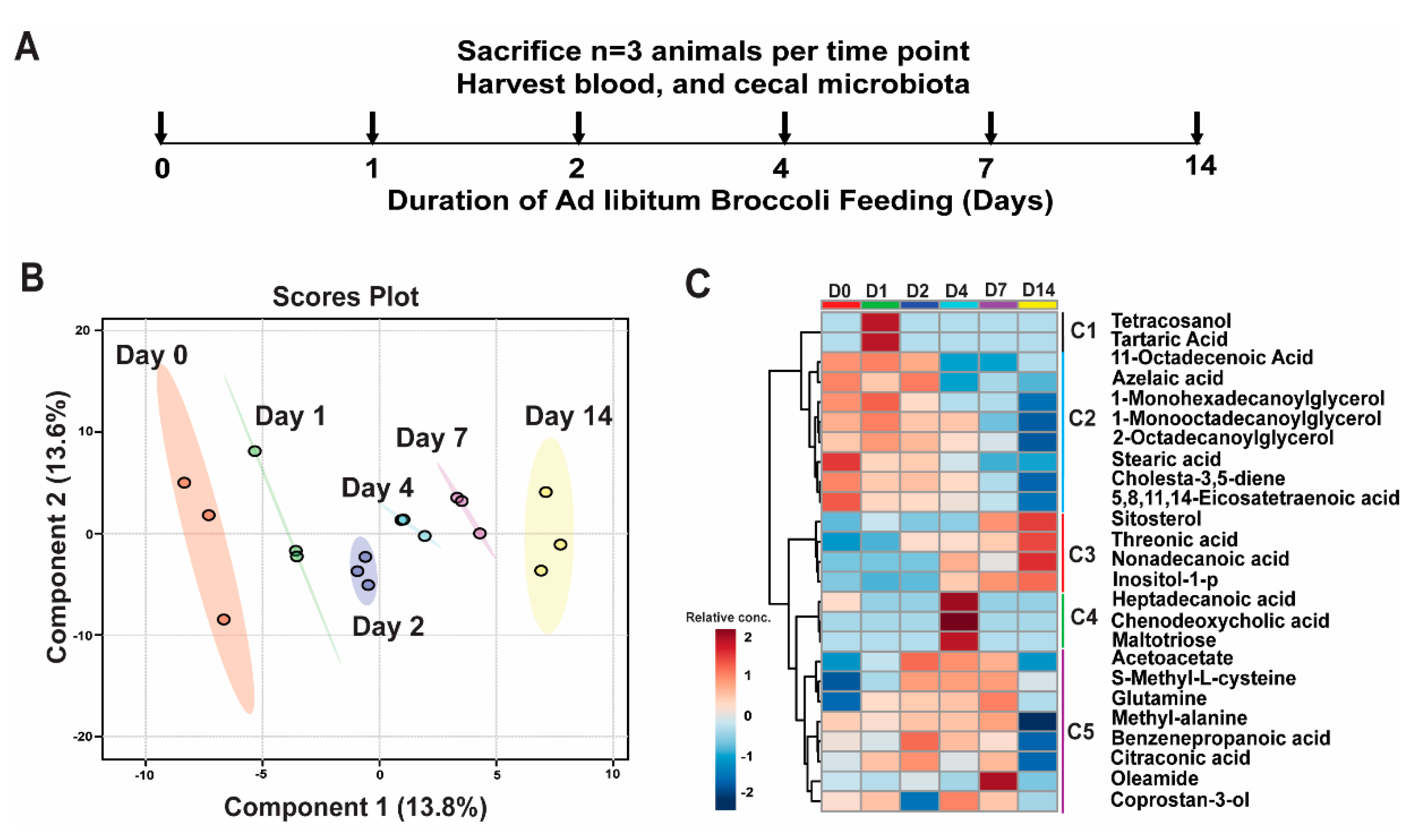
2.2. Long-Term Broccoli Feeding Study
2.3. Cecal Microbiota Profiling by 16S rRNA Sequencing
2.4. Metabolic Profiling
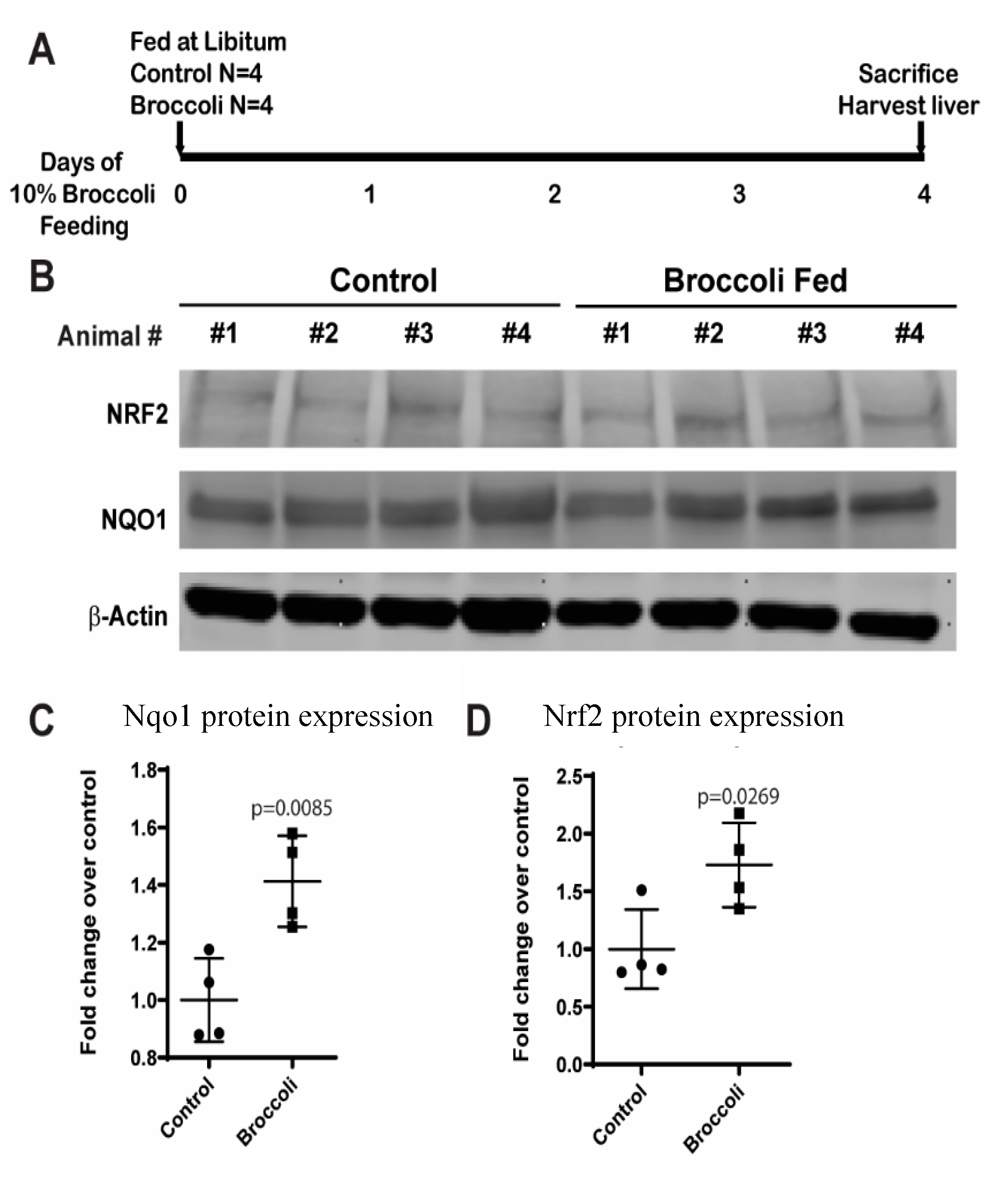
2.5. Western Blot Analysis of Hepatic Protein Expression
2.6. Statistical Analysis
3. Results
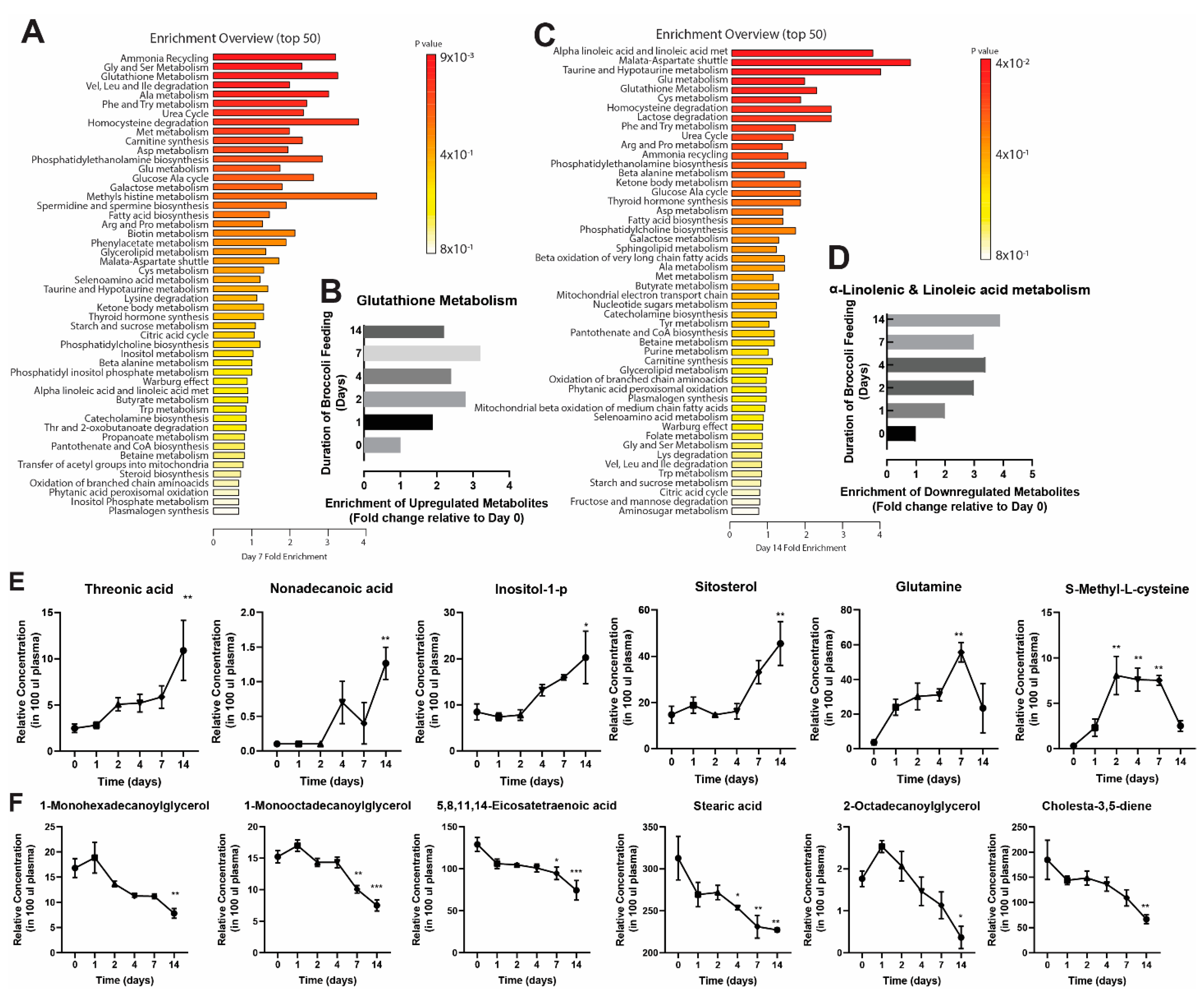
3.1. Broccoli Feeding Alters Plasma Metabolite Profile
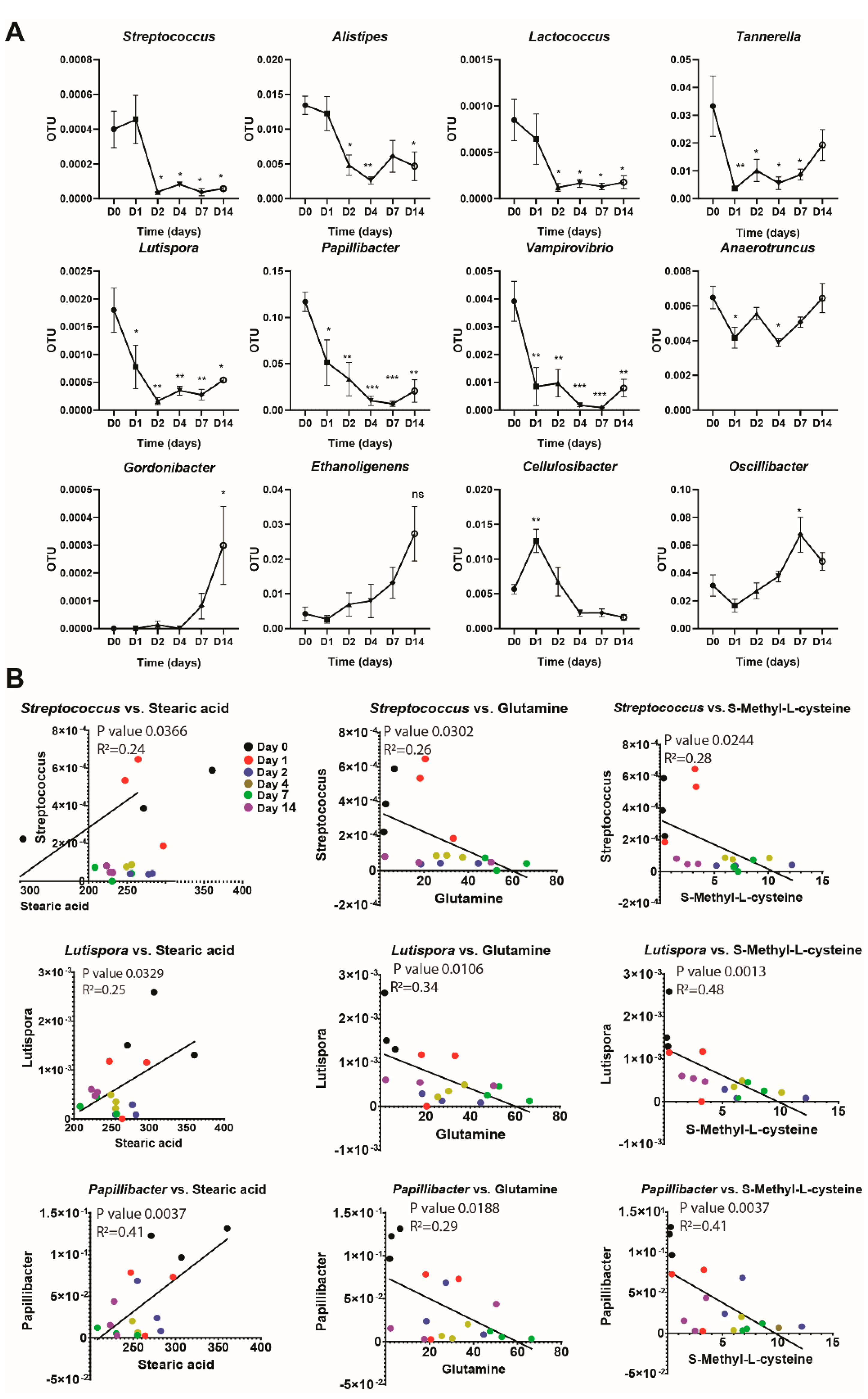
3.2. Changes in Gut Microbiota Composition Correlate with Changes in Plasma Metabolites Following Long-Term Broccoli Feeding
3.3. Broccoli Feeding Increases Protein Expression of NAD(P)H Dehydrogenase [Quinone] 1 (NQO1) and Nuclear Factor (Erythroid-Derived 2)-like 2 (Nrf2) in Rat Liver
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fowke, J.H.; Chung, F.-L.; Jin, F.; Qi, D.; Cai, Q.; Conaway, C.; Cheng, J.-R.; Shu, X.-O.; Gao, Y.-T.; Zheng, W. Urinary Isothiocyanate Levels, Brassica, and Human Breast Cancer. Cancer Res. 2003, 63, 3980–3986. [Google Scholar]
- Jeffery, E.H.; Araya, M. Physiological effects of broccoli consumption. Phytochem. Rev. 2009, 8, 283–298. [Google Scholar] [CrossRef]
- Parr, B.; Bond, J.K.; Minor, T. Vegetables and Pulses Outlook; No. (VGS-362); USDA/ERS: Washington, DC, USA, 6 May 2019; p. 43. [Google Scholar]
- Michaud, D.S.; Spiegelman, D.; Clinton, S.K.; Rimm, E.B.; Willett, W.C.; Giovannucci, E.L. Fruit and vegetable intake and incidence of bladder cancer in a male prospective cohort. J. Natl. Cancer Inst. 1999, 91, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Voorrips, L.E.; Goldbohm, R.A.; van Poppel, G.; Sturmans, F.; Hermus, R.J.J.; van den Brandt, P.A. Vegetable and fruit consumption and risks of colon and rectal cancer in a prospective cohort study—The Netherlands Cohort Study on Diet and Cancer. Am. J. Epidemiol. 2000, 152, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Terry, P.; Wolk, A.; Persson, I.; Magnusson, C. Brassica vegetables and breast cancer risk. JAMA 2001, 285, 2975–2977. [Google Scholar] [PubMed]
- Kolonel, L.N.; Hankin, J.H.; Whittemore, A.S.; Wu, A.H.; Gallagher, R.P.; Wilkens, L.R.; John, E.M.; Howe, G.R.; Dreon, D.M.; West, D.W.; et al. Vegetables, fruits, legumes and prostate cancer: A multiethnic case-control study. Cancer Epidemiol. Biomark. Prev. 2000, 9, 795–804. [Google Scholar]
- Chen, Y.-J.; Wallig, M.A.; Jeffery, E.H. Dietary Broccoli Lessens Development of Fatty Liver and Liver Cancer in Mice Given Diethylnitrosamine and Fed a Western or Control Diet. J. Nutr. 2016, 146, 542–550. [Google Scholar] [CrossRef]
- Matusheski, N.V.; Swarup, R.; Juvik, J.A.; Mithen, R.; Bennett, M.; Jeffery, E.H. Epithiospecifier protein from broccoli (Brassica oleracea L. ssp. italica) inhibits formation of the anticancer agent sulforaphane. J. Agric. Food Chem. 2006, 54, 2069–2076. [Google Scholar] [CrossRef]
- Charron, C.S.; Vinyard, B.T.; Ross, S.A.; Seifried, H.E.; Jeffery, E.H.; Novotny, J.A. Absorption and metabolism of isothiocyanates formed from broccoli glucosinolates: Effects of BMI and daily consumption in a randomised clinical trial. Br. J. Nutr. 2018, 120, 1370–1379. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Y.; Hoeflinger, J.L.; Neme, B.P.; Jeffery, E.H.; Miller, M.J. Dietary Broccoli Alters Rat Cecal Microbiota to Improve Glucoraphanin Hydrolysis to Bioactive Isothiocyanates. Nutrients 2017, 9, 262. [Google Scholar] [CrossRef]
- Paturi, G.; Butts, C.A.; Stoklosinski, H.; Herath, T.D.; Monro, J.A. Short-term feeding of fermentable dietary fibres influences the gut microbiota composition and metabolic activity in rats. Int. J. Food Sci. Technol. 2017, 52, 2572–2581. [Google Scholar] [CrossRef]
- Kaczmarek, J.L.; Charron, C.S.; Novotny, J.A.; Jeffery, E.H.; Seifried, H.E.; Ross, S.A.; Swanson, K.S.; Holscher, H.D. Broccoli Consumption Impacts the Human Gastrointestinal Microbiota. FASEB J. 2017, 31, 965-18. [Google Scholar] [CrossRef]
- Angelino, D.; Dosz, E.B.; Sun, J.; Hoeflinger, J.L.; Van Tassell, M.L.; Chen, P.; Harnly, J.M.; Miller, M.J.; Jeffery, E.H. Myrosinase-dependent and-independent formation and control of isothiocyanate products of glucosinolate hydrolysis. Front. Plant Sci. 2015, 6, 831. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Jordan, B.F. Gut microbiota-mediated inflammation in obesity: A link with gastrointestinal cancer. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Osto, M.; Geurts, L.; Everard, A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes 2012, 3, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, E.H.; Keck, A.S. Translating knowledge generated by epidemiological and in vitro studies into dietary cancer prevention. Mol. Nutr. Food Res. 2008, 52, S7–S17. [Google Scholar] [CrossRef]
- Mi, L.; Xiao, Z.; Hood, B.L.; Dakshanamurthy, S.; Wang, X.; Govind, S.; Conrads, T.P.; Veenstra, T.D.; Chung, F.-L. Covalent Binding to Tubulin by Isothiocyanates: A Mechanism of cell growth arrest and apoptosis. J. Biol. Chem. 2008, 283, 22136–22146. [Google Scholar] [CrossRef]
- Agerbirk, N.; De Nicola, G.R.; Olsen, C.E.; Müller, C.; Iori, R. Derivatization of isothiocyanates and their reactive adducts for chromatographic analysis. Phytochemistry 2015, 118, 109–115. [Google Scholar] [CrossRef]
- Steinbrecher, U.P. Oxidation of human low density lipoprotein results in derivatization of lysine residues of apolipoprotein B by lipid peroxide decomposition products. J. Biol. Chem. 1987, 262, 3603–3608. [Google Scholar]
- Conaway, C.C.; Getahun, S.M.; Liebes, L.L.; Pusateri, D.J.; Topham, D.K.; Botero-Omary, M.; Chung, F.L. Disposition of glucosinolates and sulforaphane in humans after ingestion of steamed and fresh broccoli. Nutr. Cancer 2000, 38, 168–178. [Google Scholar] [CrossRef]
- Vermeulen, M.; Klopping-Ketelaars, I.W.; van den Berg, R.; Vaes, W.H. Bioavailability and kinetics of sulforaphane in humans after consumption of cooked versus raw broccoli. J. Agric. Food Chem. 2008, 56, 10505–10509. [Google Scholar] [CrossRef] [PubMed]
- Palazoglu, M.; Fiehn, O. Metabolite Identification in Blood Plasma Using GC/MS and the Agilent Fiehn GC/MS Metabolomics RTL Library; Mine Palazoglu and Oliver FiehnUC Davis Genome CenterDavis: Davis, CA, USA, 2009. [Google Scholar]
- Borgogna, J.-L.C.; Shardell, M.D.; Yeoman, C.J.; Ghanem, K.G.; Kadriu, H.; Ulanov, A.V.; Gaydos, C.A.; Hardick, J.; Robinson, C.K.; Bavoil, P.M.; et al. The association of Chlamydia trachomatis and Mycoplasma genitalium infection with the vaginal metabolome. Sci. Rep. 2020, 10, 3420. [Google Scholar] [CrossRef] [PubMed]
- Wild, A.C.; Moinova, H.R.; Mulcahy, R.T. Regulation of gamma-glutamylcysteine synthetase subunit gene expression by the transcription factor Nrf2. J. Biol. Chem. 1999, 274, 33627–33636. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Zhang, Y. Total intracellular accumulation levels of dietary isothiocyanates determine their activity in elevation of cellular glutathione and induction of Phase 2 detoxification enzymes. Carcinogenesis 2001, 22, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Senthilkumar, G.P.; Sivaraman, K.; Bobby, Z.; Paneerselvam, S.; Harichandrakumar, K.T. Effect of s-methyl-L-cysteine on oxidative stress, inflammation and insulin resistance in male wistar rats fed with high fructose diet. Iran. J. Med. Sci. 2015, 40, 45–50. [Google Scholar]
- Shrestha, N.; Chand, L.; Han, M.K.; Lee, S.O.; Kim, C.Y.; Jeong, Y.J. Glutamine inhibits CCl4 induced liver fibrosis in mice and TGF-β1 mediated epithelial–mesenchymal transition in mouse hepatocytes. Food Chem. Toxicol. 2016, 93, 129–137. [Google Scholar] [CrossRef]
- Housley, L.; Magana, A.A.; Hsu, A.; Beaver, L.M.; Wong, C.P.; Stevens, J.F.; Choi, J.; Jiang, Y.; Bella, D.; Williams, D.E.; et al. Untargeted Metabolomic Screen Reveals Changes in Human Plasma Metabolite Profiles Following Consumption of Fresh Broccoli Sprouts. Mol. Nutr. Food Res. 2018, 62, 1700665. [Google Scholar] [CrossRef]
- Shibahara, A.; Yamamoto, K.; Takeoka, M.; Kinoshita, A.; Kajimoto, G.; Nakayama, T.; Noda, M. Novel pathways of oleic and cis-vaccenic acid biosynthesis by an enzymatic double-bond shifting reaction in higher plants. FEBS Lett. 1990, 264, 228–230. [Google Scholar] [CrossRef]
- Mazidi, M.; Katsiki, N.; Mikhailidis, D.P.; Banach, M. Link between plasma trans-fatty acid and fatty liver is moderated by adiposity. Int. J. Cardiol. 2018, 272, 316–322. [Google Scholar] [CrossRef]
- Maciejewska, D.; Palma, J.; Dec, K.; Skonieczna-Żydecka, K.; Gutowska, I.; Szczuko, M.; Jakubczyk, K.; Stachowska, E. Is the Fatty Acids Profile in Blood a Good Predictor of Liver Changes? Correlation of Fatty Acids Profile with Fatty Acids Content in the Liver. Diagnostics 2019, 9, 197. [Google Scholar] [CrossRef]
- Cansanção, K.; Silva Monteiro, L.; Carvalho Leite, N.; Dávalos, A.; Tavares do Carmo, M.D.G.; Arantes Ferreira Peres, W. Advanced Liver Fibrosis Is Independently Associated with Palmitic Acid and Insulin Levels in Patients with Non-Alcoholic Fatty Liver Disease. Nutrients 2018, 10, 1586. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Dai, Z.; Liu, A.B.; Huang, J.; Narsipur, N.; Guo, G.; Kong, B.; Reuhl, K.; Lu, W.; Luo, Z.; et al. Intake of stigmasterol and β-sitosterol alters lipid metabolism and alleviates NAFLD in mice fed a high-fat western-style diet. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 1274–1284. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Park, Y. Conjugated nonadecadienoic acid is more potent than conjugated linoleic acid on body fat reduction. J. Nutr. Biochem. 2010, 21, 764–773. [Google Scholar] [CrossRef] [PubMed]
| Sample (Rat #) | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time (Days) | 0 | 0 | 0 | 1 | 1 | 1 | 2 | 2 | 2 | 4 | 4 | 4 | 7 | 7 | 7 | 14 | 14 | 14 |
| 11-Octadecenoic acid | 38.7 | 51.6 | 44.2 | 40.9 | 45 | 51.2 | 45.2 | 46.1 | 38.3 | 31.6 | 32.5 | 28.9 | 36.8 | 29.9 | 27.2 | 39.3 | 34.4 | 32.8 |
| 1-Monohexadecanoylglycerol | 16.5 | 20.2 | 13.7 | 25 | 15.6 | 16 | 14.1 | 14.3 | 12.5 | 11.4 | 11.6 | 10.8 | 10.7 | 10.8 | 12.1 | 9.4 | 7.9 | 6.1 |
| 1-Monooctadecanoylglycerol | 14 | 17.2 | 14.5 | 17.4 | 15.3 | 18.3 | 13.9 | 15.6 | 13.5 | 13.2 | 13.8 | 16 | 8.9 | 10.8 | 10.5 | 8.8 | 7.9 | 5.8 |
| 2-Octadecanoylglycerol | 1.9 | 1.4 | 2 | 2.8 | 2.5 | 2.3 | 1.4 | 2.6 | 2.2 | 1.7 | 1.9 | 0.8 | 1.5 | 0.5 | 1.4 | 0.9 | 0.1 | 0.1 |
| 5,8,11,14-Eicosatetraenoic acid | 117.2 | 145.2 | 124.4 | 116 | 96.8 | 104.9 | 106.8 | 105.6 | 101.5 | 91.7 | 102.9 | 107.8 | 107.2 | 95 | 81.3 | 95.4 | 72.1 | 55.4 |
| Azelaic acid | 1.3 | 0.6 | 0.8 | 0.6 | 0.5 | 0.5 | 0.7 | 1 | 1 | 0.1 | 0.3 | 0.1 | 0.4 | 0.4 | 0.1 | 0.1 | 0.1 | 0.5 |
| Benzenepropanoic acid | 1.1 | 0.6 | 0.6 | 0.6 | 0.8 | 0.5 | 0.6 | 1 | 0.8 | 0.6 | 0.8 | 1 | 0.8 | 1.2 | 1 | 0.1 | 0.1 | 0.1 |
| Chenodeoxycholic acid | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 3.3 | 0.7 | 0.9 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Cholesta-3,5-diene | 237.8 | 207.4 | 108.9 | 151 | 127.2 | 154 | 150.3 | 172 | 123.6 | 124.7 | 120.3 | 163.7 | 125.9 | 124.7 | 77 | 84.4 | 57.3 | 58.7 |
| Glutamine | 1.8 | 6.5 | 2.6 | 33.2 | 18.2 | 20.5 | 18.5 | 44.6 | 27.4 | 30.3 | 25.5 | 37.4 | 66.4 | 52.9 | 47.6 | 50.4 | 17.6 | 2.2 |
| Heptadecanoic acid | 0.4 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.4 | 0.6 | 0.5 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Inositol-1-p | 7.7 | 11.8 | 5.9 | 9.1 | 6.5 | 6.6 | 5.5 | 8.6 | 9.3 | 10.9 | 13.7 | 15 | 15.2 | 17.1 | 15.6 | 26 | 25.9 | 8.9 |
| Maltotriose | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 1.3 | 1.9 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Nonadecanoic acid | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.9 | 1.1 | 1 | 0.1 | 0.1 | 1.7 | 1.2 | 0.9 |
| Oleamide | 0.1 | 0.1 | 0.4 | 0.1 | 0.3 | 0.1 | 0.1 | 0.1 | 0.6 | 0.1 | 0.2 | 0.1 | 1 | 0.9 | 2.8 | 0.1 | 0.1 | 0.1 |
| Sitosterol | 16.5 | 20 | 7.6 | 25.8 | 16 | 14.7 | 15.3 | 13.9 | 14.8 | 9.6 | 18.2 | 20.7 | 35.2 | 40.7 | 23.5 | 57.7 | 51.8 | 27 |
| S-Methyl-L-cysteine | 0.4 | 0.3 | 0.2 | 0.4 | 3.3 | 3.2 | 5.2 | 12.2 | 6.8 | 6 | 10.1 | 6.7 | 6.8 | 7.2 | 8.6 | 3.5 | 2.5 | 1.5 |
| Stearic acid | 306.8 | 360.5 | 271.3 | 296.9 | 247.2 | 264.2 | 278 | 282.6 | 255.1 | 255.9 | 255.5 | 249.1 | 255 | 230.3 | 208.1 | 227.8 | 231.1 | 223.2 |
| Tartaric acid | 0.1 | 0.1 | 0.1 | 5.2 | 3.4 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Tetracosanol | 0.1 | 0.1 | 0.1 | 0.3 | 0.3 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Threonic acid | 3.3 | 2.4 | 1.7 | 3.1 | 2.1 | 3.2 | 6.5 | 4.2 | 4.5 | 6.7 | 3.4 | 5.5 | 4.6 | 4.7 | 8.3 | 15 | 13.2 | 4.5 |
| Sample (Rat #) | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time (Days) | 0 | 0 | 0 | 1 | 1 | 1 | 2 | 2 | 2 | 4 | 4 | 4 | 7 | 7 | 7 | 14 | 14 | 14 |
| Bacteria;Actinobacteria;Actinobacteria;Coriobacteriales;Coriobacteriaceae;Gordonibacter | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00004 | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00016 | 0.00008 | 0.00000 | 0.00057 | 0.00009 | 0.00024 |
| Bacteria;Bacteroidetes;Bacteroidia;Bacteroidales;Porphyromonadaceae;Tannerella | 0.04213 | 0.04601 | 0.01162 | 0.00223 | 0.00485 | 0.00379 | 0.00252 | 0.01583 | 0.01213 | 0.00988 | 0.00238 | 0.00443 | 0.00701 | 0.01258 | 0.00639 | 0.01220 | 0.01546 | 0.03028 |
| Bacteria;Bacteroidetes;Bacteroidia;Bacteroidales;Rikenellaceae;Alistipes | 0.01398 | 0.01098 | 0.01540 | 0.01043 | 0.01712 | 0.00923 | 0.00270 | 0.00425 | 0.00758 | 0.00204 | 0.00217 | 0.00364 | 0.00151 | 0.00833 | 0.00846 | 0.00221 | 0.00880 | 0.00302 |
| Bacteria;Bacteroidetes;Flavobacteriia;Flavobacteriales;Flavobacteriaceae;Lutaonella | 0.00022 | 0.00017 | 0.00023 | 0.00022 | 0.00011 | 0.00022 | 0.00004 | 0.00000 | 0.00004 | 0.00000 | 0.00004 | 0.00000 | 0.00016 | 0.00008 | 0.00044 | 0.00005 | 0.00014 | 0.00032 |
| Bacteria;Firmicutes;Bacilli;Lactobacillales;Streptococcaceae;Lactococcus | 0.00130 | 0.00063 | 0.00062 | 0.00011 | 0.00100 | 0.00082 | 0.00004 | 0.00016 | 0.00016 | 0.00013 | 0.00026 | 0.00011 | 0.00020 | 0.00012 | 0.00007 | 0.00009 | 0.00032 | 0.00012 |
| Bacteria;Firmicutes;Bacilli;Lactobacillales;Streptococcaceae;Streptococcus | 0.00022 | 0.00059 | 0.00039 | 0.00019 | 0.00054 | 0.00065 | 0.00004 | 0.00004 | 0.00004 | 0.00009 | 0.00009 | 0.00008 | 0.00004 | 0.00000 | 0.00007 | 0.00005 | 0.00005 | 0.00008 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Gracilibacteraceae;Lutispora | 0.00259 | 0.00130 | 0.00151 | 0.00115 | 0.00118 | 0.00000 | 0.00029 | 0.00008 | 0.00012 | 0.00035 | 0.00021 | 0.00049 | 0.00012 | 0.00045 | 0.00025 | 0.00047 | 0.00054 | 0.00060 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Lachnospiraceae;Anaerostipes | 0.01658 | 0.00799 | 0.01192 | 0.00648 | 0.00696 | 0.01793 | 0.00721 | 0.01109 | 0.01320 | 0.11612 | 0.02008 | 0.02795 | 0.05762 | 0.02817 | 0.01219 | 0.02831 | 0.02562 | 0.03390 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Lachnospiraceae;Blautia | 0.00000 | 0.00004 | 0.00000 | 0.00052 | 0.00039 | 0.00056 | 0.00242 | 0.00182 | 0.01459 | 0.01594 | 0.00434 | 0.01174 | 0.00785 | 0.02475 | 0.00073 | 0.00334 | 0.00041 | 0.00511 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Lachnospiraceae;Dorea | 0.00179 | 0.00055 | 0.00096 | 0.00015 | 0.00389 | 0.00203 | 0.00231 | 0.00113 | 0.00057 | 0.00022 | 0.00009 | 0.00080 | 0.00291 | 0.00544 | 0.00290 | 0.00250 | 0.00553 | 0.00298 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Anaerotruncus | 0.00773 | 0.00622 | 0.00552 | 0.00454 | 0.00300 | 0.00496 | 0.00508 | 0.00628 | 0.00529 | 0.00403 | 0.00345 | 0.00417 | 0.00458 | 0.00561 | 0.00501 | 0.00480 | 0.00744 | 0.00709 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Butyricicoccus | 0.00634 | 0.00509 | 0.00201 | 0.00521 | 0.00278 | 0.00625 | 0.00523 | 0.00211 | 0.00221 | 0.00100 | 0.00119 | 0.00110 | 0.00199 | 0.00334 | 0.00076 | 0.00014 | 0.00073 | 0.00246 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Cellulosibacter | 0.00701 | 0.00488 | 0.00513 | 0.01426 | 0.00927 | 0.01440 | 0.01049 | 0.00340 | 0.00635 | 0.00308 | 0.00208 | 0.00159 | 0.00143 | 0.00210 | 0.00330 | 0.00207 | 0.00195 | 0.00085 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Ethanoligenens | 0.00791 | 0.00358 | 0.00139 | 0.00190 | 0.00132 | 0.00491 | 0.01248 | 0.00753 | 0.00090 | 0.01716 | 0.00608 | 0.00072 | 0.02180 | 0.01089 | 0.00693 | 0.01319 | 0.02852 | 0.04022 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Oscillibacter | 0.02015 | 0.02721 | 0.04581 | 0.00957 | 0.01455 | 0.02530 | 0.02095 | 0.03862 | 0.02172 | 0.04541 | 0.03421 | 0.03314 | 0.04610 | 0.06702 | 0.08965 | 0.06104 | 0.04217 | 0.04199 |
| Bacteria;Firmicutes;Clostridia;Clostridiales;Ruminococcaceae;Papillibacter | 0.09673 | 0.13156 | 0.12280 | 0.07307 | 0.07844 | 0.00263 | 0.02384 | 0.00822 | 0.06857 | 0.00368 | 0.00655 | 0.02019 | 0.00327 | 0.00532 | 0.01201 | 0.04371 | 0.00290 | 0.01546 |
| Bacteria;Proteobacteria;Deltaproteobacteria;Bdellovibrionales;Bdellovibrionaceae;Vampirovibrio | 0.00398 | 0.00265 | 0.00513 | 0.00007 | 0.00221 | 0.00026 | 0.00126 | 0.00000 | 0.00164 | 0.00026 | 0.00009 | 0.00019 | 0.00008 | 0.00008 | 0.00011 | 0.00108 | 0.00113 | 0.00016 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eve, A.A.; Liu, X.; Wang, Y.; Miller, M.J.; Jeffery, E.H.; Madak-Erdogan, Z. Biomarkers of Broccoli Consumption: Implications for Glutathione Metabolism and Liver Health. Nutrients 2020, 12, 2514. https://doi.org/10.3390/nu12092514
Eve AA, Liu X, Wang Y, Miller MJ, Jeffery EH, Madak-Erdogan Z. Biomarkers of Broccoli Consumption: Implications for Glutathione Metabolism and Liver Health. Nutrients. 2020; 12(9):2514. https://doi.org/10.3390/nu12092514
Chicago/Turabian StyleEve, Alicia Arredondo, Xiaoji Liu, Yanling Wang, Michael J. Miller, Elizabeth H. Jeffery, and Zeynep Madak-Erdogan. 2020. "Biomarkers of Broccoli Consumption: Implications for Glutathione Metabolism and Liver Health" Nutrients 12, no. 9: 2514. https://doi.org/10.3390/nu12092514
APA StyleEve, A. A., Liu, X., Wang, Y., Miller, M. J., Jeffery, E. H., & Madak-Erdogan, Z. (2020). Biomarkers of Broccoli Consumption: Implications for Glutathione Metabolism and Liver Health. Nutrients, 12(9), 2514. https://doi.org/10.3390/nu12092514







