Evaluation of Glycemic Index Education in People Living with Type 2 Diabetes: Participant Satisfaction, Knowledge Uptake, and Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Research Ethics Review
2.2. Participants
2.3. Intervention
2.4. Data Collection
2.5. Statistical Analysis
2.5.1. Quantitative Analysis
2.5.2. Qualitative Analysis
3. Results
3.1. Participant Characteristics
3.1.1. Satisfaction—Kirkpatrick Model L1
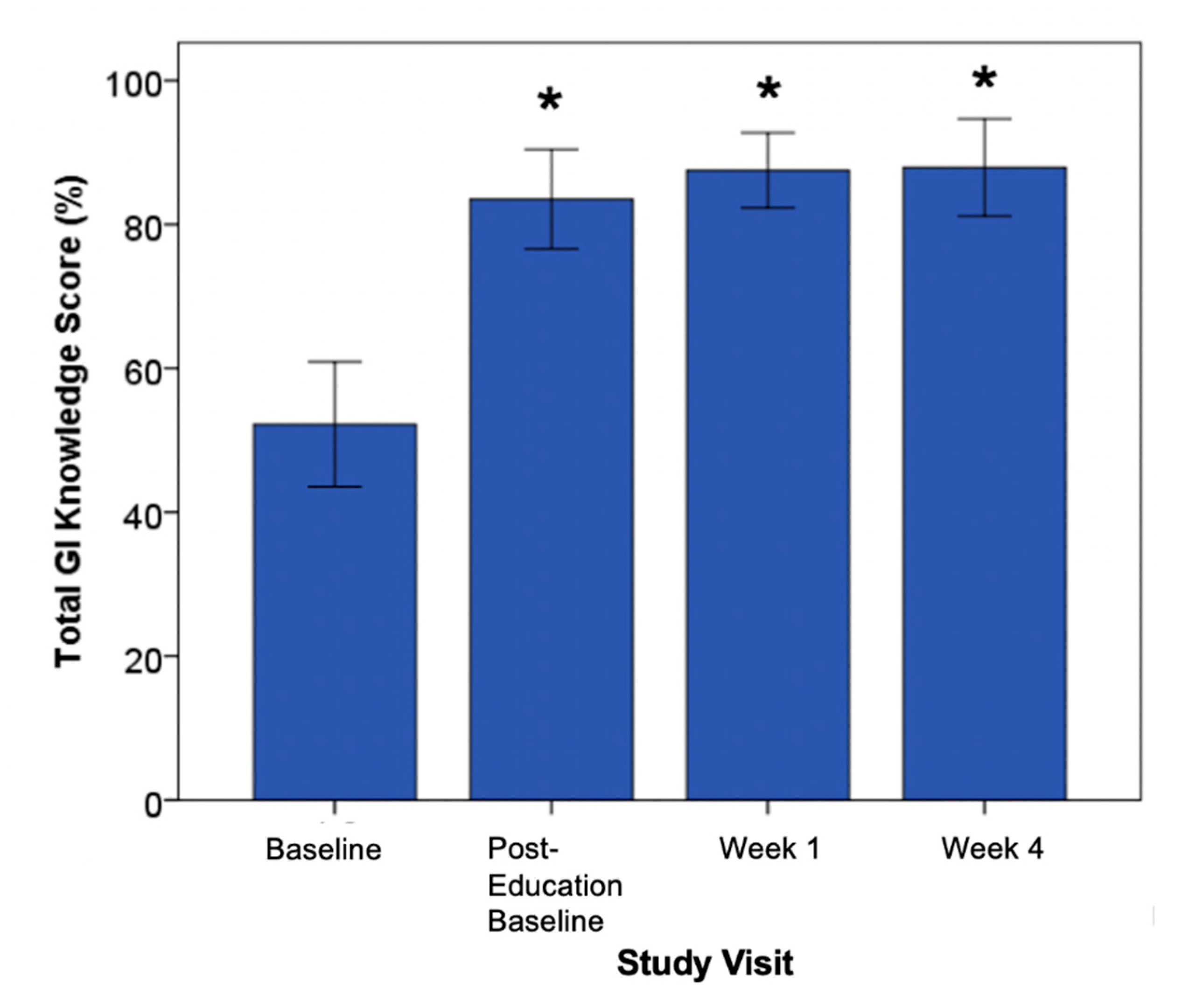
3.1.2. Knowledge Uptake—Kirkpatrick Model L2
3.1.3. Behavior Change/Transfer—Kirkpatrick L3
4. Discussion
4.1. Strengths and Limitations
4.2. Implications for Practice and Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Canadian Diabetes Association Clinical Practice Guidelines Expert Committee; Dworatzek, P.D.; Arcudi, K.; Gougeon, R.; Husein, N.; Sievenpiper, J.L.; Williams, S.L. Nutrition therapy. Can. J. Diabetes 2013, 37 (Suppl. S1), S45–S55. [Google Scholar] [CrossRef]
- Diabetes Canada Clinical Practice Guidelines Expert Committee; Sievenpiper, J.L.; Chan, C.B.; Dworatzek, P.D.; Freeze, C.; Williams, S.L. Nutrition Therapy. Can. J. Diabetes 2018, 42 (Suppl. S1), S64–S79. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 4. Lifestyle Management: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018, 41, S38–S50. [Google Scholar] [CrossRef] [PubMed]
- Dyson, P.A.; Twenefour, D.; Breen, C.; Duncan, A.; Elvin, E.; Goff, L.; Hill, A.; Kalsi, P.; Marsland, N.; McArdle, P.; et al. Diabetes UK evidence-based nutrition guidelines for the prevention and management of diabetes. Diabet. Med. 2018, 35, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.I.; De Leeuw, I.; Hermansen, K.; Karamanos, B.; Karlstrom, B.; Katsilambros, N.; Riccardi, G.; Rivellese, A.A.; Rizkalla, S.; Slama, G.; et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 373–394. [Google Scholar] [CrossRef]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias. Eur. Heart J. 2016, 37, 2999–3058. [Google Scholar] [CrossRef]
- Anderson, T.J.; Gregoire, J.; Pearson, G.J.; Barry, A.R.; Couture, P.; Dawes, M.; Francis, G.A.; Genest, J., Jr.; Grover, S.; Gupta, M.; et al. 2016 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in the Adult. Can. J. Cardiol. 2016, 32, 1263–1282. [Google Scholar] [CrossRef]
- The Royal Australian College of General Practitioners. Managemenrt of Type 2 Diabetes: A Handbook for General Practice. Available online: https://www.racgp.org.au/clinical-resources/clinical-guidelines/key-racgp-guidelines/view-all-racgp-guidelines/diabetes/introduction (accessed on 28 July 2020).
- Kalergis, M.; Pytka, E.; Yale, J.F.; Mayo, N.; Strychar, I. Canadian dietitians’ use and perceptions of glycemic index in diabetes management. Can. J. Diet. Pract. Res. 2006, 67, 21–27. [Google Scholar] [CrossRef]
- Grant, S.M.; Wolever, T.M. Perceived barriers to application of glycaemic index: Valid concerns or lost in translation? Nutrients 2011, 3, 330–340. [Google Scholar] [CrossRef]
- Kalergis, M. The role of the glycemic index in the prevention and management of diabetes: A review and discussion. Can. J. Diabetes 2004, 29, 27–38. [Google Scholar]
- Slabber, M. Complexities of consumer understanding of the glycaemic index concept and practical guidelines for incorporation in diets. S. Afr. J. Clin. Nutr. 2005, 18, 252–257. [Google Scholar] [CrossRef]
- Doak, C.D.L.; Root, J. Teaching Patients with Low Literacy Skills, 2nd ed.; Lipincott: Philadelphia, PA, USA, 1996. [Google Scholar]
- Southgate, K.; Wolever, T.M.S. Integrating the glycemic index into clinical practice: Written education materials and perception of utility. Can. J. Diabetes 2012, 36, 54–57. [Google Scholar] [CrossRef][Green Version]
- Grant, S.M.; Wolever, T.M.; O’Connor, D.L.; Nisenbaum, R.; Josse, R.G. Effect of a low glycaemic index diet on blood glucose in women with gestational hyperglycaemia. Diabetes Res. Clin. Pract. 2011, 91, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Fontvieille, A.M.; Rizkalla, S.W.; Penfornis, A.; Acosta, M.; Bornet, F.R.; Slama, G. The use of low glycaemic index foods improves metabolic control of diabetic patients over five weeks. Diabet. Med. 1992, 9, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Moses, R.G.; Casey, S.A.; Quinn, E.G.; Cleary, J.M.; Tapsell, L.C.; Milosavljevic, M.; Petocz, P.; Brand-Miller, J.C. Pregnancy and Glycemic Index Outcomes study: Effects of low glycemic index compared with conventional dietary advice on selected pregnancy outcomes. Am. J. Clin. Nutr. 2014, 99, 517–523. [Google Scholar] [CrossRef]
- Moses, R.G.; Barker, M.; Winter, M.; Petocz, P.; Brand-Miller, J.C. Can a low-glycemic index diet reduce the need for insulin in gestational diabetes mellitus? A randomized trial. Diabetes Care 2009, 32, 996–1000. [Google Scholar] [CrossRef]
- Miller, C.K.; Lindberg, D.V. Evaluation of a computer-based game about the glycemic index among college-aged students. Top. Clin. Nutr. 2007, 22, 299–306. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Kendall, C.W.; McKeown-Eyssen, G.; Josse, R.G.; Silverberg, J.; Booth, G.L.; Vidgen, E.; Josse, A.R.; Nguyen, T.H.; Corrigan, S.; et al. Effect of a low-glycemic index or a high-cereal fiber diet on type 2 diabetes: A randomized trial. JAMA 2008, 300, 2742–2753. [Google Scholar] [CrossRef]
- Mirrahimi, A.; Chiavaroli, L.; Srichaikul, K.; Augustin, L.S.; Sievenpiper, J.L.; Kendall, C.W.; Jenkins, D.J. The role of glycemic index and glycemic load in cardiovascular disease and its risk factors: A review of the recent literature. Curr. Atheroscler. Rep. 2014, 16, 381. [Google Scholar] [CrossRef]
- Zafar, M.I.; Mills, K.E.; Zheng, J.; Peng, M.M.; Ye, X.; Chen, L.L. Low glycaemic index diets as an intervention for obesity: A systematic review and meta-analysis. Obes. Rev. 2019, 20, 290–315. [Google Scholar] [CrossRef]
- Cambon, L.; Minary, L.; Ridde, V.; Alla, F. Transferability of interventions in health education: A review. BMC Public Health 2012, 12, 497. [Google Scholar] [CrossRef] [PubMed]
- Tarquinio, C.; Kivits, J.; Minary, L.; Coste, J.; Alla, F. Evaluating complex interventions: Perspectives and issues for health behaviour change interventions. Psychol. Health 2015, 30, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Bandura, A. Self-efficacy: Toward a unifying theory of behavioral change. Psychol. Rev. 1977, 84, 191–215. [Google Scholar] [CrossRef] [PubMed]
- Koska, J.; Blazicek, P.; Marko, M.; Grna, J.D.; Kvetnansky, R.; Vigas, M. Insulin, catecholamines, glucose and antioxidant enzymes in oxidative damage during different loads in healthy humans. Physiol. Res. 2000, 49 (Suppl. S1), S95–S100. [Google Scholar] [PubMed]
- Attree, M. Evaluating healthcare education: Issues and methods. Nurse Educ. Pract. 2006, 6, 332–338. [Google Scholar] [CrossRef]
- Bonell, C.; Fletcher, A.; Morton, M.; Lorenc, T.; Moore, L. Realist randomised controlled trials: A new approach to evaluating complex public health interventions. Soc. Sci. Med. 2012, 75, 2299–2306. [Google Scholar] [CrossRef]
- Knowledge Translation Canada. KT Canada. Available online: http://ktcanada.net/ (accessed on 12 January 2020).
- Canadian Institute of Health Research. A Guide to Researcher and Knowledge-User Collaboration in Health Research. Available online: https://cihr-irsc.gc.ca/e/44954.html (accessed on 12 January 2020).
- Barwick, M.; Lockett, D.M. Scientist Knowledge Translation Manual; The Hospital for Sick Children/Centre for Addictions and Mental Health: Toronto, ON, Canada, 2005. [Google Scholar]
- Berwick, D.M. Disseminating innovations in health care. JAMA 2003, 289, 1969–1975. [Google Scholar] [CrossRef]
- KirkPatrick Partners. The One and Only Kirkpatrick. Available online: https://www.kirkpatrickpartners.com/ (accessed on 12 January 2020).
- Miller, G.E. The assessment of clinical skills/competence/performance. Acad. Med. 1990, 65, S63–S67. [Google Scholar] [CrossRef]
- Kern, D.E.T.P.A.; Hughes, M.T. Curriculum Development for Medical Education: A Six-Step Approach Paperback, 2nd ed.; Hopkins Fulfullment Service: Baltimore, MD, USA, 2009. [Google Scholar]
- Leslie, K.; Baker, L.; Egan-Lee, E.; Esdaile, M.; Reeves, S. Advancing faculty development in medical education: A systematic review. Acad. Med. 2013, 88, 1038–1045. [Google Scholar] [CrossRef]
- Peterson, R.A. Constructing Effective Questionnaires; SAGE Publications Incorporated: Thousand Oaks, CA, USA, 2000. [Google Scholar]
- Burani, J.; Longo, P.J. Low-glycemic index carbohydrates: An effective behavioral change for glycemic control and weight management in patients with type 1 and 2 diabetes. Diabetes Educ. 2006, 32, 78–88. [Google Scholar] [CrossRef]
- Grant, S.M.W.T.M.S. Glycemic Index Education: Translating Knowledge to Action. Available online: https://members.dietitians.ca/DCMember/LearnProduct?id=01tf4000003j6wJAAQ (accessed on 17 January 2020).
- Frost, G.; Wilding, J.; Beecham, J. Dietary advice based on the glycaemic index improves dietary profile and metabolic control in type 2 diabetic patients. Diabet. Med. 1994, 11, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.; Mehling, C. High-carbohydrate-low-glycaemic index dietary advice improves glucose disposition index in subjects with impaired glucose tolerance. Br. J. Nutr. 2002, 87, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Frost, G.; Keogh, B.; Smith, D.; Akinsanya, K.; Leeds, A. The effect of low-glycemic carbohydrate on insulin and glucose response in vivo and in vitro in patients with coronary heart disease. Metabolism 1996, 45, 669–672. [Google Scholar] [CrossRef]
- Health Canada. Eating Well with Canada’s Food Guide. Available online: https://www.canada.ca/en/health-canada/services/canada-food-guide/about/history-food-guide.html#a2007 (accessed on 17 January 2020).
- Franz, M.J.; Bantle, J.P.; Beebe, C.A.; Brunzell, J.D.; Chiasson, J.L.; Garg, A.; Holzmeister, L.A.; Hoogwerf, B.; Mayer-Davis, E.; Mooradian, A.D.; et al. Evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. Diabetes Care 2002, 25, 148–198. [Google Scholar] [CrossRef]
- Viguiliouk, E.; Nishi, S.K.; Wolever, T.M.S.; Sievenpiper, J.L. Point: Glycemic Index—An important but oft misunderstood marker of carbohydrate quality. Cereal Foods World 2018, 63, 158–164. [Google Scholar] [CrossRef]
- The University of Sydney. The Glycemic Index. Available online: http://www.glycemicindex.com/ (accessed on 17 January 2020).
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef]
- Emrich, T.E.; Mendoza, J.E.; L’Abbe, M.R. Effectiveness of front-of-pack nutrition symbols: A pilot study with consumers. Can. J. Diet. Pract. Res. 2012, 73, 200–203. [Google Scholar] [CrossRef]
- Emrich, T.E.; Qi, Y.; Lou, W.Y.; L’Abbe, M.R. Traffic-light labels could reduce population intakes of calories, total fat, saturated fat, and sodium. PLoS ONE 2017, 12, e0171188. [Google Scholar] [CrossRef]
- Taylor-Powell, E.; Renner, M. Analyzing Qualitative Data; University of Wisconsin-Extension: Madison, WI, USA, 2003. [Google Scholar]
- Willms, D.G.; Best, J.A.; Taylor, D.W.; Gilbert, J.R.; Wilson, D.M.; Lindsay, E.A.; Singer, J. A systematic approach for using qualitative methods in primary prevention research. Med. Anthropol. Q. 1990, 4, 391–409. [Google Scholar] [CrossRef]
- LaPelle, N.R. Simplifying Qualitative Data Analysis Using General Purpose Software Tools. Available online: https://escholarship.umassmed.edu/prevbeh_pp/84 (accessed on 29 May 2020).
- Pi-Sunyer, F.X. Glycemic index and disease. Am. J. Clin. Nutr. 2002, 76, 290S–298S. [Google Scholar] [CrossRef]
- Palmer, S. Finding a Place for the Glycemic Index. Available online: https://www.todaysdietitian.com/newarchives/060109p8.shtml (accessed on 29 May 2020).
- Grant, S.; Noseworthy, R.; Thompson, A.; Glenn, A.; Seider, M.; O’connor, D.; Josse, R.; Darling, P.; Wolever, T. The effect of low glycemic index education on satisfaction, knowledge, behaviour and glycaemic control in women with gestational diabetes. Can. J. Diabetes 2017, 41. [Google Scholar] [CrossRef]
- Lipscombe, L.L.; Delos-Reyes, F.; Glenn, A.J.; de Sequeira, S.; Liang, X.; Grant, S.; Thorpe, K.E.; Price, J.A.D. The Avoiding Diabetes After Pregnancy Trial in Moms Program: Feasibility of a Diabetes Prevention Program for Women With Recent Gestational Diabetes Mellitus. Can. J. Diabetes 2019, 43, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine & Health Canada. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/healthy-eating/dietary-reference-intakes/tables.html (accessed on 17 January 2020).
- Cleary, J.; Casey, S.; Hofsteede, C.; Moses, R.G.; Milosavljevic, M.; Brand-Miller, J. Does a low glycaemic index (GI) diet cost more during pregnancy? Nutrients 2012, 4, 1759–1766. [Google Scholar] [CrossRef] [PubMed]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias: The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS) Developed with the special contribution of the European Assocciation for Cardiovascular Prevention & Rehabilitation (EACPR). Atherosclerosis 2016, 253, 281–344. [Google Scholar] [CrossRef]
- Buttriss, J.L. Food and nutrition: Attitudes, beliefs, and knowledge in the United Kingdom. Am. J. Clin. Nutr. 1997, 65, 1985S–1995S. [Google Scholar] [CrossRef]
- Paquette, M.C. Perceptions of healthy eating: State of knowledge and research gaps. Can. J. Public Health 2005, 96 (Suppl. S3), S15–S19. [Google Scholar]
- Avedzi, H.M.; Mathe, N.; Storey, K.; Johnson, J.A.; Johnson, S.T. Examining sex differences in glycemic index knowledge and intake among individuals with type 2 diabetes. Prim. Care Diabetes 2018, 12, 71–79. [Google Scholar] [CrossRef]
- Larsen, T.M.; Dalskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunesova, M.; Pihlsgard, M.; et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Kendall, C.W.; Augustin, L.S.; Franceschi, S.; Hamidi, M.; Marchie, A.; Jenkins, A.L.; Axelsen, M. Glycemic index: Overview of implications in health and disease. Am. J. Clin. Nutr. 2002, 76, 266S–273S. [Google Scholar] [CrossRef]
- Braddon, K. Evaluating Pre- and Post-Education Knowledge Scores from Women with Gestational Diabetes Mellitus Interactiing with an Online Low Glycemic Index Education Platform. Master’s Thesis, Mount Saint Vincent University, Halifax Regional Municipality, NS, Canada, 2020. [Google Scholar]
- Statistics Canada. Focus on Geography Series, 2016 Census. Available online: https://www12.statcan.gc.ca/census-recensement/2016/as-sa/fogs-spg/Facts-CSD-eng.cfm?TOPIC=7&LANG=eng&GK=CSD&GC=3520005 (accessed on 29 July 2020).
- St. Michael’s Hospital. Facts about St. Michael’s. Available online: http://www.stmichaelshospital.com/about/snapshot.php (accessed on 29 July 2020).
- Watanabe, T.; Berry, T.R.; Willows, N.D.; Bell, R.C. Assessing intentions to eat low-glycemic index foods by adults with diabetes using a new questionnaire based on the theory of planned behaviour. Can. J. Diabetes 2015, 39, 94–100. [Google Scholar] [CrossRef]
- Louie, J.C.; Markovic, T.P.; Perera, N.; Foote, D.; Petocz, P.; Ross, G.P.; Brand-Miller, J.C. A randomized controlled trial investigating the effects of a low-glycemic index diet on pregnancy outcomes in gestational diabetes mellitus. Diabetes Care 2011, 34, 2341–2346. [Google Scholar] [CrossRef] [PubMed]
- Gunn, A. Nova Scotia Lone Province Where Child Poverty Rates Rising; The Chronicle Herald: Halifax, NS, Canada, 27 February 2019. [Google Scholar]
- Wilush, M. Threat of Diabetes Looms Large for Nova Scotians. Available online: https://multibriefs.com/briefs/VON/VON042016.php (accessed on 14 April 2016).
- Government of Canada. Canada’s Food Guide. Available online: https://food-guide.canada.ca/en/ (accessed on 23 July 2020).
- Diabetes Canada Clinical Practice Guidelines Expert, C.; Feig, D.S.; Berger, H.; Donovan, L.; Godbout, A.; Kader, T.; Keely, E.; Sanghera, R. Diabetes and Pregnancy. Can. J. Diabetes 2018, 42 (Suppl. S1), S255–S282. [Google Scholar] [CrossRef]
- Kirkpatrick Partners. The New World Kirkpatrick Model. Available online: https://www.kirkpatrickpartners.com/Our-Philosophy/The-New-World-Kirkpatrick-Model (accessed on 29 July 2020).
- Diabetes Canada. The Glycemic Index Food Guide. Available online: https://www.diabetes.ca/DiabetesCanadaWebsite/media/Managing-My-Diabetes/Tools%20and%20Resources/glycemic-index-food-guide.pdf?ext=.pdf (accessed on 9 May 2020).
- Diabetes Canada. Glycemic Index Food Cards. Available online: https://orders.diabetes.ca/products/glycemic-index-food-card?variant=2584625348634 (accessed on 8 May 2020).
- Grant, S.M.; Noseworthy, R. Translating glycemic index knowledge to action. Diabetes Commun. 2018, 19–20. [Google Scholar]
| Characteristic, n (%) | All Participants (n = 29) |
|---|---|
| Time since diabetes diagnosis, mean years ± SD | 7 ± 8 |
| Sex, female | 9 (31) |
| Ethnicity | |
| European descent | 18 (62) |
| Aboriginal | 2 (7) |
| African/Caribbean | 1 (3) |
| West Indian | 1 (3) |
| South Asian | 2 (7) |
| East Asian | 2 (7) |
| Southeast Asian | 2 (7) |
| Mixed | 1 (3) |
| Born in Canada | 15 (52) |
| English main language spoken at home | 24 (83) |
| Who purchases food most often? | |
| I do | 20 (69) |
| Spouse/partner | 5 (17) |
| Housekeeper | 1 (3) |
| Who cooks meals most often? | |
| I do | 19 (66) |
| Spouse/partner | 7 (24) |
| Children | 1 (3) |
| Housekeeper | 1 (3) |
| Highest level of education received | |
| Less than high school | 2 (7) |
| High school or equivalent | 2 (7) |
| College certificate or diploma | 10 (34) |
| Undergraduate degree or higher | 16 (55) |
| How diabetes is treated | |
| Lifestyle only | 3 (10) |
| Oral medications | 15 (51) |
| Oral medications + insulin | 9 (31) |
| Insulin | 2 (7) |
| Met with a dietitian before to discuss diet | 26 (90) |
| Heard of the glycemic index before | 24 (83) |
| Know what the glycemic index is | 19 (66) |
| Been taught about the glycemic index from a health care professional before | 5 (17) |
| Dietary Intake Outcome, Mean ± SEM | Baseline | Week 1 | Week 4 |
|---|---|---|---|
| Calories (kcal) | 1965 ± 67 | 1647 ± 73 * | 1631 ± 74 * |
| Carbohydrate, total (g) | 222 ± 8 | 183 ± 9 * | 187 ± 9 * |
| Fiber (g) | 21 ± 1 | 22 ± 1 | 23 ± 1 |
| Protein (g) | 95 ± 4 | 86 ± 4 | 91 ± 4 |
| Fat (g) | 76 ± 4 | 63 ± 4 * | 58 ± 4 * |
| Glycemic Index (%) | 58 ± 1 | 54 ± 1 * | 54 ± 1 * |
| Carbohydrate, total (% of total energy), mean | 44 | 45 | 44 |
| Fat (% of total energy), mean | 36 | 35 | 34 |
| Protein (% of total energy), mean | 20 | 20 | 22 |
| GIQ Question | Week 1, n (%) | Week 4, n (%) |
|---|---|---|
| I have added low GI foods to my diet since week 1 | 18 (75) | 21 (87) |
| Percentage of total intake has been made up of low GI foods | ||
| <51% | 15 (63) | 13 (54) |
| ≥51% | 9 (37) | 11 (46) |
| Your ability to choose low GI foods in the supermarket | ||
| Good or higher | 19 (79) | 17 (71) |
| Fair or less | 5 (21) | 7 (29) |
| Your ability to choose low GI foods when eating outside of the home | ||
| Good or higher | 13 (54) | 16 (63) |
| Fair or less | 11 (46) | 9 (37) |
| Your ability to include low GI foods in meal planning | ||
| Good or higher | 16 (67) | 18 (75) |
| Fair or less | 8 (33) | 6 (25) |
| Your ability to make traditional meals with low GI foods | ||
| Good or higher | 15 (65) | 18 (75) |
| Fair or less | 8 (35) | 6 (25) |
| The people you live with been eating low GI foods since week 1 | ||
| Yes | 8 (44) | 10 (67) |
| No | 10 (56) a | 5 (33) b |
| How would your house mates rate the low GI foods? | ||
| Good or higher | 11 (58) | 4 (25) |
| Fair or less | 8 (42) c | 12 (75) d |
| Planning meals with low GI foods does not require more time | 16 (70) | 18 (75) |
| Low GI foods cost the same as other foods | 17 (74) | 18 (75) |
| I will continue to eat low GI foods after the study is over | 23 (96) | 24 (100) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
M. Grant, S.; J. Glenn, A.; M. S. Wolever, T.; G. Josse, R.; L. O’Connor, D.; Thompson, A.; D. Noseworthy, R.; Seider, M.; Sobie, M.; Bhatti, G.; et al. Evaluation of Glycemic Index Education in People Living with Type 2 Diabetes: Participant Satisfaction, Knowledge Uptake, and Application. Nutrients 2020, 12, 2416. https://doi.org/10.3390/nu12082416
M. Grant S, J. Glenn A, M. S. Wolever T, G. Josse R, L. O’Connor D, Thompson A, D. Noseworthy R, Seider M, Sobie M, Bhatti G, et al. Evaluation of Glycemic Index Education in People Living with Type 2 Diabetes: Participant Satisfaction, Knowledge Uptake, and Application. Nutrients. 2020; 12(8):2416. https://doi.org/10.3390/nu12082416
Chicago/Turabian StyleM. Grant, Shannan, Andrea J. Glenn, Thomas M. S. Wolever, Robert G. Josse, Deborah L. O’Connor, Alexandra Thompson, Rebecca D. Noseworthy, Maxine Seider, Melissa Sobie, Gurita Bhatti, and et al. 2020. "Evaluation of Glycemic Index Education in People Living with Type 2 Diabetes: Participant Satisfaction, Knowledge Uptake, and Application" Nutrients 12, no. 8: 2416. https://doi.org/10.3390/nu12082416
APA StyleM. Grant, S., J. Glenn, A., M. S. Wolever, T., G. Josse, R., L. O’Connor, D., Thompson, A., D. Noseworthy, R., Seider, M., Sobie, M., Bhatti, G., Cavanagh, J., Jones, E., & B. Darling, P. (2020). Evaluation of Glycemic Index Education in People Living with Type 2 Diabetes: Participant Satisfaction, Knowledge Uptake, and Application. Nutrients, 12(8), 2416. https://doi.org/10.3390/nu12082416





