4.1. The Present Study
There is strong evidence in the literature establishing associations between symptoms of anxiety and depression with dietary choices [
1,
15,
20,
26]. However, the magnitude of these associations has yet to be established [
15,
26]. One of the challenges associated with quantifying the effects of symptoms of anxiety and depression on diet are inconsistencies in the measurement of diet [
15,
38]. Additionally, insufficient attention has been paid to the role of biologic sex in the relationship between anxiety, depression, and diet. The current study addresses these gaps and contributes to our understanding by estimating the impact of anxiety and depression symptoms on dietary quality and choices among college students while accounting for sex differences in these relationships. Indeed, symptoms of anxiety and depression were associated with reduced total caloric intake and increased sugar consumption for all participants. Men, in particular, appeared to consume more fat, and fewer fruits and vegetables as symptoms of depression increased.
The first set of multigroup path analyses confirmed Hypotheses 1a and 1b regarding caloric intake only. That is, within the anxiety global diet quality indices and depression global diet quality indices models, GAD-7 and PHQ-9 scores had an inverse relationship with total caloric intake but not HEI total score. The analyses failed to confirm Hypotheses 2a and 2b in that no sex differences were found in the relationship between GAD-7 score with HEI total score or total caloric intake, nor in the relationship between PHQ-9 score with HEI total score or total caloric intake. Overall, the results of the multigroup path analyses with the anxiety and depression global diet quality indices models suggest that anxiety and depression decrease total caloric intake for all subjects. However, there are no significant differences in this relationship across biologic sex on these global measures.
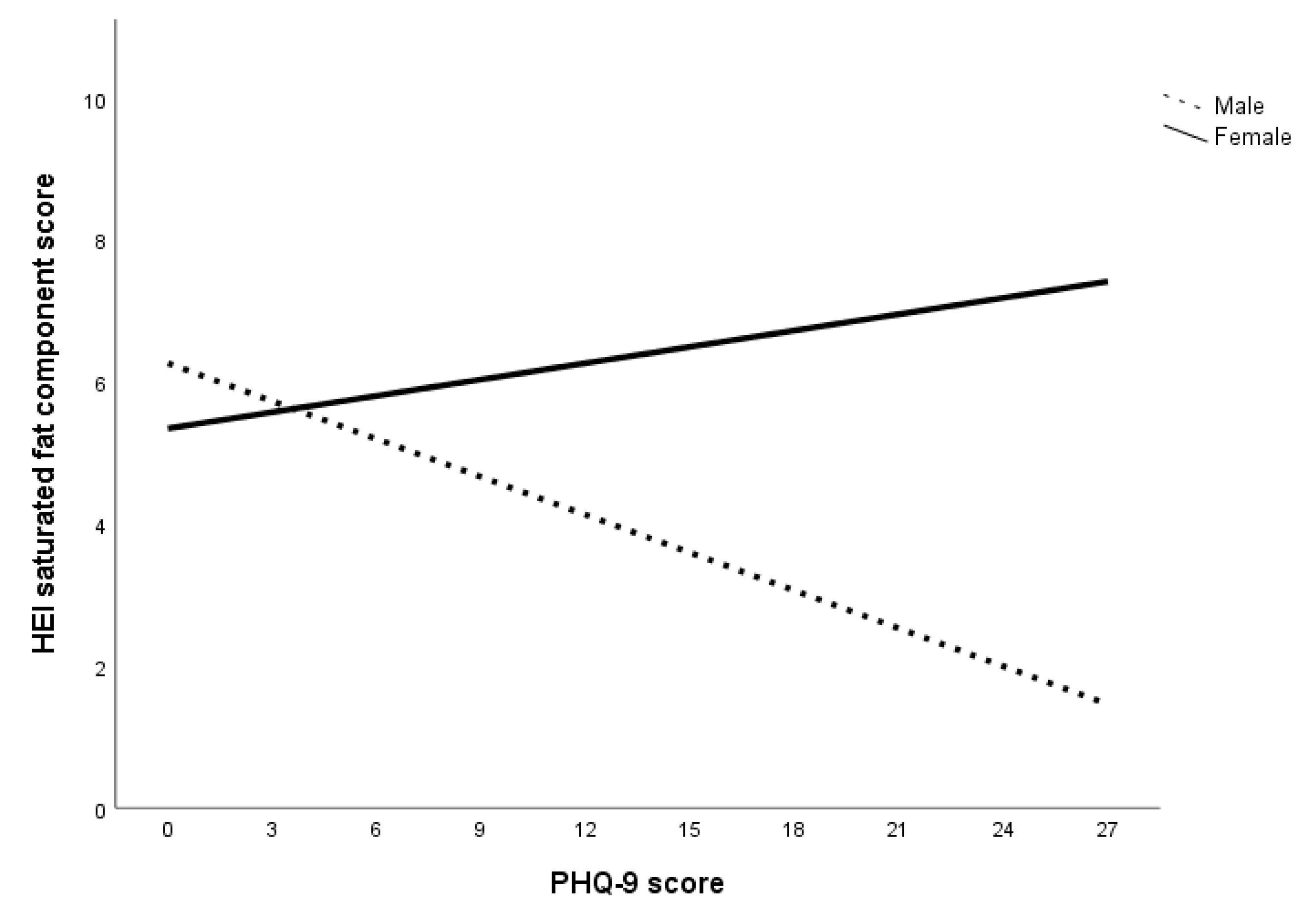
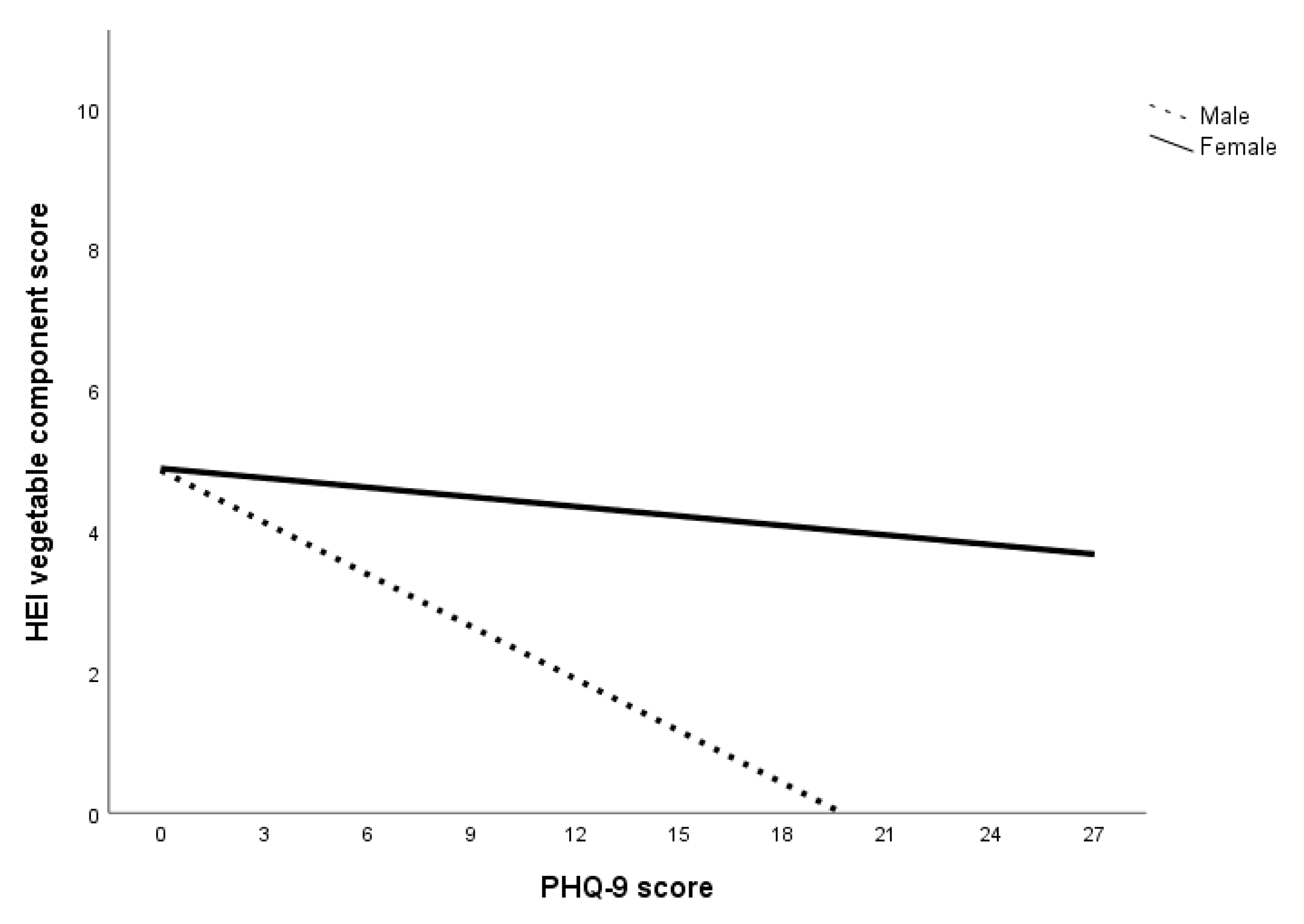
The second set of multigroup path analyses confirmed Hypotheses 3a and 3b regarding sugar only. That is, for all subjects within the anxiety, depression, and specific diet models, GAD-7 and PHQ-9 scores had an inverse relationship with HEI sugar component score but not HEI saturated fat component score. The analysis also failed to confirm Hypothesis 4a in that no sex differences were found in the relationship between GAD-7 score and HEI component scores. Within the depression specific diet model, there were sex differences in the relationship between PHQ-9 score and HEI component scores of saturated fat, fruits, and vegetables. Specifically, greater declines in these food components were observed in men compared to women. Overall, the results of the multigroup path analysis with the anxiety and depression specific diet indices models suggest that while both anxiety and depression affect sugar consumption for both sexes, depression was associated with reduced consumption of saturated fat, fruits, and vegetables in men.
The findings that total calorie consumption and HEI sugar component score decreased with higher levels of anxiety and depression are particularly interesting when examined together. Lower HEI sugar component score indicates an increase in energy contribution of sugar in the total energy consumed. In other words, sugar accounted for more energy contributions with higher levels of anxiety and depression. One explanation for these findings is that sugar consumption increases at higher levels of anxiety and depression. Anxiety can induce physiological changes that curb appetite while depression can lead to a reduction in motivation for activity, including eating [
26]. The availability and palatability of sweet foods make them an appealing and pleasurable option when individuals do not feel like eating but require energy [
67]. Parental practices that encourage the use of sweet foods to regulate mood may also explain the increase in sugar consumption during times of psychological distress [
68]. In short, while experiencing higher levels of anxiety and depression can cause a decrease in appetite, individuals may be more inclined to consume sweet foods when they do eat.
The results can also be attributed to a decrease in the consumption of low-sugar foods, especially considering the decrease in HEI component scores of saturated fat, fruits, and vegetables at higher levels of depression for men. A lower HEI saturated fat component score indicates an increase in the energy contribution of saturated fat in the total energy consumed while lower HEI component scores of fruits and vegetables indicate a decrease in fruits and vegetables consumption. In short, men who experienced higher levels of depression were more inclined to decrease total caloric intake as well as consumption of fruits and vegetables but not sugar and saturated fat. This is consistent with Grossniklaus, et al. [
69]’s suggestion that sweet and fatty foods are more palatable, as well as Boek, Bianco-Simeral, Chan and Goto [
31]’s finding that men are more motivated by taste when selecting foods.
Another interesting finding is that men with higher levels of depression were more likely to have worse diets (i.e., increased saturated fat, decreased fruit and vegetable intake) while women’s food choices were more stable. One reason for this sex discrepancy could be that women are more intrinsically motivated to select foods for health and nutrition while men’s food choices are driven by taste [
31,
32]. These motivations may endure with increased psychological distress, prompting women to continue to make healthier food choices while men select more palatable foods. Isasi, Ostrovsky and Wills [
68] suggest that emotion regulation may play a role in the association between depression and dietary choices. Specifically, individuals with better emotion regulation abilities are more likely to have increased self-efficacy and subsequently, better diet quality. Individuals with poor emotion regulation, on the other hand, are more likely to experience depressive symptoms and decreased self-efficacy, leading to a lower likelihood of sustaining diet quality [
68]. This is supported by an expectancy study done by Lyman [
70], who found that individuals tend to prefer healthy food (e.g., fruits and vegetables) when they are experiencing positive moods and unhealthy foods (e.g., sugar and fat) when experiencing negative moods. Christensen and Brooks [
71] suggest that consuming sweet and fatty foods trigger the release of opioids in the brain, improving mood and exponentially enhancing the palatability of the food. One possibility regarding the directionality of the relationship between mood and diet is that there is a cyclic effect of mood and food [
68,
70]. Specifically, while mood can affect food preferences, the foods consumed can also affect mood [
68,
70].
4.2. Clinical Impact
The findings regarding total calorie consumption are particularly important, given the implications for health. The decrease of 30.16 calories per 1-unit increase in GAD-7 score is equivalent to a decrease of up to 633.36 calories per day for someone with severe anxiety. The decrease of 27.44 calories per 1-unit increase in PHQ-9 score is equivalent to a decrease of up to 740.88 calories per day for someone with severe depression. This accounts for approximately 24%–41% of the recommended daily caloric intake for sedentary men and women (See
Table 7 for comparison) and can have a severe impact on physical wellbeing. This is particularly concerning given the associated risks for malnutrition, metabolic disturbances, and issues related to fertility [
72,
73,
74,
75]. Malnutrition and its subsequent impact on health is particularly concerning given the findings regarding increased consumption of sugar and fat as well as decreased consumption of fruits and vegetables with higher levels of anxiety and depression.
The study also showed decreased HEI component score of sugar with increased levels of anxiety and depression as well as decreased component scores of saturated fat, fruits, and vegetables with depressed men. This decrease in HEI component scores indicates an increase in sugar and fat contribution to total energy consumed as well as a decrease in fruits and vegetables consumption. The comparison of consumption of these diet indices compared to the daily intake recommended by USDA guidelines can be seen in
Tables S5 and S6 in
Supplementary Materials.
The increase in sugar and saturated fat contribution to total energy consumed is concerning considering its impact on long-term health. Increased sugar consumption has been found to be associated with increased risk of cardiovascular disease, metabolic syndrome and obesity [
76,
77,
78]. The significant decrease in fruit and vegetable consumption, on the other hand, has stronger implications on the reduction of diet-related diseases and mortality. Specifically, higher fruit and vegetable consumption has been theorized to be protective against cardiovascular diseases and certain types of cancers, including digestive cancer [
79,
80,
81]. It has also been found to be associated with a lower risk of multimorbidity (i.e., having two or more chronic diseases), specifically those involving hypertension and diabetes [
80].Though not a primary focus of this paper, we also found significant sex differences in self-reported symptoms of anxiety and depression. Specifically, women were more likely to report increased anxiety and depression compared to men. Interestingly, the higher rates of reported psychological distress among women did not affect their diet quality compared to men, who demonstrated poorer diet with increased depression. However, this sex discrepancy in reported distress and health-related issues has been well documented across a range of settings [
82]. Thus, we expect the correlations found in our study between mental health reporting and dietary intake to be similar to that found in a clinical setting.
Overall, the results of this study indicate that depression may be more of a risk factor for poor nutrition compared to anxiety and that men’s nutrition is more affected by psychological distress. The results also suggest that caloric intake may be an area to address with individuals who identify more severe levels of anxious and depressive symptoms, given the negative health implications associated with it. The findings underscore the need to involve primary care (e.g., student health centers) in screening for psychological symptoms of distress given the association between increased risk of developing chronic diseases and poor nutrition [
83]. This need is echoed in the recommendations made by the U.S. Preventative Services Task Force (USPSTF) to include screening for depression in all primary care practices [
84]. Given the associations between depression and poor nutrition found in this study, there is a need for student health centers to adopt these screening methods, as primary care providers are more likely to detect and initiate early intervention, as patients suffering from psychological distress tend to present with somatic symptoms [
85]. Within the student health network, referrals to dieticians can be made and nutritional guidelines developed to support preventative health measures during critical periods of development [
59].
4.4. Limitations and Future Directions
Though this study contributes to the literature on the effects of anxiety and depression symptoms on dietary choices among emerging adults as well as sex differences in these relationships, several limitations exist that inform directions for future studies. While the rates of moderate and severe anxiety and depression in the sample collected here (16.8% and 19.5% respectively) were comparable to incidences reported by WHO for college students [16.7% and 18.5% respectively; 6], the absolute number of participants with moderate to severe anxiety and depression was too small to make confident inferences of population-level differences. A sample four to five times larger would yield more severely anxious and depressed participants and allow greater confidence in the estimates of impact on diet quality, total caloric consumption, and dietary choices. Additionally, dietary data was collected using a self-report measure. While the ASA24 accuracy is comparable to that of the gold standard interviewer-administered 24-h recall and food journals, self-reported data is always subject to recall bias. This could be addressed in future studies by using ecological momentary assessment (EMA).
Another limitation is the cross-sectional nature of the data. While the study provides insight on directionality, effects of anxiety and depression symptoms on diet as well as sex differences among these relationships, longitudinal assessment of diet is required to establish a dietary pattern and provide conclusive results [
88]. Data was collected throughout the week and we did not account for the difference between weekday and weekend dietary intake in our analysis, due to sample size and power restrictions. This can be addressed in future studies using repeated measures that group dietary intake separately for weekday and weekend days. Additionally, our sample on average consisted of healthy weight adults (see
Table S2 in
Supplementary Materials). This lack of variability in Body Mass Index (BMI) prevented us from accounting for the effects of weight in our analysis, a factor that has known associations with depression and caloric consumption [
89,
90]. However, the average BMI (
M = 23.55) of our sample is consistent with the 2015–2019 reports released by the National College Health Assessment (NCHA; average BMI range 24.39–25.13) [
91], indicating good representation of American college students’ BMI. Our overall findings regarding depression and caloric consumption in college students are pertinent to those in the average BMI range. This relationship may prove to be different for students with elevated BMI. Future studies should include related factors such as weight, income and physical activity using a longitudinal design to fully establish the effects of these factors on college students’ dietary intake [
15,
67].
Finally, the sample was recruited at a southeastern university in the United States, thus, the findings may not be representative of the culinary diversity across different areas of the country nor among individuals of different ethnicities. Further replication with larger samples across geographic regions is needed. Replication across cultural and culinary traditions would also provide insight on factors that would influence dietary choices when experiencing psychological distress.