Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern?
Abstract
1. Introduction
2. Selection and Assessment of Vitamin C Status Publications
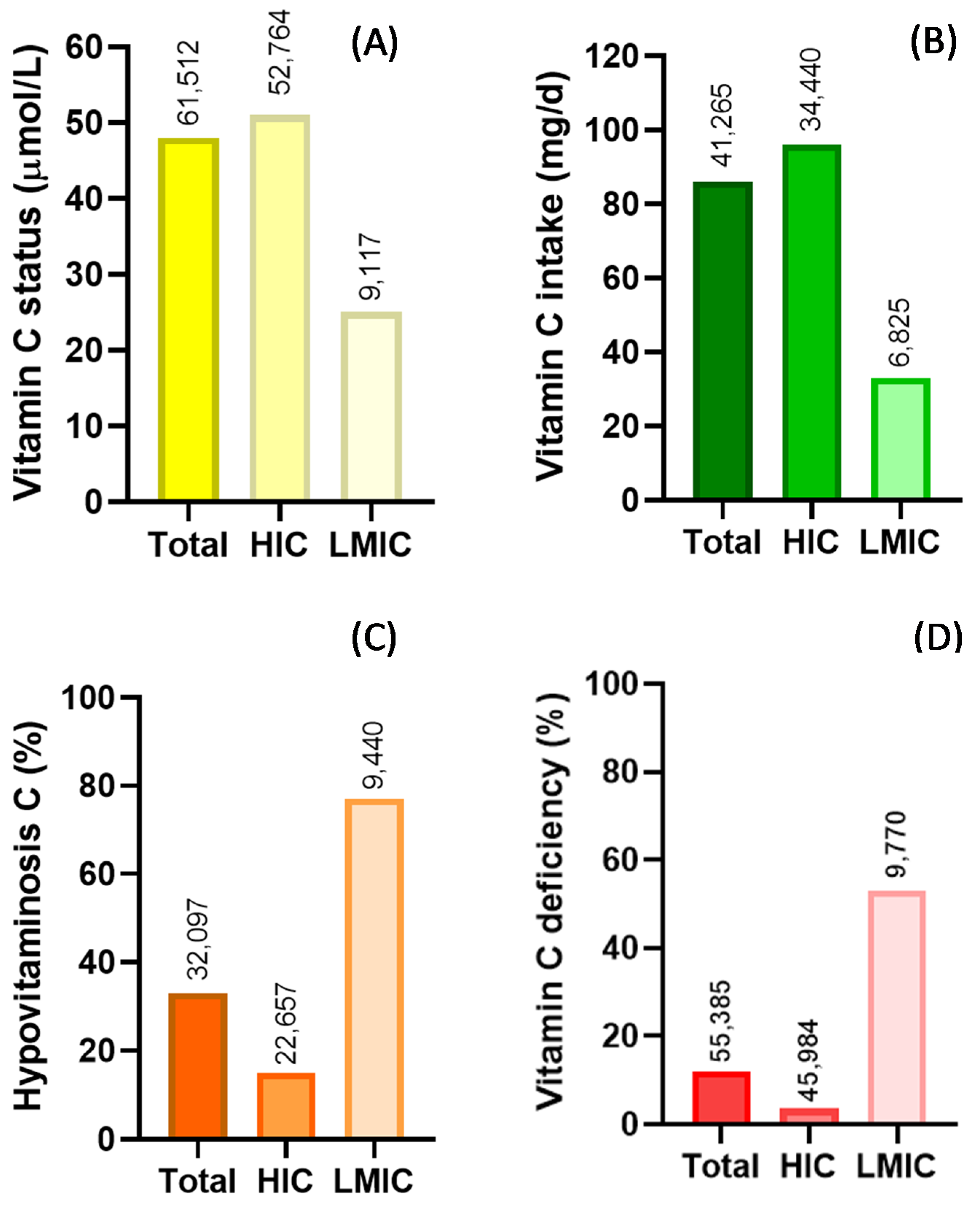
3. Global Vitamin C Status and Prevalence of Insufficiency
3.1. High Income Countries
3.2. Low and Middle Income Countries
3.3. Children and Adolescents
4. Limitations of Vitamin C Status Studies
5. Associations between Vitamin C Status and Health
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Padayatty, S.J.; Espey, M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011, 2, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M.V. SVCT1 and SVCT2: Key proteins for vitamin C uptake. Amino Acids 2008, 34, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Hornig, D. Distribution of ascorbic acid, metabolites and analogues in man and animals. Ann. N. Y. Acad. Sci. 1975, 258, 103–118. [Google Scholar] [CrossRef] [PubMed]
- Englard, S.; Seifter, S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986, 6, 365–406. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, e418. [Google Scholar] [CrossRef]
- Young, J.I.; Zuchner, S.; Wang, G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef]
- Vissers, M.C.; Kuiper, C.; Dachs, G.U. Regulation of the 2-oxoglutarate-dependent dioxygenases and implications for cancer. Biochem. Soc. Trans. 2014, 42, 945–951. [Google Scholar] [CrossRef]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative Western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef]
- Carr, A.C.; Lykkesfeldt, J. Discrepancies in global vitamin C recommendations: A review of RDA criteria and underlying health perspectives. Crit. Rev. Food Sci. Nutr. 2020, 1–14. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Johnston, C.S.; Corte, C. People with marginal vitamin C status are at high risk of developing vitamin C deficiency. J. Am. Diet. Assoc. 1999, 99, 854–856. [Google Scholar] [CrossRef]
- Khan, R.M.; Iqbal, M.P. Deficiency of Vitamin C in South Asia. Pak. J. Med. Sci. 2006, 22, 347–355. [Google Scholar]
- Dickinson, N.; Macpherson, G.; Hursthouse, A.S.; Atkinson, J. Micronutrient deficiencies in maternity and child health: A review of environmental and social context and implications for Malawi. Environ. Geochem. Health 2009, 31, 253–272. [Google Scholar] [CrossRef] [PubMed]
- The World Bank. World Bank Country and Lending Groups. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-group (accessed on 13 January 2020).
- Canoy, D.; Wareham, N.; Welch, A.; Bingham, S.; Luben, R.; Day, N.; Khaw, K.T. Plasma ascorbic acid concentrations and fat distribution in 19,068 British men and women in the European Prospective Investigation into Cancer and Nutrition Norfolk cohort study. Am. J. Clin. Nutr. 2005, 82, 1203–1209. [Google Scholar] [CrossRef]
- McCall, S.J.; Clark, A.B.; Luben, R.N.; Wareham, N.J.; Khaw, K.T.; Myint, P.K. Plasma vitamin C levels: Risk factors for deficiency and association with self-reported functional health in the European Prospective Investigation into Cancer-Norfolk. Nutrients 2019, 11, 1552. [Google Scholar] [CrossRef]
- Bates, C.J.; Prentice, A.; Cole, T.J.; Van Der Pols, J.C.; Doyle, W.; Finch, S.; Smithers, G.; Clarke, P.C. Micronutrients: Highlights and research challenges from the 1994-5 National Diet and Nutrition Survey of people aged 65 years and over. Br. J. Nutr. 1999, 82, 7–15. [Google Scholar] [CrossRef]
- Wrieden, W.L.; Hannah, M.K.; Bolton-Smith, C.; Tavendale, R.; Morrison, C.; Tunstall-Pedoe, H. Plasma vitamin C and food choice in the third Glasgow MONICA population survey. J. Epidemiol. Community Health 2000, 54, 355–360. [Google Scholar] [CrossRef]
- Faure, H.; Preziosi, P.; Roussel, A.M.; Bertrais, S.; Galan, P.; Hercberg, S.; Favier, A. Factors influencing blood concentration of retinol, alpha-tocopherol, vitamin C, and beta-carotene in the French participants of the SU.VI.MAX trial. Eur. J. Clin. Nutr. 2006, 60, 706–717. [Google Scholar] [CrossRef]
- Galan, P.; Viteri, F.E.; Bertrais, S.; Czernichow, S.; Faure, H.; Arnaud, J.; Ruffieux, D.; Chenal, S.; Arnault, N.; Favier, A.; et al. Serum concentrations of beta-carotene, vitamins C and E, zinc and selenium are influenced by sex, age, diet, smoking status, alcohol consumption and corpulence in a general French adult population. Eur. J. Clin. Nutr. 2005, 59, 1181–1190. [Google Scholar] [CrossRef]
- Birlouez-Aragon, I.; Delcourt, C.; Tessier, F.; Papoz, L. Associations of age, smoking habits and diabetes with plasma vitamin C of elderly of the POLA study. Int. J. Vitam. Nutr. Res. 2001, 71, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Rock, C.L.; Henderson, S.A.; Shore, A.B.; Fischler, C.; Galan, P.; Preziosi, P.; Hercberg, S. Serum beta-carotene and vitamin C as biomarkers of vegetable and fruit intakes in a community-based sample of French adults. Am. J. Clin. Nutr. 1997, 65, 1796–1802. [Google Scholar] [CrossRef] [PubMed]
- Marangon, K.; Herbeth, B.; Lecomte, E.; Paul-Dauphin, A.; Grolier, P.; Chancerelle, Y.; Artur, Y.; Siest, G. Diet, antioxidant status, and smoking habits in French men. Am. J. Clin. Nutr. 1998, 67, 231–239. [Google Scholar] [CrossRef]
- Paalanen, L.; Prattala, R.; Alfthan, G.; Salminen, I.; Laatikainen, T. Vegetable and fruit consumption, education and plasma vitamin C concentration in Russian and Finnish Karelia, 1992–2002. Public Health Nutr. 2014, 17, 2278–2286. [Google Scholar] [CrossRef] [PubMed]
- Matilainen, T.; Vartiainen, E.; Puska, P.; Alfthan, G.; Pokusajeva, S.; Moisejeva, N.; Uhanov, M. Plasma ascorbic acid concentrations in the Republic of Karelia, Russia and in North Karelia, Finland. Eur. J. Clin. Nutr. 1996, 50, 115–120. [Google Scholar] [PubMed]
- Nyyssonen, K.; Parviainen, M.T.; Salonen, R.; Tuomilehto, J.; Salonen, J.T. Vitamin C deficiency and risk of myocardial infarction: Prospective population study of men from eastern Finland. BMJ 1997, 314, 634–638. [Google Scholar] [CrossRef]
- Sen, A.; Marsche, G.; Freudenberger, P.; Schallert, M.; Toeglhofer, A.M.; Nagl, C.; Schmidt, R.; Launer, L.J.; Schmidt, H. Association between higher plasma lutein, zeaxanthin, and vitamin C concentrations and longer telomere length: Results of the Austrian Stroke Prevention Study. J. Am. Geriatr. Soc. 2014, 62, 222–229. [Google Scholar] [CrossRef]
- Vioque, J.; Weinbrenner, T.; Asensio, L.; Castello, A.; Young, I.S.; Fletcher, A. Plasma concentrations of carotenoids and vitamin C are better correlated with dietary intake in normal weight than overweight and obese elderly subjects. Br. J. Nutr. 2007, 97, 977–986. [Google Scholar] [CrossRef]
- Olmedilla, B.; Granado, F.; Southon, S.; Wright, A.J.; Blanco, I.; Gil-Martinez, E.; Berg, H.; Corridan, B.; Roussel, A.M.; Chopra, M.; et al. Serum concentrations of carotenoids and vitamins A, E, and C in control subjects from five European countries. Br. J. Nutr. 2001, 85, 227–238. [Google Scholar] [CrossRef]
- Jungert, A.; Neuhauser-Berthold, M. The lower vitamin C plasma concentrations in elderly men compared with elderly women can partly be attributed to a volumetric dilution effect due to differences in fat-free mass. Br. J. Nutr. 2015, 113, 859–864. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Carroll, M.D.; Ford, E.S.; Lacher, D.A. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003–2004 National Health and Nutrition Examination Survey (NHANES). Am. J. Clin. Nutr. 2009, 90, 1252–1263. [Google Scholar] [CrossRef]
- Langlois, K.; Cooper, M.; Colapinto, C.K. Vitamin C status of Canadian adults: Findings from the 2012/2013 Canadian Health Measures Survey. Health Rep. 2016, 27, 3–10. [Google Scholar]
- Cahill, L.; Corey, P.N.; El-Sohemy, A. Vitamin C deficiency in a population of young Canadian adults. Am. J. Epidemiol. 2009, 170, 464–471. [Google Scholar] [CrossRef]
- Sakai, N.; Yokoyama, T.; Date, C.; Yoshiike, N.; Matsumura, Y. An inverse relationship between serum vitamin C and blood pressure in a Japanese community. J. Nutr. Sci. Vitaminol. 1998, 44, 853–867. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saito, K.; Yokoyama, T.; Yoshida, H.; Kim, H.; Shimada, H.; Yoshida, Y.; Iwasa, H.; Shimizu, Y.; Yoshitaka, K.; Handa, S.; et al. A significant relationship between plasma vitamin C concentration and physical performance among Japanese elderly women. J. Gerontol. A Biol. Sci. Med. Sci. 2011. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.; New, A.L.; Lee, B.L.; Ong, C.N. Plasma vitamins A, C and E in the general population of Singapore, 1993 to 1995. Ann. Acad. Med. Singap. 1998, 27, 149–153. [Google Scholar] [PubMed]
- Hughes, K.; Ong, C.N. Vitamins, selenium, iron, and coronary heart disease risk in Indians, Malays, and Chinese in Singapore. J. Epidemiol. Community Health 1998, 52, 181–185. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pearson, J.F.; Pullar, J.M.; Wilson, R.; Spittlehouse, J.K.; Vissers, M.C.M.; Skidmore, P.M.L.; Willis, J.; Cameron, V.A.; Carr, A.C. Vitamin C status correlates with markers of metabolic and cognitive health in 50-year-olds: Findings of the CHALICE cohort study. Nutrients 2017, 9, 831. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Hodges, R.E. Serum levels of vitamin C in relation to dietary and supplemental intake of vitamin C in smokers and nonsmokers. Ann. N. Y. Acad. Sci. 1987, 498, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Hampl, J.S.; Taylor, C.A.; Johnston, C.S. Vitamin C deficiency and depletion in the United States: The Third National Health and Nutrition Examination Survey, 1988 to 1994. Am. J. Public Health 2004, 94, 870–875. [Google Scholar] [CrossRef]
- Hoffer, L.J. Re: “Vitamin C deficiency in a population of young Canadian adults”. Am. J. Epidemiol. 2010, 171, 387. [Google Scholar] [CrossRef]
- Pullar, J.M.; Bayer, S.; Carr, A.C. Appropriate handling, processing and analysis of blood samples is essential to avoid oxidation of vitamin C to dehydroascorbic acid. Antioxidants 2018, 7, 29. [Google Scholar] [CrossRef]
- Garcia, O.P.; Ronquillo, D.; Caamano Mdel, C.; Camacho, M.; Long, K.Z.; Rosado, J.L. Zinc, vitamin A, and vitamin C status are associated with leptin concentrations and obesity in Mexican women: Results from a cross-sectional study. Nutr. Metab. 2012, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Villalpando, S.; Montalvo-Velarde, I.; Zambrano, N.; Garcia-Guerra, A.; Ramirez-Silva, C.I.; Shamah-Levy, T.; Rivera, J.A. Vitamins A, and C and folate status in Mexican children under 12 years and women 12–49 years: A probabilistic national survey. Salud Publ. Mex. 2003, 45, S508–S519. [Google Scholar] [CrossRef]
- Hamer, D.H.; Sempertegui, F.; Estrella, B.; Tucker, K.L.; Rodriguez, A.; Egas, J.; Dallal, G.E.; Selhub, J.; Griffiths, J.K.; Meydani, S.N. Micronutrient deficiencies are associated with impaired immune response and higher burden of respiratory infections in elderly Ecuadorians. J. Nutr. 2009, 139, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Madruga de Oliveira, A.; Rondo, P.H.; Mastroeni, S.S.; Oliveira, J.M. Plasma concentrations of ascorbic acid in parturients from a hospital in Southeast Brazil. Clin. Nutr. 2008, 27, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Charlton, K.E.; Kolbe-Alexander, T.L.; Nel, J.H. Micronutrient dilution associated with added sugar intake in elderly black South African women. Eur. J. Clin. Nutr. 2005, 59, 1030–1042. [Google Scholar] [CrossRef] [PubMed]
- Ugwa, E.A.; Iwasam, E.A.; Nwali, M.I. Low Serum Vitamin C Status Among Pregnant Women Attending Antenatal Care at General Hospital Dawakin Kudu, Northwest Nigeria. Int. J. Prev. Med. 2016, 7, 40. [Google Scholar] [CrossRef]
- Nwagha, U.I.; Iyare, E.E.; Ejezie, F.E.; Ogbodo, S.O.; Dim, C.C.; Anyaehie, B.U. Parity related changes in obesity and some antioxidant vitamins in non-pregnant women of South-Eastern Nigeria. Niger. J. Clin. Pract. 2012, 15, 380–384. [Google Scholar] [CrossRef]
- Kiondo, P.; Tumwesigye, N.M.; Wandabwa, J.; Wamuyu-Maina, G.; Bimenya, G.S.; Okong, P. Plasma vitamin C assay in women of reproductive age in Kampala, Uganda, using a colorimetric method. Trop. Med. Int. Health 2012, 17, 191–196. [Google Scholar] [CrossRef]
- Halestrap, P.; Scheenstra, S. Outbreak of scurvy in Tana River County, Kenya: A case report. Afr. J. Prim. Health Care Fam. Med. 2018, 10, e1–e3. [Google Scholar] [CrossRef] [PubMed]
- Frankenfeld, C.L.; Lampe, J.W.; Shannon, J.; Gao, D.L.; Li, W.; Ray, R.M.; Chen, C.; King, I.B.; Thomas, D.B. Fruit and vegetable intakes in relation to plasma nutrient concentrations in women in Shanghai, China. Public Health Nutr. 2012, 15, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.K.; Freedman, N.D.; Fan, J.H.; Qiao, Y.L.; Dawsey, S.M.; Taylor, P.R.; Abnet, C.C. Prediagnostic plasma vitamin C and risk of gastric adenocarcinoma and esophageal squamous cell carcinoma in a Chinese population. Am. J. Clin. Nutr. 2013, 98, 1289–1297. [Google Scholar] [CrossRef]
- Ma, A.G.; Schouten, E.G.; Wang, Y.; Xu, R.X.; Zheng, M.C.; Li, Y.; Wang, Q.; Sun, Y. Micronutrient status in anemic and non-anemic Chinese women in the third trimester of pregnancy. Asia Pac. J. Clin. Nutr. 2009, 18, 41–47. [Google Scholar] [PubMed]
- Viroonudomphol, D.; Mahaisiriyodom, A.; Mingkhawn, R.; Sadomthian, P.; Korchasri, N.; Jittngamkhum, S.; Sangcakul, A. Relationship between serum antioxidant vitamins A, E, and C and lipid profiles in priest subjects at the Priest Hospital. Southeast Asian J. Trop. Med. Public Health 2005, 36, 246–253. [Google Scholar]
- Ravindran, R.D.; Vashist, P.; Gupta, S.K.; Young, I.S.; Maraini, G.; Camparini, M.; Jayanthi, R.; John, N.; Fitzpatrick, K.E.; Chakravarthy, U.; et al. Prevalence and risk factors for vitamin C deficiency in north and south India: A two centre population based study in people aged 60 years and over. PLoS ONE 2011, 6, e28588. [Google Scholar] [CrossRef]
- Dherani, M.; Murthy, G.V.; Gupta, S.K.; Young, I.S.; Maraini, G.; Camparini, M.; Price, G.M.; John, N.; Chakravarthy, U.; Fletcher, A.E. Blood levels of vitamin C, carotenoids and retinol are inversely associated with cataract in a North Indian population. Invest. Ophthalmol. Vis. Sci. 2008, 49, 3328–3335. [Google Scholar] [CrossRef]
- Chiplonkar, S.A.; Agte, V.V.; Mengale, S.S.; Tarwadi, K.V. Are lifestyle factors good predictors of retinol and vitamin C deficiency in apparently healthy adults? Eur. J. Clin. Nutr. 2002, 56, 96–104. [Google Scholar] [CrossRef]
- Amaliya, A.; Laine, M.L.; Delanghe, J.R.; Loos, B.G.; Van Wijk, A.J.; Van der Velden, U. Java project on periodontal diseases: Periodontal bone loss in relation to environmental and systemic conditions. J. Clin. Periodontol. 2015, 42, 325–332. [Google Scholar] [CrossRef]
- Cheung, E.; Mutahar, R.; Assefa, F.; Ververs, M.T.; Nasiri, S.M.; Borrel, A.; Salama, P. An epidemic of scurvy in Afghanistan: Assessment and response. Food Nutr. Bull. 2003, 24, 247–255. [Google Scholar] [CrossRef]
- Levine, M.; Wang, Y.; Padayatty, S.J.; Morrow, J. A new recommended dietary allowance of vitamin C for healthy young women. Proc. Natl. Acad. Sci. USA 2001, 98, 9842–9846. [Google Scholar] [CrossRef] [PubMed]
- Camarena, V.; Wang, G. The epigenetic role of vitamin C in health and disease. Cell. Mol. Life Sci. 2016, 73, 1645–1658. [Google Scholar] [CrossRef] [PubMed]
- Garcia, O.P.; Ronquillo, D.; del Carmen Caamano, M.; Martinez, G.; Camacho, M.; Lopez, V.; Rosado, J.L. Zinc, iron and vitamins A, C and e are associated with obesity, inflammation, lipid profile and insulin resistance in Mexican school-aged children. Nutrients 2013, 5, 5012–5030. [Google Scholar] [CrossRef] [PubMed]
- Chainani, N.; Sharma, P.; Meena, N.; Sharma, U. Pattern of vitamin deficiencies among the malnourished preschool children in ICDS blocks of Jaipur city. Indian J. Matern. Child Health 1994, 5, 109–111. [Google Scholar] [PubMed]
- Sivakumar, B.; Nair, K.M.; Sreeramulu, D.; Suryanarayana, P.; Ravinder, P.; Shatrugna, V.; Kumar, P.A.; Raghunath, M.; Rao, V.V.; Balakrishna, N.; et al. Effect of micronutrient supplement on health and nutritional status of schoolchildren: Biochemical status. Nutrition 2006, 22, S15–S25. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.G.; Toteja, G.S.; Suman, R. Plasma vitamin C status of adolescent girls in a slum of Delhi. Indian Pediatr. 2014, 51, 932–933. [Google Scholar]
- Ahmed, F.; Khan, M.R.; Banu, C.P.; Qazi, M.R.; Akhtaruzzaman, M. The coexistence of other micronutrient deficiencies in anaemic adolescent schoolgirls in rural Bangladesh. Eur. J. Clin. Nutr. 2008, 62, 365–372. [Google Scholar] [CrossRef]
- Ene-Obong, H.N.; Odoh, I.F.; Ikwuagwu, O.E. Plasma vitamin A and C status of in-school adolescents and associated factors in Enugu State, Nigeria. J. Health Popul. Nutr. 2003, 21, 18–25. [Google Scholar]
- World Health Organization. Requirements of Ascorbic Acid, Vitamin D, Vitamin B12, Folate, and Iron; World Health Organization: Geneva, Switzerland, 1970. [Google Scholar]
- Montagnac, J.A.; Davis, C.R.; Tanumihardjo, S.A. Nutritional value of cassava for use as a staple food and recent advances for improvement. Compr. Rev. Food Sci. Food Saf. 2009, 8, 181–194. [Google Scholar] [CrossRef]
- Washko, P.W.; Welch, R.W.; Dhariwal, K.R.; Wang, Y.; Levine, M. Ascorbic acid and dehydroascorbic acid analyses in biological samples. Anal. Biochem. 1992, 204, 1–14. [Google Scholar] [CrossRef]
- Lins, I.L.; Bueno, N.B.; Grotti Clemente, A.P.; Pfrimer, K.; Sawaya, A.L.; De Menezes Toledo Florencio, T.M. Energy Intake in Socially Vulnerable Women Living in Brazil: Assessment of the Accuracy of Two Methods of Dietary Intake Recording Using Doubly Labeled Water. J. Acad. Nutr. Diet. 2016, 116, 1560–1567. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority Panel on Dietetic Products Nutrition and Allergies. Scientific opinion on dietary reference values for vitamin C. EFSA J. 2013, 11, 3418. [Google Scholar]
- Carr, A.C.; Bozonet, S.M.; Vissers, M.C. A randomised cross-over pharmacokinetic bioavailability study of synthetic versus kiwifruit-derived vitamin C. Nutrients 2013, 5, 4451–4461. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rowe, S. Factors affecting vitamin C status and prevalence of deficiency: A global health perspective. Nutrients 2020, 12, 1963. [Google Scholar] [CrossRef]
- Wilson, R.; Willis, J.; Gearry, R.; Skidmore, P.; Fleming, E.; Frampton, C.; Carr, A. Inadequate vitamin C status in prediabetes and type 2 diabetes mellitus: Associations with glycaemic control, obesity, and smoking. Nutrients 2017, 9, 997. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Mokdad, A.H.; Giles, W.H.; Brown, D.W. The metabolic syndrome and antioxidant concentrations: Findings from the Third National Health and Nutrition Examination Survey. Diabetes 2003, 52, 2346–2352. [Google Scholar] [CrossRef]
- Khaw, K.T.; Bingham, S.; Welch, A.; Luben, R.; Wareham, N.; Oakes, S.; Day, N. Relation between plasma ascorbic acid and mortality in men and women in EPIC-Norfolk prospective study: A prospective population study. European Prospective Investigation into Cancer and Nutrition. Lancet 2001, 357, 657–663. [Google Scholar] [CrossRef]
- Goyal, A.; Terry, M.B.; Siegel, A.B. Serum antioxidant nutrients, vitamin A, and mortality in U.S. Adults. Cancer Epidemiol. Biomark. Prev. 2013, 22, 2202–2211. [Google Scholar] [CrossRef]
- Wang, S.M.; Fan, J.H.; Taylor, P.R.; Lam, T.K.; Dawsey, S.M.; Qiao, Y.L.; Abnet, C.C. Association of plasma vitamin C concentration to total and cause-specific mortality: A 16-year prospective study in China. J. Epidemiol. Community Health 2018, 72, 1076–1082. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Poulsen, H.E. Is vitamin C supplementation beneficial? Lessons learned from randomised controlled trials. Br. J. Nutr. 2010, 103, 1251–1259. [Google Scholar] [CrossRef]
- Hemilä, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Cook, J. Intravenous vitamin C for cancer therapy—Identifying the current gaps in our knowledge. Front. Physiol. 2018, 9, 1182. [Google Scholar] [CrossRef] [PubMed]
- May, J.M. Vitamin C transport and its role in the central nervous system. Subcell. Biochem. 2012, 56, 85–103. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef]
- Carr, A.C. Vitamin C in pneumonia and sepsis. In Vitamin C: New Biochemical and Functional Insights; Chen, Q., Vissers, M., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 115–135. [Google Scholar]
- Calder, P.C.; Carr, A.C.; Gombart, A.F.; Eggersdorfer, M. Optimal nutritional status for a well-functioning immune system is an important factor to protect against viral infections. Nutrients 2020, 12, 1181. [Google Scholar] [CrossRef]
- German Nutrition Society. New reference values for vitamin C intake. Ann. Nutr. Metab. 2015, 67, 13–20. [Google Scholar] [CrossRef]
- Carr, A.C.; Vissers, M.C. Synthetic or food-derived vitamin C—Are they equally bioavailable? Nutrients 2013, 5, 4284–4304. [Google Scholar] [CrossRef]
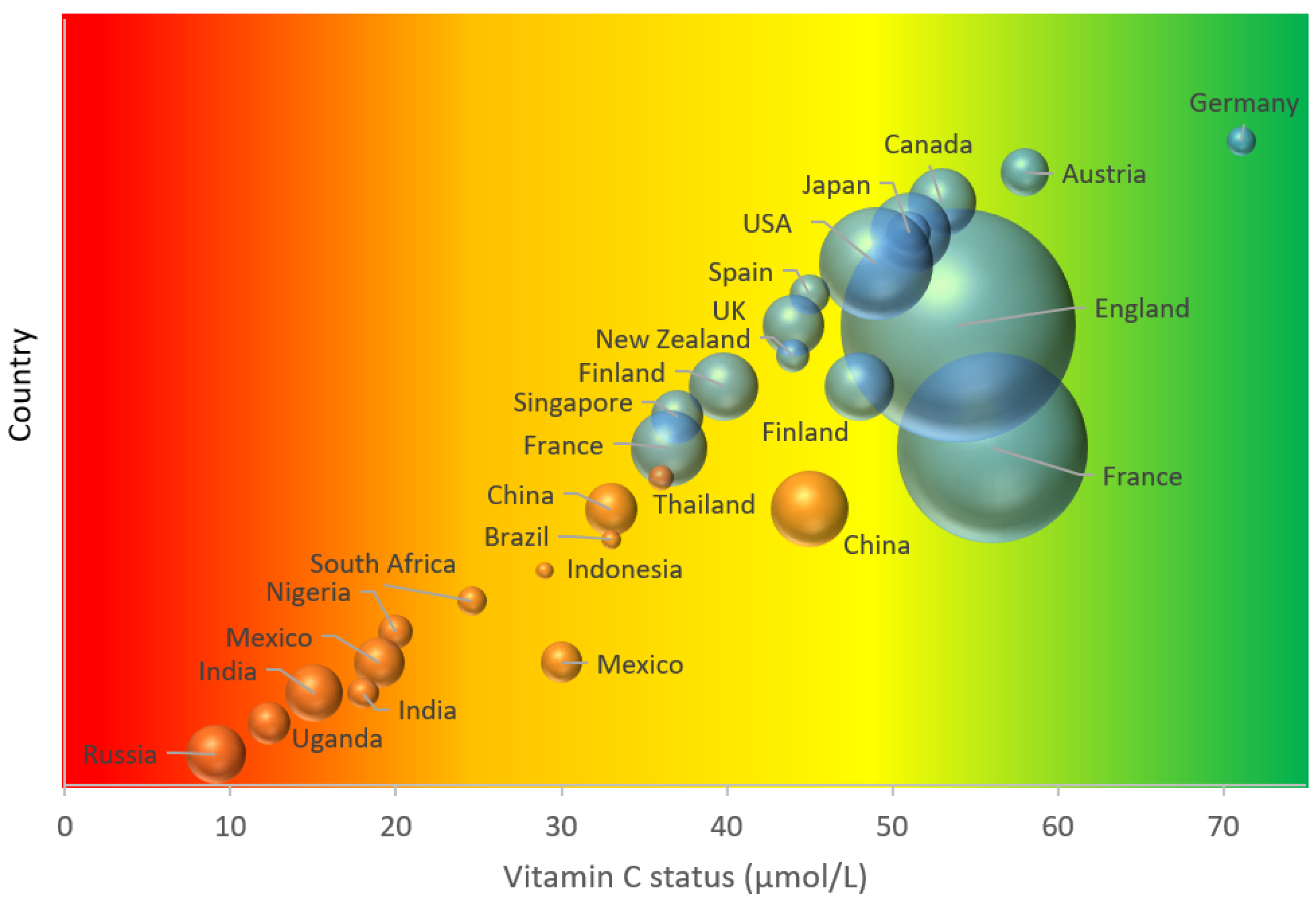
| Country (Region) Sampling Dates | Population (Age Range) | Vitamin C Status (µmol/L) a | Deficiency (% <11 µmol/L) | Hypovitaminosis C (% <23 or 28 µmol/L) | Dietary Intake (mg/day) a | References |
|---|---|---|---|---|---|---|
| Europe | ||||||
| England (Norfolk) 1993–1997 | 22,474 total (40–79 years) | 54 b | 1.4 | 12 | 85 b | [16,17] |
| 10,267 males | 48 b | 2.2 | 17 | 83 b | ||
| 12,207 females | 59 b | 0.8 | 8 | 87 b | ||
| UK (England and Scotland) 1994–1995 | 1310 elderly (≥65 years) | 44 (25) | 14 | – | – | [18] |
| Scotland (Glasgow) 1992 | 1267 total (25–74 years) | – | 20 | 44 | – | [19] |
| 632 males | – | 26 | 52 | – | ||
| 635 females | – | 14 | 36 | – | ||
| France (all regions) 1994–1995 | 12,741 total (35–60 years) | 56 b | 1.0 | – | 100 b | [20,21] |
| 5028 males | 50 (23) | 1.8 | – | 103 (48) | ||
| 7713 females | 60 (31) | 0.4 | – | 98 (44) | ||
| France (South; Sète) 1995–1997 | 1987 total (>60 years) | 36 b | 9 b | – | – | [22] |
| 874 elderly males | 32 | 13 b | – | – | ||
| 1113 elderly females | 40 | 6 b | – | – | ||
| France (Paris) <1991 | 837 total (≥18 years) | 48 b | – | – | – | [23] |
| 361 males | 41 | – | – | – | ||
| 476 females | 53 | – | – | – | ||
| France (Nancy) | 459 males (20–60 years) | 39 b | – | – | 80 b | [24] |
| Finland (North Karelia) 1992–2002 | 1616 total (25–64 years) | 40 b | – | – | – | [25,26] |
| 974 males | 37 | 2.2 | 4.4 | – | ||
| 642 females | 44 | – | – | – | ||
| Finland (Eastern) 1984–1989 | 1605 males (42–60 years) | 48 (23) | 5.7 | – | – | [27] |
| Austria (Graz) 1991–1994 | 786 total (45–86 years) | 58 (21) | – | – | – | [28] |
| 330 males | 50 (20) | – | – | – | ||
| 456 females | 64 (19) | – | – | – | ||
| Spain (Alicante province) 2000–2001 | 545 total (>65 years) | 45 b | – | – | 131 b | [29] |
| 252 elderly males | 38 (20) | – | – | 125 (64) | ||
| 293 elderly females | 51 (18) | – | – | 136 (70) | ||
| Europe (France, Ireland, Spain Netherlands) | 349 total (25–45 years) | 59 b | – | – | – | [30] |
| 175 males | 54 (13–103) d | – | – | – | ||
| 174 females | 64 (6–117) d | – | – | – | ||
| Germany (Giessen) 1994–2004 | 279 total (62–92 years) | 71 b | – | – | 90 b | [31] |
| 98 elderly males | 62 (55–74) c | – | – | 84 (61–116) c | ||
| 181 elderly females | 76 (62–89) c | – | – | 93 (70–132) c | ||
| North America | ||||||
| United States 2003–2004 | 4438 total (≥20 years) | 49 (32–71) c | 8.4 | – | – | [32] |
| 2153 males | 45 (27–66) c | 10.0 | – | – | ||
| 2285 females | 53 (38–76) c | 6.9 | – | – | ||
| Canada (16 sites countrywide) 2012–2013 | 1615 total (20–79 years) | 53 | <3 | – | – | [33] |
| 804 males | 47 | – | – | – | ||
| 811 females | 59 | – | – | – | ||
| Canada (Toronto) 2004–2008 | 979 total (20–29 years) | 31 | 14 | 47 | 140/242 b,e | [34] |
| 287 males | 29 | 16 | 37 | 228 | ||
| 692 females | 33 | 13 | 45 | 248 | ||
| Asia-Pacific | ||||||
| Japan (Shibata, Niigata Prefecture) 1977 | 2185 total (40–89 years) | 51 b | – | – | – | [35] |
| 919 males | 43 (19) | – | – | – | ||
| 1266 females | 57 (17) | – | – | – | ||
| Japan (Itabashi, Tokyo) 2006 | 655 elderly females | 51 (9) | – | – | – | [36] |
| (70–84 years) | ||||||
| Singapore 1993–1995 | 941 total (30–69 years) | 37 b | 12 b | – | – | [37,38] |
| 468 males | 32 b | 17 b | – | – | ||
| 473 females | 41 b | 6.0 b | – | – | ||
| New Zealand (Canterbury region) 2010–2013 | 369 total (50 years) | 44 | 2.4 | 13 | 110 | [39] |
| 174 males | 41 | 4.0 | 15 | 113 | ||
| 195 females | 47 | 1.0 | 11 | 107 |
| Country (Region) Sampling Dates | Population (Age Range) | Vitamin C Status (µmol/L) a | Deficiency (% <11 µmol/L) | Hypovitaminosis C (% <23 or 28 µmol/L) | Dietary Intake (mg/day) a | References |
|---|---|---|---|---|---|---|
| Upper-middle | ||||||
| Russia (Pitkäranta District, Republic of Karelia) 1992–2002 | 1191 total (25–64 years) | 9.0 b | – | – | – | [25,26] |
| 579 males | 5.0 | 79 | 90 | – | ||
| 612 females | 13 | – | – | – | ||
| China (Shanghai) 1995–2001 | 2031 females (30–64 years) | 45 (48) | – | – | – | [53] |
| China (Linxian) 1999–2000 | 948 total (~50–79 years) | 33 (14–55) c | – | – | – | [54] |
| 473 males | 27 (11–50) c | – | – | – | ||
| 475 females | 37 (16–57) c | – | – | – | ||
| China (Gansu, Guangxi, Shandong, Fujian) 1999–2001 | 734 pregnant females (20–35 years) | 19 b | – | 66 | – | [55] |
| Thailand (Bangkok) 2003 | 209 total (23–68 years) | 36 (0–102) d | – | 31 b | – | [56] |
| 90 males | 37 (0–77) d | – | 33 | – | ||
| 119 females | – | 30 | – | |||
| Mexico (multiple states) | 855 non-pregnant females (12–49 years) | 19 | 39 | – | – | [45] |
| Mexico (Central-Queretaro state) 2012 | 580 females (37 years) e | 30 (13) | 5 | 32 | 72 b | [44] |
| Ecuador (Quito) 2003–2004 | 369 total (>65 years) | 15 b | 43 b | – | – | [46] |
| 125 elderly males | 11 (9) | 60 | – | – | ||
| 224 elderly females | 17 (10) | 33 | – | – | ||
| Brazil (Sao Paulo) 2008 | 117 pregnant females (≥15 years) | 33 (2) | 6 | 31 | – | [47] |
| South Africa (Cape Town) 2015 | 285 total (≥60 years) | 25 b | – | 66 b,f | 39 b | [48] |
| 53 elderly males | 23 | – | 84 f | 27 | ||
| 232 elderly females | 25 | – | 62 f | 42 | ||
| Lower-middle | ||||||
| India (North-Haryana state; South-Tamil Nadu) 2004–2006 | 5638 total (≥60 years) | – | 59 b | 81 b | 29 b | [57] |
| 2668 North total | – | 74 | 89 | 23 b | ||
| 1283 elderly males | – | 78 | – | – | ||
| 1385 elderly females | – | 71 | – | – | ||
| 2970 South total | – | 46 | 74 | 34 b | ||
| 1407 elderly males | – | 51 | – | – | ||
| 1563 elderly females | – | 40 | – | – | ||
| India (North-Balba-garh, Faridabad district) 2002–2003 | 1112 total (≥50 years) | ~15 b | – | – | – | [58] |
| ~48% males | 13 | – | – | – | ||
| ~52% females | 17 | – | – | – | ||
| India (West-Maharashtra state) 1998–2000 | 322 total (20–45 years) | 18 b | 18 b | 71 b | 34 b | [59] |
| 214 males | 17 (7) | 20 | 75 | 40 b | ||
| 108 females | 20 (7) | 13 | 63 | 29 b | ||
| Indonesia (West Java) 2011 | 98 total (39–50 years) | 29 (19) | 11 | 45 | – | [60] |
| 45 males; 53 females | ||||||
| Nigeria (Northwest-Kano state) 2009–2011 | 400 pregnant females (<20–39 years) | 20 (29) | – | 80 | – | [49] |
| Nigeria (South-east-Enugu) 2009 | 200 non-pregnant females (29 years) b,e | 74 b | 0 | 0 | 683 b,g | [50] |
| Low income | ||||||
| Uganda (Kampala) 2008–2009 | 600 females (15–49 years) | 12 b | 56 b | – | – | [51] |
| 400 pregnant | 11 (4) | 70 | – | – | ||
| 200 non-pregnant | 15 (6) | 28 | – | – |
| Country (Region) Sampling Dates | Population (Age Range) | Vitamin C Status (µmol/L) a | Deficiency (% <11 µmol/L) | Hypovitaminosis C (% <23 or 28 µmol/L) | Dietary Intake (mg/day) a | References |
|---|---|---|---|---|---|---|
| High income | ||||||
| United States 2003–2004 | 823 children (6–11 years) | 71 b | 1.6 b | – | – | [32] |
| 400 boys | 74 (60–88) c | 1.3 | – | – | ||
| 423 girls | 69 (56–87) c | 1.8 | – | – | ||
| 2016 adolescents (12–19 years) | 53 b | 3.3 b | – | – | ||
| 1037 boys | 51 (37–68) c | 2.7 | – | – | ||
| 979 girls | 55 (38–76) c | 3.9 | – | – | ||
| Upper-middle | ||||||
| Mexico (multiple states) 1999 | 1815 children (0–11 years) | 28 | 23 | – | – | [45] |
| Mexico (Queretaro state) 2012 | 197 children (6–11 years) | 24 (9) | 8 | 38 | 44 | [64] |
| Lower-middle | ||||||
| India (Jaipur city) | 5000 well-nourished preschool children | – | 0 | – | – | [65] |
| 1000 malnourished preschool children | – | 1.1 | – | – | ||
| India (Hyderabad) | 869 children (6–16 years) | – | – | 60 d | – | [66] |
| India (Delhi slum) 2012–2013 | 775 adolescent females (11–18 years) | 43 (26) | 6.3 | 34 | 48 (26) | [67] |
| Bangladesh (Dhaka district) 2003 | 307 adolescent females (14–18 years) | 46 (20) | 2.0 | 11 | – | [68] |
| Nigeria (Enugu state) | 90 adolescents (13–20 years) | – | – | 47 e | – | [69] |
| Males | 35–43 | – | 60 e | 24–27 | ||
| Females | 47–51 | – | 40 e | 15–20 |
| Country or Authority | Males (mg/day) | Females (mg/day) |
|---|---|---|
| High Income | ||
| France | 110 | 110 |
| DACH, European Union | 110 | 95 |
| Japan | 100 | 100 |
| Italy, Singapore | 105 | 85 |
| USA, Canada | 90 | 75 |
| Nordic, Netherlands | 75 | 75 |
| Spain | 60 | 60 |
| Australia, New Zealand, FAO/WHO | 45 | 45 |
| United Kingdom | 40 | 40 |
| Low-Middle Income | ||
| China | 100 | 100 |
| South Africa | 90 | 90 |
| Thailand | 90 | 75 |
| Malaysia | 70 | 70 |
| Vietnam | 70 | 65 |
| Philippines | 70 | 60 |
| Indonesia | 60 | 60 |
| India | 40 | 40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rowe, S.; Carr, A.C. Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern? Nutrients 2020, 12, 2008. https://doi.org/10.3390/nu12072008
Rowe S, Carr AC. Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern? Nutrients. 2020; 12(7):2008. https://doi.org/10.3390/nu12072008
Chicago/Turabian StyleRowe, Sam, and Anitra C. Carr. 2020. "Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern?" Nutrients 12, no. 7: 2008. https://doi.org/10.3390/nu12072008
APA StyleRowe, S., & Carr, A. C. (2020). Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern? Nutrients, 12(7), 2008. https://doi.org/10.3390/nu12072008






