The Effects of a Low Calorie Ketogenic Diet on Glycaemic Control Variables in Hyperinsulinemic Overweight/Obese Females
Abstract
1. Introduction
2. Material and Methods
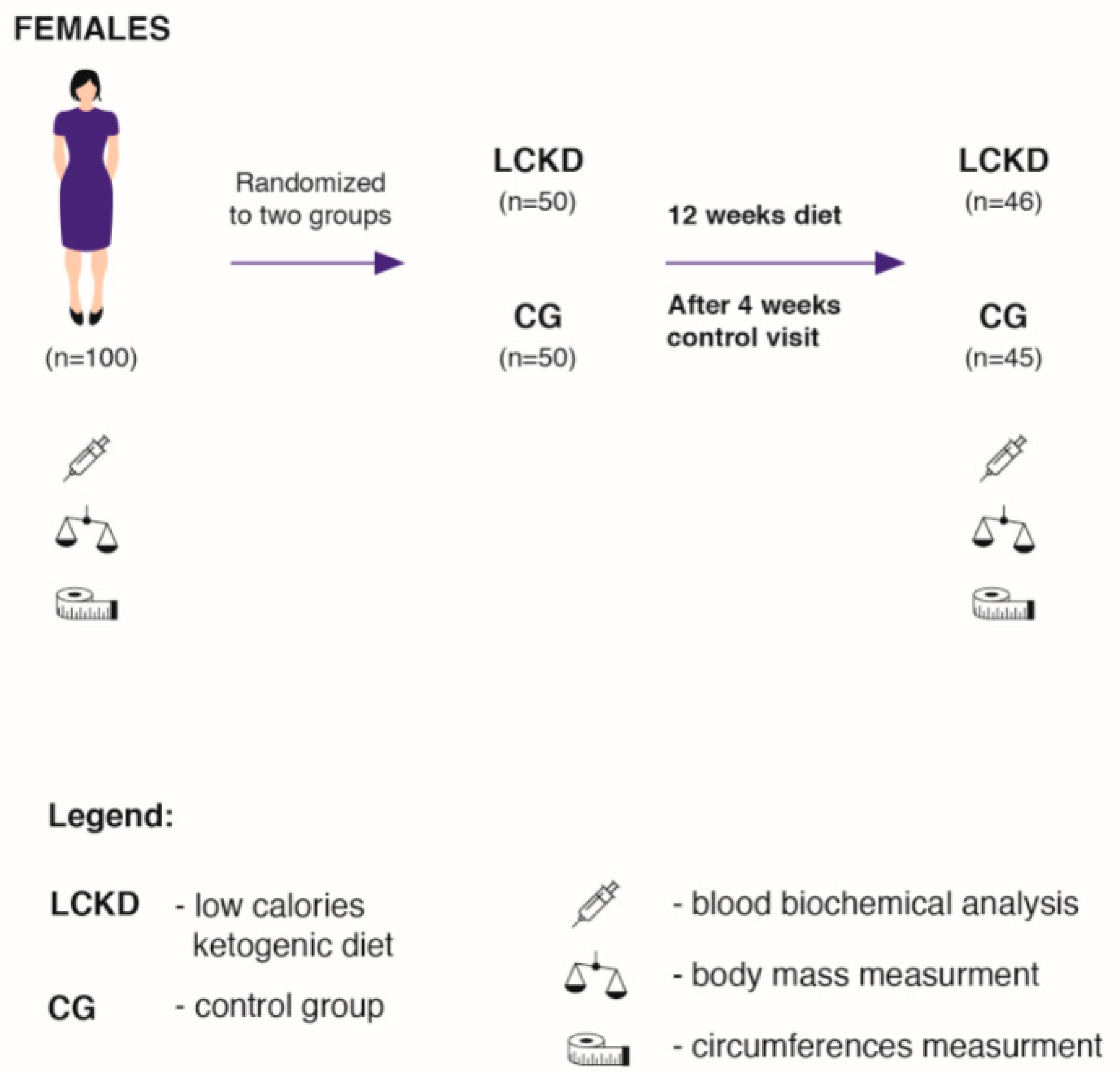
2.1. Study Sample
2.2. Dietary Procedures
2.3. Diet Composition
2.4. Diet Control
2.5. Experimental Design
2.6. Biochemical Analysis
2.7. Body Mass and Circumference Measurements
2.8. Statistical Analysis
3. Results
Biochemical Variables
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017. [Google Scholar]
- Bolla, A.M.; Caretto, A.; Laurenzi, A.; Scavini, M.; Piemonti, L. Low-Carb and Ketogenic Diets in Type 1 and Type 2 Diabetes. Nutrients 2019, 11, 962. [Google Scholar] [CrossRef] [PubMed]
- Michalczyk, M.M.; Maszczyk, A.; Stastny, P. The Effects of Low-Energy Moderate-Carbohydrate (MCD) and Mixed (MixD) Diets on Serum Lipid Profiles and Body Composition in Middle-Aged Men: A Randomized Controlled Parallel-Group Clinical Trial. Int. J. Environ. Res. Public Health 2020, 17, 1332. [Google Scholar] [CrossRef]
- Webster, C.C.; E Murphy, T.; Larmuth, K.; Noakes, T.D.; Smith, J. Diet, Diabetes Status, and Personal Experiences of Individuals with Type 2 diabetes Who Self-Selected and Followed a Low Carbohydrate High Fat diet. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 2567–2582. [Google Scholar] [CrossRef]
- Hołowko, J.; Michalczyk, M.M.; Zając, A.; Czerwińska-Rogowska, M.; Ryterska, K.; Banaszczak, M.; Jakubczyk, K.; Stachowska, E. Six Weeks of Calorie Restriction Improves Body Composition and Lipid Profile in Obese and Overweight Former Athletes. Nutrients 2019, 11, 1461. [Google Scholar] [CrossRef]
- Sonksen, P.; Sonksen, J. Insulin: Understanding its action in health and disease. Br. J. Anaesth. 2000, 85, 69–79. [Google Scholar] [CrossRef]
- Taylor, R. Type 2 Diabetes. Diabetes Care 2013, 36, 1047–1055. [Google Scholar] [CrossRef]
- Wang, L.-L.; Wang, Q.; Hong, Y.; Ojo, O.; Jiang, Q.; Hou, Y.-Y.; Huang, Y.-H.; Wang, X.-H. The Effect of Low-Carbohydrate Diet on Glycemic Control in Patients with Type 2 Diabetes Mellitus. Nutrients 2018, 10, 661. [Google Scholar] [CrossRef]
- Tay, J.; Luscombe-Marsh, N.D.; Thompson, C.H.; Noakes, M.; Buckley, J.D.; Wittert, G.A.; Yancy, W.S.; Brinkworth, G.D. Comparison of low- and high-carbohydrate diets for type 2 diabetes management: A randomized trial. Am. J. Clin. Nutr. 2015, 102, 780–790. [Google Scholar] [CrossRef]
- Michalczyk, M.M.; Zajac-Gawlak, I.; Zając, A.; Pelclová, J.; Roczniok, R.; Langfort, J. Influence of Nutritional Education on the Diet and Nutritional Behaviors of Elderly Women at the University of the Third Age. Int. J. Environ. Res. Public Health 2020, 17, 696. [Google Scholar] [CrossRef] [PubMed]
- Reaven, G.M. THE INSULIN RESISTANCE SYNDROME: Definition and Dietary Approaches to Treatment. Annu. Rev. Nutr. 2005, 25, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F. Overweight and diabetes prevention: Is a low-carbohydrate–high-fat diet recommendable? Eur. J. Nutr. 2018, 57, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Farin, H.M.; Abbasi, F.; Reaven, G.M. Comparison of Body Mass Index Versus Waist Circumference with the Metabolic Changes That Increase the Risk of Cardiovascular Disease in Insulin-Resistant Individuals. Am. J. Cardiol. 2006, 98, 1053–1056. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A. Ketogenic Diet for Obesity: Friend or Foe? Int. J. Environ. Res. Public Health 2014, 11, 2092–2107. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Dibaba, D.; Xun, P.; Song, Y.; Rosanoff, A.; Shechter, M.; He, K. The effect of magnesium supplementation on blood pressure in individuals with insulin resistance, prediabetes, or noncommunicable chronic diseases: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 921–929. [Google Scholar] [CrossRef]
- Paoli, A.; Cenci, L.; Fancelli, M.; Parmagnani, A.; Fratter, A.; Cucchi, A.; Bianco, A. Ketogenic diet and phytoextracts comparison of the efficacy of mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind. Hi Tech 2010, 21, 24–29. [Google Scholar]
- Paoli, A.; Mancin, L.; Giacona, M.C.; Bianco, A.; Caprio, M. Effects of a ketogenic diet in overweight women with polycystic ovary syndrome. J. Transl. Med. 2020, 18, 1–11. [Google Scholar] [CrossRef]
- Volek, J.S.; Sharman, M.; Gómez, A.L.; DiPasquale, C.; Roti, M.; Pumerantz, A.; Kraemer, W.J. Comparison of a very low-carbohydrate and low-fat diet on fasting lipids, LDL subclasses, insulin resistance, and postprandial lipemic responses in overweight women. J. Am. Coll. Nutr. 2004, 23, 177–184. [Google Scholar] [CrossRef]
- Volek, J.S.; Sharman, M.; Gómez, A.; Judelson, D.; Rubin; Watson, G.; Sokmen, B.; Silvestre, R.; French, D.; Kraemer, W.J. Comparison of energy-restricted very low-carbohydrate and low-fat diets on weight loss and body composition in overweight men and women. Nutr. Metab. 2004, 1, 13. [Google Scholar] [CrossRef]
- Volek, J.S.; Sharman, M.; Forsythe, C.E. Modification of Lipoproteins by Very Low-Carbohydrate Diets. J. Nutr. 2005, 135, 1339–1342. [Google Scholar] [CrossRef]
- Volek, J.S.; Phinney, S.; Forsythe, C.E.; Quann, E.E.; Wood, R.J.; Puglisi, M.J.; Kraemer, W.J.; Bibus, D.M.; Fernandez, M.-L.; Feinman, R.D. Carbohydrate Restriction has a More Favorable Impact on the Metabolic Syndrome than a Low Fat Diet. Lipids 2008, 44, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Westman, E.C.; Yancy, W.S.; Mavropoulos, J.C.; Marquart, M.; McDuffie, J.R. The effect of a low-carbohydrate, ketogenic diet versus a low-glycemic index diet on glycemic control in type 2 diabetes mellitus. Nutr. Metab. 2008, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- Westman, E.C.; Tondt, J.; Maguire, E.; Yancy, W.S. Implementing a low-carbohydrate, ketogenic diet to manage type 2 diabetes mellitus. Expert Rev. Endocrinol. Metab. 2018, 13, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.M.; Mathew, T.C.; Khadada, M.; Al-Mousawi, M.; Talib, H.; Asfar, S.K.; Behbahani, A.I.; Al-Zaid, N.S. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol. Cell. Biochem. 2007, 302, 249–256. [Google Scholar] [CrossRef]
- Wirrell, E.C. Ketogenic Ratio, Calories and Fluids: Do They Matter? Epilepsia 2008, 49 (Suppl. 8), 17–19. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, H.H.; Thiele, E.A. Low-glycemic-index treatment: A liberalized ketogenic diet for treatment of intractable epilepsy. Neurology 2005, 65, 1810–1812. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H.; Krauss, G.L.; McGrogan, J.R.; Freeman, J.M. Efficacy of the Atkins diet as therapy for intractable epilepsy. Neurology 2003, 61, 1789–1791. [Google Scholar] [CrossRef] [PubMed]
- Owen, O.E.; Morgan, A.P.; Kemp, H.G.; Sullivan, J.M.; Herrera, M.G.; Cahill, G.F. Brain Metabolism during Fasting*. J. Clin. Investig. 1967, 46, 1589–1595. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H.; McGrogan, J.R. Worldwide Use of the Ketogenic Diet. Epilepsia 2005, 46, 280–289. [Google Scholar] [CrossRef]
- Goday, A.; Bellido, D.; Sajoux, I.; Crujeiras, A.B.; Burguera, B.; García-Luna, P.P.; Oleaga, A.; Moreno, B.; Casanueva, F.F. Short-term safety, tolerability and efficacy of a very low-calorie-ketogenic diet interventional weight loss program versus hypocaloric diet in patients with type 2 diabetes mellitus. Nutr. Diabetes 2016, 6, e230. [Google Scholar] [CrossRef]
- Dashti, H.M.; Al-Zaid, N.S.; Mathew, T.C.; Al-Mousawi, M.; Talib, H.; Asfar, S.K.; Behbahani, A.I. Long Term Effects of Ketogenic Diet in Obese Subjects with High Cholesterol Level. Mol. Cell. Biochem. 2006, 286, 1–9. [Google Scholar] [CrossRef]
- Allen, B.; Bhatia, S.K.; Anderson, C.; Eichenberger-Gilmore, J.M.; Sibenaller, Z.A.; Mapuskar, K.A.; Schoenfeld, J.D.; Buatti, J.M.; Spitz, D.R.; Fath, M.A. Ketogenic diets as an adjuvant cancer therapy: History and potential mechanism. Redox Biol. 2014, 2, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.A.; Mathew, T.C.; Dashti, A.A.; Asfar, S.; Al-Zaid, N.; Dashti, H.M. Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrients 2012, 28, 1016–1021. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Sargrad, K.; Homko, C.; Mozzoli, M.; Stein, T.P. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann. Intern. Med. 2005, 142, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Jarosz, M. Standards of Nutrition for the Polish Population—Amendment; Institute of Food and Nutrition: Warsaw, Poland, 2017. [Google Scholar]
- Maciejewska, D.; Michalczyk, M.M.; Czerwińska, M.; Banaszczak, M.; Ryterska, K.; Jakubczyk, K.; Piotrwski, J.; Hołowko, J.; Drozd, A.; Wysokińki, P.; et al. Seeking Optimal Nutrition for Healthy Body Mass Reduction Among Former Athletes. J. Hum. Kinet. 2017, 60, 63–75. [Google Scholar] [CrossRef][Green Version]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef]
- Westman, E.C.; Vernon, M.C.; Wallace, T.M.; Matthews, D.R. The assessment of insulin resistance in man. Diabet. Med. 2002, 19, 527–534. [Google Scholar]
- Hopkins, W.G. Linear models and effect magnitudes for research, clinical and practical applications. Sportscience 2010, 14, 49–57. [Google Scholar]
- Michalczyk, M.; Zajac, A.; Mikolajec, K.; Zydek, G.; Langfort, J. No Modification in Blood Lipoprotein Concentration but Changes in Body Composition after 4 Weeks of Low Carbohydrate Diet (LCD) Followed by 7 Days of Carbohydrate Loading in Basketball Players. J. Hum. Kinet. 2018, 65, 125–137. [Google Scholar] [CrossRef]
- Michalczyk, M.M.; Chycki, J.; Zajac, A.; Maszczyk, A.; Zydek, G.; Langfort, J. Anaerobic Performance after a Low-Carbohydrate Diet (LCD) Followed by 7 Days of Carbohydrate Loading in Male Basketball Players. Nutrients 2019, 11, 778. [Google Scholar] [CrossRef]
- Janovská, A.; Hatzinikolas, G.; Mano, M.; Wittert, G.A. The effect of dietary fat content on phospholipid fatty acid profile is muscle fiber type dependent. Am. J. Physiol. Endocrinol. Metab. 2010, 298, E779–E786. [Google Scholar] [CrossRef] [PubMed]
- Crockett, M.J.; Fehr, E. Social brains on drugs: Tools for neuromodulation in social neu-roscience. Soc. Cogn. Affect. Neurosci. 2014, 9, 250–254. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vermeer, A.B.L.; Riečanský, I.; Eisenegger, C. Competition, testosterone, and adult neurobehavioral plasticity. Prog. Brain Res. 2016, 229, 213–238. [Google Scholar] [CrossRef]
- Asarian, L.; Geary, N. Modulation of appetite by gonadal steroid hormones. Philos. Trans. R. Soc. B Boil. Sci. 2006, 361, 1251–1263. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developingand maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Choi, B.C.K.; Pak, A.W.P. Daily step goal of 10,000 steps: A literature review. Clin. Investig. Med. 2007, 30, 146. [Google Scholar] [CrossRef] [PubMed]
- Samaha, F.F.; Iqbal, N.; Seshadri, P.; Chicano, K.L.; Daily, D.A.; McGrory, J.; Williams, T.; Williams, M.; Gracely, E.J.; Stern, L. A Low-Carbohydrate as Compared with a Low-Fat Diet in Severe Obesity. N. Engl. J. Med. 2003, 348, 2074–2081. [Google Scholar] [CrossRef]
- Yamada, Y.; Uchida, J.; Izumi, H.; Tsukamoto, Y.; Inoue, G.; Watanabe, Y.; Irie, J.; Yamada, S. A Non-calorie-restricted Low-carbohydrate Diet is Effective as an Alternative Therapy for Patients with Type 2 Diabetes. Intern. Med. 2014, 53, 13–19. [Google Scholar] [CrossRef]
- Nielsen, J.V.; Joensson, E. Low-carbohydrate diet in type 2 diabetes. Stable improvement of bodyweight and glycemic control during 22 months follow-up. Nutr. Metab. 2006, 3, 22. [Google Scholar] [CrossRef]
- Paniagua, J.; De La Sacristana, A.G.; Romero, I.; Vidal-Puig, A.; Latre, J.; Sánchez, E.; Pérez-Martínez, P.; López-Miranda, J.; Jiménez, F.P. Monounsaturated Fat-Rich Diet Prevents Central Body Fat Distribution and Decreases Postprandial Adiponectin Expression Induced by a Carbohydrate-Rich Diet in Insulin-Resistant Subjects. Diabetes Care 2007, 30, 1717–1723. [Google Scholar] [CrossRef] [PubMed]
- Hatting, M.; Tavares, C.D.J.; Sharabi, K.; Rines, A.K.; Puigserver, P. Insulin regulation of gluconeogenesis. Ann. N. Y. Acad. Sci. 2017, 1411, 21–35. [Google Scholar] [CrossRef]
- Yancy, W.S.; Foy, M.; Chalecki, A.M.; Vernon, M.C.; Westman, E.C. A low-carbohydrate, ketogenic diet to treat type 2 diabetes. Nutr. Metab. 2005, 2, 34. [Google Scholar] [CrossRef] [PubMed]
- Templeman, N.M.; Skovsø, S.; Page, M.M.; E Lim, G.; Johnson, J.D. A causal role for hyperinsulinemia in obesity. J. Endocrinol. 2017, 232, R173–R183. [Google Scholar] [CrossRef] [PubMed]
- Kosinski, C.; Jornayvaz, F.R. Effects of Ketogenic Diets on Cardiovascular Risk Factors: Evidence from Animal and Human Studies. Nutrients 2017, 9, 517. [Google Scholar] [CrossRef]
- Johnstone, A.M.; Horgan, G.W.; Murison, S.D.; Bremner, D.M.; E Lobley, G. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 2008, 87, 44–55. [Google Scholar] [CrossRef]
- Howard, B.V. Polyunsaturated fatty acids results in greater cholesterol lowering and less triglyceride elevation than monounsaturated fatty acids. Am. J. Clin. Nutr. 1995, 2, 392. [Google Scholar] [CrossRef]
- Davidson, M.H. Mechanisms for the Hypotriglyceridemic Effect of Marine Omega-3 Fatty Acids. Am. J. Cardiol. 2006, 98, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Guisado, J.; Muñoz-Serrano, A. The Effect of the Spanish Ketogenic Mediterranean Diet on Nonalcoholic Fatty Liver Disease: A Pilot Study. J. Med. Food 2011, 14, 677–680. [Google Scholar] [CrossRef]
- Gugliucci, A.; Kotani, K.; Taing, J.; Matsuoka, Y.; Sano, Y.; Yoshimura, M.; Egawa, K.; Horikawa, C.; Kitagawa, Y.; Kiso, Y.; et al. Short-Term Low Calorie Diet Intervention Reduces Serum Advanced Glycation End Products in Healthy Overweight or Obese Adults. Ann. Nutr. Metab. 2009, 54, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Westerterp-Plantenga, M.; Nieuwenhuizen, A.; Tomé, D.; Soenen, S.; Westerterp, K. Dietary Protein, Weight Loss, and Weight Maintenance. Annu. Rev. Nutr. 2009, 29, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Veldhorst, M.A.; Smeets, A.; Soenen, S.; Hochstenbach-Waelen, A.; Hursel, R.; Diepvens, K.; Lejeune, M.; Luscombe-Marsh, N.D.; Westerterp-Plantenga, M. Protein-induced satiety: Effects and mechanisms of different proteins. Physiol. Behav. 2008, 94, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, S.; Neri, I.G.; Trentani, C.; Ferraris, C.; De Amicis, R.; Battezzati, A.; Veggiotti, P.; De Giorgis, V.; Tagliabue, A. Short-term effects of ketogenic diet on anthropometric parameters, body fat distribution, and inflammatory cytokine production in GLUT1 deficiency syndrome. Nutrients 2015, 31, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Dyson, P.A.; Beatty, S.; Matthews, D.R. A low-carbohydrate diet is more effective in reducing body weight than healthy eating in both diabetic and non-diabetic subjects. Diabet. Med. 2007, 24, 1430–1435. [Google Scholar] [CrossRef] [PubMed]
- Meckling, K.A.; O’Sullivan, C.; Saari, D. Comparison of a Low-Fat Diet to a Low-Carbohydrate Diet on Weight Loss, Body Composition, and Risk Factors for Diabetes and Cardiovascular Disease in Free-Living, Overweight Men and Women. J. Clin. Endocrinol. Metab. 2004, 89, 2717–2723. [Google Scholar] [CrossRef]
| Contents | LCKD Mean ± SD | CG Mean ± SD |
|---|---|---|
| RMR, kcal TDEE | 1616 ± 154 2263 ± 216 | 1637 ± 142 2292 ± 155 |
| TEI, kcal | 1810 ± 173 | 2170 ± 211 |
| Carbohydrate, % | 8 | 50 |
| Carbohydrate, g | 36 ± 3.5 | 286 ± 32 |
| Fibre, g | 16 ± 3 | 15 ± 5 |
| Proteins, % | 20 | 18 |
| Proteins, g | 90.5 ± 9 | 97.6 ± 9.5 |
| Proteins, g/kg bm/d | 1 ± 0.6 | 1.1 ± 0.8 |
| Fat, % | 72 | 32 |
| Fat, g | 145 ± 14 | 77 ± 7.5 |
| SFA, g | 35 ± 4 | 44 ± 3.5 |
| MUFA, g | 65 ± 8 | 15 ± 2 |
| PUFA, g | 45 ± 4 | 18 ± 2 |
| n-6, g | 38 ± 3.5 | 16 ± 1.5 |
| n-3, g | 7 ± 0.5 | 1.7 ± 0.5 |
| EPA and DHA, g | 5.4 ± 0.5 | 0.8 ± 0.1 |
| n-6/n-3 | 5.5:1 | 11:1 |
| Cholesterol, g | 543 ± 74 | 685 ± 58 |
| Variables | LCKD, BASELINE Mean ± SD | LCKD, AFTER Mean ± SD | CG, BASELINE Mean ± SD | CG, AFTER Mean ± SD |
|---|---|---|---|---|
| Gl, mmol/L | 5.94 ±0.56 | 4.74 ± 0.90 | 5.86 ± 0.39 | 5.82 ± 0.90 |
| I, µg/Dl | 14.12 ± 4.75 | 6.61 ± 2.63 | 13.78 ± 4.21 | 13.80 ± 3.63 |
| HbA1c, mg/L | 5.87 ± 0.94 | 5.38 ± 0.74 | 5.86 ± 0.60 | 5.90 ± 0.74 |
| HOMA-IR | 3.73 ± 1.2 | 1.38 ± 2.63 | 3.57 ± 1.09 | 3.58 ± 1.13 |
| TG, mg/dL | 213.45 ± 63.60 | 129.13 ± 46.23 | 210.57 ± 36.45 | 206.44 ± 50.03 |
| HDL-C, mg/dL | 36.71 ± 4.42 0.15 ± 0.03 | 52.99 ± 7.77 3.46 ± 0.06 | 44.14 ± 5.07 0.11 ± 0.03 | 43.01 ± 5.03 0.10 ± 0.09 |
| β-HB, mmol/L |
| Parameters | LCKG, BASELINE Mean ± SD | LCKG, AFTER Mean ± SD | CG, BASELINE Mean ± SD | CG, AFTER Mean ± SD |
|---|---|---|---|---|
| BM, kg | 89.08 ± 14.68 | 75.36 ± 13.47 | 90.63 ± 11.04 | 89.86 ± 11.30 |
| BMI, kg/m2 | 32.52 ± 4.50 | 27.47 ± 3.92 | 33.21 ± 4.55 | 32.92 ± 4.55 |
| WC, cm | 101.04 ± 11.86 | 87.34 ± 9.50 | 102.93 ± 10.32 | 103.67 ± 9.79 |
| HC, cm | 112.82 ± 9.89 | 101.21 ± 7.42 | 112.55 ± 8.90 | 111.36 ± 9.36 |
| TC, cm | 64.57 ± 6.36 | 56,91 ± 6,36 | 65.26 ± 6.71 | 64.94 ± 7.02 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michalczyk, M.M.; Klonek, G.; Maszczyk, A.; Zajac, A. The Effects of a Low Calorie Ketogenic Diet on Glycaemic Control Variables in Hyperinsulinemic Overweight/Obese Females. Nutrients 2020, 12, 1854. https://doi.org/10.3390/nu12061854
Michalczyk MM, Klonek G, Maszczyk A, Zajac A. The Effects of a Low Calorie Ketogenic Diet on Glycaemic Control Variables in Hyperinsulinemic Overweight/Obese Females. Nutrients. 2020; 12(6):1854. https://doi.org/10.3390/nu12061854
Chicago/Turabian StyleMichalczyk, Małgorzata Magdalena, Grzegorz Klonek, Adam Maszczyk, and Adam Zajac. 2020. "The Effects of a Low Calorie Ketogenic Diet on Glycaemic Control Variables in Hyperinsulinemic Overweight/Obese Females" Nutrients 12, no. 6: 1854. https://doi.org/10.3390/nu12061854
APA StyleMichalczyk, M. M., Klonek, G., Maszczyk, A., & Zajac, A. (2020). The Effects of a Low Calorie Ketogenic Diet on Glycaemic Control Variables in Hyperinsulinemic Overweight/Obese Females. Nutrients, 12(6), 1854. https://doi.org/10.3390/nu12061854







