SNP rs6564851 in the BCO1 Gene Is Associated with Varying Provitamin a Plasma Concentrations but Not with Retinol Concentrations among Adolescents from Rural Ghana
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Physical Examination
2.3. Questionnaire-Based Interviews
2.4. Identification of Releveant BCO1 SNPs
2.5. Laboratory Analyses
2.5.1. Biomarkers of Vitamin A Metabolism and Undernutrition
2.5.2. Genotyping
2.6. Statistical Analyses
- Additive effect:
- 0 = homozygous for normal conversion efficiency;
- 1 = one allele known to decrease conversion efficiency (heterozygous);
- 2 = homozygous for decreased conversion efficiency.
- Dominant effect:
- 0 = homozygous for normal conversion efficiency;
- 1 = heterozygous or homozygous for decreased conversion efficiency.
3. Results
3.1. Socio-Demographic Characteristics
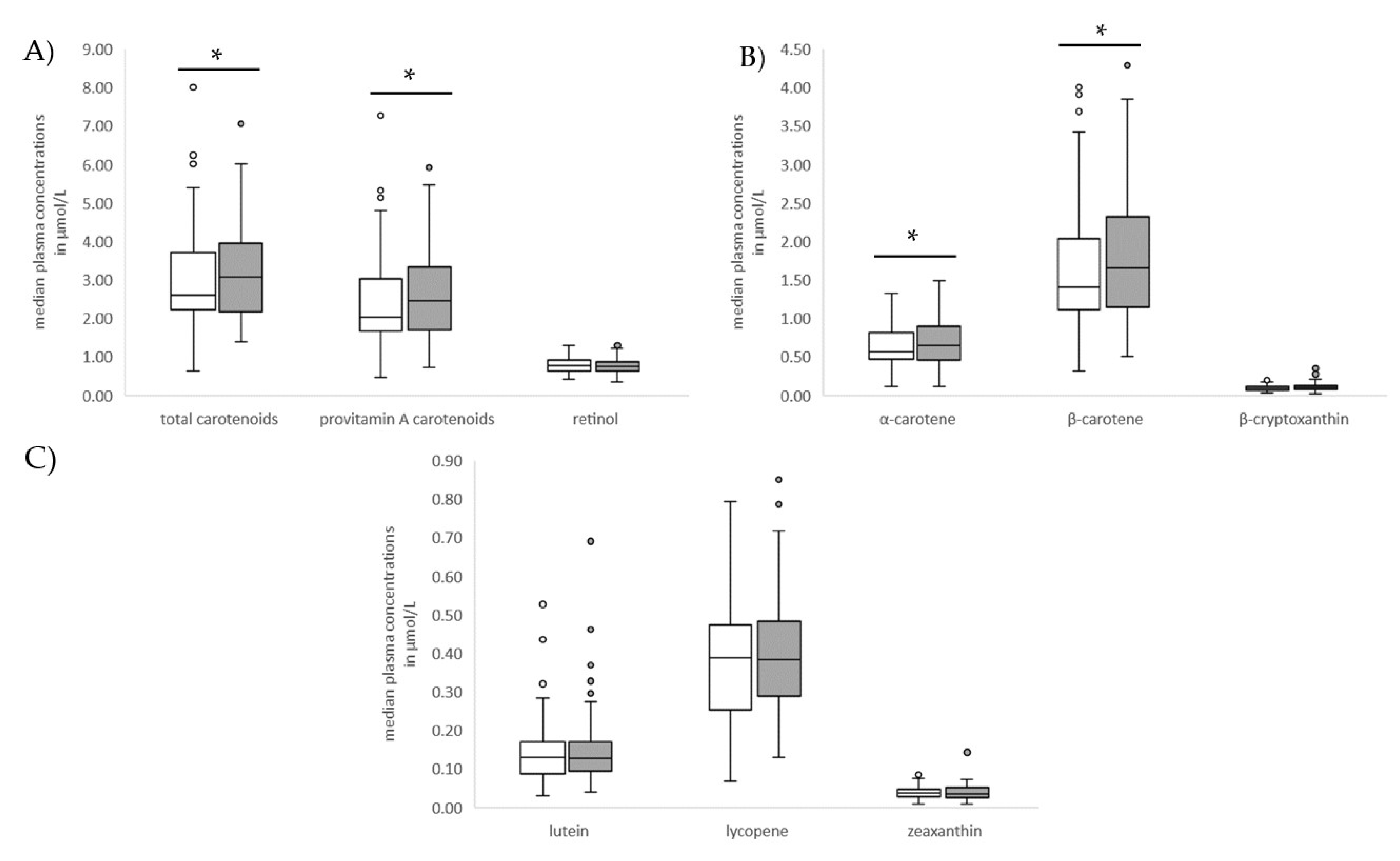
3.2. Anthropometric, Dietary and Clinical Factors
3.3. Genetic Variants and Plasma Carotenoid Concentrations
4. Discussion
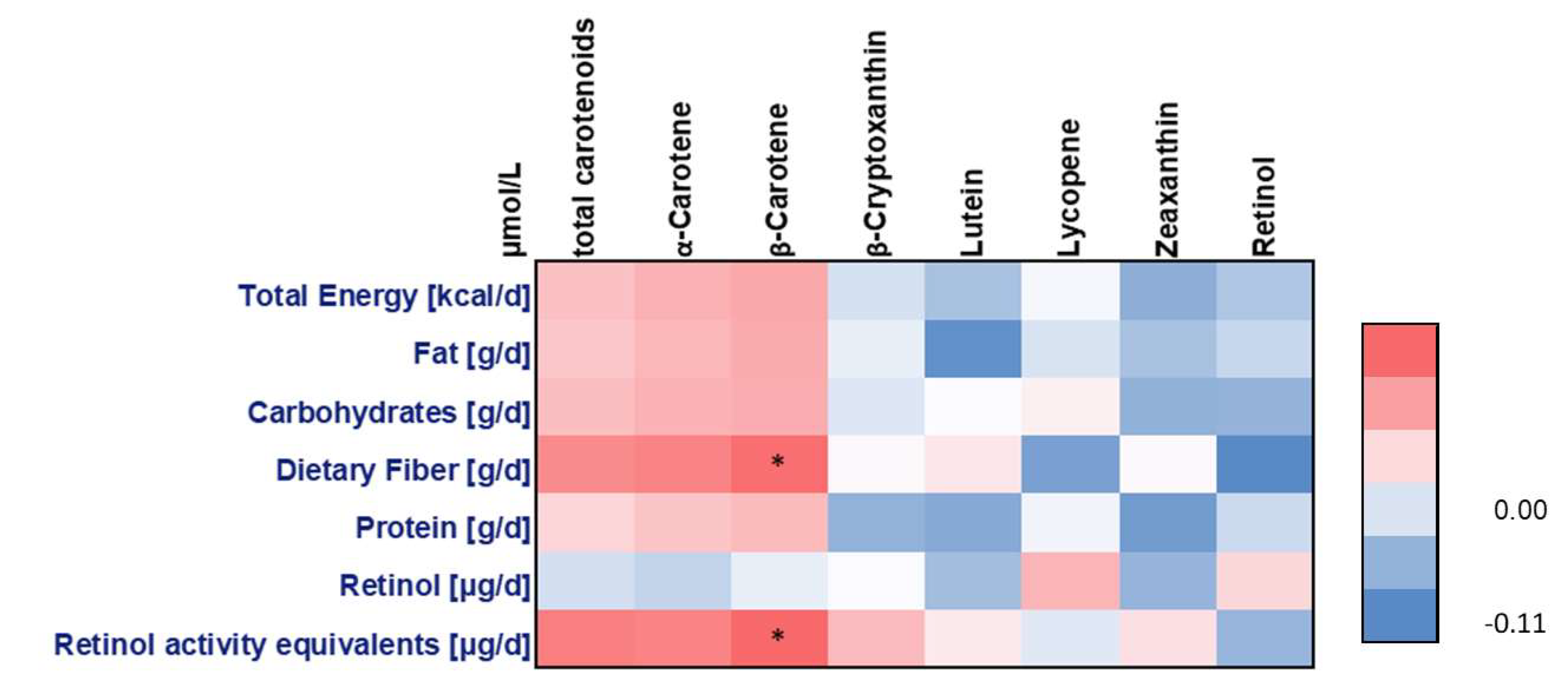
4.1. Non-Genetic Factors and Carotenoid Concentrations
4.2. BCO1 Variants and Carotenoid Concentrations
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Vitamin A Deficiency. Available online: https://www.who.int/nutrition/topics/vad/en/ (accessed on 26 August 2019).
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; De-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R.; et al. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Heal. 2015, 3, e528–e536. [Google Scholar] [CrossRef]
- World Health Organization. WHO Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin A Deficiency; World Health Organization: Geneva, Belgium, 2009. [Google Scholar]
- Mayo-Wilson, E.; Imdad, A.; Herzer, K.; Yakoob, M.Y.; Bhutta, Z.A. Vitamin A supplements for preventing mortality, illness, and blindness in children aged under 5: Systematic review and meta-analysis. BMJ 2011, 343, d5094. [Google Scholar] [CrossRef]
- Clagett-Dame, M.; Knutson, D. Vitamin A in Reproduction and Development. Nutrients 2011, 3, 385–428. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.T. IARC handbooks of cancer prevention volume 2: Carotenoids and volume 3: Vitamin A. Eur. J. Clin. Nutr. 1999, 53, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Acad. Nutr. Diet. 2001, 101, 294. [Google Scholar]
- Reboul, E. Absorption of Vitamin A and Carotenoids by the Enterocyte: Focus on Transport Proteins. Nutrients 2013, 5, 3563–3581. [Google Scholar] [CrossRef]
- Lindqvist, A.; Andersson, S. Cell type-specific expression of beta-carotene 15,15’-mono-oxygenase in human tissues. J. Histochem. Cytochem. 2004, 52, 491–499. [Google Scholar] [CrossRef]
- Yan, W.; Jang, G.F.; Haeseleer, F.; Esumi, N.; Chang, J.; Kerrigan, M.; Campochiaro, M.; Campochiaro, P.; Palczewski, K.; Zack, D.J. Cloning and characterization of a human beta,beta-carotene-15,15’-dioxygenase that is highly expressed in the retinal pigment epithelium. Genomics 2001, 72, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.H. Mechanisms of Digestion and Absorption of Dietary Vitamin A. Annu. Rev. Nutr. 2005, 25, 87–103. [Google Scholar] [CrossRef] [PubMed]
- Lobo, G.P.; Hessel, S.; Eichinger, A.; Noy, N.; Moise, A.R.; Wyss, A.; Palczewski, K.; Von Lintig, J. ISX is a retinoic acid-sensitive gatekeeper that controls intestinal β,β-carotene absorption and vitamin A production. FASEB J. 2010, 24, 1656–1666. [Google Scholar] [CrossRef] [PubMed]
- Alicke, M.; Boakye-Appiah, J.K.; Abdul-Jalil, I.; Henze, A.; Van Der Giet, M.; Schulze, M.B.; Schweigert, F.J.; Mockenhaupt, F.P.; Bedu-Addo, G.; Danquah, I. Adolescent health in rural Ghana: A cross-sectional study on the co-occurrence of infectious diseases, malnutrition and cardio-metabolic risk factors. PLoS ONE 2017, 12, e0180436. [Google Scholar] [CrossRef] [PubMed]
- Al-Delaimy, W.K.; Van Kappel, A.L.; Ferrari, P.; Slimani, N.; Steghens, J.-P.; Bingham, S.; Johansson, I.; Wallström, P.; Overvad, K.; Tjonneland, A.; et al. Plasma levels of six carotenoids in nine European countries: Report from the European Prospective Investigation into Cancer and Nutrition (EPIC). Public Heal. Nutr. 2004, 7, 713–722. [Google Scholar] [CrossRef]
- Hercberg, S. The SU.VI.MAX study, a randomized, placebo-controlled trial on the effects of antioxidant vitamins and minerals on health. Ann. Pharm. Fr. 2006, 64, 2335–2342. [Google Scholar]
- Hanson, C.; Lyden, E.R.; Anderson-Berry, A.; Kocmich, N.; Rezac, A.; Delair, S.F.; Furtado, J.; Van Ormer, M.; Izevbigie, N.; Olateju, E.; et al. Status of Retinoids and Carotenoids and Associations with Clinical Outcomes in Maternal-Infant Pairs in Nigeria. Nutrients 2018, 10, 1286. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, L.; Perry, J.R.; Matteini, A.; Perola, M.; Tanaka, T.; Silander, K.; Rice, N.; Melzer, D.; Murray, A.; Cluett, C.; et al. Common variation in the beta-carotene 15,15’-monooxygenase 1 gene affects circulating levels of carotenoids: A genome-wide association study. Am. J. Hum. Genet. 2009, 84, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, S.J.; Hazra, A.; Chen, C.; Eliassen, A.H.; Kraft, P.; Rosner, B.A.; Willett, W.C. β-Carotene 15,15’-monooxygenase 1 single nucleotide polymorphisms in relation to plasma carotenoid and retinol concentrations in women of European descent. Am. J. Clin. Nutr. 2012, 96, 1379–1389. [Google Scholar] [CrossRef]
- Mondul, A.M.; Yu, K.; Wheeler, W.; Zhang, H.; Weinstein, S.J.; Major, J.M.; Cornelis, M.C.; Männistö, S.; Hazra, A.; Hsing, A.W.; et al. Genome-wide association study of circulating retinol levels. Hum. Mol. Genet. 2011, 20, 4724–4731. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Desmarchelier, C. Genetic Variations Associated with Vitamin A Status and Vitamin A Bioavailability. Nutrients 2017, 9, 246. [Google Scholar] [CrossRef]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Rühl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600–1685. [Google Scholar] [CrossRef]
- Lietz, G.; Oxley, A.; Leung, W.; Hesketh, J. Single nucleotide polymorphisms upstream from the beta-carotene 15,15’-monoxygenase gene influence provitamin A conversion efficiency in female volunteers. J. Nutr. 2012, 142, 161S–165S. [Google Scholar] [CrossRef]
- Ford, E.S.; Gillespie, C.; Ballew, C.; Sowell, A.; Mannino, D.M. Serum carotenoid concentrations in US children and adolescents. Am. J. Clin. Nutr. 2002, 76, 818–827. [Google Scholar] [CrossRef] [PubMed]
- Neuhouser, M.L.; Rock, C.L.; Eldridge, A.L.; Kristal, A.; Patterson, R.E.; Cooper, D.A.; Neumark-Sztainer, D.; Cheskin, L.J.; Thornquist, M.D. Serum Concentrations of Retinol, α-Tocopherol and the Carotenoids Are Influenced by Diet, Race and Obesity in a Sample of Healthy Adolescents. J. Nutr. 2001, 131, 2184–2191. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Agogo Presbytarian Hospital, Ghana. Available online: https://www.who.int/buruli/events/agogo_hospital/en/ (accessed on 2 September 2019).
- Bedu-Addo, G.; Alicke, M.; Boakye-Appiah, J.K.; Abdul-Jalil, I.; Van Der Giet, M.; Schulze, M.B.; Mockenhaupt, F.P.; Danquah, I. In utero exposure to malaria is associated with metabolic traits in adolescence: The Agogo 2000 birth cohort study. J. Infect. 2017, 75, 455–463. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cunningham, F.; Achuthan, P.; Akanni, W.; Allen, J.; Amode, M.R.; Armean, I.M.; Bennett, R.; Bhai, J.; Billis, K.; Boddu, S.; et al. Ensembl 2019. Nucleic Acids Res. 2018, 47, D745–D751. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations; World Health Organization: Geneva, Belgium, 2011. [Google Scholar]
- Applied Biosystems. TaqMan (R) Genotyping Master Mix Protocol; Applied Biosystems: Foster City, CA, USA, 2010. [Google Scholar]
- Applied Biosystems. TaqMan(R) SNP Genotyping Assays User Guide; Applied Biosystems: Foster City, CA, USA, 2017. [Google Scholar]
- Rodriguez, S.; Gaunt, T.R.; Day, I.N. Hardy-Weinberg equilibrium testing of biological ascertainment for Mendelian randomization studies. Am. J. Epidemiol. 2009, 169, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Santamarĩa, J.V.; Breidenassel, C.; Folle, J.; González-Gross, M. Retinol, β-carotene, α-tocopherol and vitamin D status in European adolescents; regional differences and variability: A review. Nutr. Hosp. 2011, 26, 280–288. [Google Scholar]
- Desmarchelier, C.; Borel, P. Overview of carotenoid bioavailability determinants: From dietary factors to host genetic variations. Trends Food Sci. Technol. 2017, 69, 270–280. [Google Scholar] [CrossRef]
- World Health Organization. Serum Retinol Concentrations for Determining the Prevalence of Vitamin A Deficiency in Populations; World Health Organization: Geneva, Belgium, 2011. [Google Scholar]
- Olson, J.A. Serum Levels of Vitamin A and Carotenoids as Reflectors of Nutritional Status12. J. Natl. Cancer Inst. 1984, 73, 1439–1444. [Google Scholar] [CrossRef]
- Tanumihardjo, S.A.; Palacios, N.; Pixley, K.V. Provitamin a carotenoid bioavailability:what really matters? Int. J. Vitam. Nutr. Res. 2010, 80, 336–350. [Google Scholar] [CrossRef]
- Ribaya-Mercado, J.D.; Solon, F.S.; Solon, M.A.; Cabal-Barza, M.A.; Perfecto, C.S.; Tang, G.; Solon, J.A.; Fjeld, C.R.; Russell, R.M. Bioconversion of plant carotenoids to vitamin A in Filipino school-aged children varies inversely with vitamin A status. Am. J. Clin. Nutr. 2000, 72, 455–465. [Google Scholar] [CrossRef]
- Mason, J.B.; Greiner, T.; Shrimpton, R.; Sanders, D.; Yukich, J. Reply to West et al. Vitamin A policies need rethinking. Int. J. Epidemiol. 2015, 44, 294–296. [Google Scholar] [CrossRef]
- Biro, E.; Menon, P. Adressing the Challenge of Hidden Hunger. Available online: https://www.globalhungerindex.org/issues-in-focus/2014.html (accessed on 13 October 2019).
- University of Ghana, GroundWork; University of Wisconsin-Madison, KEMRI-WellcomeTrust, UNICEF. Ghana Micronutrient Survey 2017; University of Ghana: Accra, Republic of Ghana, 2017. [Google Scholar]
- Schweigert, F.J. Inflammation-induced changes in the nutritional biomarkers serum retinol and carotenoids. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Gan, H.K.; Pond, G.R.; Moore, M.J.; Siu, L.L. Reply to D.J. Stewart. J. Clin. Oncol. 2010, 28, e652–e653. [Google Scholar] [CrossRef]
- Kelleher, J.; Humphrey, C.S.; Homer, D.; Davison, A.M.; Giles, G.R.; Losowsky, M.S. Vitamin A and its Transport Proteins in Patients with Chronic Renal Failure Receiving Maintenance Haemodialysis and after Renal Transplantation. Clin. Sci. 1983, 65, 619–626. [Google Scholar] [CrossRef]
- Mills, J.P.; Furr, H.C.; Tanumihardjo, S.A. Retinol to Retinol-Binding Protein (RBP) Is Low in Obese Adults due to Elevatedapo-RBP. Exp. Boil. Med. 2008, 233, 1255–1261. [Google Scholar] [CrossRef]
- Zanotti, G.; Berni, R. Plasma Retinol-Binding Protein: Structure and Interactions with Retinol, Retinoids, and Transthyretin. Nitric Oxide 2004, 69, 271–295. [Google Scholar] [CrossRef]
- Amengual, J.; Widjaja-Adhi, M.A.K.; Rodriguez-Santiago, S.; Hessel, S.; Golczak, M.; Palczewski, K.; Von Lintig, J. Two Carotenoid Oxygenases Contribute to Mammalian Provitamin A Metabolism. J. Boil. Chem. 2013, 288, 34081–34096. [Google Scholar] [CrossRef]
- McLaren, D.S.; Zekian, B. Failure of enzymic cleavage of beta-carotene. The cause of vitamin A deficiency in a child. Am. J. Dis. Child. 1971, 121, 278–280. [Google Scholar] [CrossRef] [PubMed]
- West, C.E.; Eilander, A.; Van Lieshout, M. Consequences of Revised Estimates of Carotenoid Bioefficacy for Dietary Control of Vitamin A Deficiency in Developing Countries. J. Nutr. 2002, 132, 2920S–2926S. [Google Scholar] [CrossRef]
- Lobo, G.P.; Amengual, J.; Baus, D.; Shivdasani, R.A.; Taylor, D.; Von Lintig, J. Genetics and Diet Regulate Vitamin A Production via the Homeobox Transcription Factor ISX*. J. Boil. Chem. 2013, 288, 9017–9027. [Google Scholar] [CrossRef]
- Yabuta, S.; Urata, M.; Wai Kun, R.Y.; Masaki, M.; Shidoji, Y. Common SNP rs6564851 in the BCO1 Gene Affects the Circulating Levels of beta-Carotene and the Daily Intake of Carotenoids in Healthy Japanese Women. PLoS ONE 2016, 11, e0168857. [Google Scholar] [CrossRef] [PubMed]
- Hessel, S.; Eichinger, A.; Isken, A.; Amengual, J.; Hunzelmann, S.; Hoeller, U.; Elste, V.; Hunziker, W.; Goralczyk, R.; Oberhauser, V.; et al. CMO1 Deficiency Abolishes Vitamin A Production from β-Carotene and Alters Lipid Metabolism in Mice. J. Boil. Chem. 2007, 282, 33553–33561. [Google Scholar] [CrossRef] [PubMed]
| Total Carotenoid Concentration [µmol/L] | ||||
|---|---|---|---|---|
| Total (n = 189) 2.81 (2.17–3.78) | First Tertile (n = 63) 1.97 (1.61–2.17) | Second Tertile (n = 63) 2.81 (2.55–3.09) | Third Tertile (n = 63) 4.19 (3.78–4.90) | |
| Sex (m/f) | 94/95 | 32/31 | 31/32 | 31/32 |
| Age (in years) | 15.2 (15.0–15.3) | 15.2 (15.1–15.3) | 15.2 (15.0–15.3) | 15.2 (15.0–15.3) |
| Residence, Agogo % | 71.4 (135) | 68.3 (43) | 74.6 (47) | 71.4 (45) |
| Place of school, Agogo % | 57.1 (108) | 55.6 (35) | 54.0 (34) | 61.9 (39) |
| Illiteracy % | 9.0 (17) | 6.4 (4) | 6.4 (4) | 14.3 (9) |
| Education % | ||||
| Mother | ||||
| None | 4.2 (8) | 4.8 (3) | 0 (0) | 7.9 (5) |
| Primary | 18.0 (34) | 20.6 (13) | 20.6 (13) | 12.7 (8) |
| Secondary | 18.5 (35) | 25.4 (16) | 12.7 (8) | 17.5 (11) |
| Tertiary | 3.2 (6) | 4.8 (3) | 1.6 (1) | 3.2 (2) |
| Other | 56.1 (106) | 44.4 (28) | 65.1 (41) | 58.7 (37) |
| Father | ||||
| None | 3.7 (7) | 3.2 (2) | 4.8 (3) | 3.2 (2) |
| Primary | 12.2 (23) | 15.9 (10) | 14.3 (9) | 6.3 (4) |
| Secondary | 29.1 (55) | 33.3 (21) | 23.8 (15) | 30.2 (19) |
| Tertiary | 8.5 (16) | 6.3 (4) | 11.1 (7) | 7.9 (5) |
| Other | 46.5 (88) | 41.3 (26) | 46.0 (29) | 52.4 (33) |
| Occupation % | ||||
| Mother | ||||
| Manual work | 22.7 (43) | 19.1 (12) | 19.1 (12) | 30.2 (19) |
| Intellectual work | 63.5 (120) | 68.2 (43) | 69.8 (44) | 52.4 (33) |
| Other work | 10.6 (20) | 11.1 (7) | 9.5 (6) | 11.1 (7) |
| Unemployed | 3.2 (6) | 1.6 (1) | 1.6 (1) | 6.3 (4) |
| Father | ||||
| Manual work | 43.4 (82) | 39.8 (25) | 42.8 (27) | 47.6 (30) |
| Intellectual work | 30.2 (57) | 39.6 (25) | 27.0 (17) | 23.8 (15) |
| Other work | 25.9 (49) | 20.6 (13) | 28.6 (18) | 28.6 (18) |
| Unemployed | 0.5 (1) | 0 (0) | 1.6 (1) | 0 (0) |
| Number of siblings | 4 (3–5) | 4 (3–5) | 4 (3–5) | 4 (3–5) |
| People in the household | 11 (7–20) | 10 (8–20) | 11 (7–20) | 12 (7–18) |
| Wealth score | 0.55 (0.36–0.64) | 0.55 (0.36–0.64) | 0.55 (0.36–0.73) | 0.46 (0.27–0.64) |
| Total Carotenoid Concentration [µmol/L] | ||||
|---|---|---|---|---|
| Total (n = 189) 2.81 (2.17–3.78) | First Tertile (n = 63) 1.97 (1.61–2.17) | Second Tertile (n = 63) 2.81 (2.55–3.09) | Third Tertile (n = 63) 4.19 (3.78–4.90) | |
| BMI [kg/m²] | 19.1 (17.6–20.7) | 19.4 (17.9–21.7) | 19.0 (17.7–20.8) | 19.0 (17.5–20.2) |
| HAZ | −0.87 (−1.55–0.28) | −0.70 (−1.47–0.03) | −0.83 (−1.67–0.24) | −1.17 (−1.57–0.52) |
| BAZ | −0.40 (−1.07–0.19) | −0.32 (−0.96–0.59) | −0.39 (−1.01–0.13) | −0.54 (−1.11–0.02) |
| Energy intake [kcal/d] | 2872 (2424–3440) | 2748 (2380–3436) | 2921 (2445–3440) | 2961 (2424–3490) |
| CHO [energy%] | 48.2 (44.3–51.5) | 47.2 (44.7–50.7) | 49.0 (44.2–52.4) | 48.0 (44.2–51.2) |
| Fat [energy%] | 37.1 (34.6–40.2) | 37.7 (35.4–40.2) | 36.6 (34.1–40.1) | 37.9 (34.3–41.3) |
| Protein [energy%] | 14.5 (13.5–15.4) | 14.5 (13.7–15.5) | 14.6 (13.4–15.4) | 14.2 (13.5–15.3) |
| RAE [µg/d] | 1523 (1229–1879) | 1440 (1109–1879) | 1545 (1214–1825) | 1529 (1315–2015) |
| Physical activity [min/day] a | 179 (167–196) | 179 (168–198) | 181 (168–195) | 177 (164–196) |
| Blood pressure [mmHg] | ||||
| Systolic | 110 (103–117) | 110 (102–117) | 109 (102–118) | 112 (104–119) |
| Diastolic | 68 (62–74) | 68 (63–73) | 66 (61–74) | 70 (64–75) |
| Blood parameters | ||||
| α-Carotene [µmol/L] | 0.61 (0.46–0.86) | 0.39 (0.32–0.47) | 0.60 (0.54–0.66) | 0.94 (0.86–1.10) |
| β-Carotene [µmol/L] | 1.49 (1.17–2.22) | 0.97 (0.79–1.12) | 1.49 (1.37–1.77) | 2.52 (2.22–2.96) |
| β-Cryptoxanthin [µmol/L] | 0.09 (0.07–0.12) | 0.06 (0.04–0.92) | 0.10 (0.08–0.13) | 0.11 (0.09–0.15) |
| Lutein [µmol/L] | 0.13 (0.09–0.17) | 0.08 (0.07–0.12) | 0.13 (0.10–0.18) | 0.16 (0.13–0.20) |
| Lycopene [µmol/L] | 0.38 (0.28–0.48) | 0.33 (0.22–0.43) | 0.39 (0.30–0.50) | 0.42 (0.30–0.59) |
| Zeaxanthin [µmol/L] | 0.04 (0.03–0.05) | 0.03 (0.02–0.04) | 0.04 (0.03–0.06) | 0.05 (0.03–0.06) |
| Retinol [µmol/L] | 0.77 (0.64–0.91) | 0.79 (0.61–0.92) | 0.75 (0.62–0.89) | 0.75 (0.66–0.93) |
| VAD % | 35.5 (67) | 36.5 (23) | 39.7 (25) | 30.2 (19) |
| RBP4 [µmol/L] b | 1.17 (0.91–1.42) | 1.22 (0.89–1.53) | 1.18 (0.92–1.42) | 1.16 (0.92–1.34) |
| TTR [µmol/L] b | 2.79 (2.25–3.51) | 2.93 (2.27–3.55) | 2.66 (2.20–3.31) | 2.91 (2.33–3.53) |
| RBP4/TTR b | 0.41 (0.33–0.50) | 0.41 (0.36–0.47) | 0.40 (0.32–0.53) | 0.42 (0.33–0.49) |
| CRP [mg/L] b | 0.62 (0.30–2.11) | 0.57 (0.29–2.16) | 0.69 (0.42–2.18) | 0.56 (0.10–1.34) |
| Hemoglobin [g/dL] | 13.0 (12.2–14.0) | 13.2 (12.1–14.1) | 13.0 (12.4–13.9) | 12.9 (12.1–14.0) |
| Anemia (positive %) | 30.7 (58) | 30.2 (19) | 27.0 (17) | 34.9 (22) |
| Ferritin [µg/L] b | 57.8 (39.9–90.0) | 62.4 (40.1–90.0) | 50.7 (39.1–77.9) | 64.2 (40.6–96.4) |
| Fasting plasma glucose [mmol/L] | 4.3 (3.9–4.7) | 4.2 (3.8–4.6) | 4.3 (3.9–4.8) | 4.3 (3.9–4.8) |
| First Tertile (n = 63) 1.97 (1.61–2.17) | Second Tertile (n = 63) 2.81 (2.55–3.09) | Third Tertile (n = 63) 4.19 (3.78–4.90) | x² p-Value | |
|---|---|---|---|---|
| rs6564851 GT/GG | 42.9 (27) | 50.8 (32) | 65.1 (41) | 0.041 |
| rs7500996 CT/CC | 50.8 (32) | 52.4 (33) | 54.0 (34) | 0.938 |
| rs10048138 AG/AA | 58.7 (37) | 69.8 (44) | 58.7 (37) | 0.331 |
| rs6420424 AG/AA | 69.8 (44) | 63.5 (40) | 66.7 (42) | 0.752 |
| rs8044334 GT/GG | 82.5 (52) | 79.4 (50) | 82.5 (52) | 0.869 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graßmann, S.; Pivovarova-Ramich, O.; Henze, A.; Raila, J.; Ampem Amoako, Y.; King Nyamekye, R.; Bedu-Addo, G.; Mockenhaupt, F.P.; Schulze, M.B.; Danquah, I. SNP rs6564851 in the BCO1 Gene Is Associated with Varying Provitamin a Plasma Concentrations but Not with Retinol Concentrations among Adolescents from Rural Ghana. Nutrients 2020, 12, 1786. https://doi.org/10.3390/nu12061786
Graßmann S, Pivovarova-Ramich O, Henze A, Raila J, Ampem Amoako Y, King Nyamekye R, Bedu-Addo G, Mockenhaupt FP, Schulze MB, Danquah I. SNP rs6564851 in the BCO1 Gene Is Associated with Varying Provitamin a Plasma Concentrations but Not with Retinol Concentrations among Adolescents from Rural Ghana. Nutrients. 2020; 12(6):1786. https://doi.org/10.3390/nu12061786
Chicago/Turabian StyleGraßmann, Sophie, Olga Pivovarova-Ramich, Andrea Henze, Jens Raila, Yaw Ampem Amoako, Richard King Nyamekye, George Bedu-Addo, Frank P. Mockenhaupt, Matthias B. Schulze, and Ina Danquah. 2020. "SNP rs6564851 in the BCO1 Gene Is Associated with Varying Provitamin a Plasma Concentrations but Not with Retinol Concentrations among Adolescents from Rural Ghana" Nutrients 12, no. 6: 1786. https://doi.org/10.3390/nu12061786
APA StyleGraßmann, S., Pivovarova-Ramich, O., Henze, A., Raila, J., Ampem Amoako, Y., King Nyamekye, R., Bedu-Addo, G., Mockenhaupt, F. P., Schulze, M. B., & Danquah, I. (2020). SNP rs6564851 in the BCO1 Gene Is Associated with Varying Provitamin a Plasma Concentrations but Not with Retinol Concentrations among Adolescents from Rural Ghana. Nutrients, 12(6), 1786. https://doi.org/10.3390/nu12061786



.png)



