Microbiota Composition of Breast Milk from Women of Different Ethnicity from the Manawatu—Wanganui Region of New Zealand
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Microbiota Quantification
2.3. Immune and Growth Factor Assays
2.4. Statistical Analysis
3. Results
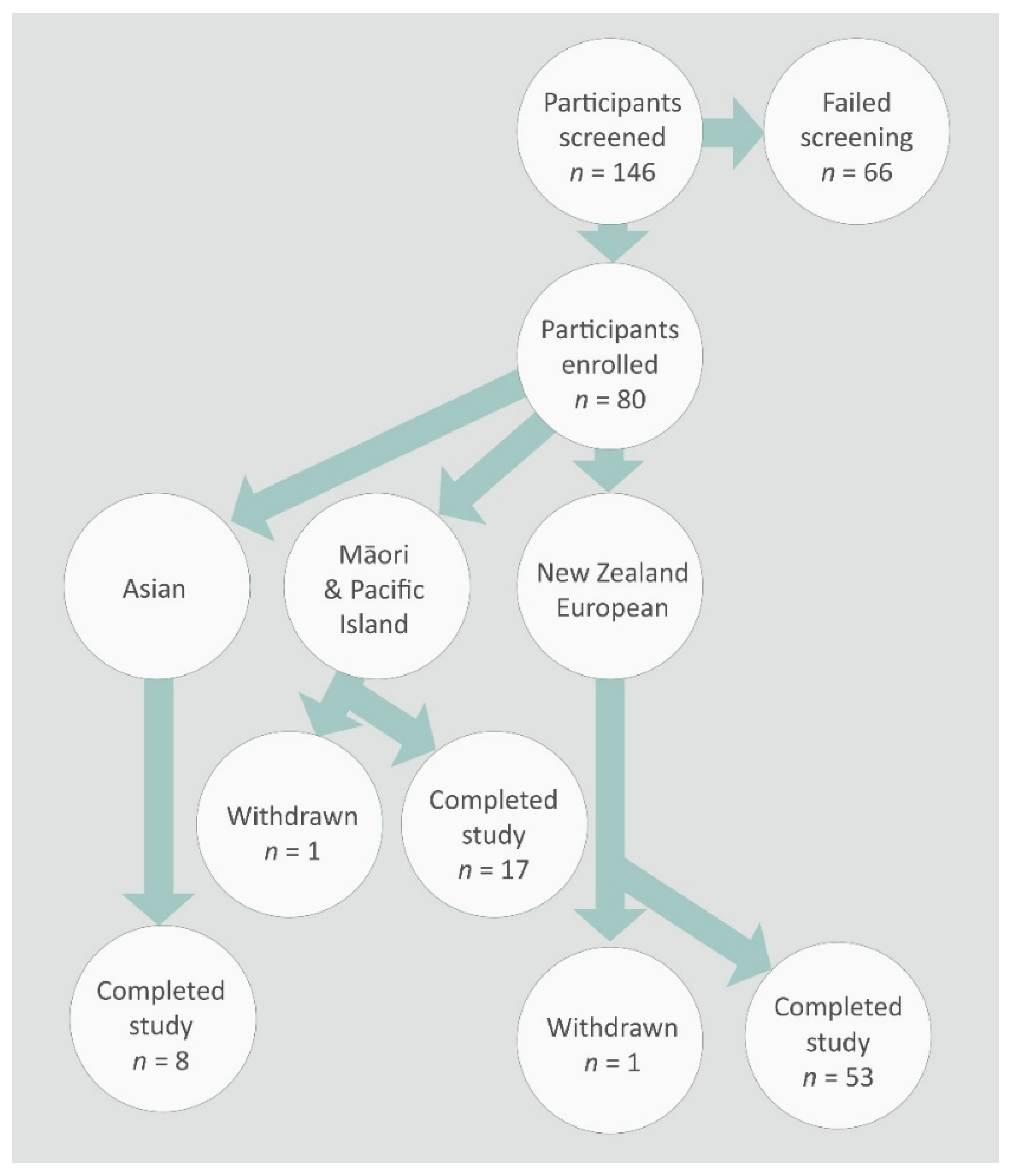
3.1. Study Participants
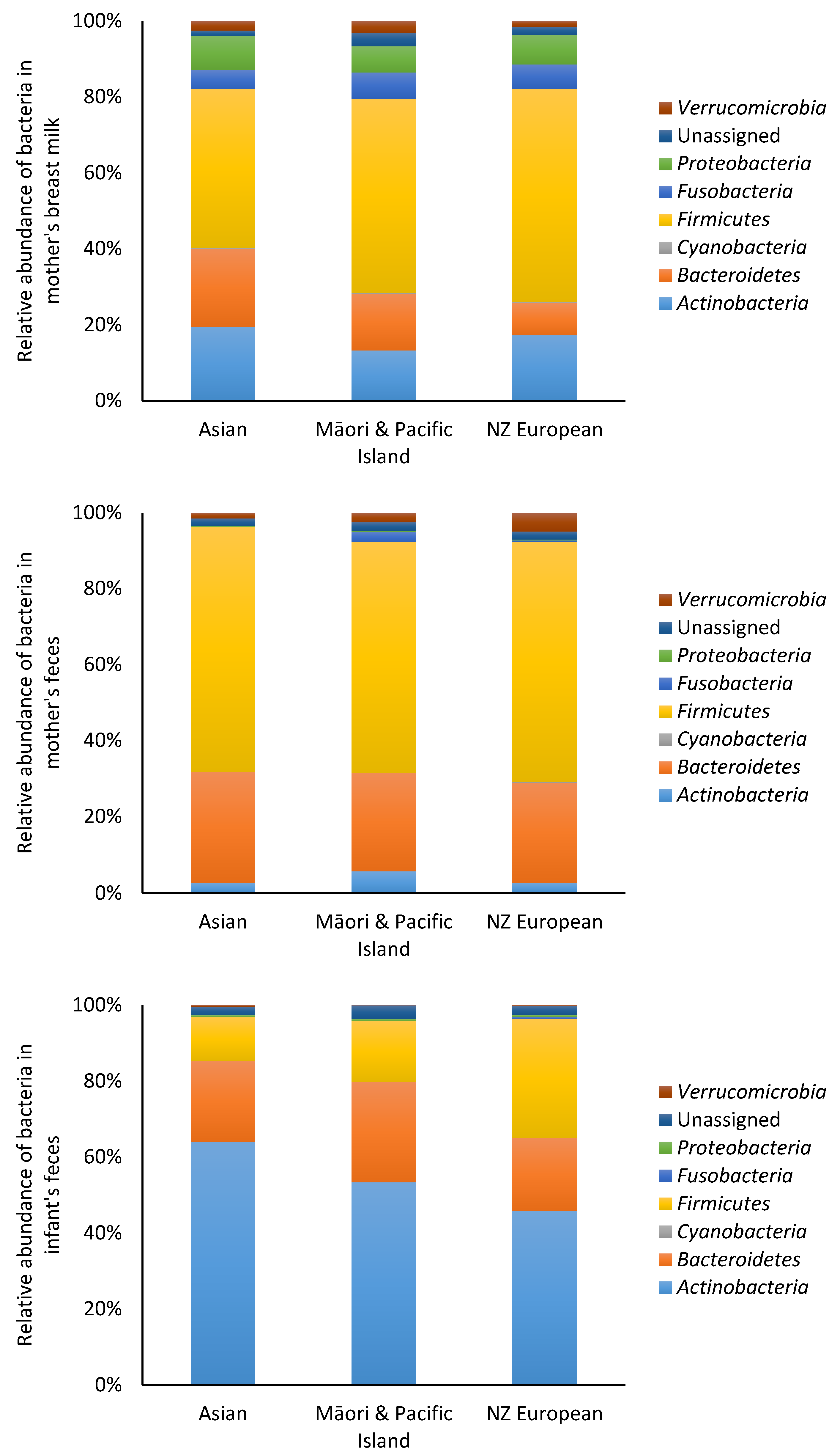
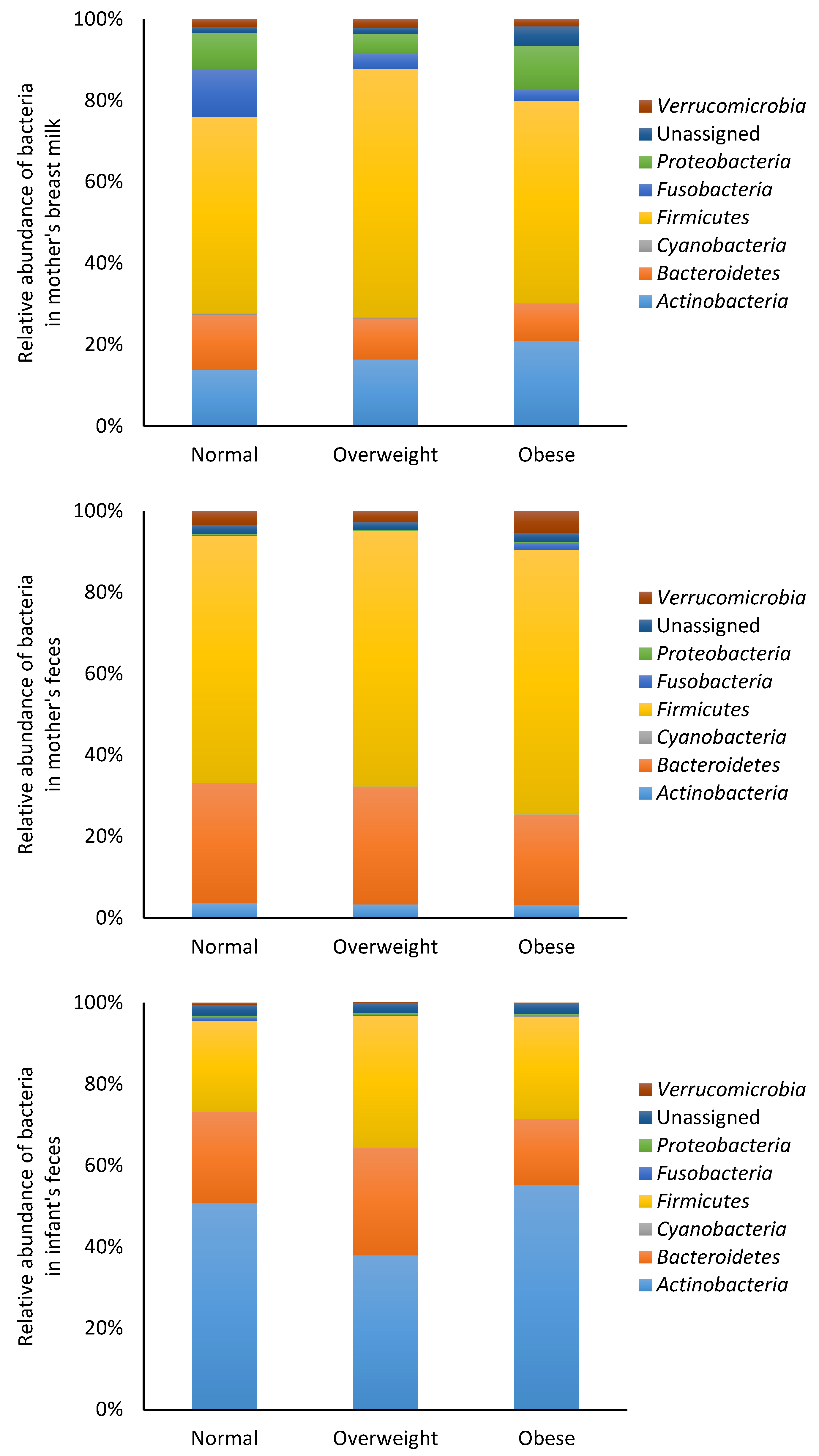
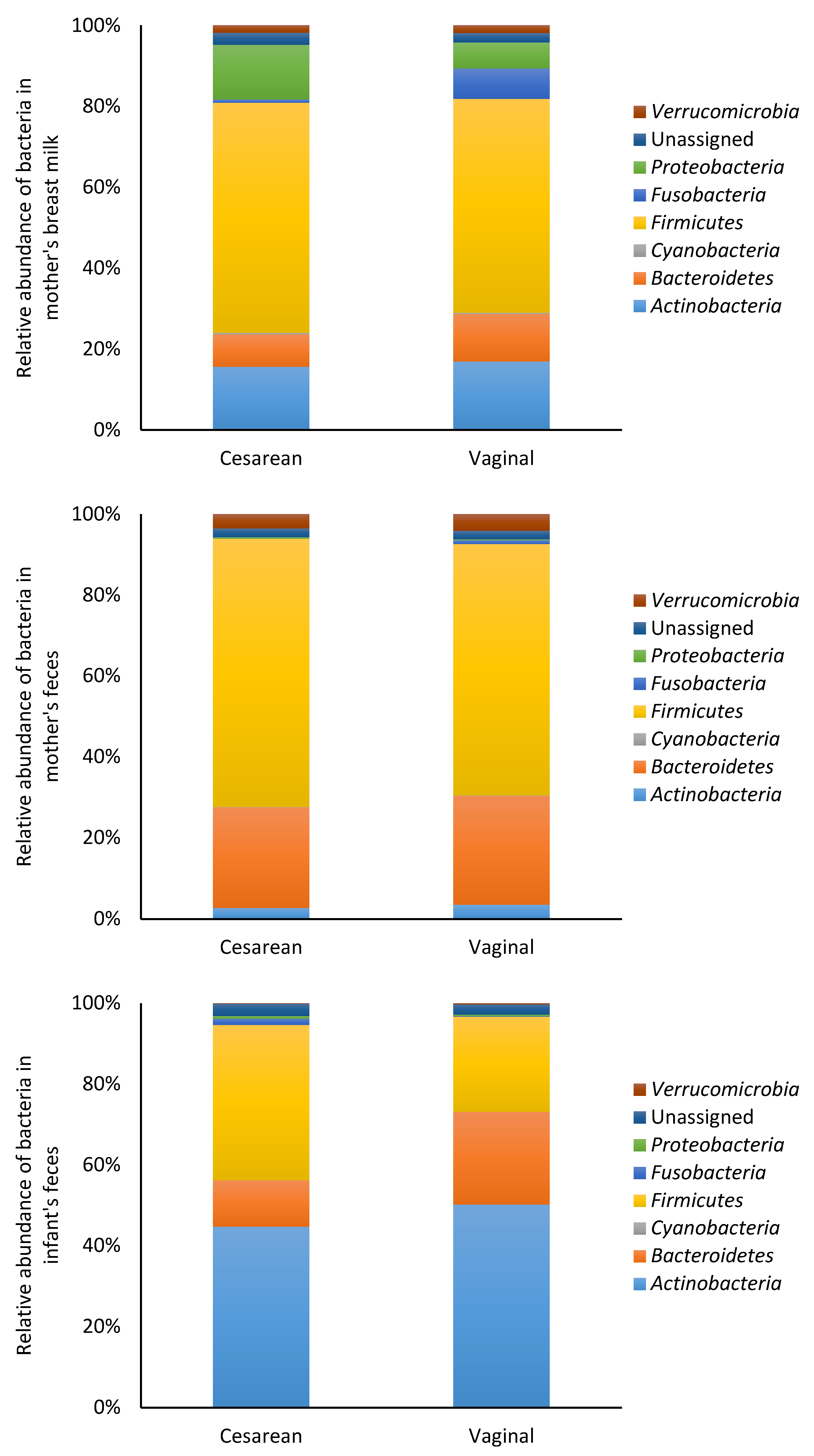
3.2. Microbiota
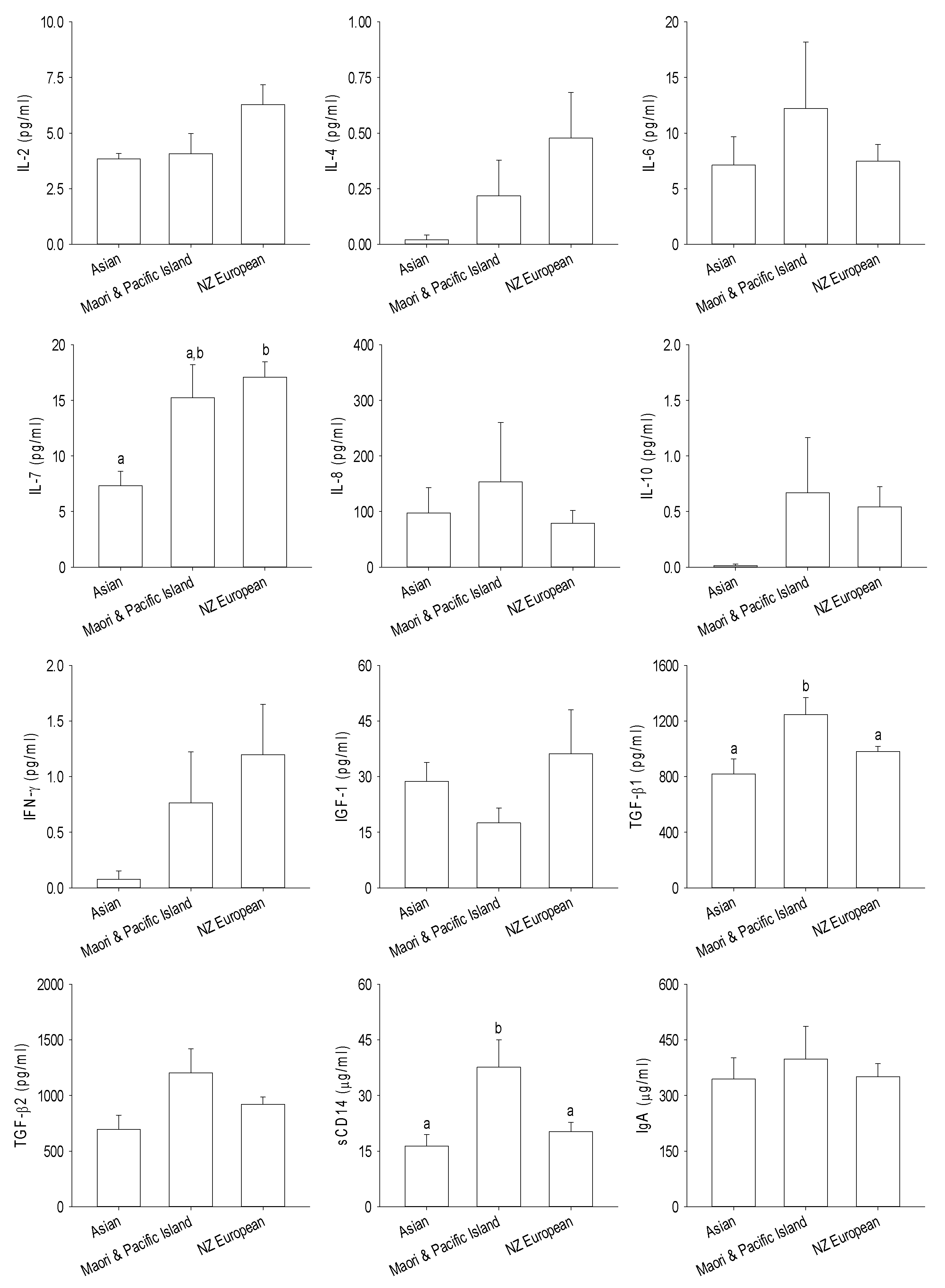
3.3. Immune and Growth Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sommer, F.; Backhed, F. The gut microbiota—Masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef]
- Di Giulio, D.B.; Romero, R.; Kusanovic, J.P.; Gómez, R.; Kim, C.J.; Seok, K.S.; Gotsch, F.; Mazaki-Tovi, S.; Vaisbuch, E.; Sanders, K.; et al. Prevalence and diversity of microbes in the amniotic fluid, the fetal inflammatory response, and pregnancy outcome in women with preterm pre-labor rupture of membranes. Am. J. Reprod. Immunol. 2010, 64, 38–57. [Google Scholar]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, P.; Pasolli, E.; Tett, A.; Asnicar, F.; Gorfer, V.; Fedi, S.; Armanini, F.; Truong, D.T.; Manara, S.; Zolfo, M.; et al. Mother to infant microbial transmission from different body sites shapes the developing infant gut microbiome. Cell Host Microbe 2018, 24, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Calatayud, M.; Koren, O.; Collado, M.C. Maternal microbiome and metabolic health program microbiome development and health of the offspring. Trends Endocrinol. Metab. 2019, 30, 735–744. [Google Scholar] [CrossRef]
- Kumar, H.; du Toit, E.; Kulkarni, A.; Aakko, J.; Linderborg, K.M.; Zhang, Y.; Nicol, M.P.; Isolauri, E.; Yang, B.; Collado, M.C.; et al. Distinct patterns in human milk microbiota and fatty acid profiles across specific geographic locations. Front. Microbiol. 2016, 7, 1619. [Google Scholar] [CrossRef] [PubMed]
- Hermansson, H.; Kumar, H.; Collado, M.C.; Salminen, S.; Isolauri, E.; Rautava, S. Breast milk microbiota is shaped by mode of delivery and intrapartum antibiotic exposure. Front. Nutr. 2019, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Pannaraj, P.S.; Li, F.; Cerini, C.; Bender, J.M.; Yang, S.; Rollie, A.; Adisetiyo, H.; Zabih, S.; Lincez, P.J.; Bittinger, K.; et al. Association between breast milk bacterial communities and establishment and development of the infant gut microbiome. JAMA Pediatr. 2017, 171, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Lackey, K.A.; Williams, J.E.; Meehan, C.L.; Zachek, J.A.; Benda, E.D.; Price, W.J.; Foster, J.A.; Sellen, D.W.; Kamau-Mbuthia, E.W.; Kamundia, E.W.; et al. What’s normal? Microbiomes in human milk and infant feces are related to each other but vary geographically: The inspire study. Front. Nutr. 2019, 6, 45. [Google Scholar] [CrossRef]
- Van den Elsen, L.W.J.; Garssen, J.; Burcelin, R.; Verhasselt, V. Shaping the gut microbiota by breastfeeding: The gateway to allergy prevention? Front. Pediatr. 2019, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Turfkruyer, M.; Verhasselt, V. Breast milk and its impact on maturation of the neonatal immune system. Curr. Opin. Infect. Dis. 2015, 28, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Frost, B.L.; Jilling, T.; Lapin, B.; Maheshwari, A.; Caplan, M.S. Maternal breast milk transforming growth factor-beta and feeding intolerance in preterm infants. Pediatr. Res. 2014, 76, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, J.T.; Lebrilla, C.B.; Mills, D.A.; German, J.B.; Freeman, S.L. Breast milk oligosaccharides: Structure-function relationships in the neonate. Annu. Rev. Nutr. 2014, 34, 143–169. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.M.; Nazar, A.M. Breastfeeding, the immune response, and long-term health. J. Am. Osteopath. Assoc. 2006, 106, 203–207. [Google Scholar] [PubMed]
- Rogier, E.W.; Frantz, A.L.; Bruno, M.E.C.; Wedlund, L.; Cohen, D.A.; Stromberg, A.J.; Kaetzel, C.S. Secretory antibodies in breast milk promote long-term intestinal homeostasis by regulating the gut microbiota and host gene expression. Proc. Natl. Acad. Sci. USA 2014, 111, 3074–3079. [Google Scholar] [CrossRef] [PubMed]
- Statistics New Zealand. 2013 Census Quickstats about Culture and Identity; Statistics New Zealand: Wellington, New Zealand, 2014.
- Butts, C.A.; Hedderley, D.I.; Herath, T.D.; Paturi, G.; Glyn-Jones, S.; Wiens, F.; Stahl, B.; Gopal, P. Human milk composition and dietary intakes of breastfeeding women of different ethnicity from the manawatu-wanganui region of new zealand. Nutrients 2018, 10, 1231. [Google Scholar] [CrossRef]
- Herlemann, D.P.R.; Labrenz, M.; Jürgens, K.; Bertilsson, S.; Waniek, J.J.; Andersson, A.F. Transitions in bacterial communities along the 2000 km salinity gradient of the baltic sea. ISME J. 2011, 5, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I. Qiime allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. Pandaseq: Paired-end assembler for illumina sequences. BMC Bioinform. 2012, 13, 31. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than blast. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. Uchime improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Bittinger, K.; Bushman, F.D.; DeSantis, T.Z.; Andersen, G.L.; Knight, R. Pynast: A flexible tool for aligning sequences to a template alignment. Bioinformatics 2010, 26, 266–267. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16s rrna gene database and workbench compatible with arb. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.; Knight, R. Unifrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Baeza, Y.; Pirrung, M.; Gonzalez, A.; Knight, R. Emperor: A tool for visualizing high-throughput microbial community data. GigaScience 2013, 2, 16. [Google Scholar] [CrossRef]
- Ministry of Health. Food and Nutrition Guidelines for Healthy Pregnant and Breastfeeding Women: A Background Paper; Ministry of Health: Wellington, New Zealand, 2006.
- Ruiz, L.; García-Carral, C.; Rodriguez, J.M. Unfolding the human milk microbiome landscape in the omics era. Front. Microbiol. 2019, 10, 1378. [Google Scholar] [CrossRef]
- Aaltonen, J.; Ojala, T.; Laitinen, K.; Poussa, T.; Ozanne, S.; Isolauri, E. Impact of maternal diet during pregnancy and breastfeeding on infant metabolic programming: A prospective randomized controlled study. Eur. J. Clin. Nutr. 2011, 65, 10–19. [Google Scholar] [CrossRef]
- Rodriguez, J.M.; Murphy, K.; Stanton, C.; Ross, R.P.; Kober, O.I.; Juge, N.; Avershina, E.; Rudi, K.; Narbad, A.; Jenmalm, M.C.; et al. The composition of the gut microbiota throughout life, with an emphasis on early life. Microb. Ecol. Health Dis. 2015, 26, 26050. [Google Scholar] [CrossRef]
- Jeurink, P.V.; Knipping, K.; Wiens, F.; Baranska, K.; Stahl, B.; Garssen, J.; Krolak-Olejnik, B. Importance of maternal diet in the training of the infant’s immune system during gestation and lactation. Crit. Rev. Food Sci. Nutr. 2019, 59, 1311–1319. [Google Scholar] [CrossRef]
- Lee, S.A.; Lim, J.Y.; Kim, B.-S.; Cho, S.J.; Kim, N.Y.; Kim, O.B.; Kim, Y. Comparison of the gut microbiota profile in breast-fed and formula-fed Korean infants using pyrosequencing. Nutr. Res. Pract. 2015, 9, 242–248. [Google Scholar] [CrossRef]
- Guaraldi, F.; Salvatori, G. Effect of breast and formula feeding on gut microbiota shaping in newborns. Front. Cell Infect. Microbiol. 2012, 2, 94. [Google Scholar] [CrossRef] [PubMed]
- Dogra, S.; Sakwinska, O.; Soh, S.-E.; Ngom-Bru, C.; Brück, W.M.; Berger, B.; Brüssow, H.; Lee, Y.S.; Yap, F.; Chong, Y.-S.; et al. Dynamics of infant gut microbiota are influenced by delivery mode and gestational duration and are associated with subsequent adiposity. mBio 2015, 6, e02419. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Arango, L.F.; Barrett, H.L.; Wilkinson, S.A.; Callaway, L.K.; McIntyre, H.D.; Morrison, M.; Dekker Nitert, M. Low dietary fiber intake increases collinsella abundance in the gut microbiota of overweight and obese pregnant women. Gut Microbes 2018, 9, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Gallardo-Becerra, L.; Cornejo-Granados, F.; García-López, R.; Valdez-Lara, A.; Bikel, S.; Canizales-Quinteros, S.; López-Contreras, B.E.; Mendoza-Vargas, A.; Nielsen, H.; Ochoa-Leyva, A. Metatranscriptomic analysis to define the secrebiome, and 16s rrna profiling of the gut microbiome in obesity and metabolic syndrome of mexican children. Microb. Cell Factories 2020, 19, 61. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Karmaus, W.; Davis, S.; Gangur, V. Immune markers in breast milk and fetal and maternal body fluids: A systematic review of perinatal concentrations. J. Hum. Lact. 2011, 27, 171–186. [Google Scholar] [CrossRef]
- Dawod, B.; Marshall, J.S. Cytokines and soluble receptors in breast milk as enhancers of oral tolerance development. Front. Immunol. 2019, 10, 16. [Google Scholar] [CrossRef]
- Bottcher, M.F.; Jenmalm, M.C.; Garofalo, R.P.; Bjorksten, B. Cytokines in breast milk from allergic and nonallergic mothers. Pediatr. Res. 2000, 47, 157–162. [Google Scholar] [CrossRef]
- Castellote, C.; Casillas, R.; Ramírez-Santana, C.; Pérez-Cano, F.J.; Castell, M.; Moretones, M.G.; López-Sabater, M.C.; Franch, A. Premature delivery influences the immunological composition of colostrum and transitional and mature human milk. J. Nutr. 2011, 141, 1181–1187. [Google Scholar] [CrossRef]
- Collado, M.C.; Laitinen, K.; Salminen, S.; Isolauri, E. Maternal weight and excessive weight gain during pregnancy modify the immunomodulatory potential of breast milk. Pediatr. Res. 2012, 72, 77–85. [Google Scholar] [CrossRef]
- Ohtsuka, Y.; Sanderson, I.R. Transforming growth factor-β: An important cytokine in the mucosal immune response. Curr. Opin. Gastroenterol. 2000, 16, 541–545. [Google Scholar] [CrossRef]
- Snijders, B.E.P.; Stelma, F.F.; Reijmerink, N.E.; Thijs, C.; Van Der Steege, G.; Damoiseaux, J.G.M.C.; Van Den Brandt, P.A.; Van Ree, R.; Postma, D.S.; Koppelman, G.H. Cd14 polymorphisms in mother and infant, soluble cd14 in breast milk and atopy development in the infant (koala study). Pediatr. Allergy Immunol. 2010, 21, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Amoudruz, P.; Holmlund, U.; Schollin, J.; Sverremark-Ekström, E.; Montgomery, S.M. Maternal country of birth and previous pregnancies are associated with breast milk characteristics. Pediatr. Allergy Immunol. 2009, 20, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Holmlund, U.; Amoudruz, P.; Johansson, M.A.; Haileselassie, Y.; Ongoiba, A.; Kayentao, K.; Traoré, B.; Doumbo, S.; Schollin, J.; Doumbo, O.; et al. Maternal country of origin, breast milk characteristics and potential influences on immunity in offspring. Clin. Exp. Allergy 2010, 162, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. N. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed]
- Ngom, P.T.; Collinson, A.C.; Pido-Lopez, J.; Henson, S.M.; Prentice, A.M.; Aspinall, R. Improved thymic function in exclusively breastfed infants is associated with higher interleukin 7 concentrations in their mothers’ breast milk. Am. J. Clin. Nutr. 2004, 80, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Aspinall, R.; Prentice, A.M.; Ngom, P.T. Interleukin 7 from maternal milk crosses the intestinal barrier and modulates t-cell development in offspring. PLoS ONE 2011, 6, e20812. [Google Scholar] [CrossRef] [PubMed]
| Asian | Māori and Pacific Island | New Zealand European | Benjamini–Hochberg-Adjusted p Value | |
|---|---|---|---|---|
| Actinomyces | 0.48 ± 0.47 | 0.12 ± 0.10 | 0.46 ± 0.25 | 0.969 |
| Agrobacterium | 1.20 ± 0.95 | 0.09 ± 0.07 | 0.85 ± 0.64 | 0.858 |
| Akkermansia | 2.34 ± 1.14 | 3.01 ± 0.68 | 1.41 ± 0.29 | 0.376 |
| Anaerococcus | 0.15 ± 0.15 | 0.11 ± 0.07 | 0.26 ± 0.14 | 0.969 |
| Anaerostipes | 0 | 0.10 ± 0.10 | 0.03 ± 0.03 | 0.376 |
| Atopobium | 0.66 ± 0.65 | 0.54 ± 0.53 | 0.19 ± 0.11 | 0.980 |
| Bacteroidales_S24_7 | 0.02 ± 0.02 | 0.12 ± 0.07 | 0.03 ± 0.01 | 0.773 |
| Bacteroides | 1.63 ± 0.80 | 3.88 ± 1.04 | 2.61 ± 0.50 | 0.748 |
| Barnesiellaceae | 0.04 ± 0.03 | 0.04 ± 0.03 | 0.05 ± 0.03 | 0.969 |
| Bifidobacteriaceae | 0.01 ± 0.01 | 0.25 ± 0.20 | 0.06 ± 0.04 | 0.969 |
| Bifidobacterium | 6.38 ± 2.26 | 6.08 ± 1.10 | 7.87 ± 1.01 | 0.897 |
| Blautia | 2.68 ± 0.90 | 3.97 ± 0.60 | 2.15 ± 0.27 | 0.376 |
| Brevundimonas | 0.01 ± 0.01 | 0.15 ± 0.14 | 0.21 ± 0.20 | 0.748 |
| Caulobacteraceae | 1.12 ± 0.95 | 0.88 ± 0.69 | 1.28 ± 0.51 | 0.531 |
| Christensenella | 0 | 0 | 0.04 ± 0.04 | 0.865 |
| Christensenellaceae | 0.39 ± 0.37 | 0.48 ± 0.15 | 0.21 ± 0.05 | 0.390 |
| Chryseobacterium | 0.06 ± 0.03 | 2.90 ± 2.81 | 2.39 ± 1.22 | 0.969 |
| Clostridiaceae | 0.70 ± 0.46 | 1.24 ± 0.35 | 0.93 ± 0.17 | 0.682 |
| Clostridiaceae_Other | 0.02 ± 0.02 | 0.24 ± 0.12 | 0.15 ± 0.05 | 0.969 |
| Clostridiales | 3.41 ± 1.38 | 4.38 ± 0.74 | 2.53 ± 0.36 | 0.402 |
| Clostridiales_Other | 0.89 ± 0.47 | 0.47 ± 0.11 | 0.48 ± 0.10 | 0.782 |
| Clostridium | 1.12 ± 0.74 | 2.00 ± 1.05 | 0.88 ± 0.25 | 0.460 |
| Collinsella | 0.92 ± 0.35 | 1.09 ± 0.23 | 1.23 ± 0.17 | 0.969 |
| Coprococcus | 2.13 ± 0.85 | 1.89 ± 0.38 | 1.73 ± 0.28 | 0.865 |
| Coriobacteriaceae | 0.56 ± 0.33 | 0.57 ± 0.17 | 0.25 ± 0.07 | 0.396 |
| Corynebacterium | 0.89 ± 0.34 | 2.92 ± 2.13 | 4.02 ± 1.20 | 0.969 |
| Dorea | 0.51 ± 0.37 | 1.00 ± 0.36 | 0.66 ± 0.26 | 0.376 |
| Elizabethkingia | 16.2 ± 10.6 | 0.03 ± 0.02 | 0.02 ± 0.01 | 0.403 |
| Enterobacteriaceae | 0.15 ± 0.15 | 0.04 ± 0.02 | 0.20 ± 0.13 | 0.969 |
| Epulopiscium | 0 | 0 | 0.02 ± 0.02 | 0.969 |
| Faecalibacterium | 0.52 ± 0.27 | 1.70 ± 0.28 | 0.89 ± 0.15 | 0.376 |
| Finegoldia | 0.03 ± 0.03 | 0.03 ± 0.03 | 0.16 ± 0.08 | 0.460 |
| Fusobacterium | 0.75 ± 0.45 | 0.61 ± 0.46 | 0.49 ± 0.19 | 0.624 |
| Lachnospira | 1.27 ± 0.63 | 1.45 ± 0.62 | 0.78 ± 0.17 | 0.518 |
| Lachnospiraceae | 4.71 ± 2.55 | 6.80 ± 0.93 | 5.05 ± 0.82 | 0.461 |
| Lachnospiraceae_Other | 0.14 ± 0.05 | 1.11 ± 0.37 | 0.29 ± 0.07 | 0.177 |
| Leucobacter | 0 | 0 | 0.26 ± 0.23 | 0.773 |
| Micrococcaceae | 0 | 0.01 ± 0.00 | 0.04 ± 0.04 | 0.461 |
| Micrococcus | 0.01 ± 0.01 | 0 | 0.07 ± 0.05 | 0.773 |
| Mogibacteriaceae | 0.16 ± 0.12 | 0 | 0.10 ± 0.07 | 0.714 |
| Odoribacter | 0 | 0 | 0.04 ± 0.04 | 0.461 |
| Oscillospira | 0.63 ± 0.46 | 0.65 ± 0.20 | 0.56 ± 0.14 | 0.599 |
| Parabacteroides | 0.26 ± 0.17 | 0.33 ± 0.12 | 0.29 ± 0.09 | 0.858 |
| Peptostreptococcus | 0.29 ± 0.19 | 0.04 ± 0.04 | 0.39 ± 0.14 | 0.402 |
| Porphyromonas | 0.17 ± 0.17 | 0 | 0.13 ± 0.09 | 0.682 |
| Prevotella | 0.01 ± 0.01 | 0.08 ± 0.03 | 0.27 ± 0.13 | 0.748 |
| Propionibacterium | 6.07 ± 5.15 | 0.35 ± 0.10 | 0.27 ± 0.09 | 0.390 |
| Pseudomonas | 0.28 ± 0.16 | 4.31 ± 3.04 | 0.96 ± 0.56 | 0.854 |
| Rikenellaceae | 1.92 ± 1.52 | 6.98 ± 1.92 | 2.01 ± 0.60 | 0.710 |
| Roseburia | 0.52 ± 0.28 | 0.56 ± 0.22 | 0.46 ± 0.12 | 0.969 |
| Rothia | 3.10 ± 1.78 | 0.83 ± 0.40 | 1.94 ± 0.76 | 0.969 |
| Ruminococcaceae | 10.7 ± 4.13 | 17.2 ± 2.05 | 10.2 ± 1.51 | 0.390 |
| [Ruminococcus] | 1.17 ± 0.80 | 1.32 ± 0.37 | 0.83 ± 0.30 | 0.376 |
| Ruminococcus | 2.05 ± 0.84 | 3.03 ± 0.68 | 1.49 ± 0.26 | 0.376 |
| Sphingobium | 0.11 ± 0.10 | 0.10 ± 0.05 | 0.83 ± 0.43 | 0.390 |
| Sphingomonas | 0.21 ± 0.09 | 0.41 ± 0.26 | 0.52 ± 0.15 | 0.969 |
| Streptophyta | 0.22 ± 0.21 | 0.10 ± 0.06 | 0.24 ± 0.09 | 0.748 |
| Unassigned | 1.51 ± 0.30 | 3.62 ± 1.24 | 2.20 ± 0.47 | 0.682 |
| Veillonella | 0.07 ± 0.05 | 0.02 ± 0.01 | 0.27 ± 0.08 |
| Asian | Māori and Pacific Island | New Zealand European | Benjamini–Hochberg-Adjusted p Value | |
|---|---|---|---|---|
| Actinomyces | 0 | 0 | 0.01 ± 0.01 | 0.471 |
| Agrobacterium | 0 | 0 | 0.04 ± 0.04 | 0.499 |
| Akkermansia | 1.42 ± 0.84 | 2.42 ± 0.78 | 3.17 ± 1.02 | 0.453 |
| Anaerococcus | 0 | 0.01 ± 0.01 | 0.03 ± 0.03 | 0.257 |
| Anaerostipes | 0.07 ± 0.05 | 0.08 ± 0.03 | 0.08 ± 0.04 | 0.453 |
| Bacteroidales | 0 | 0.33 ± 0.33 | 0.10 ± 0.10 | 0.894 |
| Bacteroidales_S24_7 | 0 | 0.09 ± 0.08 | 0.83 ± 0.43 | 0.629 |
| Bacteroides | 20.5 ± 7.53 | 16.6 ± 4.30 | 17.4 ± 2.25 | 0.894 |
| Barnesiellaceae | 0.38 ± 0.19 | 1.19 ± 0.51 | 0.96 ± 0.19 | 0.931 |
| Bifidobacterium | 2.22 ± 0.88 | 3.65 ± 1.42 | 1.57 ± 0.31 | 0.240 |
| Blautia | 1.94 ± 0.46 | 3.25 ± 1.11 | 2.73 ± 0.33 | 0.748 |
| Butyricimonas | 0.10 ± 0.04 | 0.08 ± 0.04 | 0.13 ± 0.05 | 0.629 |
| Caulobacteraceae | 0.07 ± 0.07 | 0 | 0.31 ± 0.09 | 0.240 |
| Christensenellaceae | 0.50 ± 0.47 | 0.48 ± 0.16 | 0.55 ± 0.09 | 0.240 |
| Clostridiaceae | 0.16 ± 0.08 | 0.60 ± 0.21 | 0.34 ± 0.07 | 0.240 |
| Clostridiaceae_Other | 0.03 ± 0.02 | 0.11 ± 0.07 | 0.03 ± 0.01 | 0.894 |
| Clostridiales | 4.90 ± 1.61 | 3.25 ± 0.89 | 5.49 ± 0.54 | 0.240 |
| Clostridiales_Other | 0.85 ± 0.25 | 0.49 ± 0.10 | 0.98 ± 0.16 | 0.453 |
| Clostridium | 0.40 ± 0.13 | 0.22 ± 0.04 | 0.34 ± 0.03 | 0.407 |
| Collinsella | 0.36 ± 0.09 | 1.44 ± 0.53 | 0.64 ± 0.10 | 0.352 |
| Coprococcus | 3.10 ± 0.45 | 2.63 ± 0.60 | 2.81 ± 0.42 | 0.580 |
| Coriobacteriaceae | 0.11 ± 0.06 | 0.32 ± 0.07 | 0.37 ± 0.08 | 0.294 |
| Dorea | 0.45 ± 0.14 | 1.35 ± 0.45 | 0.58 ± 0.10 | 0.240 |
| Eggerthella | 0.03 ± 0.01 | 0.04 ± 0.02 | 0.06 ± 0.02 | 0.986 |
| Enterobacteriaceae | 0 | 0.01 ± 0.01 | 0.10 ± 0.04 | 0.453 |
| Epulopiscium | 0 | 0 | 0.11 ± 0.11 | 0.580 |
| Faecalibacterium | 2.39 ± 0.87 | 1.69 ± 0.33 | 1.44 ± 0.28 | 0.352 |
| Finegoldia | 0.21 ± 0.21 | 0.02 ± 0.02 | 0.20 ± 0.04 | 0.453 |
| Fusobacterium | 0 | 2.71 ± 2.69 | 0.02± 0.01 | 0.629 |
| Lachnospira | 2.62 ± 0.96 | 2.01 ± 1.00 | 1.45 ± 0.38 | 0.320 |
| Lachnospiraceae | 7.24 ± 1.61 | 6.20 ± 1.15 | 6.05 ± 0.88 | 0.664 |
| Lachnospiraceae_Other | 0.49 ± 0.14 | 0.75 ± 0.13 | 0.42 ± 0.07 | 0.240 |
| Mogibacteriaceae | 0.04 ± 0.04 | 0.10 ± 0.05 | 0.05 ± 0.01 | 0.748 |
| Odoribacter | 0.10 ± 0.06 | 0.19 ± 0.09 | 0.19 ± 0.04 | 0.663 |
| Oscillospira | 0.47 ± 0.17 | 0.84 ± 0.17 | 0.88 ± 0.20 | 0.664 |
| Parabacteroides | 1.03 ± 0.45 | 1.71 ± 0.53 | 1.67 ± 0.39 | 0.926 |
| Paraprevotella | 0 | 0.49 ± 0.29 | 0.46 ± 0.37 | 0.240 |
| Porphyromonas | 0.02 ± 0.01 | 0 | 0.03 ± 0.02 | 0.472 |
| Prevotella | 5.58 ± 5.57 | 2.38 ± 2.16 | 0.826 ± 0.26 | 0.748 |
| Rikenellaceae | 1.05 ± 0.34 | 2.64 ± 0.86 | 3.51 ± 0.58 | 0.626 |
| Rikenellaceae_Other | 0 | 0 | 0.02 ± 0.01 | 0.240 |
| Roseburia | 2.13 ± 1.75 | 0.75 ± 0.40 | 0.40 ± 0.12 | 0.664 |
| Ruminococcaceae | 26.1 ± 6.54 | 26.4 ± 3.88 | 19.1 ± 2.70 | 0.406 |
| [Ruminococcus] | 0.41 ± 0.14 | 2.38 ± 1.52 | 0.86 ± 0.19 | 0.294 |
| Ruminococcus | 4.05 ± 1.71 | 3.93 ± 1.09 | 3.26 ± 0.61 | 0.627 |
| Sphingomonas | 0.01 ± 0.01 | 0 | 0.03 ± 0.01 | 0.240 |
| Sutterella | 0.12 ± 0.06 | 0.02 ± 0.01 | 0.09 ± 0.05 | 0.453 |
| Unassigned | 2.06 ± 0.21 | 2.29 ± 0.27 | 2.11 ± 0.17 | 0.580 |
| Veillonella | 0.95 ± 0.95 | 0.09 ± 0.09 | 2.25 ± 0.63 | 0.254 |
| Asian | Māori and Pacific Island | New Zealand European | Benjamini–Hochberg-Adjusted p Value | |
|---|---|---|---|---|
| Actinomyces | 0.01 ± 0.006 | 0.02 ± 0.02 | 1.31 ± 0.86 | 0.706 |
| Agrobacterium | 0 | 0.005 ± 0.005 | 0.07 ± 0.05 | 0.706 |
| Akkermansia | 0.39 ± 0.37 | 0.07 ± 0.05 | 0.01 ± 0.01 | 0.706 |
| Anaerotruncus | 0 | 0 | 0.02 ± 0.02 | 0.706 |
| Bacteroides | 20.1 ± 10.8 | 24.12± 7.52 | 16.01 ± 3.57 | 0.706 |
| Bifidobacterium | 60.1 ± 12.4 | 50.4 ± 8.270 | 41.2 ± 4.94 | 0.706 |
| Blautia | 0.23 ± 0.19 | 0.12 ± 0.06 | 0.48 ± 0.24 | 0.965 |
| Caulobacteraceae | 0 | 0 | 1.74 ± 1.16 | 0.706 |
| Clostridiaceae | 6.13 ± 6.00 | 2.80 ± 2.69 | 3.14 ± 1.84 | 0.706 |
| Clostridiaceae_Other | 0 | 0.01 ± 0.01 | 0.02 ± 0.02 | 0.936 |
| Clostridiales | 0.43 ± 0.37 | 0.12 ± 0.08 | 0.45 ± 0.31 | 0.706 |
| Clostridiales_Other | 0.02 ± 0.01 | 0.02 ± 0.01 | 0.08 ± 0.04 | 0.706 |
| Clostridium | 0.18 ± 0.09 | 6.21 ± 5.78 | 14.3 ± 4.16 | 0.706 |
| Collinsella | 3.70 ± 3.42 | 2.47 ± 1.05 | 2.49 ± 1.08 | 0.706 |
| Coprococcus | 0.32 ± 0.28 | 0.08 ± 0.05 | 0.23 ± 0.10 | 0.706 |
| Coriobacteriaceae | 0.05 ± 0.03 | 0.23 ± 0.16 | 0.43 ± 0.36 | 0.706 |
| Dorea | 0.08 ± 0.06 | 0.07 ± 0.07 | 0.63 ± 0.49 | 0.719 |
| Eggerthella | 0.05 ± 0.04 | 0.12 ± 0.08 | 0.13 ± 0.06 | 0.706 |
| Enterobacteriaceae | 0.36 ± 0.30 | 0.35 ± 0.13 | 0.33 ± 0.07 | 0.706 |
| Faecalibacterium | 0.22 ± 0.19 | 0.09 ± 0.07 | 0.03 ± 0.01 | 0.706 |
| Finegoldia | 0.01 ± 0.01 | 0 | 0.04 ± 0.03 | 0.706 |
| Fusobacterium | 0.03 ± 0.03 | 0.03 ± 0.02 | 0.34 ± 0.33 | 0.706 |
| Lachnospiraceae | 1.11 ± 0.82 | 2.32 ± 1.65 | 0.80 ± 0.31 | 0.706 |
| Lachnospiraceae_Other | 0.02 ± 0.02 | 0.06 ± 0.02 | 0.68 ± 0.63 | 0.724 |
| Megasphaera | 0 | 0.36 ± 0.26 | 0.04 ± 0.04 | 0.706 |
| Odoribacter | 0 | 0 | 0.07 ± 0.05 | 0.706 |
| Oscillospira | 0.31 ± 0.27 | 0.01 ± 0.01 | 0.26 ± 0.13 | 0.706 |
| Parabacteroides | 0.22 ± 0.14 | 2.05 ± 1.07 | 2.57 ± 1.37 | 0.936 |
| Peptostreptococcus | 0.03 ± 0.03 | 0.13 ± 0.13 | 0.03 ± 0.03 | 0.936 |
| Porphyromonas | 0 | 0 | 0.06 ± 0.05 | 0.706 |
| Prevotella | 0.01 ± 0.01 | 0.03 ± 0.02 | 0.12 ± 0.08 | 0.936 |
| Pseudoramibacter | 0 | 0.22 ± 0.22 | 0.03 ± 0.03 | 0.833 |
| Rikenellaceae | 0.83 ± 0.83 | 0.01 ± 0.00 | 0.40 ± 0.18 | 0.706 |
| Ruminococcaceae | 0.92 ± 0.66 | 1.04 ± 0.61 | 1.39 ± 0.95 | 0.706 |
| [Ruminococcus] | 0.79 ± 0.73 | 1.85 ± 1.15 | 3.72 ± 1.83 | 0.706 |
| Ruminococcus | 0.51 ± 0.43 | 0.25 ± 0.18 | 0.09 ± 0.02 | 0.706 |
| Sphingomonas | 0.01 ± 0.01 | 0 | 0.27 ± 0.26 | 0.706 |
| Unassigned | 2.25 ± 0.60 | 3.56 ± 0.91 | 2.40 ± 0.26 | 0.706 |
| Veillonella | 0.01 ± 0.01 | 0.03 ± 0.01 | 0.54 ± 0.20 | 0.706 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butts, C.A.; Paturi, G.; Blatchford, P.; Bentley-Hewitt, K.L.; Hedderley, D.I.; Martell, S.; Dinnan, H.; Eady, S.L.; Wallace, A.J.; Glyn-Jones, S.; et al. Microbiota Composition of Breast Milk from Women of Different Ethnicity from the Manawatu—Wanganui Region of New Zealand. Nutrients 2020, 12, 1756. https://doi.org/10.3390/nu12061756
Butts CA, Paturi G, Blatchford P, Bentley-Hewitt KL, Hedderley DI, Martell S, Dinnan H, Eady SL, Wallace AJ, Glyn-Jones S, et al. Microbiota Composition of Breast Milk from Women of Different Ethnicity from the Manawatu—Wanganui Region of New Zealand. Nutrients. 2020; 12(6):1756. https://doi.org/10.3390/nu12061756
Chicago/Turabian StyleButts, Christine A., Gunaranjan Paturi, Paul Blatchford, Kerry L. Bentley-Hewitt, Duncan I. Hedderley, Sheridan Martell, Hannah Dinnan, Sarah L. Eady, Alison J. Wallace, Sarah Glyn-Jones, and et al. 2020. "Microbiota Composition of Breast Milk from Women of Different Ethnicity from the Manawatu—Wanganui Region of New Zealand" Nutrients 12, no. 6: 1756. https://doi.org/10.3390/nu12061756
APA StyleButts, C. A., Paturi, G., Blatchford, P., Bentley-Hewitt, K. L., Hedderley, D. I., Martell, S., Dinnan, H., Eady, S. L., Wallace, A. J., Glyn-Jones, S., Wiens, F., Stahl, B., & Gopal, P. (2020). Microbiota Composition of Breast Milk from Women of Different Ethnicity from the Manawatu—Wanganui Region of New Zealand. Nutrients, 12(6), 1756. https://doi.org/10.3390/nu12061756







