Pitfalls in the Diagnosis of Coeliac Disease and Gluten-Related Disorders
Abstract
1. Introduction
2. Coeliac Disease
2.1. Clinical Issues Leading to Misdiagnoses of CD
2.1.1. Diagnostic Errors Related to Incorrect Timing of a Gluten-Free Diet
2.1.2. Serology-Related Diagnostic Errors
2.2. Endoscopy/Histology-Related Diagnostic Mistakes
2.3. Diagnostic Errors Related to HLA Typing
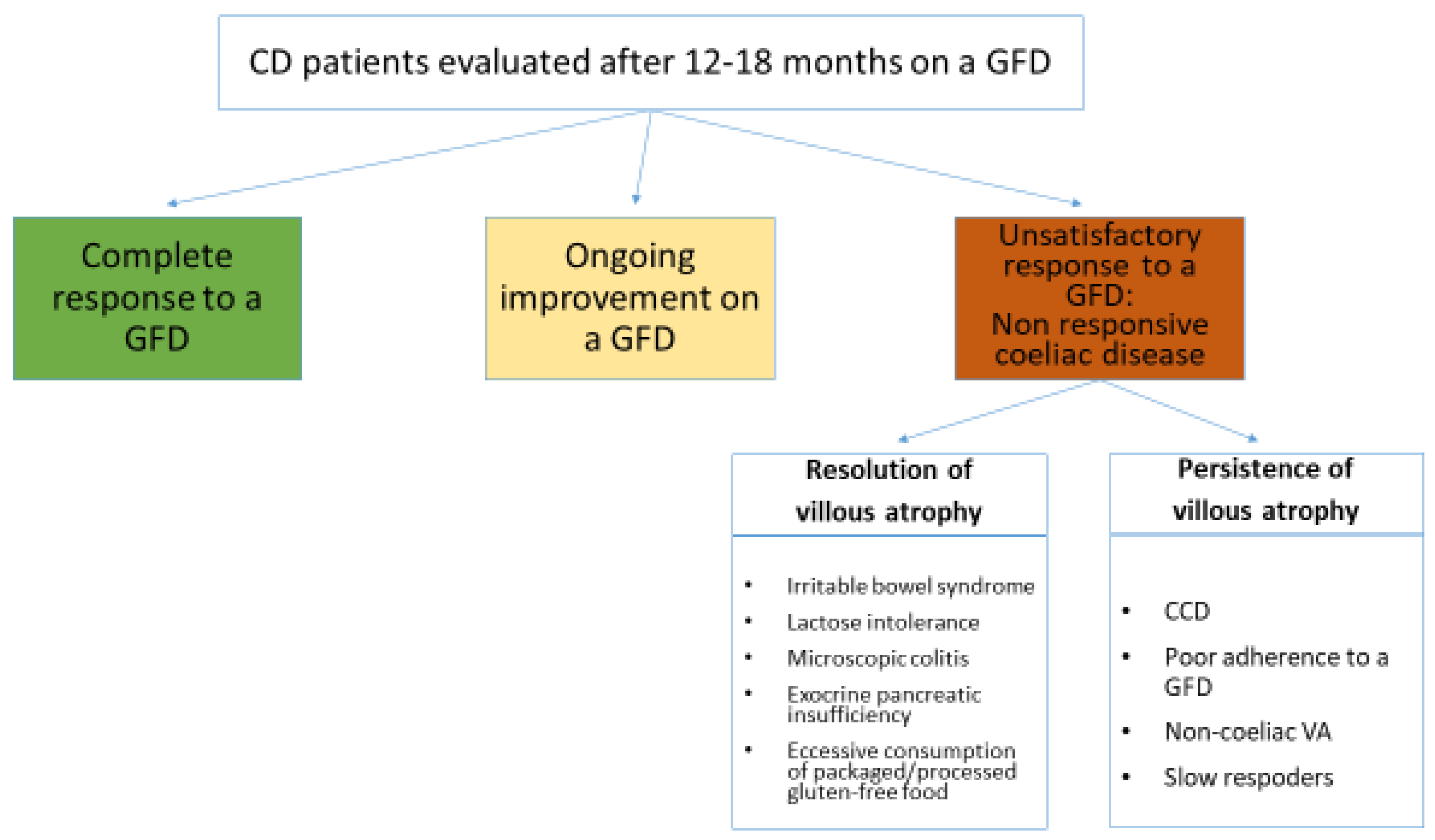
3. Non-Responsive Coeliac Disease
4. Dermatitis Herpetiformis
5. Wheat Allergy
5.1. IgE-Mediated Wheat Allergy
5.2. Non-IgE-Mediated Wheat Allergy
6. Non-Coeliac Gluten Sensitivity
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Sanders, D.S.; Green, P.H.R. Coeliac disease. Lancet 2018, 391, 70–81. [Google Scholar] [CrossRef]
- Jakes, A.D.; Bradley, S.; Donlevy, L. Dermatitis herpetiformis. BMJ 2014, 348, g2557. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M.; Sanders, D.D.; Aeschlimann, D.P. Gluten-related disorders: Gluten ataxia. Dig. Dis. 2015, 33, 264–268. [Google Scholar] [CrossRef]
- Cianferoni, A. JAA-81550-Wheat allergy: Diagnosis and management. J. Asthma Allergy 2016, 9, 13–25. [Google Scholar] [CrossRef]
- Catassi, C.; Bai, J.C.; Bonaz, B.; Bouma, G.; Calabrò, A.; Carroccio, A.; Castillejo, G.; Ciacci, C.; Cristofori, F.; Dolinsek, J.; et al. Non-celiac gluten sensitivity: The new frontier of gluten related disorders. Nutrients 2013, 5, 3839–3853. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.; Sharrack, B.; Sanders, D.; Lobo, A.; Williamson, C.; Woodroofe, N.; Wood, N.; Davies-Jones, A. Gluten ataxia in perspective: Epidemiology, genetic susceptibility and clinical characteristics. Brain 2003, 126, 685–691. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Rao, D.G.; Gr, R.A.; Aeschlimann, D.P.; Sarrigiannis, P.G.; Hoggard, N.; Aeschlimann, P.; Mooney, P.D.; Sanders, D.S. Neurological Dysfunction in Coeliac Disease and Non-Coeliac Gluten Sensitivity. AM. J. Gastroenterol. 2016, 111, 561. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small Amounts of Gluten in Subjects With Suspected Nonceliac Gluten Sensitivity: A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1604–1612. [Google Scholar] [CrossRef]
- Elli, L.; Tomba, C.; Branchi, F.; Roncoroni, L.; Lombardo, V.; Bardella, M.T.; Ferretti, F.; Conte, D.; Valiante, F.; Fini, L.; et al. Evidence for the presence of non-celiac gluten sensitivity in patients with functional gastrointestinal symptoms: Results from a multicenter randomized double-blind placebo-controlled gluten challenge. Nutrients 2016, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Rej, A.; Sanders, D.S. Gluten-free diet and its ‘cousins’ in irritable bowel syndrome. Nutrients 2018, 10, 1727. [Google Scholar] [CrossRef] [PubMed]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients With Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539.e2. [Google Scholar] [CrossRef] [PubMed]
- Bucci, C.; Zingone, F.; Russo, I.; Morra, I.; Tortora, R.; Pogna, N.; Scalia, G.; Lovino, P.; Ciacci, C. Gliadin does not induce mucosal inflammation or basophil activation in patients with nonceliac gluten sensitivity. Clin. Gastroenterol. Hepatol. 2013, 11, 1294–1299. [Google Scholar] [CrossRef] [PubMed]
- Junker, Y.; Zeissig, S.; Kim, S.-J.; Barisani, D.; Wieser, H.; Leffler, D.A.; Zevallos, V.; Libermann, T.A.; Dillon, S.; Freitag, T.L.; et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J. Exp. Med. 2012, 209, 2395–2408. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Shepherd, S.J. Food choice as a key management strategy for functional gastrointestinal symptoms. Am. J. Gastroenterol. 2012, 107, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Biesiekierski, J.R.; Newnham, E.D.; Irving, P.M.; Barrett, J.S.; Haines, M.; Doecke, J.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Gluten Causes gastrointestinal symptoms in subjects without celiac disease: A double-blind randomized placebo-controlled trial. Am. J. Gastroenterol. 2011, 106, 508–514. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Peters, S.L.; Newnham, E.D.; Rosella, O.; Muir, J.G.; Gibson, P.R. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013, 145, 320–328. [Google Scholar] [CrossRef]
- Carroccio, A.; Mansueto, P.; Iacono, G.; Soresi, M.; D’Alcamo, A.; Cavataio, F.; Brusca, I.; Florena, A.M.; Ambrosiano, G.; Seidita, A.; et al. Non-celiac wheat sensitivity diagnosed by double-blind placebo-controlled challenge: Exploring a new clinical entity. Am. J. Gastroenterol. 2012, 107, 1898–1906. [Google Scholar] [CrossRef]
- Long, K.H.; Rubio-Tapia, A.; Wagie, A.E.; Melton, L.J.; Lahr, B.D.; Van Dyke, C.T.; Murray, J.A. The economics of coeliac disease: A population-based study. Aliment. Pharmacol. Ther. 2010, 32, 261–269. [Google Scholar] [CrossRef]
- Fuchs, V.; Kurppa, K.; Huhtala, H.; Mäki, M.; Kekkonen, L.; Kaukinen, K. Delayed celiac disease diagnosis predisposes to reduced quality of life and incremental use of health care services and medicines: A prospective nationwide study. United Eur. Gastroenterol. J. 2018, 6, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Corrao, G.; Corazza, G.R.; Bagnardi, V.; Brusco, G.; Ciacci, C.; Cottone, M.; Sategna Guidetti, C.; Usai, P.; Cesari, P.; Pelli, M.A.; et al. Mortality in patients with coeliac disease and their relatives: A cohort study. Lancet 2001, 358, 356–361. [Google Scholar] [CrossRef]
- Holmes, G.K.T.; Prior, P.; Lane, M.R.; Pope, D.; Allan, R.N. Malignancy in coeliac disease—Effect of a gluten free diet. Gut 1989, 30, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Green, P.H.; Fleischauer, A.T.; Bhagat, G.; Goyal, R.; Jabri, B.; Neugut, A.I. Risk of malignancy in patients with celiac disease. Am. J. Med. 2003, 115, 191–195. [Google Scholar] [CrossRef]
- Biagi, F.; Schiepatti, A.; Maiorano, G.; Fraternale, G.; Agazzi, S.; Zingone, F.; Ciacci, C.; Volta, U.; Caio, G.; Tortora, R.; et al. Risk of complications in coeliac patients depends on age at diagnosis and type of clinical presentation. Dig. Liver Dis. 2018, 50, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Biagi, F.; Marchese, A.; Ferretti, F.; Ciccocioppo, R.; Schiepatti, A.; Volta, U.; Caio, G.; Ciacci, C.; Zingone, F.; D’Odorico, A.; et al. A multicentre case control study on complicated coeliac disease: Two different patterns of natural history, two different prognoses. BMC Gastroenterol. 2014, 14, 139. [Google Scholar] [CrossRef]
- Biagi, F.; Schiepatti, A.; Malamut, G.; Marchese, A.; Cellier, C.; Bakker, S.F.; Mulder, C.J.J.; Volta, U.; Zingone, F.; Ciacci, C.; et al. PROgnosticating COeliac patieNts SUrvivaL: The PROCONSUL score. PLoS ONE 2014, 9, e84163. [Google Scholar] [CrossRef]
- Tanpowpong, P.; Ingham, T.R.; Lampshire, P.K.; Kirchberg, F.F.; Epton, M.J.; Crane, J.; Camargo, C.A.; The New Zealand Asthma and Allergy Cohort Study Group. Coeliac disease and gluten avoidance in New Zealand children. Arch. Dis. Child. 2012, 97, 12–16. [Google Scholar] [CrossRef]
- Aziz, I.; Lewis, N.R.; Hadjivassiliou, M.; Winfield, S.N.; Rugg, N.; Kelsall, A.; Newrick, L.; Sanders, D.S. A UK study assessing the population prevalence of self-reported gluten sensitivity and referral characteristics to secondary care. Eur. J. Gastroenterol. Hepatol. 2014, 26, 33–39. [Google Scholar] [CrossRef]
- Jones, A.L. The Gluten-Free Diet: Fad or Necessity? Diabetes Spectr. 2017, 30, 118–123. [Google Scholar] [CrossRef]
- Aziz, I.; Sanders, D.S. Patients who avoid wheat and gluten: Is that health or lifestyle? Dig. Dis. Sci. 2014, 59, 1080–1082. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Croall, I.D.; Trott, N.; Rej, A.; Aziz, I.; O’Brien, D.J.; George, H.A.; Hossain, M.Y.; Marks, L.J.S.; Richardson, J.I.; Rigby, R.; et al. A population survey of dietary attitudes towards Gluten. Nutrients 2019, 11, 1276. [Google Scholar] [CrossRef] [PubMed]
- Croall, I.D.; Aziz, I.; Trott, N.; Tosi, P.; Hoggard, N.; Sanders, D.S. Gluten Does Not Induce Gastrointestinal Symptoms in Healthy Volunteers: A Double-Blind Randomized Placebo Trial. Gastroenterology 2019, 157, 881–883. [Google Scholar] [CrossRef] [PubMed]
- Graber, M.L.; Franklin, N.; Gordon, R. Diagnostic error in internal medicine. Arch. Intern. Med. 2005, 165, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Giardina, T.D.; Meyer, A.N.D.; Forjuoh, S.N.; Reis, M.D.; Thomas, E.J. Types and origins of diagnostic errors in primary care settings. JAMA Intern. Med. 2013, 173, 418–425. [Google Scholar] [CrossRef]
- Megiorni, F.; Pizzuti, A. HLA-DQA1 and HLA-DQB1 in Celiac disease predisposition: Practical implications of the HLA molecular typing. J. Biomed. Sci. 2012, 19, 88. [Google Scholar] [CrossRef]
- Singh, P.; Arora, S.; Singh, A.; Strand, T.A.; Makharia, G.K. Prevalence of celiac disease in Asia: A systematic review and meta-analysis. J. Gastroenterol. Hepatol. 2016, 31, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Kryszak, D.; Bhatti, B.; Sturgeon, C.; Helzlsouer, K.; Clipp, S.L.; Gelfond, D.; Puppa, E.; Sferruzza, A.; Fasano, A. Natural history of celiac disease autoimmunity in a USA cohort followed since 1974. Ann. Med. 2010, 42, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Lohi, S.; Mustalahti, K.; Kaukinen, K.; Laurila, K.; Collin, P.; Rissanen, H.; Lohi, O.; Bravi, E.; Gasparin, M.; Reunanen, A.; et al. Increasing prevalence of coeliac disease over time. Aliment. Pharmacol. Ther. 2007, 26, 1217–1225. [Google Scholar] [CrossRef]
- Sanders, D.S.; Hurlstone, D.P.; Stokes, R.O.; Rashid, F.; Milford-Ward, A.; Hadjivassiliou, M.; Lobo, A.J. Changing face of adult coeliac disease: Experience of a single university hospital in South Yorkshire. Postgrad. Med. J. 2002, 78, 31–33. [Google Scholar] [CrossRef]
- Fasano, A.; Berti, I.; Gerarduzzi, T.; Not, T.; Colletti, R.B.; Drago, S.; Elitsur, Y.; Green, P.H.R.; Guandalini, S.; Hill, I.D.; et al. Prevalence of Celiac disease in at-risk and not-at-risk groups in the United States: A large multicenter study. Arch. Intern. Med. 2003, 163, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Kyle, R.A.; Kaplan, E.L.; Johnson, D.R.; Page, W.; Erdtmann, F.; Brantner, T.L.; Kim, W.R.; Phelps, T.K.; Lahr, B.D.; et al. Increased Prevalence and Mortality in Undiagnosed Celiac Disease. Gastroenterology 2009, 137, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Biagi, F.; Corazza, G.R. Mortality in celiac disease. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Bai, J.C.; Biagi, F.; Card, T.R.; Ciacci, C.; Ciclitira, P.J.; Green, P.H.R.; Hadjivassiliou, M.; Holdoway, A.; Van Heel, D.A.; et al. Diagnosis and management of adult coeliac disease: Guidelines from the British society of gastroenterology. Gut 2014, 63, 1210–1228. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. ACG clinical guidelines: Diagnosis and management of celiac disease. Am. J. Gastroenterol. 2013, 108, 656–676. [Google Scholar] [CrossRef]
- Downey, L.; Houten, R.; Murch, S.; Longson, D.; Abuzakouk, M.; Briggs, S.; Burden, S.; Dadswell, M.; Ferry, B.; Forrest, M.; et al. Recognition, assessment, and management of coeliac disease: Summary of updated NICE guidance. BMJ 2015, 351, h4513. [Google Scholar] [CrossRef]
- Al-Toma, A.; Volta, U.; Auricchio, R.; Castillejo, G.; Sanders, D.S.; Cellier, C.; Mulder, C.J.; Lundin, K.E.A. European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United Eur. Gastroenterol. J. 2019, 7, 583–613. [Google Scholar] [CrossRef]
- Biagi, F.; Bianchi, P.I.; Campanella, J.; Zanellati, G.; Corazza, G.R. The impact of misdiagnosing celiac disease at a referral centre. Can. J. Gastroenterol. Hepatol. 2009, 23, 543–545. [Google Scholar] [CrossRef]
- Ianiro, G.; Bibbò, S.; Bruno, G.; Ricci, R.; Arena, V.; Gasbarrini, A.; Cammarota, G. Prior misdiagnosis of celiac disease is common among patients referred to a tertiary care center: A prospective cohort study. Clin. Transl. Gastroenterol. 2016, 7, e139. [Google Scholar] [CrossRef]
- Sánchez, M.I.P.; Smecuol, E.; Vázquez, H.; Mazure, R.; Mauriño, E.; Bai, J.C. Very high rate of misdiagnosis of celiac disease in clinical practice. Acta Gastroenterol. Latinoam. 2009, 39, 250–253. [Google Scholar]
- Corazza, G.R.; Brusco, G.; Andreani, M.L.; Biagi, F.; Di Stefano, M.; Gasbarrini, G. Previous misdiagnosis and diagnostic delay in adult celiac sprue. J. Clin. Gastroenterol. 1996, 22, 324–325. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Caio, G.; De Giorgio, R. Mistakes in coeliac disease diagnosis and how to avoid them. UEG Educ. 2016, 16, 1–3. [Google Scholar]
- Vavricka, S.R.; Stelzer, T.; Lattmann, J.; Stotz, M.; Lehmann, R.; Zeitz, J.; Scharl, M.; Misselwitz, B.; Pohl, D.; Fried, M.; et al. Celiac Disease is Misdiagnosed Based on Serology only in a Substantial Proportion of Patients. J. Clin. Gastroenterol. 2017, 52, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Barratt, S.M.; Leeds, J.S.; Robinson, K.; Lobo, A.J.; McAlindon, M.E.; Sanders, D.S. Prodromal irritable bowel syndrome may be responsible for delays in diagnosis in patients presenting with unrecognized Crohn’s disease and celiac disease, but not ulcerative colitis. Dig. Dis. Sci. 2011, 56, 3270–3275. [Google Scholar] [CrossRef] [PubMed]
- Freeman, H.J. Pearls and pitfalls in the diagnosis of adult celiac disease. Can. J. Gastroenterol. Hepatol. 2008, 22, 273–280. [Google Scholar] [CrossRef]
- Rostami-Nejad, M.; Villanacci, V.; Hogg-Kollars, S.; Volta, U.; Manenti, S.; Reza-Zali, M.; Caio, G.; Giovenali, P.; Barakauskiene, A.; Kazenaite, E.; et al. Endoscopic and histological pitfalls in the diagnosis of celiac disease: A multicentre study assessing the current practice. Rev. Esp. Enferm. Dig. 2013, 105, 326–333. [Google Scholar] [CrossRef]
- Fuchs, V.; Kurppa, K.; Huhtala, H.; Collin, P.; Mäki, M.; Kaukinen, K. Factors associated with long diagnostic delay in celiac disease. Scand. J. Gastroenterol. 2014, 49, 1304–1310. [Google Scholar] [CrossRef]
- Vavricka, S.R.; Vadasz, N.; Stotz, M.; Lehmann, R.; Studerus, D.; Greuter, T.; Frei, P.; Zeitz, J.; Scharl, M.; Misselwitz, B.; et al. Celiac disease diagnosis still significantly delayed—Doctor’s but not patients’ delay responsive for the increased total delay in women. Dig. Liver Dis. 2016, 48, 1148–1154. [Google Scholar] [CrossRef]
- Mansikka, E.; Salmi, T.T.; Kaukinen, K.; Collin, P.; Huhtala, H.; Reunala, T.; Hervonen, K. Diagnostic delay in dermatitis herpetiformis in a high-prevalence area. Acta Derm. Venereol. 2018, 98, 195–199. [Google Scholar] [CrossRef]
- Card, T.R.; Siffledeen, J.; West, J.; Fleming, K.M. An excess of prior irritable bowel syndrome diagnoses or treatments in Celiac disease: Evidence of diagnostic delay. Scand. J. Gastroenterol. 2013, 48, 801–807. [Google Scholar] [CrossRef]
- Bakker, S.F.; Tushuizen, M.E.; Stokvis-Brantsma, W.H.; Aanstoot, H.J.; Winterdijk, P.; Van Setten, P.A.; Von Blomberg, B.M.; Mulder, C.J.; Simsek, S. Frequent delay of coeliac disease diagnosis in symptomatic patients with type 1 diabetes mellitus: Clinical and genetic characteristics. Eur. J. Intern. Med. 2013, 24, 456–460. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Fleming, K.M.; Tata, L.J.; Card, T.R.; Crooks, C.J. Incidence and prevalence of celiac disease and dermatitis herpetiformis in the UK over two decades: Population-based study. Am. J. Gastroenterol. 2014, 109, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. European society for pediatric gastroenterology, hepatology, and nutrition guidelines for the diagnosis of coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 136–160. [Google Scholar] [CrossRef] [PubMed]
- Campanella, J.; Biagi, F.; Bianchi, P.I.; Zanellati, G.; Marchese, A.; Corazza, G.R. Clinical response to gluten withdrawal is not an indicator of coeliac disease. Scand. J. Gastroenterol. 2008, 43, 1311–1314. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.; Schuppan, D.; Pallav, K.; Najarian, R.; Goldsmith, J.D.; Hansen, J.; Kabbani, T.; Dennis, M.; Kelly, C.P. Kinetics of the histological, serological and symptomatic responses to gluten challenge in adults with coeliac disease. Gut 2013, 62, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Lähdeaho, M.L.; Mäki, M.; Laurila, K.; Huhtala, H.; Kaukinen, K. Small-bowel mucosal changes and antibody responses after low- and moderate-dose gluten challenge in celiac disease. BMC Gastroenterol. 2011, 11, 129. [Google Scholar] [CrossRef]
- Wahab, P.; Crusius, J.B.A.; Meijer, J.W.R.; Mulder, C.J.J. Gluten challenge in borderline gluten-sensitive enteropathy. Am. J. Gastroenterol. 2001, 96, 1464–1469. [Google Scholar] [CrossRef]
- Sarna, V.K.; Lundin, K.E.A.; Mørkrid, L.; Qiao, S.W.; Sollid, L.M.; Christophersen, A. HLA-DQ–Gluten Tetramer Blood Test Accurately Identifies Patients With and Without Celiac Disease in Absence of Gluten Consumption. Gastroenterology 2018, 154, 886–896. [Google Scholar] [CrossRef]
- Leffler, D.A.; Schuppan, D. Update on serologic testing in celiac disease. Am. J. Gastroenterol. 2010, 105, 2520–2524. [Google Scholar] [CrossRef] [PubMed]
- Hill, I.D. What are the sensitivity and specificity of serologic tests for celiac disease? Do sensitivity and specificity vary in different populations? Gastroenterology 2005, 128, S25–S32. [Google Scholar] [CrossRef]
- Lewis, N.R.; Scott, B.B. Meta-analysis: Deamidated gliadin peptide antibody and tissue transglutaminase antibody compared as screening tests for coeliac disease. Aliment. Pharmacol. Ther. 2010, 31, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Hoerter, N.A.; Shannahan, S.E.; Suarez, J.; Lewis, S.K.; Green, P.H.R.; Leffler, D.A.; Lebwohl, B. Diagnostic Yield of Isolated Deamidated Gliadin Peptide Antibody Elevation for Celiac Disease. Dig. Dis. Sci. 2017, 62, 1272–1276. [Google Scholar] [CrossRef] [PubMed]
- Beltran, L.; Koenig, M.; Egner, W.; Howard, M.; Butt, A.; Austin, M.R.; Patel, D.; Sanderson, R.R.; Goubet, S.; Saleh, F.; et al. High-titre circulating tissue transglutaminase-2 antibodies predict small bowel villous atrophy, but decision cut-off limits must be locally validated. Clin. Exp. Immunol. 2014, 176, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Abrams, J.A.; Diamond, B.; Rotterdam, H.; Green, P.H.R. Seronegative celiac disease: Increased prevalence with lesser degrees of villous atrophy. Dig. Dis. Sci. 2004, 49, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Dickey, W.; Hughes, D.F.; McMillan, S.A. Disappearance of endomysial antibodies in treated celiac disease does not indicate histological recovery. Am. J. Gastroenterol. 2000, 95, 712–714. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Neovius, M.; Hammarström, L. Association between IgA deficiency & other autoimmune conditions: A population-based matched cohort study. J. Clin. Immunol. 2014, 34, 444–451. [Google Scholar]
- Cataldo, F.; Lio, D.; Marino, V.; Picarelli, A.; Ventura, A.; Corazza, G.R. IgG(1) antiendomysium and IgG antitissue transglutaminase (anti-tTG) antibodies in coeliac patients with selective IgA deficiency. Working Groups on Celiac Disease of SIGEP and Club del Tenue. Gut 2000, 47, 366–369. [Google Scholar] [CrossRef]
- Villalta, D.; Tonutti, E.; Prause, C.; Koletzko, S.; Uhlig, H.H.; Vermeersch, P.; Bossuyt, X.; Stern, M.; Laass, M.W.; Ellis, J.H.; et al. IgG antibodies against deamidated gliadin peptides for diagnosis of celiac disease in patients with IgA deficiency. Clin. Chem. 2010, 56, 464–468. [Google Scholar] [CrossRef]
- Levinson, J.D.; Nastro, L.J. Giardiasis with total villous atrophy. Gastroenterology 1978, 74, 271–275. [Google Scholar] [CrossRef]
- Biagi, F.; Bianchi, P.I.; Zilli, A.; Marchese, A.; Luinetti, O.; Lougaris, V.; Plebani, A.; Villanacci, V.; Corazza, G.R. The significance of duodenal mucosal atrophy in patients with common variable immunodeficiency: A clinical and histopathologic study. Am. J. Clin. Pathol. 2012, 138, 185–189. [Google Scholar] [CrossRef]
- Schiepatti, A.; Biagi, F.; Fraternale, G.; Vattiato, C.; Balduzzi, D.; Agazzi, S.; Alpini, C.; Klersy, C.; Corazza, G.R. Short article: Mortality and differential diagnoses of villous atrophy without coeliac antibodies. Eur. J. Gastroenterol. Hepatol. 2017, 29, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Schiepatti, A.; Sanders, D.S.; Zuffada, M.; Luinetti, O.; Iraqi, A.; Biagi, F. Overview in the clinical management of patients with seronegative villous atrophy. Eur. J. Gastroenterol. Hepatol. 2019, 31, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Henry, D.X.; Campbell, J.A.; Basu, K.; Schiepatti, A.; Sanders, D.S. Seronegative coeliac disease masquerading as irritable bowel syndrome type symptoms. J. Gastrointest. Liver Dis. 2020, 29, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Hopper, A.D.; Cross, S.S.; Hurlstone, D.P.; McAlindon, M.E.; Lobo, A.J.; Hadjivassiliou, M.; Sloan, M.E.; Dixon, S.; Sanders, D.S. Pre-endoscopy serological testing for coeliac disease: Evaluation of a clinical decision tool. BMJ 2007, 334, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Aziz, I.; Peerally, M.F.; Barnes, J.H.; Kandasamy, V.; Whiteley, J.C.; Partridge, D.; Vergani, P.; Cross, S.S.; Green, P.H.; Sanders, D.S. The clinical and phenotypical assessment of seronegative villous atrophy; A prospective UK centre experience evaluating 200 adult cases over a 15-year period (2000–2015). Gut 2017, 66, 1563–1572. [Google Scholar] [CrossRef] [PubMed]
- Schiepatti, A.; Sanders, D.S.; Biagi, F. Seronegative coeliac disease: Clearing the diagnostic dilemma. Curr. Opin. Gastroenterol. 2018, 34, 154–158. [Google Scholar] [CrossRef]
- Volta, U.; Caio, G.; Boschetti, E.; Giancola, F.; Rhoden, K.J.; Ruggeri, E.; Paterini, P.; De Giorgio, R. Seronegative celiac disease: Shedding light on an obscure clinical entity. Dig. Liver Dis. 2016, 48, 1018–1022. [Google Scholar] [CrossRef]
- Salmi, T.T.; Collin, P.; Korponay-Szabó, I.R.; Laurila, K.; Partanen, J.; Huhtala, H.; Király, R.; Lorand, L.; Reunala, T.; Mäki, M.; et al. Endomysial antibody-negative coeliac disease: Clinical characteristics and intestinal autoantibody deposits. Gut 2006, 55, 1746–1753. [Google Scholar] [CrossRef]
- Lebwohl, B.; Bhagat, G.; Markoff, S.; Lewis, S.K.; Smukalla, S.; Neugut, A.I.; Green, P.H.R. Prior endoscopy in patients with newly diagnosed celiac disease: A missed opportunity? Dig. Dis. Sci. 2013, 58, 1293–1298. [Google Scholar] [CrossRef]
- Taavela, J.; Koskinen, O.; Huhtala, H.; Lähdeaho, M.L.; Popp, A.; Laurila, K.; Collin, P.; Kaukinen, K.; Kurppa, K.; Mäki, M. Validation of Morphometric Analyses of Small-Intestinal Biopsy Readouts in Celiac Disease. PLoS ONE 2013, 8, e76163. [Google Scholar] [CrossRef] [PubMed]
- Pais, W.P.; Duerksen, D.R.; Pettigrew, N.M.; Bernstein, C.N. How many duodenal biopsy specimens are required to make a diagnosis of celiac disease? Gastrointest. Endosc. 2008, 67, 1082–1087. [Google Scholar] [CrossRef]
- Brocchi, E.; Corazza, G.R.; Caletti, G.; Treggiari, E.A.; Barbara, L.; Gasbarrini, G. Endoscopic Demonstration of Loss of Duodenal Folds in the Diagnosis of Celiac Disease. N. Engl. J. Med. 1988, 319, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Ravelli, A.M.; Tobanelli, P.; Minelli, L.; Villanacci, V.; Cestari, R. Endoscopic features of celiac disease in children. Gastrointest. Endosc. 2001, 54, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Latorre, M.; Lagana, S.M.; Freedberg, D.E.; Lewis, S.K.; Lebwohl, B.; Bhagat, G.; Green, P.H.R. Endoscopic biopsy technique in the diagnosis of celiac disease: One bite or two? Gastrointest. Endosc. 2015, 81, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Ravelli, A.; Villanacci, V. Tricks of the trade: How to avoid histological Pitfalls in celiac disease. Pathol. Res. Pract. 2012, 208, 197–202. [Google Scholar] [CrossRef]
- Collin, P.; Kaukinen, K.; Vogelsang, H.; Korponay-Szabó, I.; Sommer, R.; Schreier, E.; Volta, U.; Granito, A.; Veronesi, L.; Mascart, F.; et al. Antiendomysial and antihuman recombinant tissue transglutaminase antibodies in the diagnosis of coeliac disease: A biopsy-proven European multicentre study. Eur. J. Gastroenterol. Hepatol. 2005, 17, 85–91. [Google Scholar] [CrossRef]
- Pallav, K.; Leffler, D.A.; Tariq, S.; Kabbani, T.; Hansen, J.; Peer, A.; Bhansali, A.; Najarian, R.; Kelly, C.P. Noncoeliac enteropathy: The differential diagnosis of villous atrophy in contemporary clinical practice. Aliment. Pharmacol. Ther. 2012, 35, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Corazza, G.R.; Villanacci, V.; Zambelli, C.; Milione, M.; Luinetti, O.; Vindigni, C.; Chioda, C.; Albarello, L.; Bartolini, D.; Donato, F. Comparison of the interobserver reproducibility with different histologic criteria used in celiac disease. Clin. Gastroenterol. Hepatol. 2007, 5, 838–843. [Google Scholar] [CrossRef]
- Biagi, F.; Vattiato, C.; Burrone, M.; Schiepatti, A.; Agazzi, S.; Maiorano, G.; Luinetti, O.; Alvisi, C.; Klersy, C.; Corazza, G.R. Is a detailed grading of villous atrophy necessary for the diagnosis of enteropathy? J. Clin. Pathol. 2016, 69, 1051–1054. [Google Scholar] [CrossRef]
- Oberhuber, G.; Granditsch, G.; Vogelsang, H. The histopathology of coeliac disease: Time for a standardized report scheme for pathologists. Eur. J. Gastroenterol. Hepatol. 1999, 11, 1185–1194. [Google Scholar] [CrossRef]
- Aziz, I.; Evans, K.E.; Hopper, A.D.; Smillie, D.M.; Sanders, D.S. A prospective study into the aetiology of lymphocytic duodenosis. Aliment. Pharmacol. Ther. 2010, 32, 1392–1397. [Google Scholar] [CrossRef] [PubMed]
- Biagi, F.; Trotta, L.; Alfano, C.; Balduzzi, D.; Staffieri, V.; Bianchi, P.I.; Marchese, A.; Vattiato, C.; Zilli, A.; Luinetti, O.; et al. Prevalence and natural history of potential celiac disease in adult patients. Scand. J. Gastroenterol. 2013, 48, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Caio, G.; Giancola, F.; Rhoden, K.J.; Ruggeri, E.; Boschetti, E.; Stanghellini, V.; De Giorgio, R. Features and Progression of Potential Celiac Disease in Adults. Clin. Gastroenterol. Hepatol. 2016, 14, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Wolters, V.M.; Wijmenga, C. Genetic Background of Celiac Disease and Its Clinical Implications. Am. J. Gastroenterol. 2008, 103, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Dennis, M.; Hyett, B.; Kelly, E.; Schuppan, D.; Kelly, C.P. Etiologies and predictors of diagnosis in nonresponsive celiac disease. Clin. Gastroenterol. Hepatol. 2007, 5, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Penny, H.A.; Baggus, E.M.R.; Rej, A.; Snowden, J.A.; Sanders, D.S. Non-responsive coeliac disease: A comprehensive review from the NHS england national centre for refractory coeliac disease. Nutrients 2020, 12, 216. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef]
- Rubio-Tapia, A.; Murray, J.A. Classification and management of refractory coeliac disease. Gut 2010, 59, 547–557. [Google Scholar] [CrossRef]
- Malamut, G.; Cellier, C. Editorial: Is refractory celiac disease more severe in old Europe. Am. J. Gastroenterol. 2011, 106, 929–932. [Google Scholar] [CrossRef]
- West, J. Celiac Disease and Its Complications: A Time Traveller’s Perspective. Gastroenterology 2009, 136, 32–34. [Google Scholar] [CrossRef]
- Roshan, B.; Leffler, D.A.; Jamma, S.; Dennis, M.; Sheth, S.; Falchuk, K.; Najarian, R.; Goldsmith, J.; Tariq, S.; Schuppan, D.; et al. The incidence and clinical spectrum of refractory celiac disease in a north american referral center. Am. J. Gastroenterol. 2011, 106, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Biagi, F.; Gobbi, P.; Marchese, A.; Borsotti, E.; Zingone, F.; Ciacci, C.; Volta, U.; Caio, G.; Carroccio, A.; Ambrosiano, G.; et al. Low incidence but poor prognosis of complicated coeliac disease: A retrospective multicentre study. Dig. Liver Dis. 2014, 46, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Ilus, T.; Kaukinen, K.; Virta, L.J.; Huhtala, H.; Mäki, M.; Kurppa, K.; Heikkinen, M.; Heikura, M.; Hirsi, E.; Jantunen, K.; et al. Refractory coeliac disease in a country with a high prevalence of clinically-diagnosed coeliac disease. Aliment. Pharmacol. Ther. 2014, 39, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Schiepatti, A.; Sanders, D.S.; Aziz, I.; De Silvestri, A.; Goodwin, J.; Key, T.; Quaye, L.; Giuffrida, P.; Vanoli, A.; Paulli, M.; et al. Clinical phenotype and mortality in patients with idiopathic small bowel villous atrophy. Eur. J. Gastroenterol. Hepatol. 2020, 1. [Google Scholar] [CrossRef] [PubMed]
- Schiepatti, A.; Biagi, F.; Cumetti, D.; Luinetti, O.; Sonzogni, A.; Mugellini, A.; Corazza, G.R. Olmesartan-associated enteropathy: New insights on the natural history? Report of two cases. Scand. J. Gastroenterol. 2016, 51, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Salmi, T.T.; Hervonen, K.; Kautiainen, H.; Collin, P.; Reunala, T. Prevalence and incidence of dermatitis herpetiformis: A 40-year prospective study from Finland. Br. J. Dermatol. 2011, 165, 354–359. [Google Scholar] [CrossRef]
- Reunala, T.; Salmi, T.T.; Hervonen, K.; Kaukinen, K.; Collin, P. Dermatitis herpetiformis: A common extraintestinal manifestation of coeliac disease. Nutrients 2018, 10, 602. [Google Scholar] [CrossRef]
- Zone, J.J.; Meyer, L.J.; Petersen, M.J. Deposition of granular IgA relative to clinical lesions in dermatitis herpetiformis. Arch. Dermatol. 1996, 132, 912–918. [Google Scholar] [CrossRef]
- SEAH, P.P.; FRY, L. Immunoglobulins in the skin in dermatitis herpetiformis and their relevance in diagnosis. Br. J. Dermatol. 1975, 92, 157–166. [Google Scholar] [CrossRef]
- Viljamaa, M.; Kaukinen, K.; Pukkala, E.; Hervonen, K.; Reunala, T.; Collin, P. Malignancies and mortality in patients with coeliac disease and dermatitis herpetiformis: 30-year population-based study. Dig. Liver Dis. 2006, 38, 374–380. [Google Scholar] [CrossRef]
- Hervonen, K.; Vornanen, M.; Kautiainen, H.; Collin, P.; Reunala, T. Lymphoma in patients with dermatitis herpetiformis and their first-degree relatives. Br. J. Dermatol. 2005, 152, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Longo, G.; Berti, I.; Burks, A.W.; Krauss, B.; Barbi, E. IgE-mediated food allergy in children. Lancet 2013, 382, 1656–1664. [Google Scholar] [CrossRef]
- Inomata, N. Wheat allergy. Curr. Opin. Allergy Clin. Immunol. 2009, 9, 238–243. [Google Scholar] [CrossRef]
- Tatham, A.S.; Shewry, P.R. Allergens to wheat and related cereals. Clin. Exp. Allergy 2008, 38, 1712–1726. [Google Scholar] [PubMed]
- Salcedo, G.; Quirce, S.; Diaz-Perales, A. Wheat allergens associated with Baker’s asthma. J. Investig. Allergol. Clin. Immunol. 2011, 21, 81–92. [Google Scholar] [PubMed]
- Kagalwalla, A.F.; Sentongo, T.A.; Ritz, S.; Hess, T.; Nelson, S.P.; Emerick, K.M.; Melin-Aldana, H.; Li, B.U.K. Effect of Six-Food Elimination Diet on Clinical and Histologic Outcomes in Eosinophilic Esophagitis. Clin. Gastroenterol. Hepatol. 2006, 4, 1097–1102. [Google Scholar] [CrossRef]
- Walker, M.M.; Potter, M.; Talley, N.J. Eosinophilic gastroenteritis and other eosinophilic gut diseases distal to the oesophagus. Lancet Gastroenterol. Hepatol. 2018, 3, 271–280. [Google Scholar] [CrossRef]
- Sicherer, S.H. Food allergy. Lancet 2002, 360, 701–710. [Google Scholar] [CrossRef]
- Loh, W.; Tang, M.L.K. The epidemiology of food allergy in the global context. Int. J. Environ. Res. Public Health 2018, 15, 2043. [Google Scholar] [CrossRef]
- Rona, R.J.; Keil, T.; Summers, C.; Gislason, D.; Zuidmeer, L.; Sodergren, E.; Sigurdardottir, S.T.; Lindner, T.; Goldhahn, K.; Dahlstrom, J.; et al. The prevalence of food allergy: A meta-analysis. J. Allergy Clin. Immunol. 2007, 120, 638–646. [Google Scholar] [CrossRef]
- Keet, C.A.; Matsui, E.C.; Dhillon, G.; Lenehan, P.; Paterakis, M.; Wood, R.A. The natural history of wheat allergy. Ann. Allergy Asthma Immunol. 2009, 102, 410–415. [Google Scholar] [CrossRef]
- Czaja-Bulsa, G.; Bulsa, M. What do we know now about IgE-mediated wheat allergy in children? Nutrients 2017, 9, 35. [Google Scholar] [CrossRef] [PubMed]
- Salvatori, N.; Reccardini, F.; Convento, M.; Purinan, A.; Colle, R.; De Carli, S.; Garzoni, M.; Lafiandra, D.; De Carli, M. Asthma induced by inhalation of flour in adults with food allergy to wheat. Clin. Exp. Allergy 2008, 38, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Eigenmann, P.A.; Sampson, H.A. Interpreting skin prick tests in the evaluation of food allergy in children. Pediatr. Allergy Immunol. 1998, 9, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Mehl, A.; Verstege, A.; Staden, U.; Kulig, M.; Nocon, M.; Beyer, K.; Niggemann, B. Utility of the ratio of food-specific IgE/total IgE in predicting symptomatic food allergy in children. Allergy Eur. J. Allergy Clin. Immunol. 2005, 60, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Palosuo, K.; Varjonen, E.; Klemola, T.; Reunala, T.; Palosuo, K.; Alenius, H.; Kekki, O.M.; Reunala, T.; Kalkkinen, N. Wheat ω-gliadin is a major allergen in children with immediate allergy to ingested wheat. J. Allergy Clin. Immunol. 2001, 108, 634–638. [Google Scholar] [CrossRef]
- Perry, T.T.; Matsui, E.C.; Conover-Walker, M.K.; Wood, R.A. Risk of oral food challenges. J. Allergy Clin. Immunol. 2004, 114, 1164–1168. [Google Scholar] [CrossRef]
- Cianferoni, A.; Khullar, K.; Saltzman, R.; Fiedler, J.; Garrett, J.P.; Naimi, D.R.; Spergel, J.M. Oral food challenge to wheat: A near-fatal anaphylaxis and review of 93 food challenges in children. World Allergy Organ. J. 2013, 6. [Google Scholar] [CrossRef]
- Brockow, K.; Kneissl, D.; Valentini, L.; Zelger, O.; Grosber, M.; Kugler, C.; Werich, M.; Darsow, U.; Matsuo, H.; Morita, E.; et al. Using a gluten oral food challenge protocol to improve diagnosis of wheat-dependent exercise-induced anaphylaxis. J. Allergy Clin. Immunol. 2015, 135, 977–984. [Google Scholar] [CrossRef]
- Wiszniewska, M.; Nowakowska-Świrta, E.; Pałczyński, C.; Walusiak-Skorupa, J. Diagnosing of bakers’ respiratory allergy: Is specific inhalation challenge test essential? Allergy Asthma Proc. 2011, 32, 111–118. [Google Scholar] [CrossRef]
- Hoffmann, H.J.; Santos, A.F.; Mayorga, C.; Nopp, A.; Eberlein, B.; Ferrer, M.; Rouzaire, P.; Ebo, D.G.; Sabato, V.; Sanz, M.L.; et al. The clinical utility of basophil activation testing in diagnosis and monitoring of allergic disease. Allergy Eur. J. Allergy Clin. Immunol. 2015, 70, 1393–1405. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, R.; Nagao, M.; Hiraguchi, Y.; Hosoki, K.; Matsuda, T.; Kouno, K.; Morita, E.; Fujisawa, T. Antigen-induced expression of CD203C on basophils predicts IgE-mediated wheat allergy. Allergol. Int. 2009, 58, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Wang, J.; Leung, N.; Wang, L.X.; Lisann, L.; Sicherer, S.H.; Scurlock, A.M.; Pesek, R.; Perry, T.T.; Jones, S.M.; et al. Correlations between basophil activation, allergen-specific IgE with outcome and severity of oral food challenges. Ann. Allergy Asthma Immunol. 2015, 114, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Shreffler, W.G. Road map for the clinical application of the basophil activation test in food allergy. Clin. Exp. Allergy 2017, 47, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Sander, I.; Raulf-Heimsoth, M.; Düser, M.; Flagge, A.; Czuppon, A.B.; Baur, X. Differentiation between cosensitization and cross-reactivity in wheat flour and grass pollen-sensitized subjects. Int. Arch. Allergy Immunol. 1997, 112, 378–385. [Google Scholar] [CrossRef]
- Sampson, H.A. Utility of food-specific IgE concentrations in predicting symptomatic food allergy. J. Allergy Clin. Immunol. 2001, 107, 891–896. [Google Scholar] [CrossRef]
- Spergel, J.M.; Brown-Whitehorn, T.F.; Cianferoni, A.; Shuker, M.; Wang, M.L.; Verma, R.; Liacouras, C.A. Identification of causative foods in children with eosinophilic esophagitis treated with an elimination diet. J. Allergy Clin. Immunol. 2012, 130, 461–467. [Google Scholar] [CrossRef]
- Spergel, J.M. Eosinophilic esophagitis in adults and children: Evidence for a food allergy component in many patients. Curr. Opin. Allergy Clin. Immunol. 2007, 7, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Gonsalves, N.; Yang, G.Y.; Doerfler, B.; Ritz, S.; Ditto, A.M.; Hirano, I. Elimination diet effectively treats eosinophilic esophagitis in adults; Food reintroduction identifies causative factors. Gastroenterology 2012, 142. [Google Scholar] [CrossRef]
- Molina-Infante, J.; Arias, A.; Barrio, J.; Rodríguez-Sánchez, J.; Sanchez-Cazalilla, M.; Lucendo, A.J. Four-food group elimination diet for adult eosinophilic esophagitis: A prospective multicenter study. J. Allergy Clin. Immunol. 2014, 134, 1093–1099. [Google Scholar] [CrossRef]
- Cooper, B.T.; Holmes, G.K.T.; Ferguson, R.; Thompson, R.A.; Allan, R.N.; Cooke, W.T. Gluten-sensitive diarrhea without evidence of celiac disease. Gastroenterology 1980, 79, 801–806. [Google Scholar] [CrossRef]
- Digiacomo, D.V.; Tennyson, C.A.; Green, P.H.; Demmer, R.T. Prevalence of gluten-free diet adherence among individuals without celiac disease in the USA: Results from the continuous national health and nutrition examination survey 2009–2010. Scand. J. Gastroenterol. 2013, 48, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Bardella, M.T.; Calabrò, A.; Troncone, R.; Corazza, G.R.; Bagnato, C.; Belcari, C.; Bellantoni, A.; Caio, G.; Calella, F.; et al. An Italian prospective multicenter survey on patients suspected of having non-celiac gluten sensitivity. BMC Med. 2014, 12. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Biesiekierski, J.R.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomised clinical trial: Gluten may cause depression in subjects with non-coeliac gluten sensitivity—An exploratory clinical study. Aliment. Pharmacol. Ther. 2014, 39, 1104–1112. [Google Scholar] [CrossRef] [PubMed]

| Coeliac Disease | Dermatitis Herpetiformis | Gluten Ataxia | Wheat Allergy | NCGS | |
|---|---|---|---|---|---|
| Prevalence in the general population |
| 30–75 per 100,000 |
| Prevalence assessed by OFD still unknown |
|
| Pathogenesis |
| Role of TG3 enzyme | AGA cross-react with epitopes on Purkinje cells |
|
|
| Genetics | HLA-DQ2 and DQ8 restricted | HLA-DQ2 and DQ8 restricted | Not HLA restricted | Not HLA restricted | Not HLA restricted |
| Serum antibodies |
|
|
| - positive serum IgE to wheat |
|
| Small bowel histology |
| Increased IEL in almost 100% but frank VA in only 70%–75% of patients | Duodenal VA in up to 40% patients | Normal duodenal histology | Normal duodenal architecture |
| Clinical picture |
| Itchy blistering rash involving elbows, extensor surfaces of forearms, knees and buttocks | - gait and lower limb ataxia ± other GI or extra-GI symptoms | Intestinal and extra-intestinal symptoms within minutes to 1–3 h after exposure to wheat | Intestinal and extra-intestinal symptoms |
| Risk of complications |
| Not increased | Progression of neurological dysfunction | Increased (anaphylaxis) | Unknown |
| Morbidity | Increased | Not increased | Increased | Increased | Unknown |
| Mortality | Increased | Not increased | Increased | Increased | Unknown |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiepatti, A.; Savioli, J.; Vernero, M.; Borrelli de Andreis, F.; Perfetti, L.; Meriggi, A.; Biagi, F. Pitfalls in the Diagnosis of Coeliac Disease and Gluten-Related Disorders. Nutrients 2020, 12, 1711. https://doi.org/10.3390/nu12061711
Schiepatti A, Savioli J, Vernero M, Borrelli de Andreis F, Perfetti L, Meriggi A, Biagi F. Pitfalls in the Diagnosis of Coeliac Disease and Gluten-Related Disorders. Nutrients. 2020; 12(6):1711. https://doi.org/10.3390/nu12061711
Chicago/Turabian StyleSchiepatti, Annalisa, Jessica Savioli, Marta Vernero, Federica Borrelli de Andreis, Luca Perfetti, Antonio Meriggi, and Federico Biagi. 2020. "Pitfalls in the Diagnosis of Coeliac Disease and Gluten-Related Disorders" Nutrients 12, no. 6: 1711. https://doi.org/10.3390/nu12061711
APA StyleSchiepatti, A., Savioli, J., Vernero, M., Borrelli de Andreis, F., Perfetti, L., Meriggi, A., & Biagi, F. (2020). Pitfalls in the Diagnosis of Coeliac Disease and Gluten-Related Disorders. Nutrients, 12(6), 1711. https://doi.org/10.3390/nu12061711





