Withdrawal from Extended, Intermittent Access to A Highly Palatable Diet Impairs Hippocampal Memory Function and Neurogenesis: Effects of Memantine
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Drugs
2.3. Ad libitum Diet Alternation
2.4. Object Location (OL) Task
2.5. Novel Object Recognition (NOR) Task
2.6. Novel Cue vs. Novel Place Preference (NC-NP Preference) Task
2.7. Spontaneous Alternation Behavior (SAB) Task
2.8. Food Intake Experiments
2.9. Perfusions and Immunohistochemistry
2.10. Quantification
2.11. Statistical Analysis
3. Results
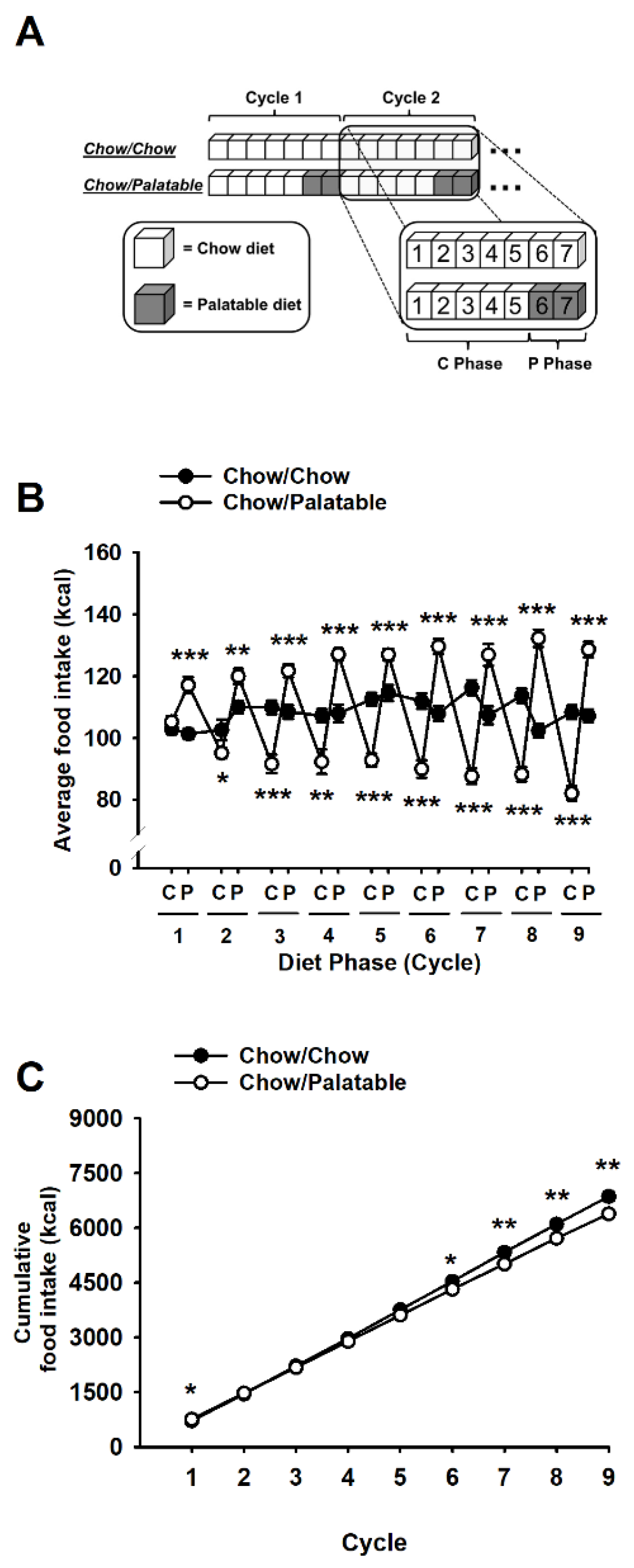
3.1. Effects of Palatable Diet Alternation on Food Intake and Body Weight
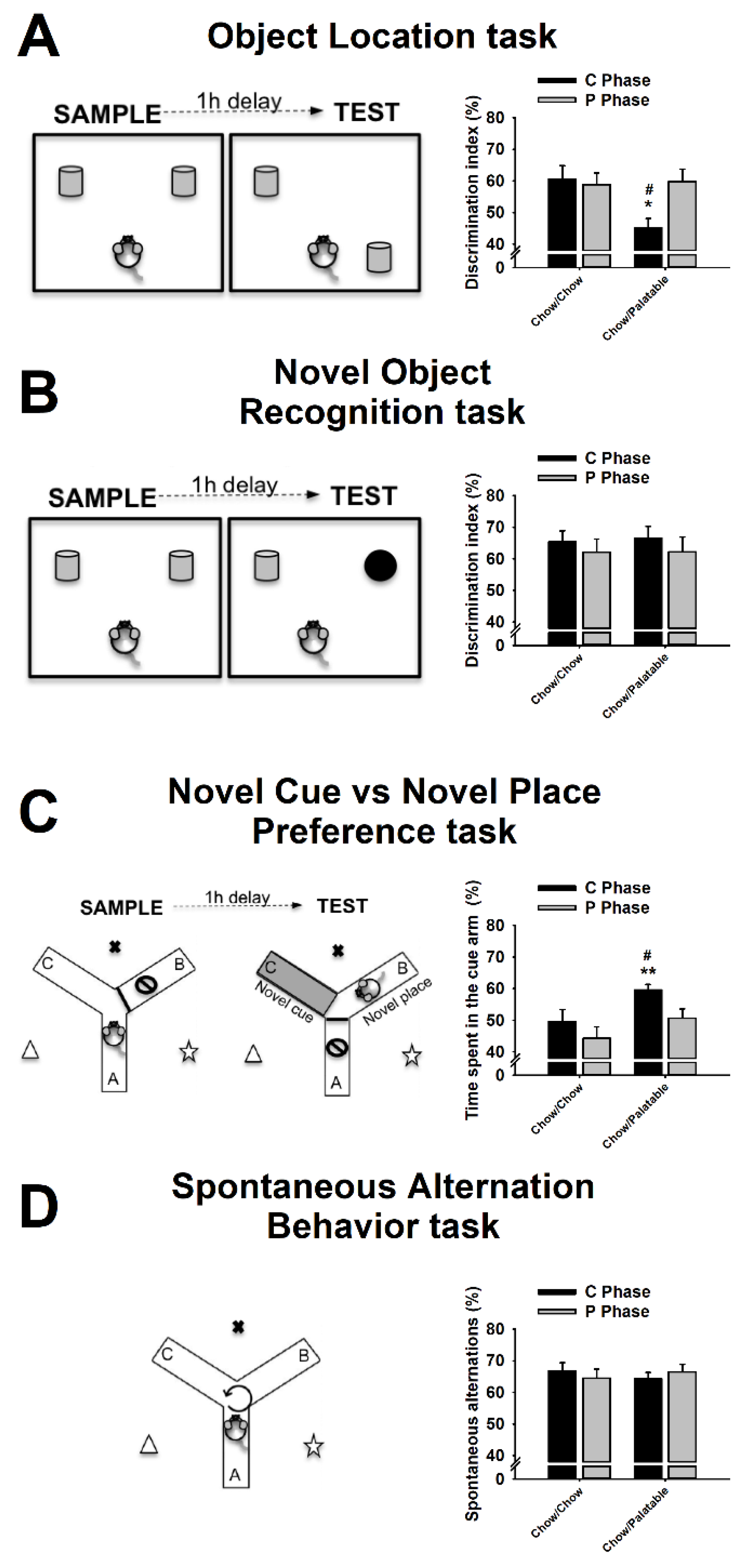
3.2. Effects of Palatable Diet Alternation on Spatial Memory Using the Object Location (OL) Task
3.3. Effects of Palatable Diet Alternation on Recognition Memory Using the Novel Object Recognition (NOR) Task
3.4. Effects of Palatable Diet Alternation on the Preference for Stimulus—vs. Spatial—Response Learning Using the Novel Cue vs. Novel Place Preference (NC-NP Preference) Task
3.5. Effects of Palatable Diet Alternation on Working Memory Using the Spontaneous Alternation Behavior (SAB) Task
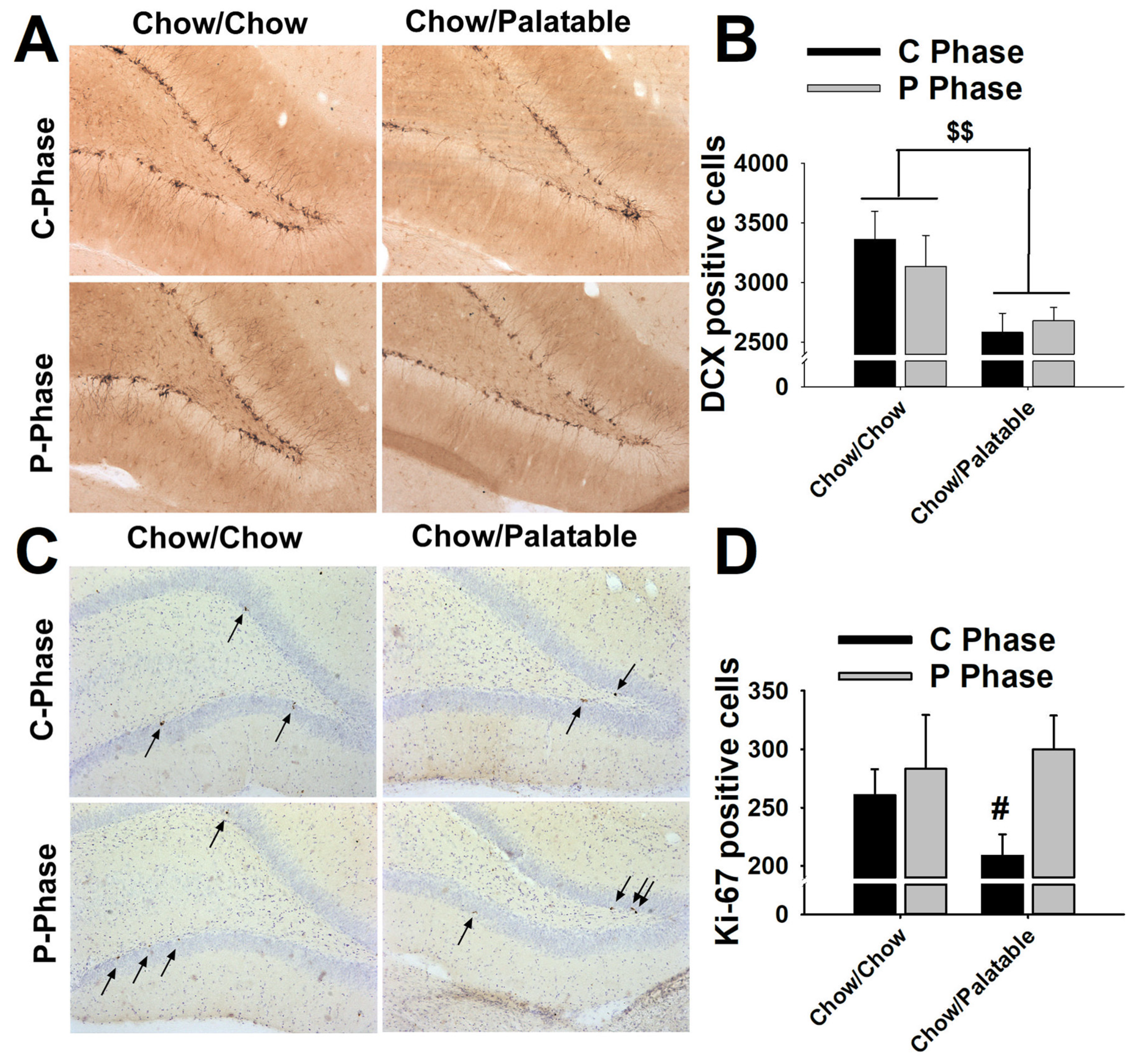
3.6. Effects of Palatable Diet Alternation on Cell Proliferation and Neurogenesis in the Dentate Gyrus of the Hippocampus
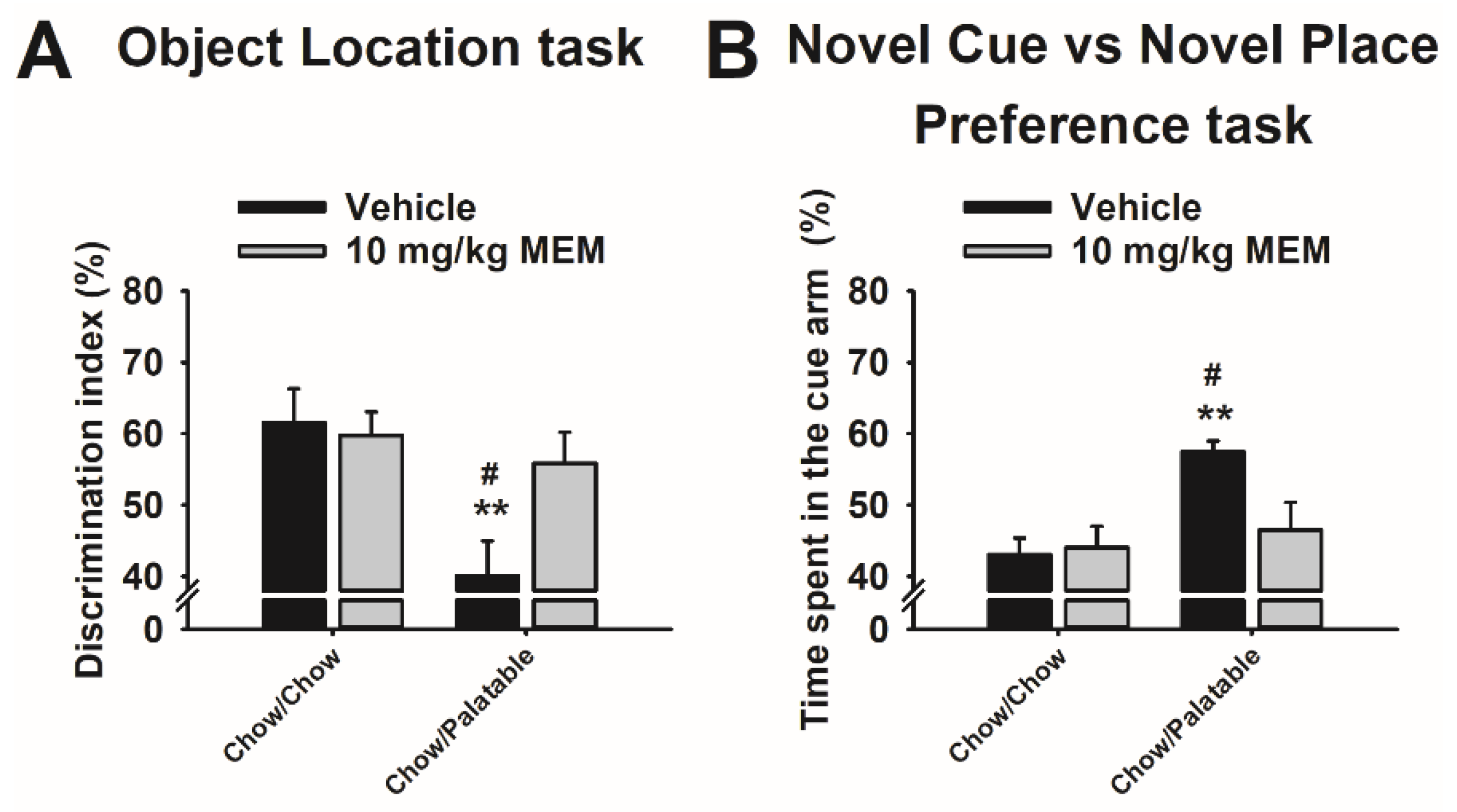
3.7. Effects of Memantine Treatment on Spatial Memory in Diet Cycled Rats
3.8. Effects of Memantine Treatment on the Preference for Stimulus—vs. Spatial—Response Learning in Diet Cycled Rats
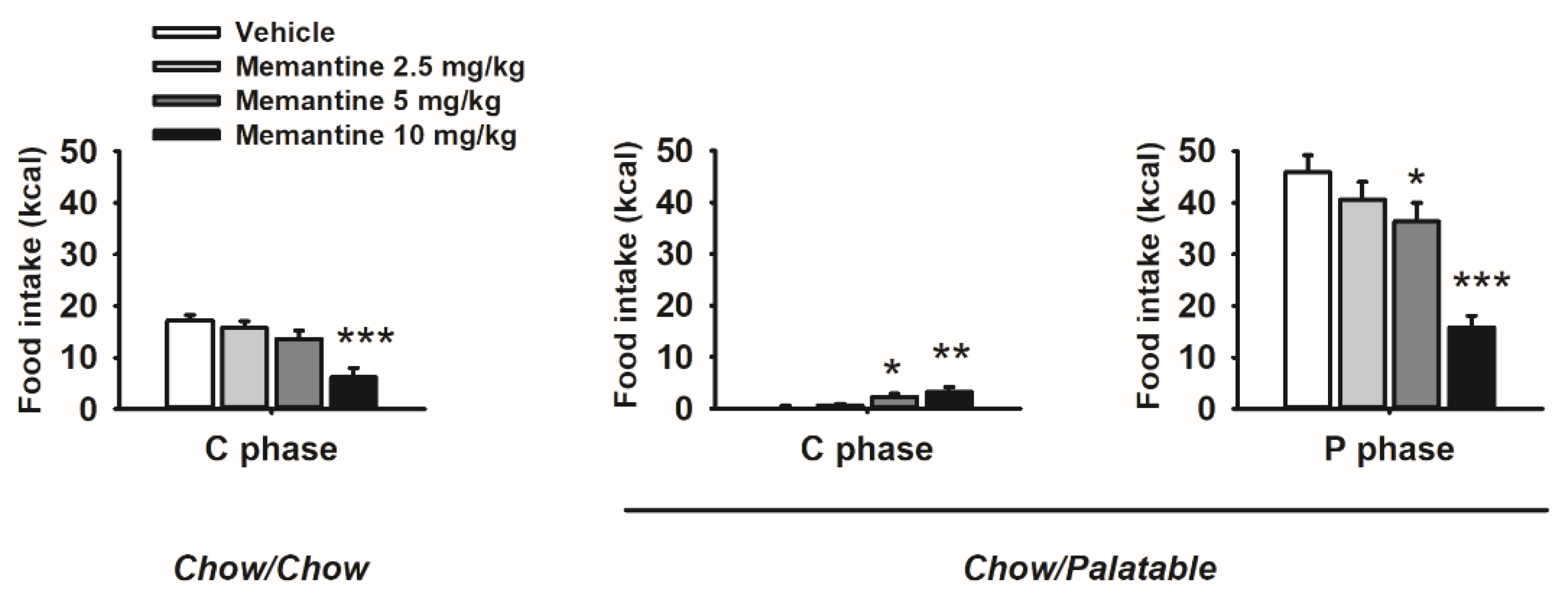
3.9. Effects of Memantine on Food Intake and Body Weight of Diet Cycled Rats
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dulloo, A.G.; Jacquet, J.; Montani, J.P.; Schutz, Y. How dieting makes the lean fatter: From a perspective of body composition autoregulation through adipostats and proteinstats awaiting discovery. Obes. Rev. 2015, 16, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Rouhani, M.H.; Haghighatdoost, F.; Surkan, P.J.; Azadbakht, L. Associations between dietary energy density and obesity: A systematic review and meta-analysis of observational studies. Nutrition 2016, 32, 1037–1047. [Google Scholar] [CrossRef] [PubMed]
- Polivy, J.; Herman, C.P. Dieting and binging. A causal analysis. Am. Psychol. 1985, 40, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.F.; Sabino, V.; Koob, G.F.; Cottone, P. Pathological Overeating: Emerging Evidence for a Compulsivity Construct. Neuropsychopharmacology 2017, 42, 1375–1389. [Google Scholar] [CrossRef]
- Kanoski, S.E.; Davidson, T.L. Western diet consumption and cognitive impairment: Links to hippocampal dysfunction and obesity. Physiol. Behav. 2011, 103, 59–68. [Google Scholar] [CrossRef]
- Weider, S.; Indredavik, M.S.; Lydersen, S.; Hestad, K. Neuropsychological function in patients with anorexia nervosa or bulimia nervosa. Int. J. Eat. Disord. 2015, 48, 397–405. [Google Scholar] [CrossRef]
- Mathias, J.L.; Kent, P.S. Neuropsychological consequences of extreme weight loss and dietary restriction in patients with anorexia nervosa. J. Clin. Exp. Neuropsychol. 1998, 20, 548–564. [Google Scholar] [CrossRef]
- Holland, P.C.; Bouton, M.E. Hippocampus and context in classical conditioning. Curr. Opin. Neurobiol. 1999, 9, 195–202. [Google Scholar] [CrossRef]
- Davidson, T.L.; Chan, K.; Jarrard, L.E.; Kanoski, S.E.; Clegg, D.J.; Benoit, S.C. Contributions of the hippocampus and medial prefrontal cortex to energy and body weight regulation. Hippocampus 2009, 19, 235–252. [Google Scholar] [CrossRef]
- Snyder, J.S.; Soumier, A.; Brewer, M.; Pickel, J.; Cameron, H.A. Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature 2011, 476, 458–461. [Google Scholar] [CrossRef]
- Leuner, B.; Gould, E. Structural plasticity and hippocampal function. Annu. Rev. Psychol. 2010, 61, 111–140. [Google Scholar] [CrossRef] [PubMed]
- Aimone, J.B.; Li, Y.; Lee, S.W.; Clemenson, G.D.; Deng, W.; Gage, F.H. Regulation and function of adult neurogenesis: From genes to cognition. Physiol. Rev. 2014, 94, 991–1026. [Google Scholar] [CrossRef] [PubMed]
- Revest, J.M.; Dupret, D.; Koehl, M.; Funk-Reiter, C.; Grosjean, N.; Piazza, P.V.; Abrous, N. Adult hippocampal neurogenesis is involved in anxiety-related behaviors. Mol. Psychiatry 2009, 14, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Mandyam, C.D.; Koob, G.F. The addicted brain craves new neurons: Putative role for adult-born progenitors in promoting recovery. Trends Neurosci. 2012, 35, 250–260. [Google Scholar] [CrossRef]
- Cottone, P.; Sabino, V.; Roberto, M.; Bajo, M.; Pockros, L.; Frihauf, J.B.; Fekete, E.M.; Steardo, L.; Rice, K.C.; Grigoriadis, D.E.; et al. CRF system recruitment mediates dark side of compulsive eating. Proc. Natl. Acad. Sci. USA 2009, 106, 20016–20020. [Google Scholar] [CrossRef]
- Iemolo, A.; Blasio, A.; St Cyr, S.A.; Jiang, F.; Rice, K.C.; Sabino, V.; Cottone, P. CRF-CRF1 receptor system in the central and basolateral nuclei of the amygdala differentially mediates excessive eating of palatable food. Neuropsychopharmacology 2013, 38, 2456–2466. [Google Scholar] [CrossRef][Green Version]
- Cottone, P.; Iemolo, A.; Narayan, A.R.; Kwak, J.; Momaney, D.; Sabino, V. The uncompetitive NMDA receptor antagonists ketamine and memantine preferentially increase the choice for a small, immediate reward in low-impulsive rats. Psychopharmacology 2013, 226, 127–138. [Google Scholar] [CrossRef]
- Sabino, V.; Narayan, A.R.; Zeric, T.; Steardo, L.; Cottone, P. mTOR activation is required for the anti-alcohol effect of ketamine, but not memantine, in alcohol-preferring rats. Behav. Brain Res. 2013, 247, 9–16. [Google Scholar] [CrossRef]
- Smith, K.L.; Rao, R.R.; Velázquez-Sánchez, C.; Valenza, M.; Giuliano, C.; Everitt, B.J.; Sabino, V.; Cottone, P. The uncompetitive N-methyl-D-aspartate antagonist memantine reduces binge-like eating, food-seeking behavior, and compulsive eating: Role of the nucleus accumbens shell. Neuropsychopharmacology 2015, 40, 1163–1171. [Google Scholar] [CrossRef]
- Iemolo, A.; Valenza, M.; Tozier, L.; Knapp, C.M.; Kornetsky, C.; Steardo, L.; Sabino, V.; Cottone, P. Withdrawal from chronic, intermittent access to a highly palatable food induces depressive-like behavior in compulsive eating rats. Behav. Pharmacol. 2012, 23, 593–602. [Google Scholar] [CrossRef]
- Blasio, A.; Iemolo, A.; Sabino, V.; Petrosino, S.; Steardo, L.; Rice, K.C.; Orlando, P.; Iannotti, F.A.; Di Marzo, V.; Zorrilla, E.P.; et al. Rimonabant precipitates anxiety in rats withdrawn from palatable food: Role of the central amygdala. Neuropsychopharmacology 2013, 38, 2498–2507. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.F.; Leonard, M.Z.; Micovic, N.M.; Miczek, K.A.; Sabino, V.; Cottone, P. Reward sensitivity deficits in a rat model of compulsive eating behavior. Neuropsychopharmacology 2020, 45, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Cottone, P.; Sabino, V.; Steardo, L.; Zorrilla, E.P. Consummatory, anxiety-related and metabolic adaptations in female rats with alternating access to preferred food. Psychoneuroendocrinology 2009, 34, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Dix, S.L.; Aggleton, J.P. Extending the spontaneous preference test of recognition: Evidence of object-location and object-context recognition. Behav. Brain Res. 1999, 99, 191–200. [Google Scholar] [CrossRef]
- Antunes, M.; Biala, G. The novel object recognition memory: Neurobiology, test procedure, and its modifications. Cogn. Process. 2012, 13, 93–110. [Google Scholar] [CrossRef]
- Ennaceur, A.; Delacour, J. A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav. Brain Res. 1988, 31, 47–59. [Google Scholar] [CrossRef]
- Schwabe, L.; Dalm, S.; Schächinger, H.; Oitzl, M.S. Chronic stress modulates the use of spatial and stimulus-response learning strategies in mice and man. Neurobiol. Learn. Mem. 2008, 90, 495–503. [Google Scholar] [CrossRef]
- Ferragud, A.; Haro, A.; Sylvain, A.; Velázquez-Sánchez, C.; Hernández-Rabaza, V.; Canales, J.J. Enhanced habit-based learning and decreased neurogenesis in the adult hippocampus in a murine model of chronic social stress. Behav. Brain Res. 2010, 210, 134–139. [Google Scholar] [CrossRef]
- McDonald, R.J.; White, N.M. A triple dissociation of memory systems: Hippocampus, amygdala, and dorsal striatum. Behav. Neurosci. 1993, 107, 3–22. [Google Scholar] [CrossRef]
- Packard, M.G.; Goodman, J. Factors that influence the relative use of multiple memory systems. Hippocampus 2013, 23, 1044–1052. [Google Scholar] [CrossRef]
- Conrad, C.D.; Galea, L.A.; Kuroda, Y.; McEwen, B.S. Chronic stress impairs rat spatial memory on the Y maze, and this effect is blocked by tianeptine pretreatment. Behav. Neurosci. 1996, 110, 1321–1334. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.S.; Shetty, A.K. Efficacy of doublecortin as a marker to analyse the absolute number and dendritic growth of newly generated neurons in the adult dentate gyrus. Eur. J. Neurosci. 2004, 19, 234–246. [Google Scholar] [CrossRef] [PubMed]
- Kee, N.; Sivalingam, S.; Boonstra, J.M.; Wojtowicz, J.M. The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J. Neurosci. Methods 2002, 115, 97–105. [Google Scholar] [CrossRef]
- Hernandez-Rabaza, V.; Navarro-Mora, G.; Velazquez-Sanchez, C.; Ferragud, A.; Marin, M.P.; Garcia-Verdugo, J.M.; Renau-Piqueras, J.; Canales, J.J. Neurotoxicity and persistent cognitive deficits induced by combined MDMA and alcohol exposure in adolescent rats. Addict. Biol. 2010, 15, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Rabaza, V.; Llorens-Martín, M.; Velázquez-Sánchez, C.; Ferragud, A.; Arcusa, A.; Gumus, H.G.; Gómez-Pinedo, U.; Pérez-Villalba, A.; Roselló, J.; Trejo, J.L.; et al. Inhibition of adult hippocampal neurogenesis disrupts contextual learning but spares spatial working memory, long-term conditional rule retention and spatial reversal. Neuroscience 2009, 159, 59–68. [Google Scholar] [CrossRef]
- Mumby, D.G.; Gaskin, S.; Glenn, M.J.; Schramek, T.E.; Lehmann, H. Hippocampal damage and exploratory preferences in rats: Memory for objects, places, and contexts. Learn. Mem. 2002, 9, 49–57. [Google Scholar] [CrossRef]
- Murray, E.A.; Richmond, B.J. Role of perirhinal cortex in object perception, memory, and associations. Curr. Opin. Neurobiol. 2001, 11, 188–193. [Google Scholar] [CrossRef]
- D’Esposito, M.; Postle, B.R. The cognitive neuroscience of working memory. Annu. Rev. Psychol. 2015, 66, 115–142. [Google Scholar] [CrossRef]
- Murphy, T.; Dias, G.P.; Thuret, S. Effects of diet on brain plasticity in animal and human studies: Mind the gap. Neural. Plast. 2014, 2014, 563160. [Google Scholar] [CrossRef]
- Cottone, P.; Sabino, V.; Steardo, L.; Zorrilla, E.P. Opioid-dependent anticipatory negative contrast and binge-like eating in rats with limited access to highly preferred food. Neuropsychopharmacology 2008, 33, 524–535. [Google Scholar] [CrossRef]
- Quaedflieg, C.; Schwabe, L. Memory dynamics under stress. Memory 2018, 26, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Schwabe, L. Stress and the engagement of multiple memory systems: Integration of animal and human studies. Hippocampus 2013, 23, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Francis, H.M.; Stevenson, R.J. Higher reported saturated fat and refined sugar intake is associated with reduced hippocampal-dependent memory and sensitivity to interoceptive signals. Behav. Neurosci. 2011, 125, 943–955. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Maconie, A.; Williams, C. The influence of the glycaemic load of breakfast on the behaviour of children in school. Physiol. Behav. 2007, 92, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Eskelinen, M.H.; Ngandu, T.; Helkala, E.L.; Tuomilehto, J.; Nissinen, A.; Soininen, H.; Kivipelto, M. Fat intake at midlife and cognitive impairment later in life: A population-based CAIDE study. Int. J. Geriatr. Psychiatry 2008, 23, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Gunstad, J.; Paul, R.H.; Cohen, R.A.; Tate, D.F.; Gordon, E. Obesity is associated with memory deficits in young and middle-aged adults. Eat. Weight Disord. 2006, 11, e15–e19. [Google Scholar] [CrossRef]
- Beilharz, J.E.; Maniam, J.; Morris, M.J. Short exposure to a diet rich in both fat and sugar or sugar alone impairs place, but not object recognition memory in rats. Brain Behav. Immun. 2014, 37, 134–141. [Google Scholar] [CrossRef]
- Kendig, M.D.; Westbrook, R.F.; Morris, M.J. Pattern of access to cafeteria-style diet determines fat mass and degree of spatial memory impairments in rats. Sci. Rep. 2019, 9, 13516. [Google Scholar] [CrossRef]
- Goncalves, J.T.; Schafer, S.T.; Gage, F.H. Adult Neurogenesis in the Hippocampus: From Stem Cells to Behavior. Cell 2016, 167, 897–914. [Google Scholar] [CrossRef]
- Jessberger, S.; Clark, R.E.; Broadbent, N.J.; Clemenson, G.D., Jr.; Consiglio, A.; Lie, D.C.; Squire, L.R.; Gage, F.H. Dentate gyrus-specific knockdown of adult neurogenesis impairs spatial and object recognition memory in adult rats. Learn. Mem. 2009, 16, 147–154. [Google Scholar] [CrossRef]
- Mandyam, C.D.; Harburg, G.C.; Eisch, A.J. Determination of key aspects of precursor cell proliferation, cell cycle length and kinetics in the adult mouse subgranular zone. Neuroscience 2007, 146, 108–122. [Google Scholar] [CrossRef]
- Snyder, J.S.; Choe, J.S.; Clifford, M.A.; Jeurling, S.I.; Hurley, P.; Brown, A.; Kamhi, J.F.; Cameron, H.A. Adult-born hippocampal neurons are more numerous, faster maturing, and more involved in behavior in rats than in mice. J. Neurosci. 2009, 29, 14484–14495. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.I.; Hiripi, E.; Pope, H.G.; Kessler, R.C. The prevalence and correlates of eating disorders in the National Comorbidity Survey Replication. Biol. Psychiatry 2007, 61, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Bale, T.L.; Epperson, C.N. Sex differences and stress across the lifespan. Nat. Neurosci. 2015, 18, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Nazareth, A.; Huang, X.; Voyer, D.; Newcombe, N. A meta-analysis of sex differences in human navigation skills. Psychon. Bull. Rev. 2019, 26, 1503–1528. [Google Scholar] [CrossRef] [PubMed]
- Hamson, D.K.; Roes, M.M.; Galea, L.A. Sex Hormones and Cognition: Neuroendocrine Influences on Memory and Learning. Compr. Physiol. 2016, 6, 1295–1337. [Google Scholar]
- Yang, Z.; Zhou, X.; Zhang, Q. Effectiveness and safety of memantine treatment for Alzheimer’s disease. J. Alzheimers Dis. 2013, 36, 445–458. [Google Scholar] [CrossRef]
- Brennan, B.P.; Roberts, J.L.; Fogarty, K.V.; Reynolds, K.A.; Jonas, J.M.; Hudson, J.I. Memantine in the treatment of binge eating disorder: An open-label, prospective trial. Int. J. Eat. Disord. 2008, 41, 520–526. [Google Scholar] [CrossRef]
- Hermanussen, M.; Tresguerres, J.A. A new anti-obesity drug treatment: First clinical evidence that, antagonising glutamate-gated Ca2+ ion channels with memantine normalises binge-eating disorders. Econ. Hum. Biol. 2005, 3, 329–337. [Google Scholar] [CrossRef]
- Bisaga, A.; Danysz, W.; Foltin, R.W. Antagonism of glutamatergic NMDA and mGluR5 receptors decreases consumption of food in baboon model of binge-eating disorder. Eur. Neuropsychopharmacol. 2008, 18, 794–802. [Google Scholar] [CrossRef]
- Moore, C.F.; Panciera, J.I.; Sabino, V.; Cottone, P. Neuropharmacology of compulsive eating. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170024. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferragud, A.; Velázquez-Sánchez, C.; Abdullatif, A.A.; Sabino, V.; Cottone, P. Withdrawal from Extended, Intermittent Access to A Highly Palatable Diet Impairs Hippocampal Memory Function and Neurogenesis: Effects of Memantine. Nutrients 2020, 12, 1520. https://doi.org/10.3390/nu12051520
Ferragud A, Velázquez-Sánchez C, Abdullatif AA, Sabino V, Cottone P. Withdrawal from Extended, Intermittent Access to A Highly Palatable Diet Impairs Hippocampal Memory Function and Neurogenesis: Effects of Memantine. Nutrients. 2020; 12(5):1520. https://doi.org/10.3390/nu12051520
Chicago/Turabian StyleFerragud, Antonio, Clara Velázquez-Sánchez, Ali Al Abdullatif, Valentina Sabino, and Pietro Cottone. 2020. "Withdrawal from Extended, Intermittent Access to A Highly Palatable Diet Impairs Hippocampal Memory Function and Neurogenesis: Effects of Memantine" Nutrients 12, no. 5: 1520. https://doi.org/10.3390/nu12051520
APA StyleFerragud, A., Velázquez-Sánchez, C., Abdullatif, A. A., Sabino, V., & Cottone, P. (2020). Withdrawal from Extended, Intermittent Access to A Highly Palatable Diet Impairs Hippocampal Memory Function and Neurogenesis: Effects of Memantine. Nutrients, 12(5), 1520. https://doi.org/10.3390/nu12051520




