Effects of a High-Protein Diet on Cardiometabolic Health, Vascular Function, and Endocannabinoids—A PREVIEW Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Cardiometabolic Risk Markers
2.2.1. Ambulatory and Office Blood Pressure
2.2.2. Serum Lipids and Lipoproteins
2.3. Vascular Function Measurements
2.4. Endocannabinoids and Endocannabinoid-Related Compounds
2.5. Statistical Analyses
3. Results
3.1. Baseline Characteristics
3.2. Cardiometabolic Risk Markers
3.2.1. Office and Ambulatory Blood Pressure
3.2.2. Serum Lipids and Lipoproteins
3.3. Endothelial Function, Arterial Stiffness, and Retinal Microvascular Structure
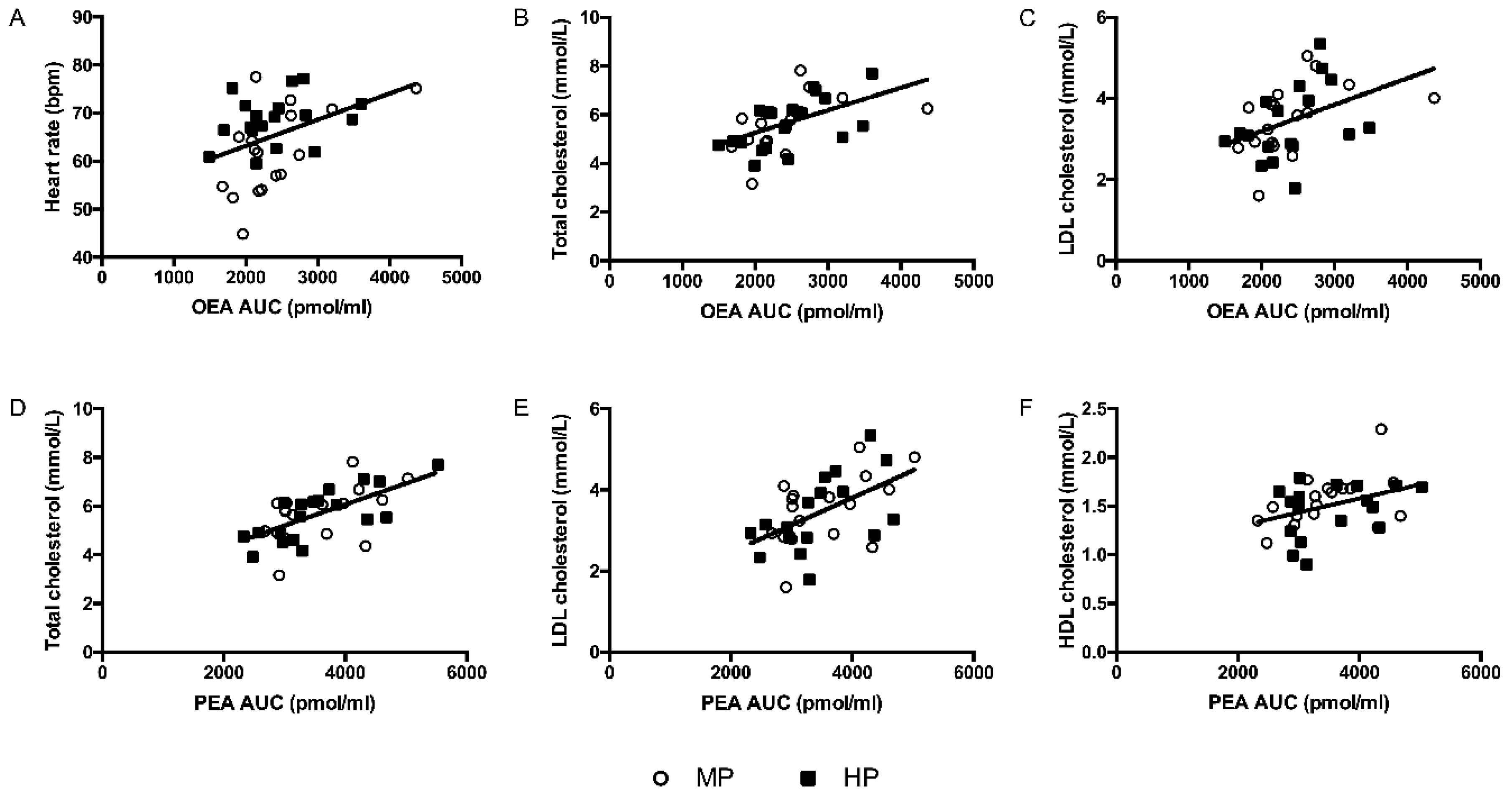
3.4. Endocannabinoids and Endocannabinoid-Related Compounds
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Cardiovascular Diseases (CVDs) Fact Sheet. Available online: http://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 19 November 2019).
- Mendis, S.; Puska, P.; Norrving, B.; World Health Organization; World Heart Federation. Global Atlas on Cardiovascular Disease Prevention and Control; World Health Organization: Geneva, Switzerland, 2011; pp. 3–18. [Google Scholar]
- Drummen, M.; Tischmann, L.; Gatta-Cherifi, B.; Adam, T.; Westerterp-Plantenga, M. Dietary Protein and Energy Balance in Relation to Obesity and Co-morbidities. Front. Endocrinol. (Lausanne) 2018, 9, 443. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, A.N.; Kondrup, J.; Borsheim, E. Health effects of protein intake in healthy adults: A systematic literature review. Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef] [PubMed]
- Huntriss, R.; Campbell, M.; Bedwell, C. The interpretation and effect of a low-carbohydrate diet in the management of type 2 diabetes: A systematic review and meta-analysis of randomised controlled trials. Eur. J. Clin. Nutr. 2018, 72, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Santesso, N.; Akl, E.A.; Bianchi, M.; Mente, A.; Mustafa, R.; Heels-Ansdell, D.; Schunemann, H.J. Effects of higher- versus lower-protein diets on health outcomes: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2012, 66, 780–788. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Long-term effects of low-fat diets either low or high in protein on cardiovascular and metabolic risk factors: A systematic review and meta-analysis. Nutr. J. 2013, 12, 48. [Google Scholar] [CrossRef]
- Teunissen-Beekman, K.F.; Dopheide, J.; Geleijnse, J.M.; Bakker, S.J.; Brink, E.J.; de Leeuw, P.W.; Serroyen, J.; van Baak, M.A. Blood pressure decreases more after high-carbohydrate meals than after high-protein meals in overweight adults with elevated blood pressure, but there is no difference after 4 weeks of consuming a carbohydrate-rich or protein-rich diet. J. Nutr. 2013, 143, 424–429. [Google Scholar] [CrossRef]
- Cote, M.; Matias, I.; Lemieux, I.; Petrosino, S.; Almeras, N.; Despres, J.P.; Di Marzo, V. Circulating endocannabinoid levels, abdominal adiposity and related cardiometabolic risk factors in obese men. Int. J. Obes. (Lond) 2007, 31, 692–699. [Google Scholar] [CrossRef]
- Kunos, G.; Jarai, Z.; Batkai, S.; Goparaju, S.K.; Ishac, E.J.; Liu, J.; Wang, L.; Wagner, J.A. Endocannabinoids as cardiovascular modulators. Chem. Phys. Lipids 2000, 108, 159–168. [Google Scholar] [CrossRef]
- Sarzani, R. Endocannabinoids, blood pressure and the human heart. J. Neuroendocrinol. 2008, 20 (Suppl. 1), 58–62. [Google Scholar] [CrossRef]
- Stanley, C.; O’Sullivan, S.E. Vascular targets for cannabinoids: Animal and human studies. Br. J. Pharmacol. 2014, 171, 1361–1378. [Google Scholar] [CrossRef]
- Scheen, A.J.; Finer, N.; Hollander, P.; Jensen, M.D.; Van Gaal, L.F.; Group, R.I.-D.S. Efficacy and tolerability of rimonabant in overweight or obese patients with type 2 diabetes: A randomised controlled study. Lancet 2006, 368, 1660–1672. [Google Scholar] [CrossRef]
- Van Gaal, L.F.; Rissanen, A.M.; Scheen, A.J.; Ziegler, O.; Rossner, S.; Group, R.I.-E.S. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet 2005, 365, 1389–1397. [Google Scholar] [CrossRef]
- Woods, S.C. Role of the endocannabinoid system in regulating cardiovascular and metabolic risk factors. Am. J. Med. 2007, 120, S19–S25. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.; Kristensen, P.K.; Bartels, E.M.; Bliddal, H.; Astrup, A. Efficacy and safety of the weight-loss drug rimonabant: A meta-analysis of randomised trials. Lancet 2007, 370, 1706–1713. [Google Scholar] [CrossRef]
- Engeli, S.; Lehmann, A.C.; Kaminski, J.; Haas, V.; Janke, J.; Zoerner, A.A.; Luft, F.C.; Tsikas, D.; Jordan, J. Influence of dietary fat intake on the endocannabinoid system in lean and obese subjects. Obesity (Silver Spring) 2014, 22, E70–E76. [Google Scholar] [CrossRef] [PubMed]
- Tischmann, L.; Drummen, M.; Gatta-Cherifi, B.; Raben, A.; Fogelholm, M.; Hartmann, B.; Holst, J.J.; Matias, I.; Cota, D.; Mensink, R.P.; et al. Effects of a High-Protein/Moderate-Carbohydrate Diet on Appetite, Gut Peptides, and Endocannabinoids-A Preview Study. Nutrients 2019, 11, 2269. [Google Scholar] [CrossRef]
- Fogelholm, M.; Larsen, T.M.; Westerterp-Plantenga, M.; Macdonald, I.; Martinez, J.A.; Boyadjieva, N.; Poppitt, S.; Schlicht, W.; Stratton, G.; Sundvall, J.; et al. PREVIEW: Prevention of Diabetes through Lifestyle Intervention and Population Studies in Europe and around the World. Design, Methods, and Baseline Participant Description of an Adult Cohort Enrolled into a Three-Year Randomised Clinical Trial. Nutrients 2017, 9, 632. [Google Scholar] [CrossRef]
- Schoffelen, P.F.; Westerterp, K.R.; Saris, W.H.; Ten Hoor, F. A dual-respiration chamber system with automated calibration. J. Appl. Physiol. (1985) 1997, 83, 2064–2072. [Google Scholar] [CrossRef]
- Joris, P.J.; Plat, J.; Bakker, S.J.; Mensink, R.P. Long-term magnesium supplementation improves arterial stiffness in overweight and obese adults: Results of a randomized, double-blind, placebo-controlled intervention trial. Am. J. Clin. Nutr. 2016, 103, 1260–1266. [Google Scholar] [CrossRef]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Joris, P.J.; Plat, J.; Kusters, Y.H.; Houben, A.J.; Stehouwer, C.D.; Schalkwijk, C.G.; Mensink, R.P. Diet-induced weight loss improves not only cardiometabolic risk markers but also markers of vascular function: A randomized controlled trial in abdominally obese men. Am. J. Clin. Nutr. 2017, 105, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Gatta-Cherifi, B.; Matias, I.; Vallee, M.; Tabarin, A.; Marsicano, G.; Piazza, P.V.; Cota, D. Simultaneous postprandial deregulation of the orexigenic endocannabinoid anandamide and the anorexigenic peptide YY in obesity. Int. J. Obes. (Lond) 2012, 36, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Kingsley, P.J.; Marnett, L.J. LC-MS-MS analysis of neutral eicosanoids. Methods Enzym. 2007, 433, 91–112. [Google Scholar] [CrossRef]
- Vallee, M.; Vitiello, S.; Bellocchio, L.; Hebert-Chatelain, E.; Monlezun, S.; Martin-Garcia, E.; Kasanetz, F.; Baillie, G.L.; Panin, F.; Cathala, A.; et al. Pregnenolone can protect the brain from cannabis intoxication. Science 2014, 343, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Drummen, M.; Tischmann, L.; Gatta-Cherifi, B.; Fogelholm, M.; Raben, A.; Adam, T.C.; Westerterp-Plantenga, M.S. High Compared with Moderate Protein Intake Reduces Adaptive Thermogenesis and Induces a Negative Energy Balance during Long-term Weight-Loss Maintenance in Participants with Prediabetes in the Postobese State: A PREVIEW Study. J. Nutr. 2020, 150, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.N.; Altman, D.G.; Campbell, M.J.; Royston, P. Analysis of serial measurements in medical research. BMJ 1990, 300, 230–235. [Google Scholar] [CrossRef]
- Drummen, M.; Tischmann, L.; Gatta-Cherifi, B.; Fogelholm, M.; Raben, A.; Adam, T.C.; Westerterp-Plantenga, M. Role of endocannabinoids in energy balance regulation in participants in the post-obese state —A PREVIEW study. J. Clin. Endocrinol. Metab. 2020, dgaa193. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Soenen, S.; Martens, E.A.; Hochstenbach-Waelen, A.; Lemmens, S.G.; Westerterp-Plantenga, M.S. Normal protein intake is required for body weight loss and weight maintenance, and elevated protein intake for additional preservation of resting energy expenditure and fat free mass. J. Nutr. 2013, 143, 591–596. [Google Scholar] [CrossRef]
- Dasinger, J.H.; Fehrenbach, D.J.; Abais-Battad, J.M. Dietary Protein: Mechanisms Influencing Hypertension and Renal Disease. Curr. Hypertens. Rep. 2020, 22, 13. [Google Scholar] [CrossRef]
- Soenen, S.; Bonomi, A.G.; Lemmens, S.G.; Scholte, J.; Thijssen, M.A.; van Berkum, F.; Westerterp-Plantenga, M.S. Relatively high-protein or ’low-carb’ energy-restricted diets for body weight loss and body weight maintenance? Physiol. Behav. 2012, 107, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Veldhorst, M.; Smeets, A.; Soenen, S.; Hochstenbach-Waelen, A.; Hursel, R.; Diepvens, K.; Lejeune, M.; Luscombe-Marsh, N.; Westerterp-Plantenga, M. Protein-induced satiety: Effects and mechanisms of different proteins. Physiol. Behav. 2008, 94, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Schrack, J.A.; Zipunnikov, V.; Goldsmith, J.; Bandeen-Roche, K.; Crainiceanu, C.M.; Ferrucci, L. Estimating energy expenditure from heart rate in older adults: A case for calibration. PLoS ONE 2014, 9, e93520. [Google Scholar] [CrossRef] [PubMed]
- Kanakis, C., Jr.; Pouget, J.M.; Rosen, K.M. The effects of delta-9-tetrahydrocannabinol (cannabis) on cardiac performance with and without beta blockade. Circulation 1976, 53, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, B.M.; Piche, L.A. Replacement of carbohydrate by protein in a conventional-fat diet reduces cholesterol and triglyceride concentrations in healthy normolipidemic subjects. Clin. Investig. Med. 1999, 22, 140–148. [Google Scholar]
- Due, A.; Toubro, S.; Stender, S.; Skov, A.R.; Astrup, A. The effect of diets high in protein or carbohydrate on inflammatory markers in overweight subjects. Diabetes Obes. Metab. 2005, 7, 223–229. [Google Scholar] [CrossRef]
- Krebs, J.D.; Elley, C.R.; Parry-Strong, A.; Lunt, H.; Drury, P.L.; Bell, D.A.; Robinson, E.; Moyes, S.A.; Mann, J.I. The Diabetes Excess Weight Loss (DEWL) Trial: A randomised controlled trial of high-protein versus high-carbohydrate diets over 2 years in type 2 diabetes. Diabetologia 2012, 55, 905–914. [Google Scholar] [CrossRef]
- Anderson, J.W.; Bush, H.M. Soy protein effects on serum lipoproteins: A quality assessment and meta-analysis of randomized, controlled studies. J. Am. Coll. Nutr. 2011, 30, 79–91. [Google Scholar] [CrossRef]
- Harland, J.I.; Haffner, T.A. Systematic review, meta-analysis and regression of randomised controlled trials reporting an association between an intake of circa 25 g soya protein per day and blood cholesterol. Atherosclerosis 2008, 200, 13–27. [Google Scholar] [CrossRef]
- Simon, V.; Cota, D. Mechanisms in Endocrinology: Endocannabinoids and metabolism: Past, present and future. Eur. J. Endocrinol. 2017, 176, R309–R324. [Google Scholar] [CrossRef]
- Nesto, R.W.; Mackie, K. Endocannabinoid system and its implications for obesity and cardiometabolic risk. Eur. Heart J. 2008, 10, B34–B41. [Google Scholar] [CrossRef]
- Matias, I.; Gatta-Cherifi, B.; Tabarin, A.; Clark, S.; Leste-Lasserre, T.; Marsicano, G.; Piazza, P.V.; Cota, D. Endocannabinoids measurement in human saliva as potential biomarker of obesity. PLoS ONE 2012, 7, e42399. [Google Scholar] [CrossRef] [PubMed]
- Abdulnour, J.; Yasari, S.; Rabasa-Lhoret, R.; Faraj, M.; Petrosino, S.; Piscitelli, F.; Prud’ Homme, D.; Di Marzo, V. Circulating endocannabinoids in insulin sensitive vs. insulin resistant obese postmenopausal women. A MONET group study. Obesity (Silver Spring) 2014, 22, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Oveisi, F.; Gaetani, S.; Lin, E.; Piomelli, D. Oleoylethanolamide, an endogenous PPAR-alpha agonist, lowers body weight and hyperlipidemia in obese rats. Neuropharmacology 2005, 48, 1147–1153. [Google Scholar] [CrossRef]
- Pataky, Z.; Carrard, I.; Gay, V.; Thomas, A.; Carpentier, A.; Bobbioni-Harsch, E.; Golay, A. Effects of a Weight Loss Program on Metabolic Syndrome, Eating Disorders and Psychological Outcomes: Mediation by Endocannabinoids? Obes. Facts 2018, 11, 144–156. [Google Scholar] [CrossRef]
- Ferrara, L.A.; Innelli, P.; Palmieri, V.; Limauro, S.; De Luca, G.; Ferrara, F.; Liccardo, E.; Celentano, A. Effects of different dietary protein intakes on body composition and vascular reactivity. Eur. J. Clin. Nutr. 2006, 60, 643–649. [Google Scholar] [CrossRef]
- Fekete, A.A.; Giromini, C.; Chatzidiakou, Y.; Givens, D.I.; Lovegrove, J.A. Whey protein lowers systolic blood pressure and Ca-caseinate reduces serum TAG after a high-fat meal in mildly hypertensive adults. Sci. Rep. 2018, 8, 5026. [Google Scholar] [CrossRef]
- Maki, K.C.; Rains, T.M.; Schild, A.L.; Dicklin, M.R.; Park, K.M.; Lawless, A.L.; Kelley, K.M. Effects of low-fat dairy intake on blood pressure, endothelial function, and lipoprotein lipids in subjects with prehypertension or stage 1 hypertension. Vasc. Health Risk Manag. 2013, 9, 369–379. [Google Scholar] [CrossRef]
- Yaron, M.; Roach, V.; Izkhakov, E.; Ish-Shalom, M.; Sack, J.; Sofer, Y.; Azzam, I.; Ray, A.; Stern, N.; Tordjman, K.M. Effects of a typical acute oral calcium load on arterial properties and endothelial function in healthy subjects. Eur. J. Clin. Nutr. 2014, 68, 608–612. [Google Scholar] [CrossRef]
- Reverri, E.J.; LaSalle, C.D.; Franke, A.A.; Steinberg, F.M. Soy provides modest benefits on endothelial function without affecting inflammatory biomarkers in adults at cardiometabolic risk. Mol. Nutr. Food Res. 2015, 59, 323–333. [Google Scholar] [CrossRef]
- Pal, S.; Ellis, V. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity (Silver Spring) 2010, 18, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Quercioli, A.; Carbone, F.; Bonaventura, A.; Liberale, L.; Pataky, Z.; Thomas, A.; Lenglet, S.; Lauer, E.; Golay, A.; Dallegri, F.; et al. Plasma palmitoylethanolamide (PEA) as a potential biomarker for impaired coronary function. Int. J. Cardiol. 2017, 231, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, A.; Oddi, S.; Maccarrone, M. The endocannabinoid system in ageing: A new target for drug development. Curr. Drug Targets 2006, 7, 1539–1552. [Google Scholar] [CrossRef] [PubMed]
| Moderate Protein (n = 18) | High Protein (n = 20) | ||
|---|---|---|---|
| 24 h | SBP (mmHg) | 133 ± 12 | 131 ± 12 |
| DBP (mmHg) | 82 ± 8 | 79 ± 7 | |
| MAP (mmHg) | 106 ± 9 | 103 ± 9 | |
| PP (mmHg) | 51 ± 9 | 52 ± 9 | |
| HR (bpm) | 62 ± 9 | 67 ± 5 * | |
| Daytime | SBP (mmHg) | 139 ± 13 | 136 ± 13 |
| DBP (mmHg) | 86 ± 9 | 83 ± 8 | |
| MAP (mmHg) | 111 ± 10 | 107 ± 9 | |
| PP (mmHg) | 53 ± 10 | 53 ± 9 | |
| HR (bpm) | 63 ± 9 | 69 ± 5 * | |
| Nighttime | SBP (mmHg) | 116 ± 13 | 117 ± 15 |
| DBP (mmHg) | 68 ± 6 | 67 ± 8 | |
| MAP (mmHg) | 90 ± 9 | 90 ± 11 | |
| PP (mmHg) | 48 ± 10 | 48 ± 7 | |
| HR (bpm) | 58 ± 8 | 61 ± 7 | |
| Dipping | SBP (%) | 16 ± 9 | 16 ± 3 |
| DBP (%) | 21 ± 6 | 19 ± 7 | |
| Moderate Protein (n = 18) | High Protein (n = 20) | Treatment Effect | |||
|---|---|---|---|---|---|
| Pre 1 | Post 1 | Pre 1 | Post 1 | Difference in Change 2 | |
| Total cholesterol (mmol/L) | 5.6 ± 1.1 | 5.6 ± 1.0 | 5.6 ± 1.0 | 5.5 ± 1.0 | −0.1 (−0.3; 0.2) |
| HDL cholesterol (mmol/L) | 1.4 ± 0.3 | 1.3 ± 0.2 | 1.5 ± 0.3 | 1.4 ± 0.3 | 0.0 (−0.1; 0.1) |
| LDL cholesterol (mmol/L) | 3.5 ± 0.9 | 3.4 ± 0.8 | 3.4 ± 0.9 | 3.3 ± 0.8 | 0.0 (−0.2; 0.2) |
| Total cholesterol/HDL cholesterol ratio | 4.0 ± 0.9 | 4.4 ± 1.1 | 3.6 ± 0.7 | 3.9 ± 0.7 | −0.1 (−0.3; 0.1) |
| Moderate Protein (n = 18) | High Protein (n = 20) | Treatment Effect | |||
|---|---|---|---|---|---|
| Pre 1 | Post 1 | Pre 1 | Post 1 | Difference in Change 2 | |
| Vascular function | |||||
| Baseline brachial diameter (cm) | 0.58 ± 0.14 | 0.58 ± 0.10 | 0.57 ± 0.12 | 0.56 ± 0.14 | −0.02 (−0.05; 0.01) |
| FMD (%) | 3.4 ± 2.1 | 3.6 ± 2.3 | 4.4 ± 3.2 | 4.4 ± 3.3 | 0.4 (−1.5; 2.3) |
| Arterial stiffness | |||||
| PWVc–f (m/s) 3 | 8.8 ± 1.5 | 8.6 ± 1.1 | 8.8 ± 1.3 | 8.8 ± 1.8 | 0.2 (−0.6; 0.9) |
| cAIxHR75 (%) | 22.1 ± 8.2 | 21.5 ± 7.1 | 25.4 ± 7.0 | 23.3 ± 8.8 | −0.7 (−4.6; 3.2) |
| Retinal microvascular structure | |||||
| Arteriolar width (μm) 4 | 128 ± 19 | 127 ± 20 | 124 ± 23 | 125 ± 19 | 2.4 (−1.9; 6.8) |
| Venular width (μm) 4 | 222 ± 25 | 220 ± 24 | 214 ± 29 | 215 ± 29 | 2.2 (−2.1; 6.5) |
| Arteriolar-to-venular ratio 2 | 0.58 ± 0.09 | 0.58 ± 0.09 | 0.58 ± 0.1 | 0.59 ± 0.08 | 0.01 (−0.02; 0.04) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tischmann, L.; Drummen, M.; Joris, P.J.; Gatta-Cherifi, B.; Raben, A.; Fogelholm, M.; Matias, I.; Cota, D.; Mensink, R.P.; Westerterp-Plantenga, M.S.; et al. Effects of a High-Protein Diet on Cardiometabolic Health, Vascular Function, and Endocannabinoids—A PREVIEW Study. Nutrients 2020, 12, 1512. https://doi.org/10.3390/nu12051512
Tischmann L, Drummen M, Joris PJ, Gatta-Cherifi B, Raben A, Fogelholm M, Matias I, Cota D, Mensink RP, Westerterp-Plantenga MS, et al. Effects of a High-Protein Diet on Cardiometabolic Health, Vascular Function, and Endocannabinoids—A PREVIEW Study. Nutrients. 2020; 12(5):1512. https://doi.org/10.3390/nu12051512
Chicago/Turabian StyleTischmann, Lea, Mathijs Drummen, Peter J. Joris, Blandine Gatta-Cherifi, Anne Raben, Mikael Fogelholm, Isabelle Matias, Daniela Cota, Ronald P. Mensink, Margriet S. Westerterp-Plantenga, and et al. 2020. "Effects of a High-Protein Diet on Cardiometabolic Health, Vascular Function, and Endocannabinoids—A PREVIEW Study" Nutrients 12, no. 5: 1512. https://doi.org/10.3390/nu12051512
APA StyleTischmann, L., Drummen, M., Joris, P. J., Gatta-Cherifi, B., Raben, A., Fogelholm, M., Matias, I., Cota, D., Mensink, R. P., Westerterp-Plantenga, M. S., & Adam, T. C. (2020). Effects of a High-Protein Diet on Cardiometabolic Health, Vascular Function, and Endocannabinoids—A PREVIEW Study. Nutrients, 12(5), 1512. https://doi.org/10.3390/nu12051512








