The Role of Soluble Corn Fiber on Glycemic and Insulin Response
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Protocol
2.3. Test Meals
2.4. Statistical Analysis
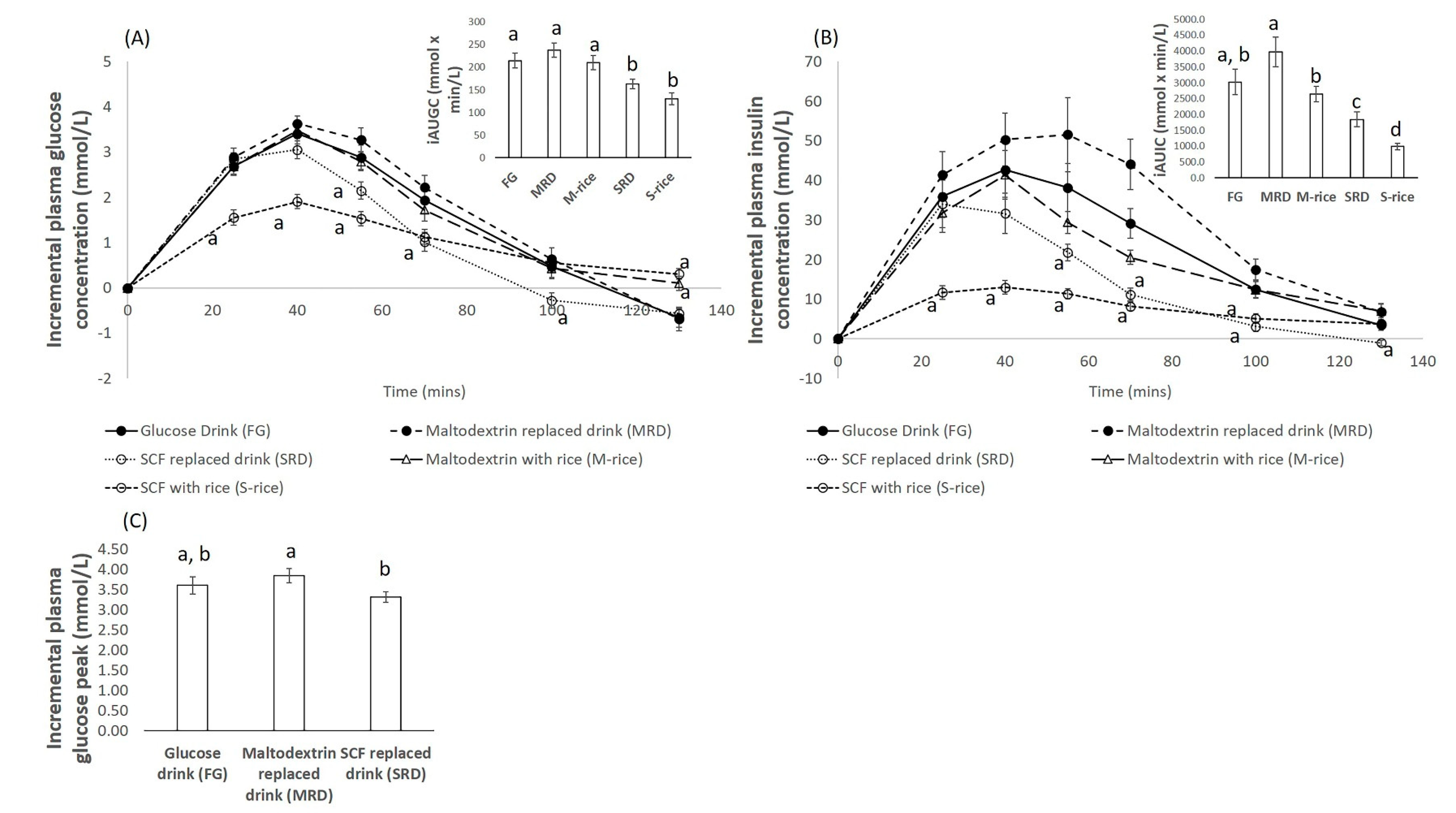
3. Results
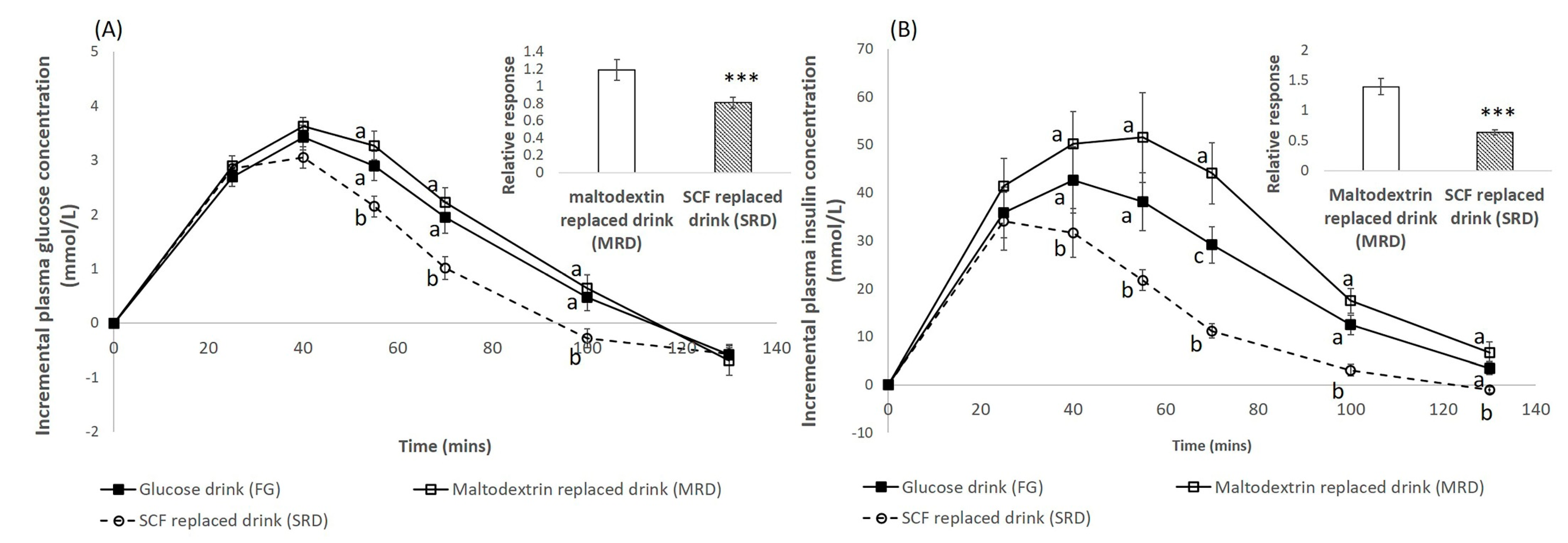
3.1. Blood Glucose Response
3.2. Blood Insulin Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ramachandran, A.; Snehalatha, C.; Shetty, A.S.; Nanditha, A. Trends in prevalence of diabetes in Asian countries. World J. Diabetes 2012, 3, 110. [Google Scholar] [CrossRef] [PubMed]
- Sheard, N.F.; Clark, N.G.; Brand-Miller, J.C.; Franz, M.J.; Pi-Sunyer, F.X.; Mayer-Davis, E.; Kulkarni, K.; Geil, P. Dietary carbohydrate (amount and type) in the prevention and management of diabetes: A statement by the American Diabetes Association. Diabetes Care 2004, 27, 2266–2271. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Liu, S.; Rimm, E.B.; Manson, J.E.; Willett, W.C.; Hu, F.B. Glycemic index, glycemic load, and dietary fiber intake and incidence of type 2 diabetes in younger and middle-aged women. Am. J. Clin. Nutr. 2004, 80, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Brunner, S.; Holub, I.; Theis, S.; Gostner, A.; Melcher, R.; Wolf, P.; Amann-Gassner, U.; Scheppach, W.; Hauner, H. Metabolic effects of replacing sucrose by isomaltulose in subjects with type 2 diabetes: A randomized double-blind trial. Diabetes Care 2012, 35, 1249–1251. [Google Scholar] [CrossRef]
- Andrade, E.F.; Vieira Lobato, R.; Vasques de Araújo, T.; Zangerônimo, M.G.; de Sousa, R.V.; Pereira, L.J. Effect of beta-glucans in the control of blood glucose levels of diabetic patients: A systematic review. Nutr. Hosp. 2015, 31, 170–177. [Google Scholar]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and glycemic control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef]
- van de Laar, F.A. Alpha-glucosidase inhibitors in the early treatment of type 2 diabetes. Vasc. Health Risk Manag. 2008, 4, 1189. [Google Scholar] [CrossRef]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Anderson, J.W.; Deakins, D.A.; Floore, T.L.; Smith, B.M.; Whitis, S.E. Dietary fiber and coronary heart disease. Crit. Rev. Food Sci. Nutr. 1990, 29, 95–147. [Google Scholar] [CrossRef]
- Gunness, P.; Gidley, M.J. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010, 1, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.; Ying, W. A review of physiological effects of soluble and insoluble dietary fibers. J. Nutr. Food Sci. 2016, 6, 476–482. [Google Scholar]
- Angioloni, A.; Collar, C. Physicochemical and nutritional properties of reduced-caloric density high-fibre breads. LWT Food Sci. Technol. 2011, 44, 747–758. [Google Scholar] [CrossRef]
- Dhingra, D.; Michael, M.; Rajput, H.; Patil, R. Dietary fibre in foods: A review. J. Food Sci. Technol. 2012, 49, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Knapp, B.K. Select Novel Carbohydrates Affect Glycemic and Insulinemic Response, Energy Value, and Indices of Gut Health as Measured Using Canine, Avian, Rodent, and in Vitro Model Systems. Ph.D. Thesis, University of Illinois at Urbana-Champaign, Champaign, IL, USA, 2011. [Google Scholar]
- Maathuis, A.; Hoffman, A.; Evans, A.; Sanders, L.; Venema, K. The effect of maize based fibers on the activity and composition of the microbiota determined in a dynamic in vitro model of the human large intestine. J. Am. Coll. Nutr. 2009, 28, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Kendall, C.W.; Esfahani, A.; Hoffman, A.J.; Evans, A.; Sanders, L.M.; Josse, A.R.; Vidgen, E.; Potter, S.M. Effect of novel maize-based dietary fibers on postprandial glycemia and insulinemia. J. Am. Coll. Nutr. 2008, 27, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Whisner, C.M.; Martin, B.R.; Nakatsu, C.H.; McCabe, G.P.; McCabe, L.D.; Peacock, M.; Weaver, C.M. Soluble maize fibre affects short-term calcium absorption in adolescent boys and girls: A randomised controlled trial using dual stable isotopic tracers. Br. J. Nutr. 2014, 112, 446–456. [Google Scholar] [CrossRef]
- Bruhwyler, J.; Carreer, F.; Demanet, E.; Jacobs, H. Digestive tolerance of inulin-type fructans: A double-blind, placebo-controlled, cross-over, dose-ranging, randomized study in healthy volunteers. Int. J. Food Sci. Nutr. 2009, 60, 165–175. [Google Scholar] [CrossRef]
- Boler, B.M.V.; Serao, M.C.R.; Bauer, L.L.; Staeger, M.A.; Boileau, T.W.; Swanson, K.S.; Fahey, G.C. Digestive physiological outcomes related to polydextrose and soluble maize fibre consumption by healthy adult men. Br. J. Nutr. 2011, 106, 1864–1871. [Google Scholar] [CrossRef]
- Jakeman, S.A.; Henry, C.N.; Martin, B.R.; McCabe, G.P.; McCabe, L.D.; Jackson, G.S.; Peacock, M.; Weaver, C.M. Soluble corn fiber increases bone calcium retention in postmenopausal women in a dose-dependent manner: A randomized crossover trial. Am. J. Clin. Nutr. 2016, 104, 837–843. [Google Scholar] [CrossRef]
- Konings, E.; Schoffelen, P.F.; Stegen, J.; Blaak, E.E. Effect of polydextrose and soluble maize fibre on energy metabolism, metabolic profile and appetite control in overweight men and women. Br. J. Nutr. 2014, 111, 111–121. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. FAO/WHO/UNU, Energy and Protein Requirements; Report of a joint FAO/WHO/UNU Expert Consultation; World Health Organization: Geneva, Switzerland, 1985. [Google Scholar]
- Wolever, T.M. Determining the GI of Foods—Methodological Considerations. In The Glycaemic Index: A Physiological Classification of Dietary Carbohydrate; Wolever, T.M., Ed.; CABI: Wallingford, UK, 2006; pp. 12–42. [Google Scholar]
- Lindström, J.; Ilanne-Parikka, P.; Peltonen, M.; Aunola, S.; Eriksson, J.G.; Hemiö, K.; Hämäläinen, H.; Härkönen, P.; Keinänen-Kiukaanniemi, S.; Laakso, M. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: Follow-up of the Finnish Diabetes Prevention Study. Lancet 2006, 368, 1673–1679. [Google Scholar] [CrossRef]
- Laaksonen, D.E.; Lindström, J.; Lakka, T.A.; Eriksson, J.G.; Niskanen, L.; Wikström, K.; Aunola, S.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Valle, T.T. Physical activity in the prevention of type 2 diabetes: The Finnish diabetes prevention study. Diabetes 2005, 54, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Achour, L.; Meance, S.; Briend, A. Comparison of gastric emptying of a solid and a liquid nutritional rehabilitation food. Eur. J. Clin. Nutr. 2001, 55, 769. [Google Scholar] [CrossRef]
- Mourot, J.; Thouvenot, P.; Couet, C.; Antoine, J.; Krobicka, A.; Debry, G. Relationship between the rate of gastric emptying and glucose and insulin responses to starchy foods in young healthy adults. Am. J. Clin. Nutr. 1988, 48, 1035–1040. [Google Scholar] [CrossRef]
- Cervantes-Pahm, S.; Kim, B.; Stein, H. Digestible energy in resistant starch and dietary fiber sources fed to pigs. J. Anim. Sci. 2009, 87, 547. [Google Scholar]
- Ropert, A.; Cherbut, C.; Roze, C.; Le Quellec, A.; Holst, J.; Fu-Cheng, X.; Des Varannes, S.B.; Galmiche, J. Colonic fermentation and proximal gastric tone in humans. Gastroenterology 1996, 111, 289–296. [Google Scholar] [CrossRef]
- Chronakis, I.S. On the molecular characteristics, compositional properties, and structural-functional mechanisms of maltodextrins: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 599–637. [Google Scholar] [CrossRef]
| Test Food | Serving Size (g) | Energy (kJ) | Energy (kcal) | Fat (g) | Protein (g) | Dietary Fiber (g) |
|---|---|---|---|---|---|---|
| SCF 1 glutinous rice (S-rice) | 101 | 883 | 211 | 0.2 | 2 | 25 |
| Maltodextrin glutinous rice (M-rice) | 101 | 883 | 211 | 0.2 | 2 | 1.8 |
| SCF beverage (SRD) | 282 | 812 | 194 | 0 | 0 | 24.1 |
| Maltodextrin beverage (MRD) | 282 | 812 | 194 | 0 | 0 | 0.8 |
| Anthropometric and Physiological Parameters | Mean ± SEM | Range |
|---|---|---|
| Age (years) | 36.7 ± 2.8 | 23.8–60.8 |
| Height (cm) | 172.1 ± 1.5 | 158.4–181.1 |
| Weight (kg) | 70.2 ± 1.7 | 52.5–78.9 |
| BMI (kg/m2) | 23.6 ± 0.3 | 20.3–26.7 |
| Waist circumference (cm) | 83.0 ± 1.4 | 71.1–94.1 |
| Hip circumference (cm) | 96.4 ± 1.3 | 79.0–105.8 |
| Fasting blood glucose (mmol/L) | 4.9 ± 0.1 | 4.0–5.7 |
| Systolic Blood pressure (mmHg) | 125.1 ± 2.1 | 102.0–146.0 |
| Diastolic Blood pressure (mmHg) | 79.5 ± 2.1 | 58.0–100.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, W.S.K.; Chia, P.F.W.; Ponnalagu, S.; Karnik, K.; Henry, C.J. The Role of Soluble Corn Fiber on Glycemic and Insulin Response. Nutrients 2020, 12, 961. https://doi.org/10.3390/nu12040961
Tan WSK, Chia PFW, Ponnalagu S, Karnik K, Henry CJ. The Role of Soluble Corn Fiber on Glycemic and Insulin Response. Nutrients. 2020; 12(4):961. https://doi.org/10.3390/nu12040961
Chicago/Turabian StyleTan, Wei Shuan Kimberly, Pei Fen Winnie Chia, Shalini Ponnalagu, Kavita Karnik, and Christiani Jeyakumar Henry. 2020. "The Role of Soluble Corn Fiber on Glycemic and Insulin Response" Nutrients 12, no. 4: 961. https://doi.org/10.3390/nu12040961
APA StyleTan, W. S. K., Chia, P. F. W., Ponnalagu, S., Karnik, K., & Henry, C. J. (2020). The Role of Soluble Corn Fiber on Glycemic and Insulin Response. Nutrients, 12(4), 961. https://doi.org/10.3390/nu12040961





