The Effects of Low-Nickel Diet Combined with Oral Administration of Selected Probiotics on Patients with Systemic Nickel Allergy Syndrome (SNAS) and Gut Dysbiosis
Abstract
1. Introduction
2. Materials and Methods
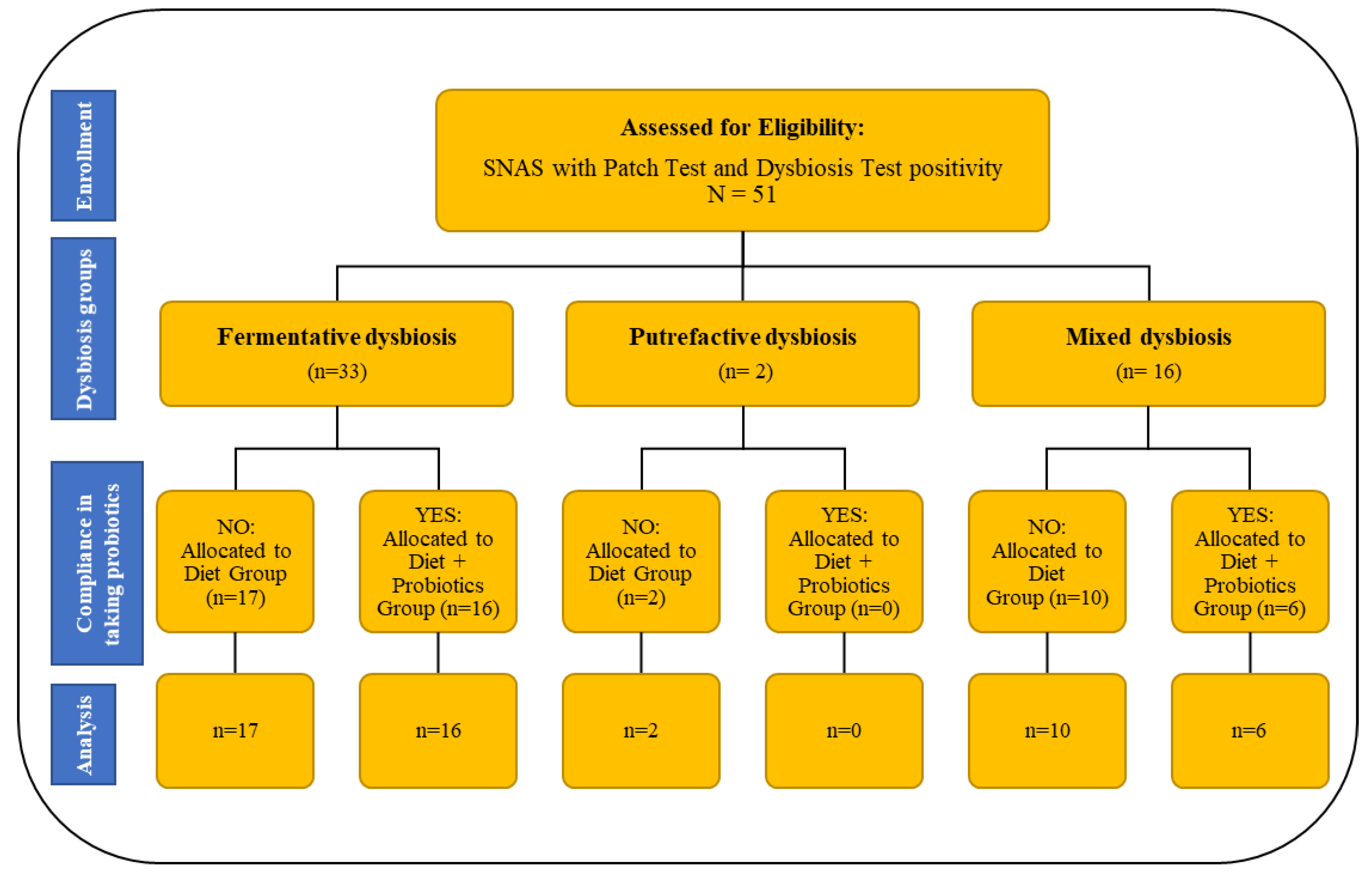
2.1. Patients’ Enrolment and Exclusion Criteria
2.2. Dysbiosis Test
2.3. Study Design
2.4. Statistical Analysis
3. Results
3.1. Characteristics of Patient Samples Stratified by Dysbiosis Type
3.2. Comprehensive Assessment of the Effects of Treatment with Low-Ni Diet Alone or Combined with Probiotic Assumption
3.3. Comparison between Effects of Low-Ni Diet Alone or in Combination with Probiotic Assumption
4. Discussion
5. Limitations of the Study
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thyssen, J.P.; Menne, T. Metal allergy—A review on exposures, penetration, genetics, prevalence, and clinical implications. Chem. Res. Toxicol. 2010, 23, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Ahlstrom, M.G.; Thyssen, J.P.; Wennervaldt, M.; Menne, T.; Johansen, J.D. Nickel allergy and allergic contact dermatitis: A clinical review of immunology, epidemiology, exposure, and treatment. Contact Dermat. 2019, 81, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Torres, F.; das Gracas, M.; Melo, M.; Tosti, A. Management of contact dermatitis due to nickel allergy: An update. Clin. Cosmet. Investig. Dermatol. 2009, 2, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Bechara, R.; Pollastro, S.; Azoury, M.E.; Szely, N.; Maillere, B.; de Vries, N.; Pallardy, M. Identification and Characterization of Circulating Naive CD4+ and CD8+ T Cells Recognizing Nickel. Front. Immunol. 2019, 10, 1331. [Google Scholar] [CrossRef] [PubMed]
- Falagiani, P.; Di Gioacchino, M.; Ricciardi, L.; Minciullo, P.L.; Saitta, S.; Carní, A.; Santoro, G.; Gangemi, S.; Minelli, M.; Bozzetti, M.P.; et al. Systemic nickel allergy syndrome [SNAS]: A review. Rev. Port. Immunoallergol. 2008, 16, 135–147. [Google Scholar]
- Menne, T.; Hjorth, N. Reactions from systemic exposure to contact allergens. Semin. Dermatol. 1982, 1, 15–24. [Google Scholar]
- Di Gioacchino, M.; Masci, S.; Cavallucci, E.; Pavone, G.; Andreassi, M.; Gravante, M.; Pizzicannella, G.; Boscolo, P. Immuno-histopathologic changes in the gastrointestinal mucosa in patients with nickel contact allergy. G. Ital. Med. Lav. 1995, 17, 33–36. [Google Scholar]
- Minelli, M.; Schiavino, D.; Musca, F.; Bruno, M.E.; Falagiani, P.; Mistrello, G.; Riva, G.; Braga, M.; Turi, M.C.; Di Rienzo, V.; et al. Oral hyposensitization to nickel induces clinical improvement and a decrease in TH1 and TH2 cytokines in patients with systemic nickel allergy syndrome. Int. J. Immunopathol. Pharmacol. 2010, 23, 193–201. [Google Scholar] [CrossRef]
- Pizzutelli, S. Systemic nickel hypersensitivity and diet: Myth or reality? Eur. Ann. Allergy Clin. Immunol. 2011, 43, 5–18. [Google Scholar]
- Di Gioacchino, M.; Ricciardi, L.; De Pita, O.; Minelli, M.; Patella, V.; Voltolini, S.; Di Rienzo, V.; Braga, M.; Ballone, E.; Mangifesta, R.; et al. Nickel oral hyposensitization in patients with systemic nickel allergy syndrome. Ann. Med. 2014, 46, 31–37. [Google Scholar] [CrossRef]
- Ricciardi, L.; Arena, A.; Arena, E.; Zambito, M.; Ingrassia, A.; Valenti, G.; Loschiavo, G.; D’Angelo, A.; Saitta, S. Systemic Nickel Allergy Syndrome: Epidemiological Data from Four Italian Allergy Units. Int. J. Immunopath. Pharmacol. 2014, 27, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Veien, N.K.; Menne, T. Nickel contact allergy and a nickel-restricted diet. Semin. Dermatol. 1990, 9, 197–205. [Google Scholar] [PubMed]
- Randazzo, C.L.; Pino, A.; Ricciardi, L.; Romano, C.; Comito, D.; Arena, E.; Saitta, S.; Caggia, C. Probiotic supplementation in systemic nickel allergy syndrome patients: Study of its effects on lactic acid bacteria population and on clinical symptoms. J. Appl. Microbiol. 2015, 118, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.B.; Dancy, B.C.R.; Horton, C.L.; Lee, Y.S.; Madejczyk, M.S.; Xu, Z.Z.; Ackermann, G.; Humphrey, G.; Palacios, G.; Knight, R.; et al. Exposure to toxic metals triggers unique responses from the rat gut microbiota. Sci. Rep. 2018, 8, 6578. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Cui, H.; Peng, X.; Pan, K.; Fang, J.; Zuo, Z.; Deng, J.; Wang, X.; Huang, J. Toxicological effects of dietary nickel chloride on intestinal microbiota. Ecotoxicol. Environ. Saf. 2014, 109, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Hugon, P.; Dufour, J.C.; Colson, P.; Fournier, P.E.; Sallah, K.; Raoult, D. A comprehensive repertoire of prokaryotic species identified in human beings. Lancet Infect. Dis. 2015, 15, 1211–1219. [Google Scholar] [CrossRef]
- Toor, D.; Wsson, M.K.; Kumar, P.; Karthikeyan, G.; Kaushik, N.K.; Goel, C.; Singh, S.; Kumar, A.; Prakash, H. Dysbiosis Disrupts Gut Immune Homeostasis and Promotes Gastric Diseases. Int. J. Mol. Sci. 2019, 20, 2432. [Google Scholar] [CrossRef]
- Chong, P.P.; Chin, V.K.; Looi, C.Y.; Wong, W.F.; Madhavan, P.; Yong, V.C. The Microbiome and Irritable Bowel Syndrome—A Review on the Pathophysiology, Current Research and Future Therapy. Front. Microbiol. 2019, 10, 1136. [Google Scholar] [CrossRef]
- Noce, A.; Marrone, G.; Di Daniele, F.; Ottaviani, E.; Wilson Jones, G.; Bernini, R.; Romani, A.; Rovella, V. Impact of Gut Microbiota Composition on Onset and Progression of Chronic Non-Communicable Diseases. Nutrients 2019, 11, 1073. [Google Scholar] [CrossRef]
- Vaziri, N.D. CKD impairs barrier function and alters microbial flora of the intestine: A major link to inflammation and uremic toxicity. Curr. Opin. Nephrol. Hypertens. 2012, 21, 587–592. [Google Scholar] [CrossRef]
- Vitetta, L.; Gobe, G. Uremia and chronic kidney disease: The role of the gut microflora and therapies with pro- and prebiotics. Mol. Nutr. Food Res. 2013, 57, 824–832. [Google Scholar] [CrossRef] [PubMed]
- King, C.E.; Toskes, P.P. Small intestine bacterial overgrowth. Gastroenterology 1979, 76, 1035–1055. [Google Scholar] [CrossRef]
- Lord, R.S.; Bralley, J.A. Clinical applications of urinary organic acids. Part 2. Dysbiosis markers. Altern. Med. Rev. 2008, 13, 292–306. [Google Scholar] [PubMed]
- Powell-Jackson, P.R.; Maudgal, D.P.; Sharp, D.; Goldie, A.; Maxwell, J.D. Intestinal bacterial metabolism of protein and bile acids: Role in pathogenesis of hepatic disease after jejuno-ileal bypass surgery. Br. J. Surg. 1979, 66, 772–775. [Google Scholar] [CrossRef]
- Simeoni, M.; Citraro, M.L.; Cerantonio, A.; Deodato, F.; Provenzano, M.; Cianfrone, P.; Capria, M.; Corrado, S.; Libri, E.; Comi, A.; et al. An open-label, randomized, placebo-controlled study on the effectiveness of a novel probiotics administration protocol (ProbiotiCKD) in patients with mild renal insufficiency (stage 3a of CKD). Eur. J. Nutr. 2019, 58, 2145–2156. [Google Scholar] [CrossRef]
- Tohyama, K.; Kobayashi, Y.; Kan, T.; Yazawa, K.; Terashima, T.; Mutai, M. Effect of lactobacilli on urinary indican excretion in gnotobiotic rats and in man. Microbiol. Immunol. 1981, 25, 101–112. [Google Scholar] [CrossRef]
- Adike, A.; DiBaise, J.K. Small Intestinal Bacterial Overgrowth: Nutritional Implications, Diagnosis, and Management. Gastroenterol. Clin. North Am. 2018, 47, 193–208. [Google Scholar] [CrossRef]
- Bryan, G.T. Quantitative studies on the urinary excretion of indoxyl sulfate (indican) in man following administration of L-tryptophan and acetyl-L-tryptophan. Am. J. Clin. Nutr. 1966, 19, 105–112. [Google Scholar] [CrossRef]
- Cassani, E.; Barichella, M.; Cancello, R.; Cavanna, F.; Iorio, L.; Cereda, E.; Bolliri, C.; Zampella Maria, P.; Bianchi, F.; Cestaro, B.; et al. Increased urinary indoxyl sulfate (indican): New insights into gut dysbiosis in Parkinson’s disease. Parkinsonism Relat. Disord. 2015, 21, 389–393. [Google Scholar] [CrossRef]
- Conlon, M.A.; Bird, A.R. The impact of diet and lifestyle on gut microbiota and human health. Nutrients 2014, 7, 17–44. [Google Scholar] [CrossRef]
- Hawrelak, J.A.; Myers, S.P. The causes of intestinal dysbiosis: A review. Altern. Med. Rev. 2004, 9, 180–197. [Google Scholar] [PubMed]
- Tidjani Alou, M.; Lagier, J.-C.; Raoult, D. Diet influence on the gut microbiota and dysbiosis related to nutritional disorders. Hum. Microbiome J. 2016, 1, 3–11. [Google Scholar] [CrossRef]
- Yokoyama, M.T.; Carlson, J.R. Microbial metabolites of tryptophan in the intestinal tract with special reference to skatole. Am. J. Clin. Nutr. 1979, 32, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Berni Canani, R.; Paparo, L.; Nocerino, R.; Di Scala, C.; Della Gatta, G.; Maddalena, Y.; Buono, A.; Bruno, C.; Voto, L.; Ercolini, D. Gut Microbiome as Target for Innovative Strategies Against Food Allergy. Front. Immunol. 2019, 10, 191. [Google Scholar] [CrossRef]
- Gagliardi, A.; Totino, V.; Cacciotti, F.; Iebba, V.; Neroni, B.; Bonfiglio, G.; Trancassini, M.; Passariello, C.; Pantanella, F.; Schippa, S. Rebuilding the Gut Microbiota Ecosystem. Int. J. Environ. Res. Public Health 2018, 15, 1679. [Google Scholar] [CrossRef]
- Roager, H.M.; Licht, T.R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 2018, 9, 3294. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Sanders, M.E.; Merenstein, D.; Merrifield, C.A.; Hutkins, R. Probiotics for human use. Nutr. Bull. 2018, 43, 212–225. [Google Scholar] [CrossRef]
- Sharma, G.; Im, S.H. Probiotics as a Potential Immunomodulating Pharmabiotics in Allergic Diseases: Current Status and Future Prospects. Allergy Asthma. Immunol. Res. 2018, 10, 575–590. [Google Scholar] [CrossRef]
- Darkoh, C.; Chappell, C.; Gonzales, C.; Okhuysen, P. A rapid and specific method for the detection of indole in complex biological samples. Appl. Environ. Microbiol. 2015, 81, 8093–8097. [Google Scholar] [CrossRef]
- Rizzi, A.; Nucera, E.; Laterza, L.; Gaetani, E.; Valenza, V.; Corbo, G.M.; Inchingolo, R.; Buonomo, A.; Schiavino, D.; Gasbarrini, A. Irritable Bowel Syndrome and Nickel Allergy: What Is the Role of the Low Nickel Diet? J. Neurogastroenterol. Motil. 2017, 23, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Lusi, E.A.; Di Ciommo, V.M.; Patrissi, T.; Guarascio, P. High prevalence of nickel allergy in an overweight female population: A pilot observational analysis. PLoS ONE 2015, 10, e0123265. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Masieri, S.; Costantini, D.; Tozzi, R.; De Giorgi, F.; Gangitano, E.; Tuccinardi, D.; Poggiogalle, E.; Mariani, S.; Basciani, S.; et al. Overweight and obese patients with nickel allergy have a worse metabolic profile compared to weight matched non-allergic individuals. PLoS ONE 2018, 13, e0202683. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Scarpellini, E.; Colica, C.; Boccuto, L.; Salehi, B.; Sharifi-Rad, J.; Aiello, V.; Romano, B.; De Lorenzo, A.; Izzo, A.A.; et al. Gut Microbiota and Obesity: A Role for Probiotics. Nutrients 2019, 11, 2690. [Google Scholar] [CrossRef]
- Curro, D.; Ianiro, G.; Pecere, S.; Bibbo, S.; Cammarota, G. Probiotics, fibre and herbal medicinal products for functional and inflammatory bowel disorders. Br. J. Pharmacol. 2017, 174, 1426–1449. [Google Scholar] [CrossRef]
- Hiippala, K.; Jouhten, H.; Ronkainen, A.; Hartikainen, A.; Kainulainen, V.; Jalanka, J.; Satokari, R. The Potential of Gut Commensals in Reinforcing Intestinal Barrier Function and Alleviating Inflammation. Nutrients 2018, 10, 988. [Google Scholar] [CrossRef]
- Plaza-Diaz, J.; Ruiz-Ojeda, F.J.; Gil-Campos, M.; Gil, A. Mechanisms of Action of Probiotics. Adv. Nutr. 2019, 10, S49–S66. [Google Scholar] [CrossRef]
- Salem, I.; Ramser, A.; Isham, N.; Ghannoum, M.A. The Gut Microbiome as a Major Regulator of the Gut-Skin Axis. Front. Microbiol. 2018, 9, 1459. [Google Scholar] [CrossRef]
- Cinque, B.; La Torre, C.; Lombardi, F.; Palumbo, P.; Evtoski, Z.; Jr Santini, S.; Falone, S.; Cimini, A.; Amicarelli, F.; Cifone, M.G. VSL#3 probiotic differently influences IEC-6 intestinal epithelial cell status and function. J. Cell. Physiol. 2017, 232, 3530–3539. [Google Scholar] [CrossRef]
- Palumbo, P.; Lombardi, F.; Cifone, M.G.; Cinque, B. The Epithelial Barrier Model Shows That the Properties of VSL#3 Depend from Where it is Manufactured. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 199–206. [Google Scholar] [CrossRef]
- Shu, S.A.; Yuen, A.W.T.; Woo, E.; Chu, K.H.; Kwan, H.S.; Yang, G.X.; Yang, Y.; Leung, P.S.C. Microbiota and Food Allergy. Clin. Rev. Allergy Immunol. 2019, 57, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, P.; Mahallei, M.; Pormohammad, A.; Varshochi, M.; Ganbarov, K.; Zeinalzadeh, E.; Yousefi, B.; Bastami, M.; Tanomand, A.; Mahmood, S.S.; et al. Microbial balance in the intestinal microbiota and its association with diabetes, obesity and allergic disease. Microb. Pathog. 2019, 127, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Atarashi, K.; Tanoue, T.; Shima, T.; Imaoka, A.; Kuwahara, T.; Momose, Y.; Cheng, G.; Yamasaki, S.; Saito, T.; Ohba, Y.; et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 2011, 331, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Berni Canani, R.; De Filippis, F.; Nocerino, R.; Paparo, L.; Di Scala, C.; Cosenza, L.; Della Gatta, G.; Calignano, A.; De Caro, C.; Laiola, M.; et al. Gut microbiota composition and butyrate production in children affected by non-IgE-mediated cow’s milk allergy. Sci. Rep. 2018, 8, 12500. [Google Scholar] [CrossRef] [PubMed]
- Geuking, M.B.; Cahenzli, J.; Lawson, M.A.; Ng, D.C.; Slack, E.; Hapfelmeier, S.; McCoy, K.D.; Macpherson, A.J. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity 2011, 34, 794–806. [Google Scholar] [CrossRef]
- Lathrop, S.K.; Bloom, S.M.; Rao, S.M.; Nutsch, K.; Lio, C.W.; Santacruz, N.; Peterson, D.A.; Stappenbeck, T.S.; Hsieh, C.S. Peripheral education of the immune system by colonic commensal microbiota. Nature 2011, 478, 250–254. [Google Scholar] [CrossRef]
- Mazmanian, S.K.; Round, J.L.; Kasper, D.L. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature 2008, 453, 620–625. [Google Scholar] [CrossRef]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly, Y.M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Stefka, A.T.; Feehley, T.; Tripathi, P.; Qiu, J.; McCoy, K.; Mazmanian, S.K.; Tjota, M.Y.; Seo, G.Y.; Cao, S.; Theriault, B.R.; et al. Commensal bacteria protect against food allergen sensitization. Proc. Natl. Acad. Sci. USA 2014, 111, 13145–13150. [Google Scholar] [CrossRef]
- Biagioli, M.; Laghi, L.; Carino, A.; Cipriani, S.; Distrutti, E.; Marchiano, S.; Parolin, C.; Scarpelli, P.; Vitali, B.; Fiorucci, S. Metabolic Variability of a Multispecies Probiotic Preparation Impacts on the Anti-inflammatory Activity. Front. Pharmacol. 2017, 8, 505. [Google Scholar] [CrossRef]
- Cinque, B.; La Torre, C.; Lombardi, F.; Palumbo, P.; Van der Rest, M.; Cifone, M.G. Production Conditions Affect the In Vitro Anti-Tumoral Effects of a High Concentration Multi-Strain Probiotic Preparation. PLoS ONE 2016, 11, e0163216. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Klaenhammer, T.R.; Ouwehand, A.C.; Pot, B.; Johansen, E.; Heimbach, J.T.; Marco, M.L.; Tennila, J.; Ross, R.P.; Franz, C.; et al. Effects of genetic, processing, or product formulation changes on efficacy and safety of probiotics. Ann. N. Y. Acad. Sci. 2014, 1309, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, V.; Laghi, L.; Vitali, B.; Parolin, C.; Giusti, I.; Capobianco, D.; Mastromarino, P.; De Simone, C. Efficacy and Safety of a Multistrain Probiotic Formulation Depends from Manufacturing. Front. Immunol. 2017, 8, 1474. [Google Scholar] [CrossRef]
- Zacarias, M.F.; Souza, T.C.; Zaburlin, N.; Carmona Cara, D.; Reinheimer, J.; Nicoli, J.; Vinderola, G. Influence of Technological Treatments on the Functionality of Bifidobacterium lactis INL1, a Breast Milk-Derived Probiotic. J. Food Sci. 2017, 82, 2462–2470. [Google Scholar] [CrossRef] [PubMed]
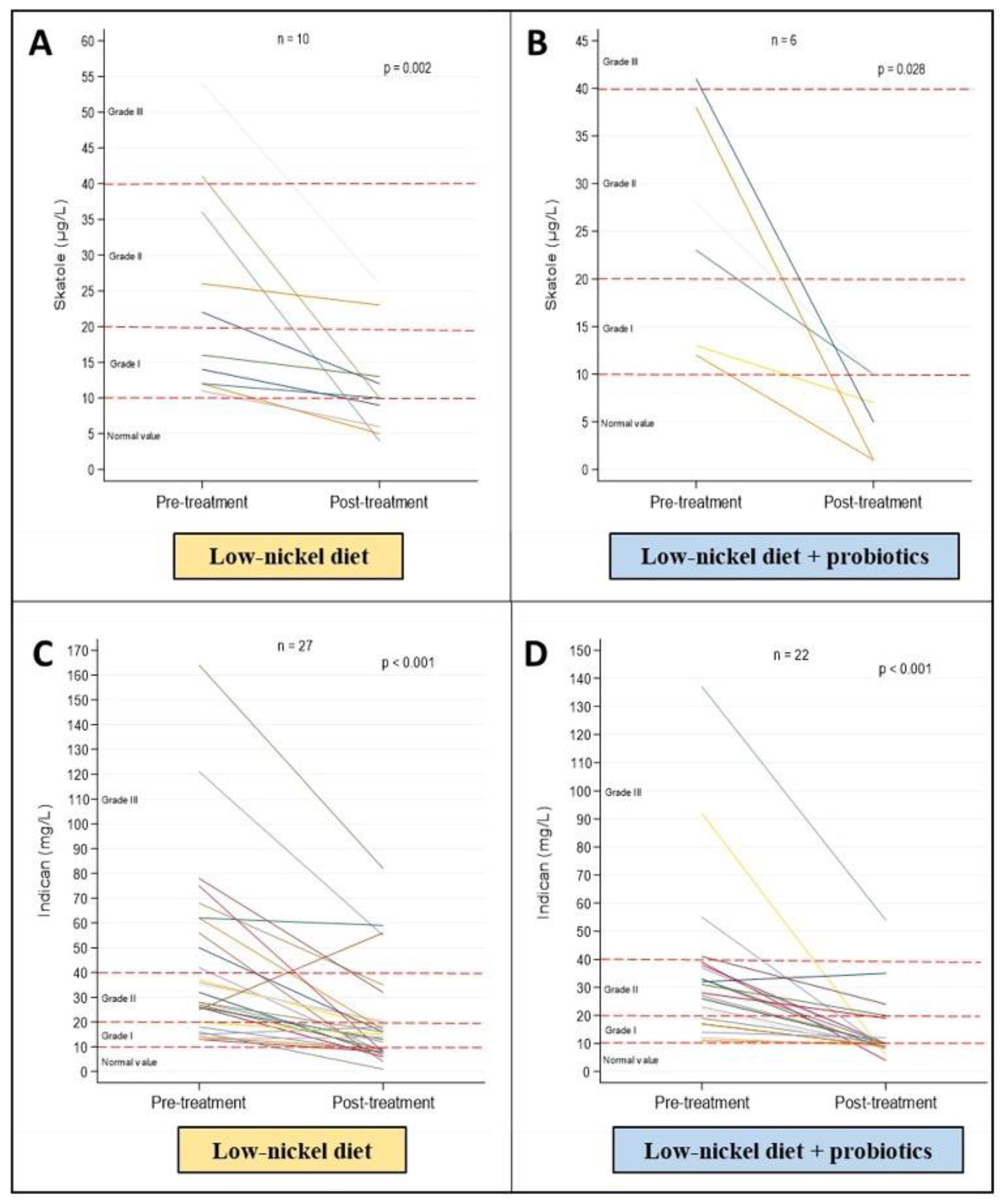
| Skatole |
| 0–10 μg/L (normal value) |
| 11–20 μg/L (grade I dysbiosis) |
| 21–40 μg/L (grade II dysbiosis) |
| >40 μg/L (grade III dysbiosis) |
| Indican |
| 0–10 mg/L (normal value) |
| 11–20 mg/L (grade I dysbiosis) |
| 21–40 mg/L (grade II dysbiosis) |
| >40 mg/L (grade III dysbiosis) |
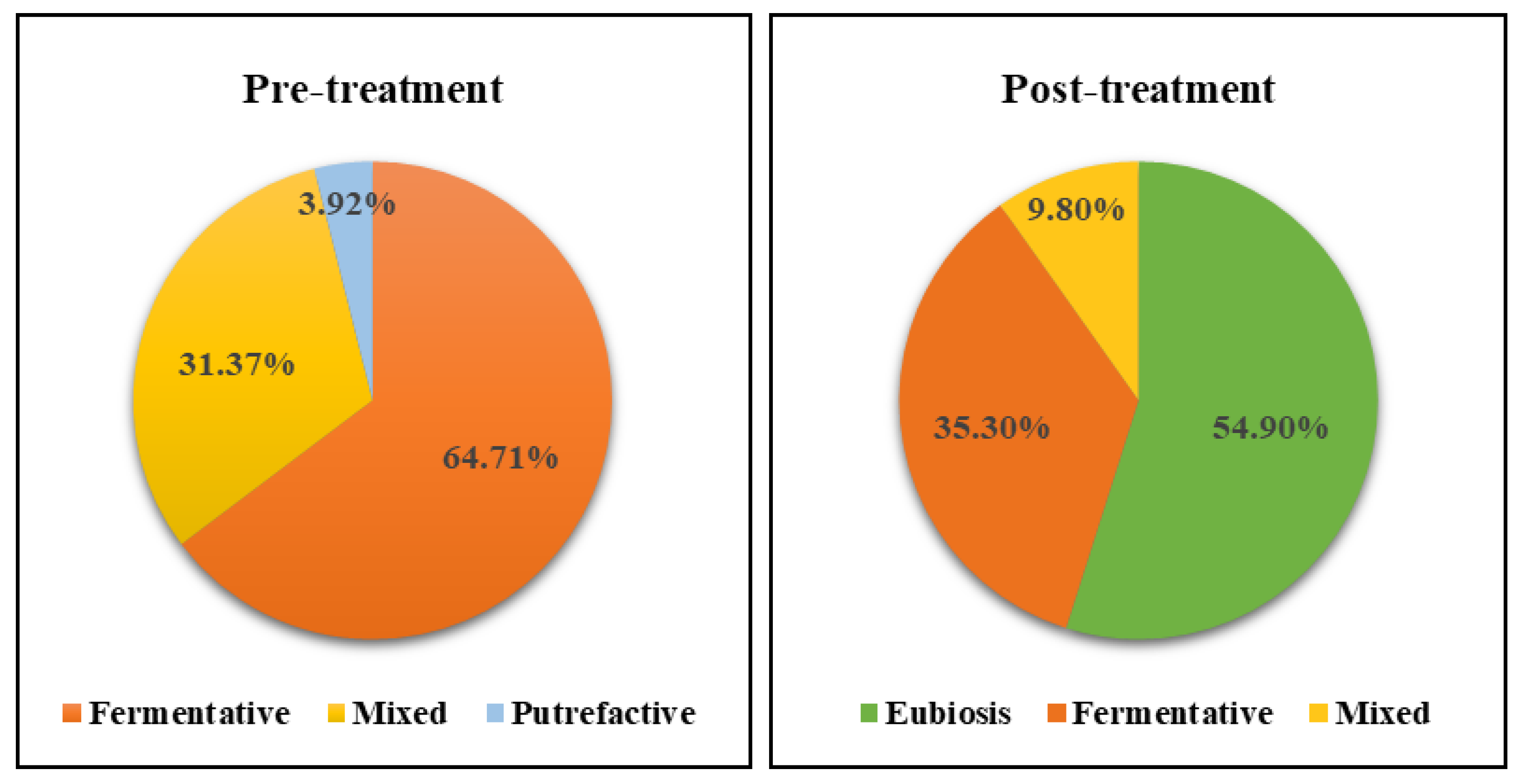
| Total n = 51 | Fermentative n (%) 33 (64.71) | Putrefactive n (%) 2 (3.92) | Mixed n (%) 16 (31.37) | p-Value | |
|---|---|---|---|---|---|
| Age, median (IQR) | 37 (28–49) | 38 (28–51) | 32.5 (29–36) | 34.5 (28.5–46) | 0.689 * |
| Sex, n (%) | 0.799 ** | ||||
| Males | 9 (17.65) | 6 (18.18) | 0 (0.00) | 3 (18.75) | |
| Females | 42 (82.35) | 27 (81.82) | 2 (100) | 13 (81.25) | |
| Ni-OST (mg), median (IQR) | 1.8 (1.5–4) | 1.75 (1.5–4) | 1.5 (1.25–1.75) | 2 (1.5–5) | 0.405 * |
| Patch test, median (IQR) | 2 (1–2) | 2 (1–2) | 2.5 (2–3) | 2 (1–2.5) | 0.472 * |
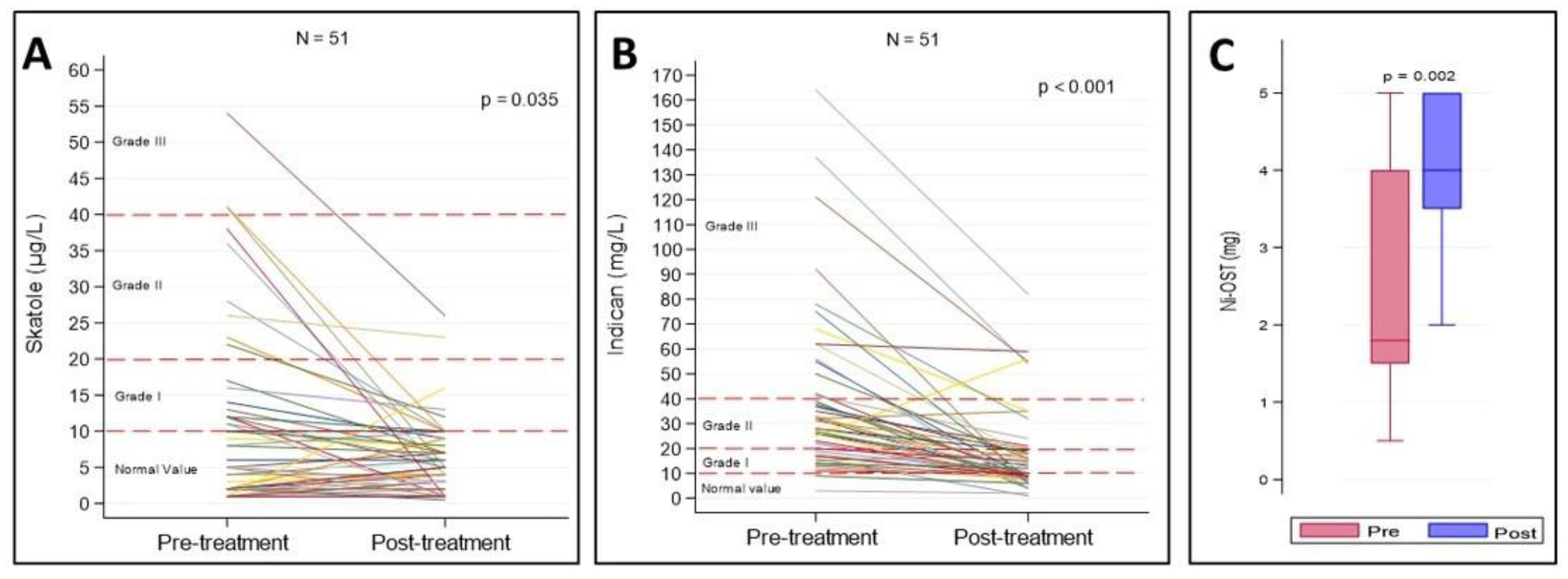
| Skatole (µg/L), median (IQR) | 5 (2–13) | 2 (1–5) | 14 (11–17) | 22.5 (12.5–37) | <0.001 * |
| Indican (mg/L), median (IQR) | 28 (18–42) | 31 (23–42) | 6 (3–9) | 30 (16.5–42.5) | <0.048 * |
| Skatole | n (%) |
| 0–10 μg/L (normal value) | 33 (64.71) |
| 11–20 μg/L (grade I dysbiosis) | 9 (17.65) |
| 21–40 μg/L (grade II dysbiosis) | 6 (11.76) |
| >40 μg/L (grade III dysbiosis) | 3 (5.88) |
| Indican | n (%) |
| 0–10 mg/L (normal value) | 2 (3.92) |
| 11–20 mg/L (grade I dysbiosis) | 13 (25.49) |
| 21–40 mg/L (grade II dysbiosis) | 22 (43.14) |
| >40 mg/L (grade III dysbiosis) | 14 (27.45) |
| Pre-Treatment | Post-Treatment | p-Value * | |
|---|---|---|---|
| Skatole (µg/L), median (IQR) | |||
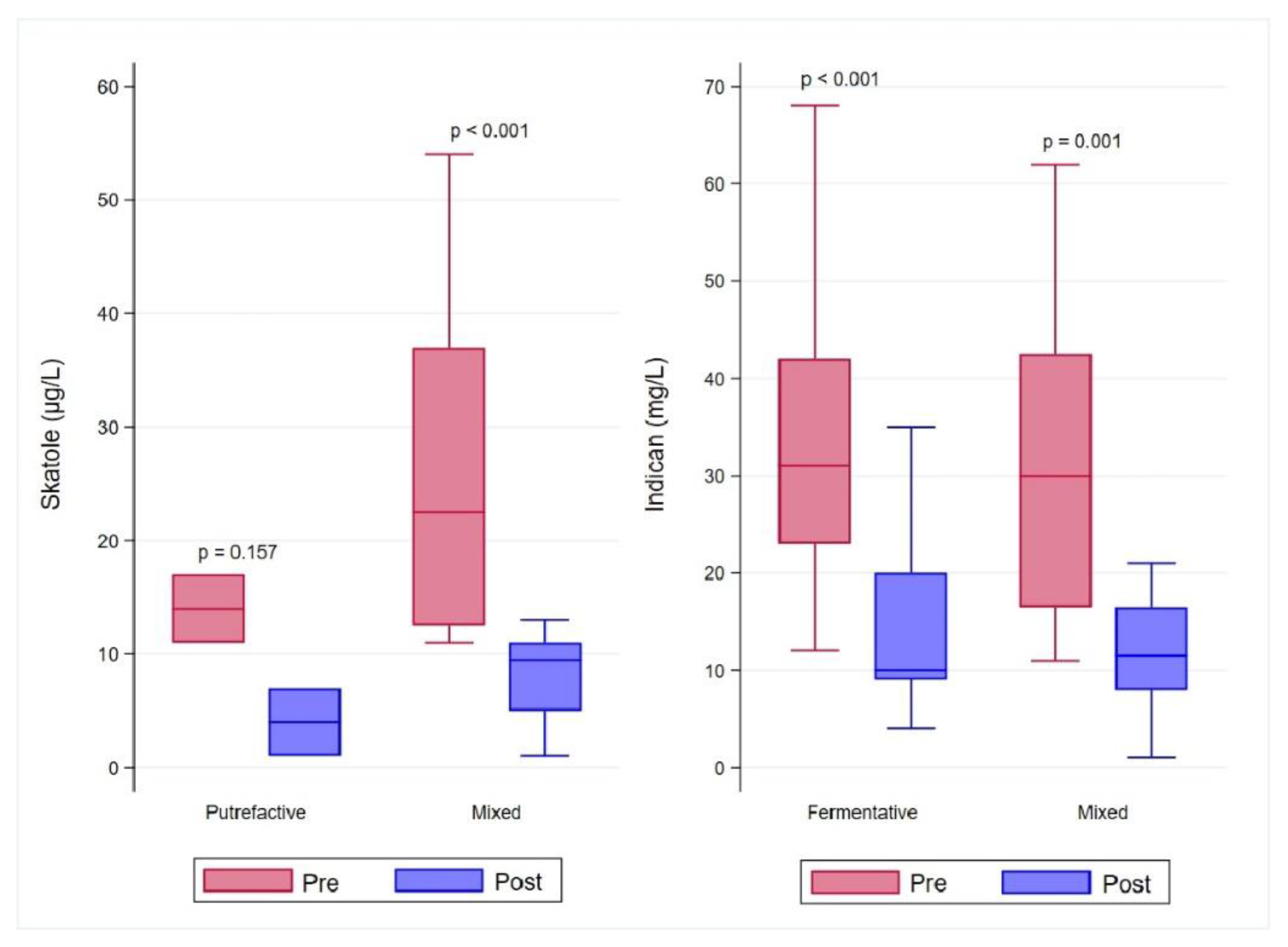
| Putrefactive | 14 (11–17) | 4 (1–7) | 0.157 |
| Mixed | 22.5 (12.5–37) | 9.5 (5–11) | <0.001 |
| Indican (mg/L), median (IQR) | |||
| Fermentative | 31 (23–42) | 10 (9–20) | <0.001 |
| Mixed | 30 (16.5–42.5) | 11.5 (8–16.5) | 0.001 |
| Ni-OST (mg), median (IQR) | 1.8 (1.5–4) | 4 (3.5–5) | 0.002 |
| Groups | Diet | p-Value Pre-Post | Diet + Probiotics | p-Value Pre-Post | p-Value Post-Post | ||
|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | ||||
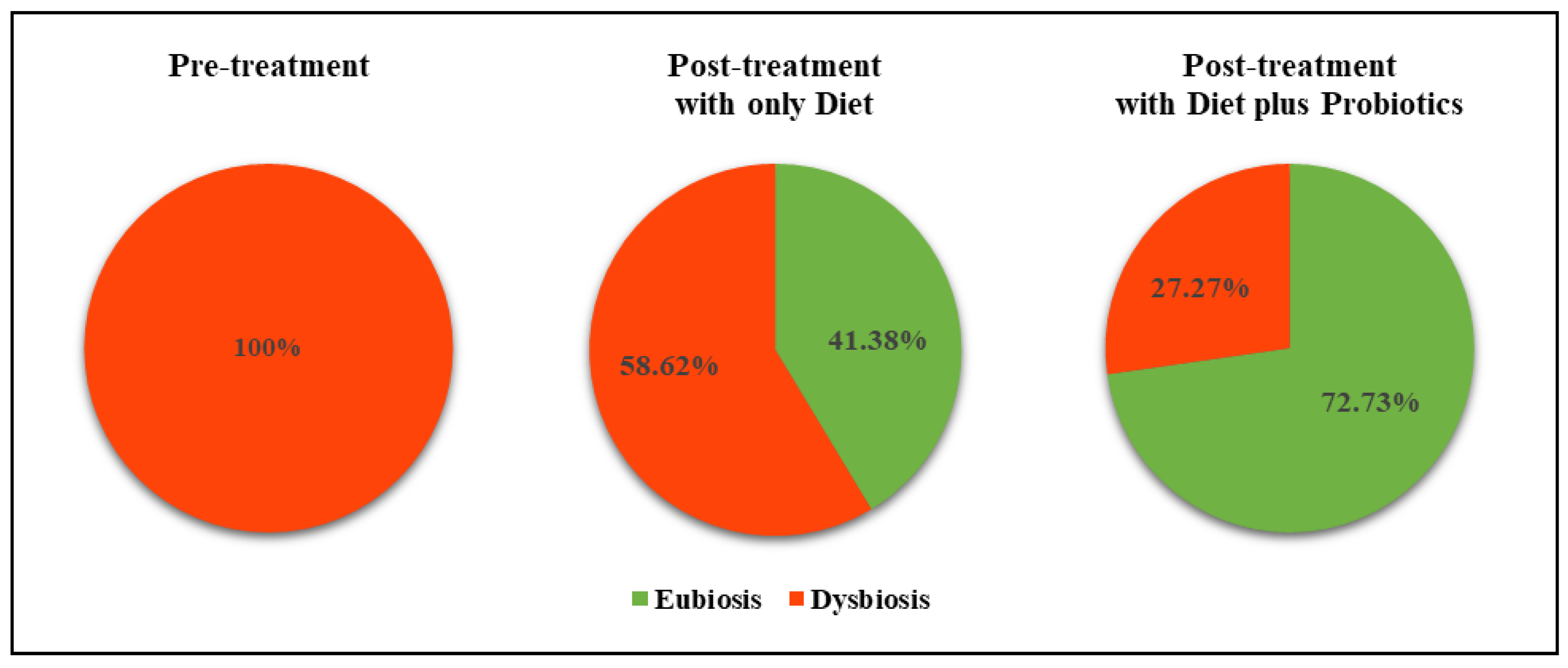
| Shift to eubiosis, n. (%) | 0 (0.00) | 12 (41.38) | <0.001 * | 0 (0.00) | 16 (72.73) | 0.031 * | 0.026 ** |
| Shift to a lower dysbiosis grade, n. (%) | 29 (100.00) | 17 (58.62) | 22 (100.00) | 6 (27.27) | |||
| Fermentative, n. (%) | 17 (58.62) | 12 (41.38) | 0.062 * | 16 (72.72) | 6 (27.27) | 0.002 * | 0.380 ** |
| Mixed, n. (%) | 10 (34.48) | 5 (17.24) | 0.063 * | 6 (27.27) | 0 (0.00) | 0.031 * | |
| Ni-OST (mg), median (IQR) | 1.80 | 4.00 | 0.031 *** | 1.75 | 4.00 | 0.028 *** | 0.765 |
| (1.38–5.00) | (3.00–5.00) | (1.70–3.00) | (3.50–5.00) | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lombardi, F.; Fiasca, F.; Minelli, M.; Maio, D.; Mattei, A.; Vergallo, I.; Cifone, M.G.; Cinque, B.; Minelli, M. The Effects of Low-Nickel Diet Combined with Oral Administration of Selected Probiotics on Patients with Systemic Nickel Allergy Syndrome (SNAS) and Gut Dysbiosis. Nutrients 2020, 12, 1040. https://doi.org/10.3390/nu12041040
Lombardi F, Fiasca F, Minelli M, Maio D, Mattei A, Vergallo I, Cifone MG, Cinque B, Minelli M. The Effects of Low-Nickel Diet Combined with Oral Administration of Selected Probiotics on Patients with Systemic Nickel Allergy Syndrome (SNAS) and Gut Dysbiosis. Nutrients. 2020; 12(4):1040. https://doi.org/10.3390/nu12041040
Chicago/Turabian StyleLombardi, Francesca, Fabiana Fiasca, Martina Minelli, Dominga Maio, Antonella Mattei, Ilaria Vergallo, Maria Grazia Cifone, Benedetta Cinque, and Mauro Minelli. 2020. "The Effects of Low-Nickel Diet Combined with Oral Administration of Selected Probiotics on Patients with Systemic Nickel Allergy Syndrome (SNAS) and Gut Dysbiosis" Nutrients 12, no. 4: 1040. https://doi.org/10.3390/nu12041040
APA StyleLombardi, F., Fiasca, F., Minelli, M., Maio, D., Mattei, A., Vergallo, I., Cifone, M. G., Cinque, B., & Minelli, M. (2020). The Effects of Low-Nickel Diet Combined with Oral Administration of Selected Probiotics on Patients with Systemic Nickel Allergy Syndrome (SNAS) and Gut Dysbiosis. Nutrients, 12(4), 1040. https://doi.org/10.3390/nu12041040











