Presence of Hypertension Is Reduced by Mediterranean Diet Adherence in All Individuals with a More Pronounced Effect in the Obese: The Hellenic National Nutrition and Health Survey (HNNHS)
Abstract
1. Introduction
2. Materials & Methods
2.1. Study Design
2.2. Parameters Investigated
2.2.1. Sociodemographic Data
2.2.2. Lifestyle & Anthropometric Factors
2.2.3. Blood Pressure Status and Measurement
2.2.4. Additional Parameters Measured
2.3. Data Analysis
2.4. Statistical Methodology
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability Statement
References
- WHO Organization. A Global Brief on Hypertension: Silent Killer, Global Public Health Crisis. Available online: http://ish-world.com/downloads/pdf/global_brief_hypertension.pdf (accessed on 28 August 2018).
- Global Burden of Metabolic Risk Factors for Chronic Diseases Collaboration. Cardiovascular disease, chronic kidney disease, and diabetes mortality burden of cardiometabolic risk factors from 1980 to 2010: A comparative risk assessment. Lancet Diabetes Endocrinol. 2014, 2, 634–647. [Google Scholar] [CrossRef]
- Danaei, G.; Finucane, M.M.; Lin, J.K.; Singh, G.M.; Paciorek, C.J.; Cowan, M.J.; Farzadfar, F.; Stevens, G.A.; Lim, S.S.; Riley, L.M.; et al. National, regional, and global trends in systolic blood pressure since 1980: Systematic analysis of health examination surveys and epidemiological studies with 786 country-years and 5.4 million participants. Lancet 2011, 377, 568–577. [Google Scholar] [CrossRef]
- Collaboration, N.R.F. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19.1 million participants. Lancet 2017, 389, 37–55. [Google Scholar]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.-T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 european guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Millett, C. Social epidemiology of hypertension in middle-income countries: Determinants of prevalence, diagnosis, treatment, and control in the who sage study. Hypertension 2013, 62, 18–26. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart disease and stroke statistics—2018 update: A report from the american heart association. Circulation 2018, 137. [Google Scholar] [CrossRef]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Covas, M.I.; Fiol, M.; Gomez-Gracia, E.; Lopez-Sabater, M.C.; Vinyoles, E.; et al. Effects of a mediterranean-style diet on cardiovascular risk factors: A randomized trial. Ann. Intern Med. 2006, 145, 1–11. [Google Scholar] [CrossRef]
- Michas, G.; Karvelas, G.; Trikas, A. Cardiovascular disease in greece; the latest evidence on risk factors. Hell. J. Cardiol. 2018, 60, 271–275. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.H.; Chrysohoou, C.; Skoumas, J.; Papadimitriou, L.; Stefanadis, C.; Toutouzas, P.K. Status and management of hypertension in greece: Role of the adoption of a mediterranean diet: The attica study. J. Hypertens. 2003, 21, 1483–1489. [Google Scholar] [CrossRef]
- Magriplis, E.; Dimakopoulos, I.; Karageorgou, D.; Mitsopoulou, A.V.; Bakogianni, I.; Micha, R.; Michas, G.; Ntouroupi, T.; Tsaniklidou, S.M.; Argyri, K.; et al. Aims, design and preliminary findings of the hellenic national nutrition and health survey (hnnhs). BMC Med. Res. Methodol. 2019, 19, 37. [Google Scholar] [CrossRef]
- Questionnaire, I.P.A. Guidelines for Data Processing and Analysis of the International Physical Activity Questionnaire (IPAQ). Revised on Novemeber 2005. Available online: http://www.ipaq.ki.se/scoring.pdf (accessed on 20 December 2019).
- European Society of Hypertension-European and the Society of Cardiology Guidelines Committee. 2003 european society of hypertension-european society of cardiology guidelines for the management of arterial hypertension. J. Hypertens. 2003, 21, 1011–1053. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2018, 71, e127–e248. [Google Scholar]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Magriplis, E.; Panagiotakos, D.; Tsioufis, C.; Mitsopoulou, A.V.; Karageorgou, D.; Dimakopoulos, I.; Bakogianni, I.; Chourdakis, M.; Micha, R.; Michas, G.; et al. Hypertension prevalence in a representative sample of Greek adults: The Hellenic National Nutrition and Health Survey (HNNHS). Hell. J. Cardiol. 2019. [Google Scholar] [CrossRef]
- Pitsavos, C.; Milias, G.A.; Panagiotakos, D.B.; Xenaki, D.; Panagopoulos, G.; Stefanadis, C. Prevalence of self-reported hypertension and its relation to dietary habits, in adults; a nutrition & health survey in greece. BMC Public Health 2006, 6, 206. [Google Scholar]
- Efstratopoulos, A.D.; Voyaki, S.M.; Baltas, A.A.; Vratsistas, F.A.; Kirlas, D.E.; Kontoyannis, J.T.; Sakellariou, J.G.; Triantaphyllou, G.B.; Alokrios, G.A.; Lianas, D.N.; et al. Prevalence, awareness, treatment and control of hypertension in hellas, greece: The hypertension study in general practice in hellas (hypertenshell) national study. Am. J. Hypertens. 2006, 19, 53–60. [Google Scholar] [CrossRef]
- Wolf-Maier, K.; Cooper, R.S.; Banegas, J.R.; Giampaoli, S.; Hense, H.W.; Joffres, M.; Kastarinen, M.; Poulter, N.; Primatesta, P.; Rodriguez-Artalejo, F.; et al. Hypertension prevalence and blood pressure levels in 6 european countries, canada, and the united states. JAMA 2003, 289, 2363–2369. [Google Scholar] [CrossRef]
- Asmar, R.; Vol, S.; Pannier, B.; Brisac, A.M.; Tichet, J.; El Hasnaoui, A. High blood pressure and associated cardiovascular risk factors in france. J. Hypertens. 2001, 19, 1727–1732. [Google Scholar] [CrossRef]
- Wong, N.D.; Franklin, S.S. Epidemiology of hypertension. J. Am. Soc. Hypertens. 2014, 8, 760–763. [Google Scholar] [CrossRef]
- Di Angelantonio, E.; Bhupathiraju, S.N.; Wormser, D.; Gao, P.; Kaptoge, S.; De Gonzalez, A.B.; Cairns, B.J.; Huxley, R.; Jackson, C.L.; Joshy, G.; et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016, 388, 776–786. [Google Scholar] [CrossRef]
- Hall, J.E.; Do Carmo, J.M.; Da Silva, A.A.; Wang, Z.; Hall, M.E. Obesity-induced hypertension: Interaction of neurohumoral and renal mechanisms. Circ. Res. 2015, 116, 991–1006. [Google Scholar] [CrossRef] [PubMed]
- Neter, J.E.; Stam, B.E.; Kok, F.J.; Grobbee, D.E.; Geleijnse, J.M. Influence of weight reduction on blood pressure: A meta-analysis of randomized controlled trials. Hypertension 2003, 42, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Gay, H.C.; Rao, S.G.; Vaccarino, V.; Ali, M.K. Effects of different dietary interventions on blood pressure: Systematic review and meta-analysis of randomized controlled trials. Hypertension 2016, 67, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Ravera, A.; Carubelli, V.; Sciatti, E.; Bonadei, I.; Gorga, E.; Cani, D.; Vizzardi, E.; Metra, M.; Lombardi, C. Nutrition and cardiovascular disease: Finding the perfect recipe for cardiovascular health. Nutrients 2016, 8, 363. [Google Scholar] [CrossRef] [PubMed]
- Carlos, S.; De La Fuente-Arrillaga, C.; Bes-Rastrollo, M.; Razquin, C.; Rico-Campa, A.; Martinez-Gonzalez, M.A.; Ruiz-Canela, M. Mediterranean diet and health outcomes in the sun cohort. Nutrients 2018, 10, 439. [Google Scholar] [CrossRef]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The mediterranean diet, its components, and cardiovascular disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef]
- Rees, K.; Hartley, L.; Clarke, A.; Thorogood, M.; Stranges, S. ‘Mediterranean’ dietary pattern for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2012, 2012, CD009825. [Google Scholar]
- Wilkins, E.W.L.; Wickramasinghe, K.; Bhatnagar, P.; Leal, J.; Luengo-Fernandez, R.; Burns, R.; Rayner, M.; Townsend, N. European Cardiovascular Disease Statistics; European Heart Network: Brussels, Belgium, 2017. [Google Scholar]
- Mancia, G.; Facchetti, R.; Bombelli, M.; Friz, H.P.; Grassi, G.; Giannattasio, C.; Sega, R. Relationship of office, home, and ambulatory blood pressure to blood glucose and lipid variables in the pamela population. Hypertension 2005, 45, 1072–1077. [Google Scholar] [CrossRef]
- Burt, V.L.; Whelton, P.; Roccella, E.J.; Brown, C.; Cutler, J.A.; Higgins, M.; Horan, M.J.; Labarthe, D. Prevalence of hypertension in the us adult population. Results from the third national health and nutrition examination survey, 1988–1991. Hypertension 1995, 25, 305–313. [Google Scholar] [CrossRef]
- Sofi, F.; Capalbo, A.; Cesari, F.; Abbate, R.; Gensini, G.F. Physical activity during leisure time and primary prevention of coronary heart disease: An updated meta-analysis of cohort studies. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 247–257. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E. The lancet commission on hypertension: Addressing the global burden of raised blood pressure on current and future generations. J. Clin. Hypertens. (Greenwich Conn.) 2017, 19, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., 3rd; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the dietary approaches to stop hypertension (dash) diet. Dash-sodium collaborative research group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- O’Halloran, S.A.; Lacy, K.E.; Grimes, C.A.; Woods, J.; Campbell, K.J.; Nowson, C.A. A novel processed food classification system applied to australian food composition databases. J. Hum. Nutr. Diet. 2017, 30, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Bibbins-Domingo, K.; Chertow, G.M.; Coxson, P.G.; Moran, A.; Lightwood, J.M.; Pletcher, M.J.; Goldman, L. Projected effect of dietary salt reductions on future cardiovascular disease. N. Engl. J. Med. 2010, 362, 590–599. [Google Scholar] [CrossRef]
- Powles, J.; Fahimi, S.; Micha, R.; Khatibzadeh, S.; Shi, P.; Ezzati, M.; Engell, R.E.; Lim, S.S.; Danaei, G.; Mozaffarian, D. Global, regional and national sodium intakes in 1990 and 2010: A systematic analysis of 24 h urinary sodium excretion and dietary surveys worldwide. BMJ Open 2013, 3, e003733. [Google Scholar] [CrossRef]
- Hasenegger, V.; Rust, P.; König, J.; Purtscher, A.; Erler, J.; Ekmekcioglu, C. Main sources, socio-demographic and anthropometric correlates of salt intake in austria. Nutrients 2018, 10, 311. [Google Scholar] [CrossRef]
- Crim, M.T.; Yoon, S.S.; Ortiz, E.; Wall, H.K.; Schober, S.; Gillespie, C.; Sorlie, P.; Keenan, N.; Labarthe, D.; Hong, Y. National surveillance definitions for hypertension prevalence and control among adults. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 343–351. [Google Scholar] [CrossRef]
- Domenech, M.; Roman, P.; Lapetra, J.; de la Garcia Corte, F.J.; Sala-Vila, A.; de la Torre, R.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Lamuela-Raventos, R.M.; et al. Mediterranean diet reduces 24-hour ambulatory blood pressure, blood glucose, and lipids: One-year randomized, clinical trial. Hypertension 2014, 64, 69–76. [Google Scholar] [CrossRef]
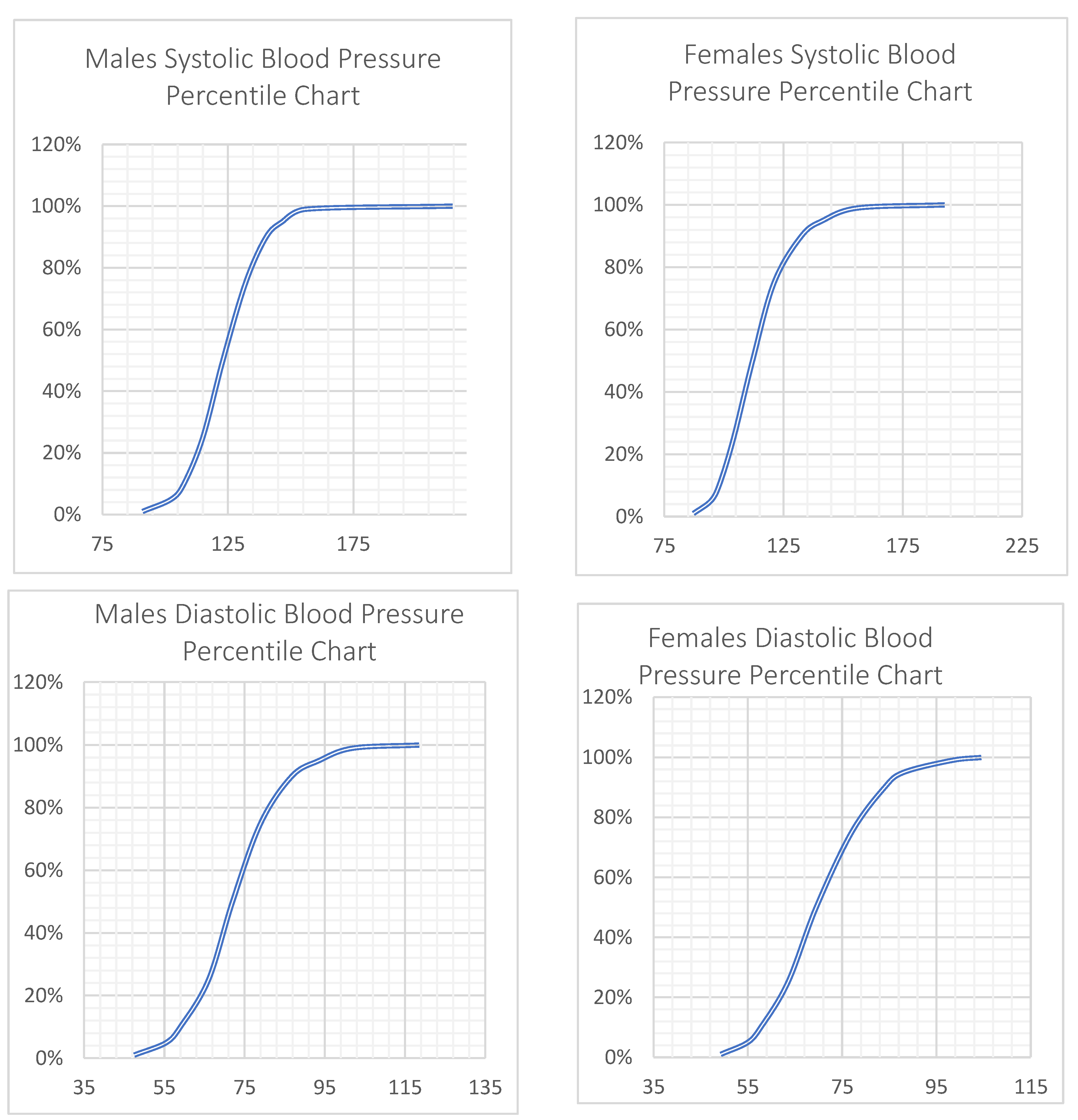
| Total (N = 3775) | Males (N = 1541) | Females (N = 2234) | p Value for Sex Differences | |
|---|---|---|---|---|
| Age (years), mean (sd) | 43.6 (18.7) | 43.1 (18,4) | 44.0 (18.8) | <0.001 |
| Weight (kg), mean (sd) | 73.2 (15.5) | 82.8 (13.6) | 66.7 (13.2) | <0.001 |
| Weight Status, n (%) | ||||
| Healthy Weight | 1906 (52.3) | 625 (41.8) | 1281 (59.7) | <0.001 |
| Overweight | 1159 (31.8) | 623 (41.6) | 536 (25.0) | |
| Obese | 577 (15.8) | 248 (16.6) | 329 (15.3) | |
| MedDiet Score, mean (sd) | 28.7 (6.5) | 28.1 (6.7) | 29.0 (6.3) | <0.001 |
| MedDiet Score, status, n (%) | 0.004 | |||
| 0–22 | 616 (17.1) | 286 (19.5) | 330 (15.5) | |
| 23–34 | 2219 (61.5) | 896 (61.0) | 1323 (62.2) | |
| 35–55 | 771 (21.4 | 288 (19.6) | 473 (22.3) | |
| Educational Status (level), n (%) | <0.001 | |||
| Low | 468 (12.4) | 153 (9.9) | 315 (14.1) | |
| Medium | 1377 (36.6) | 626 (40.7) | 751 (33.7) | |
| High | 1921 (51.0) | 758 (49.3) | 1163 (52.2) | |
| Smoking status, n (%) | <0.001 | |||
| Non-smokers | 2510 (66.5) | 959 (62.2) | 1551 (69.4) | |
| Smoker (all) | 1265 (33.5) | 582 (37.8) | 683 (30.6) | |
| Physical activity status, n (%) | <0.001 | |||
| Sedentary | 286 (7.8) | 136 (9.0) | 150 (6.9) | |
| Light | 515 (14.0) | 233 (15.4) | 282 (13.0) | |
| Moderate | 1415 (38.5) | 527 (34.8) | 888 (41.0) | |
| Active | 1463 (39.8) | 617 (40.8) | 846 (39.1) | |
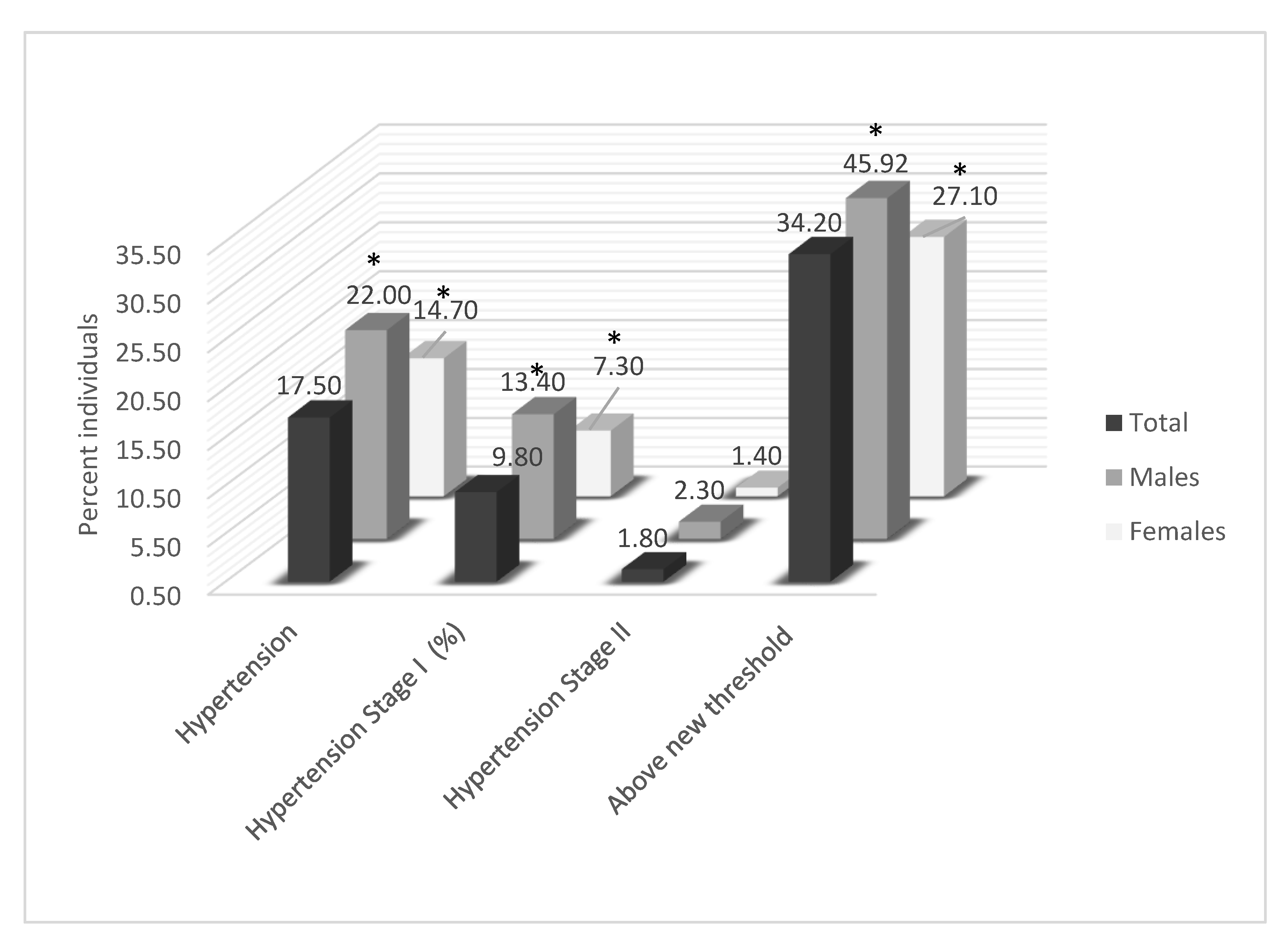
| Hypertension, %(SE) | 16.6 (0.01) | 16.2 (0.01) | 16.9 (0.01) | 0.521 |
| Other comorbidities, %(SE) | ||||
| Dyslipidemia * | 20.2 (0.01) | 19.3 (0.01) | 20.9 (0.01) | 0.208 |
| Diabetes * | 4.4 (0.0) | 4.8 (0.01) | 4.1 (0.00) | 0.336 |
| Age Group | Age Group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | 20–39 | 40–59 | 60+ | 20–39 | 40–59 | 60+ | |||
| Males | p-Value 1 | Females | p-Value 1 | ||||||
| Mean SBP, mean (SD) | 118.6 (15.3) | 124.8 (10.6) | 126.9 (13.9) | 128.9 (19.6) | 0.082 * | 109.3 (10.2) a,c | 116.8 (15.8) a,b | 126.2 (19.1) b,c | <0.001 * |
| Mean DBP, mean (SE) | 72.2 (10.6) | 72.3 (10.1) a | 78.5 (10.8) a | 76.1 (11.1) | <0.001 * | 68.6 (9.7) a,c | 73.4 (10.8) a | 72.2 (10.3) c | <0.001 * |
| Total measured Hypertension 2, % | 17.5 | 10.5 | 30.6 | 54.6 | <0.001 * | 2.5 | 15.2 | 55.8 | <0.001 * |
| Odds Ratio (SE) * | 95% Confidence Interval | Odds Ratio (SE) * | 95% Confidence Interval | |
|---|---|---|---|---|
| Simple Logistic Regression 1 | Final Model 2 | |||
| Age (per 1 year) | 1.1 (0.01) | 1.089, 1.107 | 1.1 (0.01) | 1.072, 1.102 |
| Females vs. Males | 1.1 (0.13) | 0.850, 1.236 | 0.92 (0.13) | 0.694, 1.213 |
| Smokers vs. non-smokers | 1.02 (0.06) | 0.894, 1.164 | 1.04 (0.08) | 0.892, 1.205 |
| Educational level | ||||
| Up to 6 years | base | - | base | |
| 7–12 years | 0.15 (0.02) | 0.111, 0.190 | 0.92 (0.90) | 0.756, 1.112 |
| >12 years | 0.09 (0.01) | 0.071, 0.122 | 1.01 (0.15) | 0.762, 1.340 |
| Weight Status | ||||
| Healthy weight | base | - | base | - |
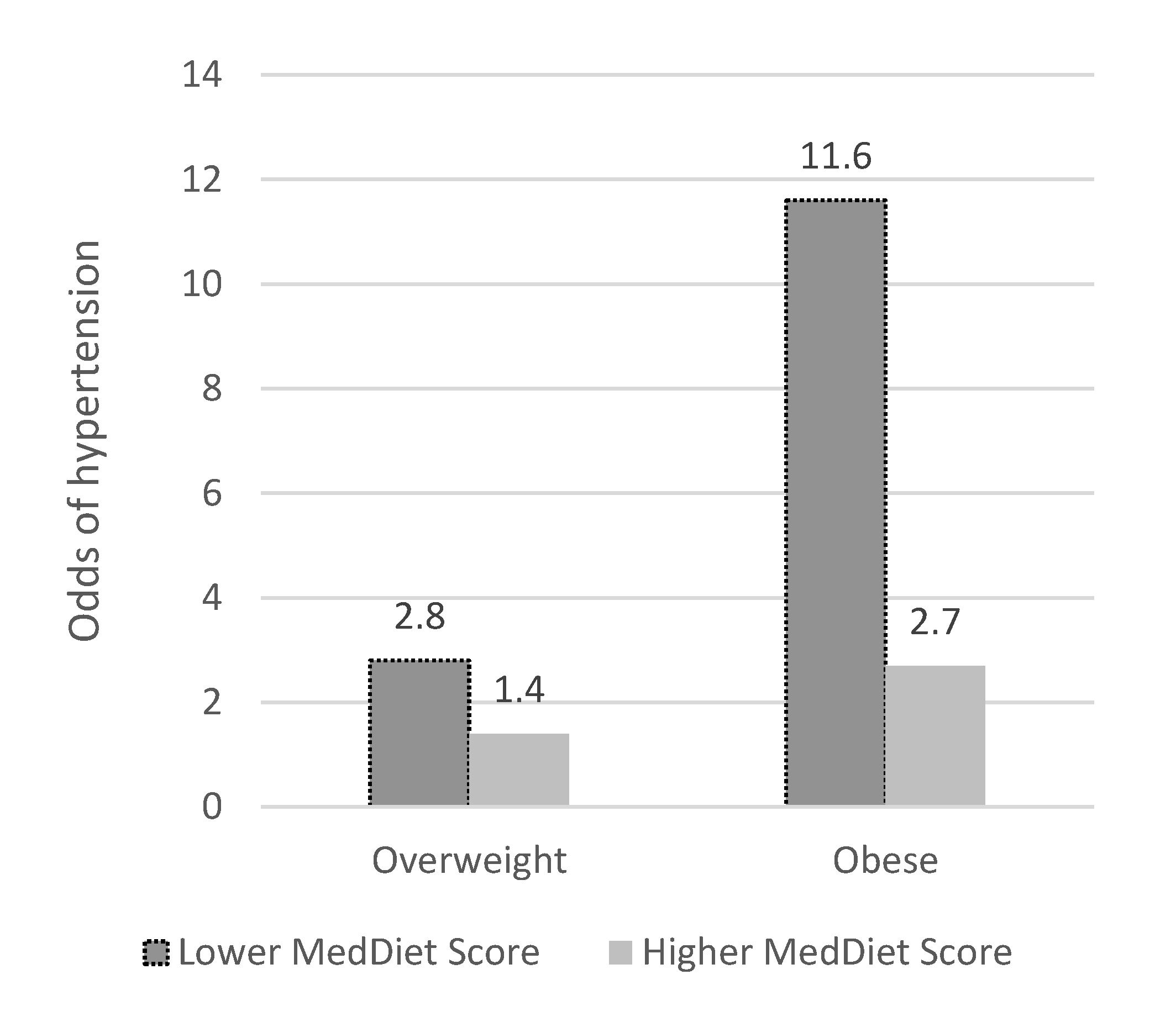
| Overweight | 3.60 (0.48) | 2.770, 4.677 | 1.89 (0.33) | 1.342, 2.679 |
| Obese | 7.06 (1.03) | 5.305, 9.387 | 3.79 (0.71) | 2.264, 5.481 |
| MedDiet score (≥23 compared to <23) | 1.25 (0.19) | 0.930, 1.681 | 0.64 (0.13) | 0.439, 0.943 |
| Weight status × MedDiet Score (>23 compared to <23) 3 | 0.51 (0.20) | 0.235, 1.096 | 0.40 (0.17) | 0.170, 0.937 |
| Employment status | ||||
| Employed | base | - | base | |
| Unemployed | 1.01 (0.19) | 0.703, 1.461 | 0.98 (0.20) | 0.655, 1.463 |
| Pension | 1.10 (0.22) | 0.746, 1.634 | 1.11 (0.24) | 0.723, 1.709 |
| Physical activity level | ||||
| Sedentary | base | - | base | - |
| Low | 0.43 (0.09) | 0.286, 0.645 | 1.03 (0.32) | 0.560, 1.911 |
| Medium | 0.44 (0.08) | 0.314, 0.623 | 0.93 (0.26) | 0.540, 1.607 |
| High | 0.37 (0.07) | 0.258, 0.526 | 1.01 (0.28) | 0.586, 1.745 |
| Sodium intake (>1500 mg/day vs. <1500 mg/day) | 1.04 (0.14) | 0.802, 1.355 | 1.01 (0.15) | 0.762, 1.340 |
| Hyperlipidemia (presence vs. non-presence) | 5.33 (0.58) | 4.318, 6.582 | 2.30 (0.33) | 1.729, 3.055 |
| Diabetes (presence vs. non-presence) | 7.0 (1.26) | 4.887, 9.902 | 1.78 (0.47) | 1.057, 2.004 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magriplis, E.; Panagiotakos, D.; Kyrou, I.; Tsioufis, C.; Mitsopoulou, A.-V.; Karageorgou, D.; Dimakopoulos, I.; Bakogianni, I.; Chourdakis, M.; Micha, R.; et al. Presence of Hypertension Is Reduced by Mediterranean Diet Adherence in All Individuals with a More Pronounced Effect in the Obese: The Hellenic National Nutrition and Health Survey (HNNHS). Nutrients 2020, 12, 853. https://doi.org/10.3390/nu12030853
Magriplis E, Panagiotakos D, Kyrou I, Tsioufis C, Mitsopoulou A-V, Karageorgou D, Dimakopoulos I, Bakogianni I, Chourdakis M, Micha R, et al. Presence of Hypertension Is Reduced by Mediterranean Diet Adherence in All Individuals with a More Pronounced Effect in the Obese: The Hellenic National Nutrition and Health Survey (HNNHS). Nutrients. 2020; 12(3):853. https://doi.org/10.3390/nu12030853
Chicago/Turabian StyleMagriplis, Emmanuella, Demosthenes Panagiotakos, Ioannis Kyrou, Costas Tsioufis, Anastasia-Vasiliki Mitsopoulou, Dimitra Karageorgou, Ioannis Dimakopoulos, Ioanna Bakogianni, Michalis Chourdakis, Renata Micha, and et al. 2020. "Presence of Hypertension Is Reduced by Mediterranean Diet Adherence in All Individuals with a More Pronounced Effect in the Obese: The Hellenic National Nutrition and Health Survey (HNNHS)" Nutrients 12, no. 3: 853. https://doi.org/10.3390/nu12030853
APA StyleMagriplis, E., Panagiotakos, D., Kyrou, I., Tsioufis, C., Mitsopoulou, A.-V., Karageorgou, D., Dimakopoulos, I., Bakogianni, I., Chourdakis, M., Micha, R., Michas, G., Ntouroupi, T., Tsaniklidou, S.-M., Argyri, K., & Zampelas, A. (2020). Presence of Hypertension Is Reduced by Mediterranean Diet Adherence in All Individuals with a More Pronounced Effect in the Obese: The Hellenic National Nutrition and Health Survey (HNNHS). Nutrients, 12(3), 853. https://doi.org/10.3390/nu12030853







