Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Interventions
2.3. Data Collection
2.4. Efficacy Measures
2.5. Adverse Events
2.6. Correlations
2.7. Statistical Analysis
3. Results
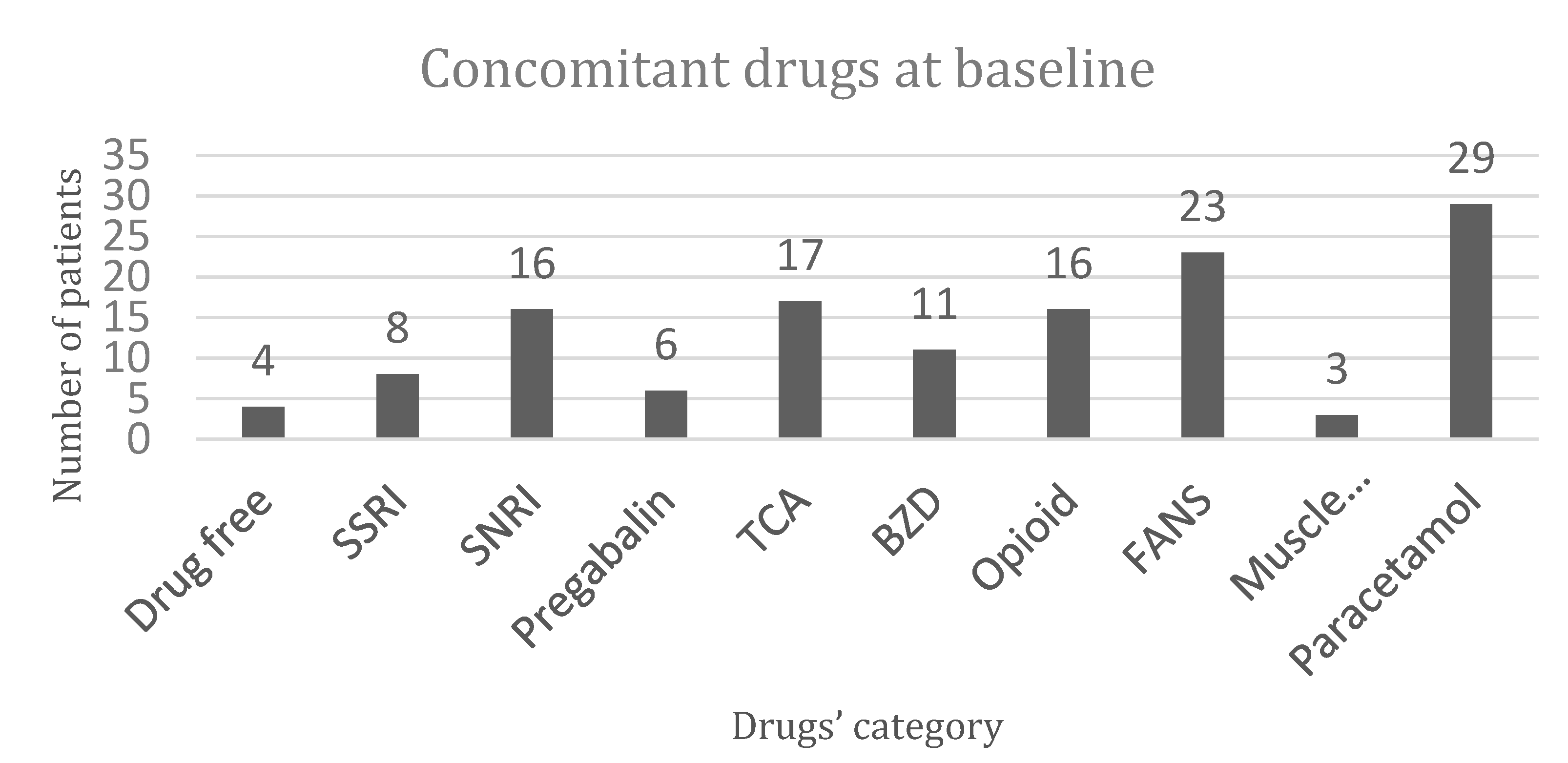
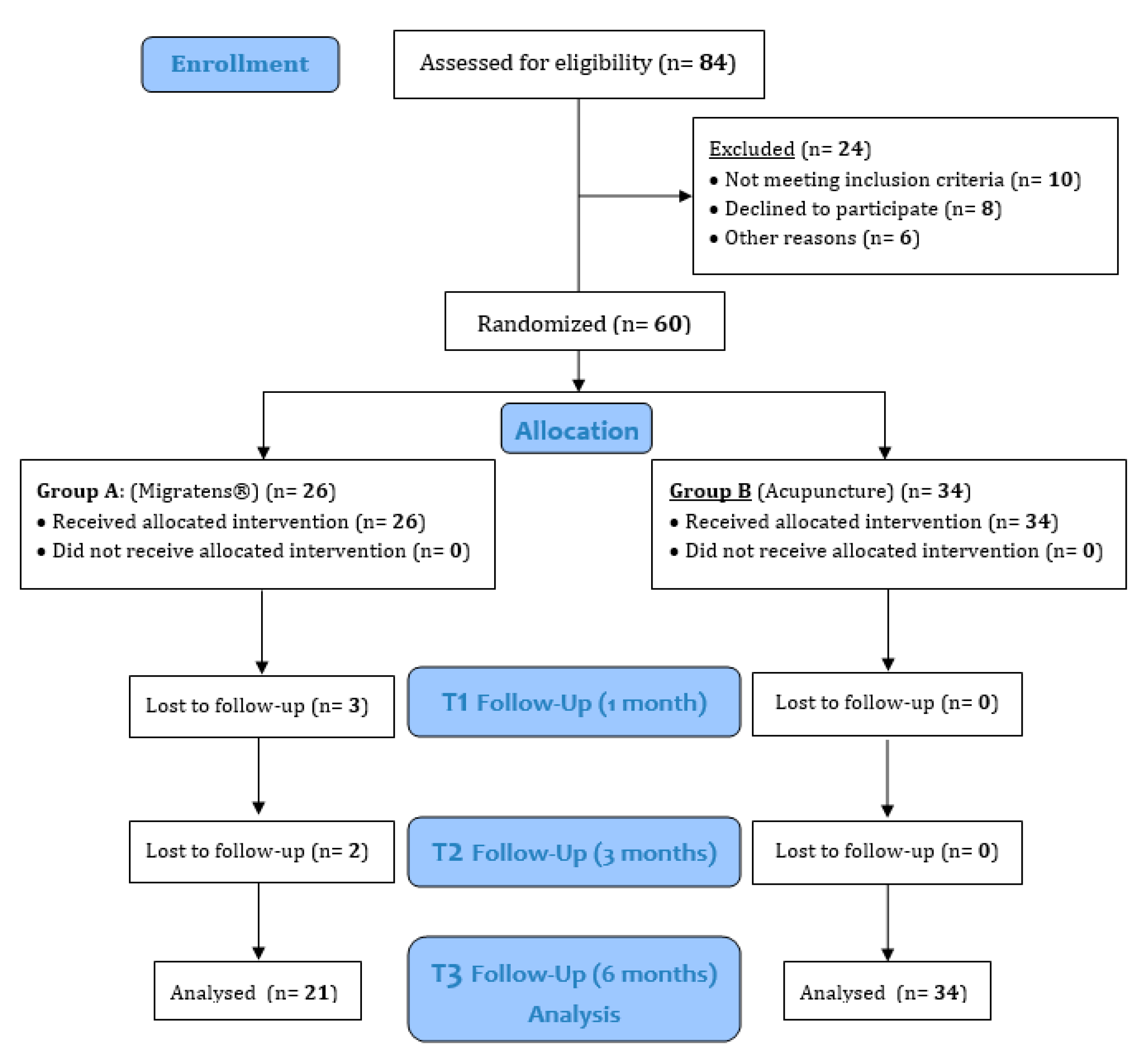
3.1. Study Population
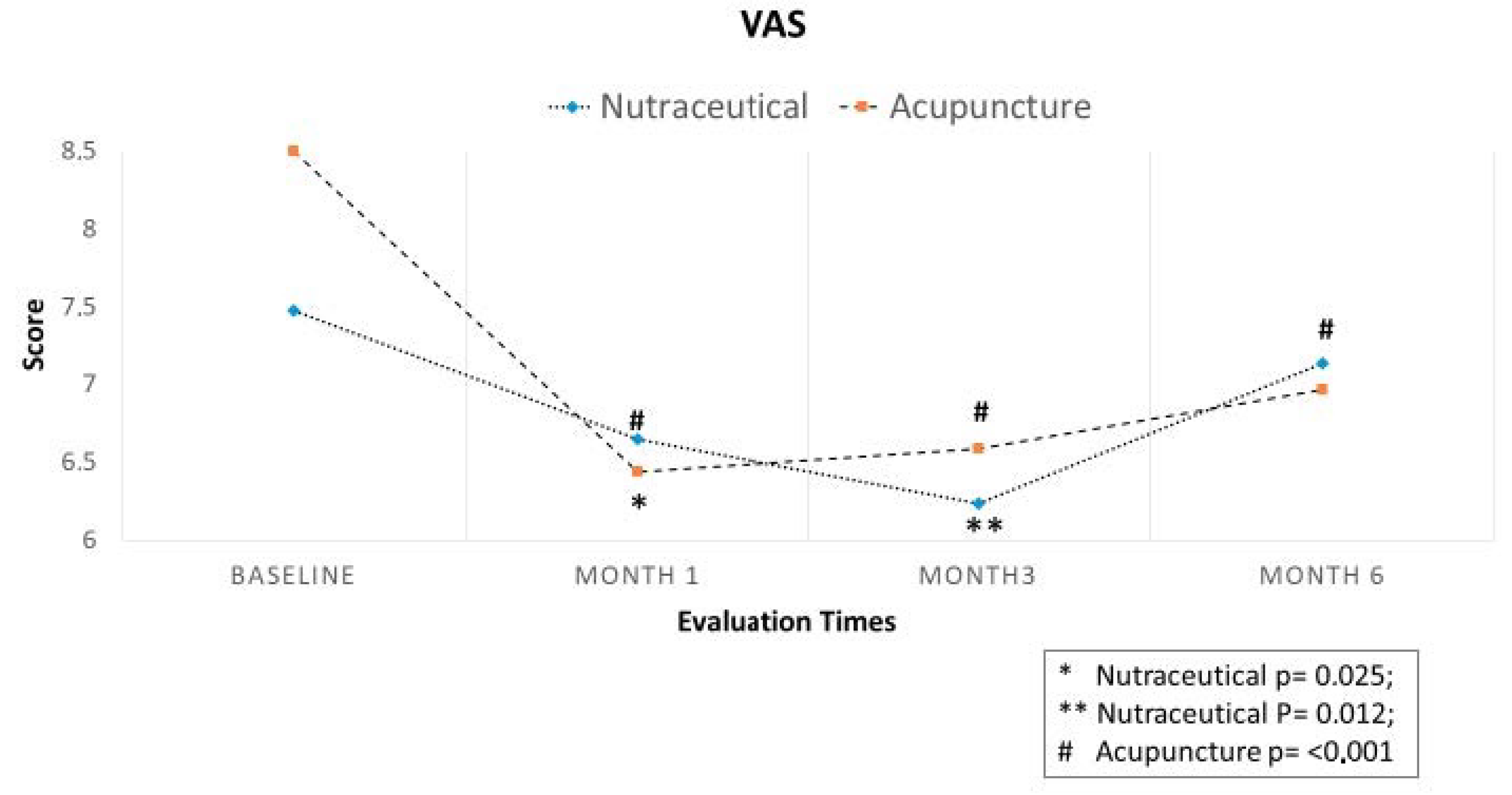
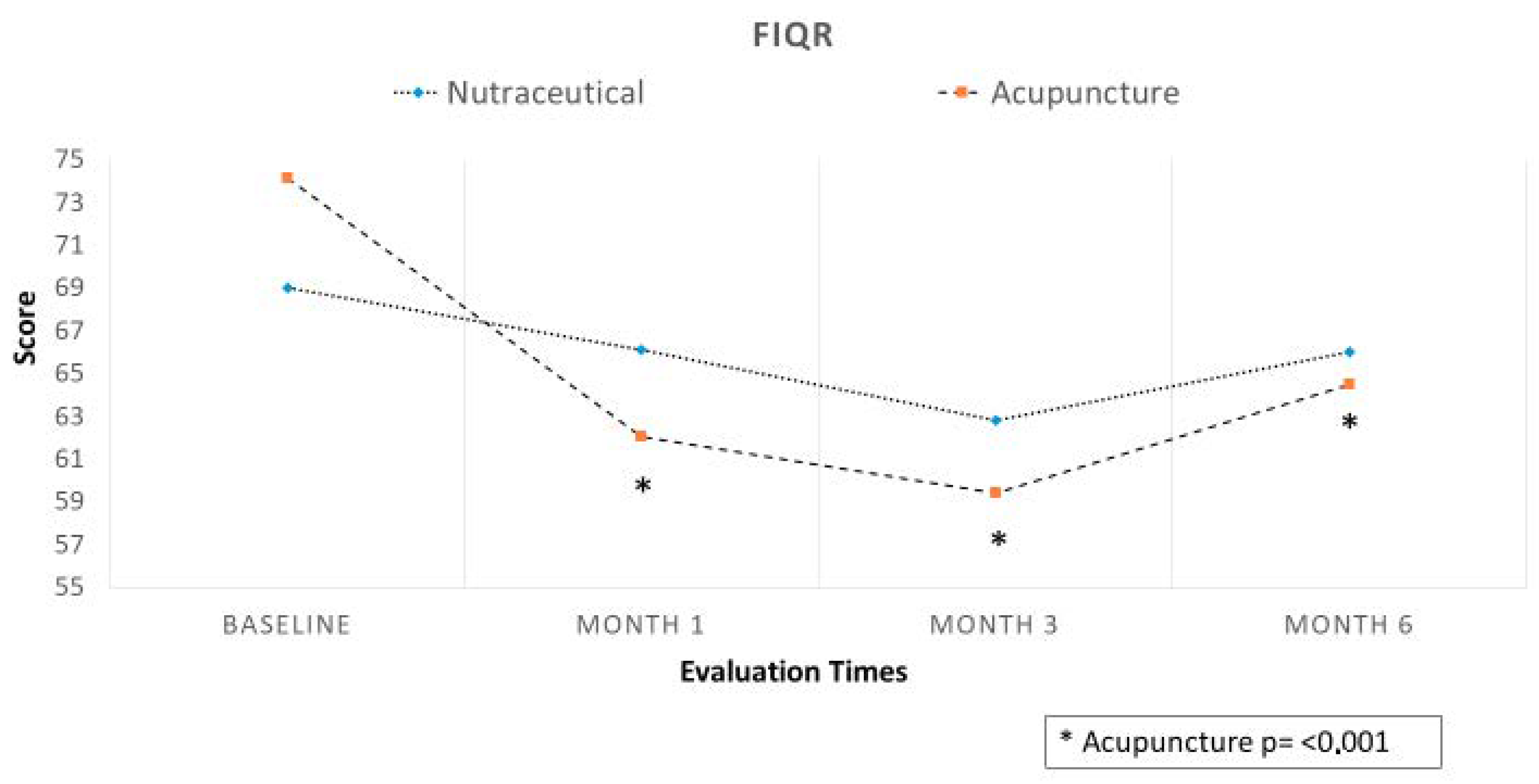
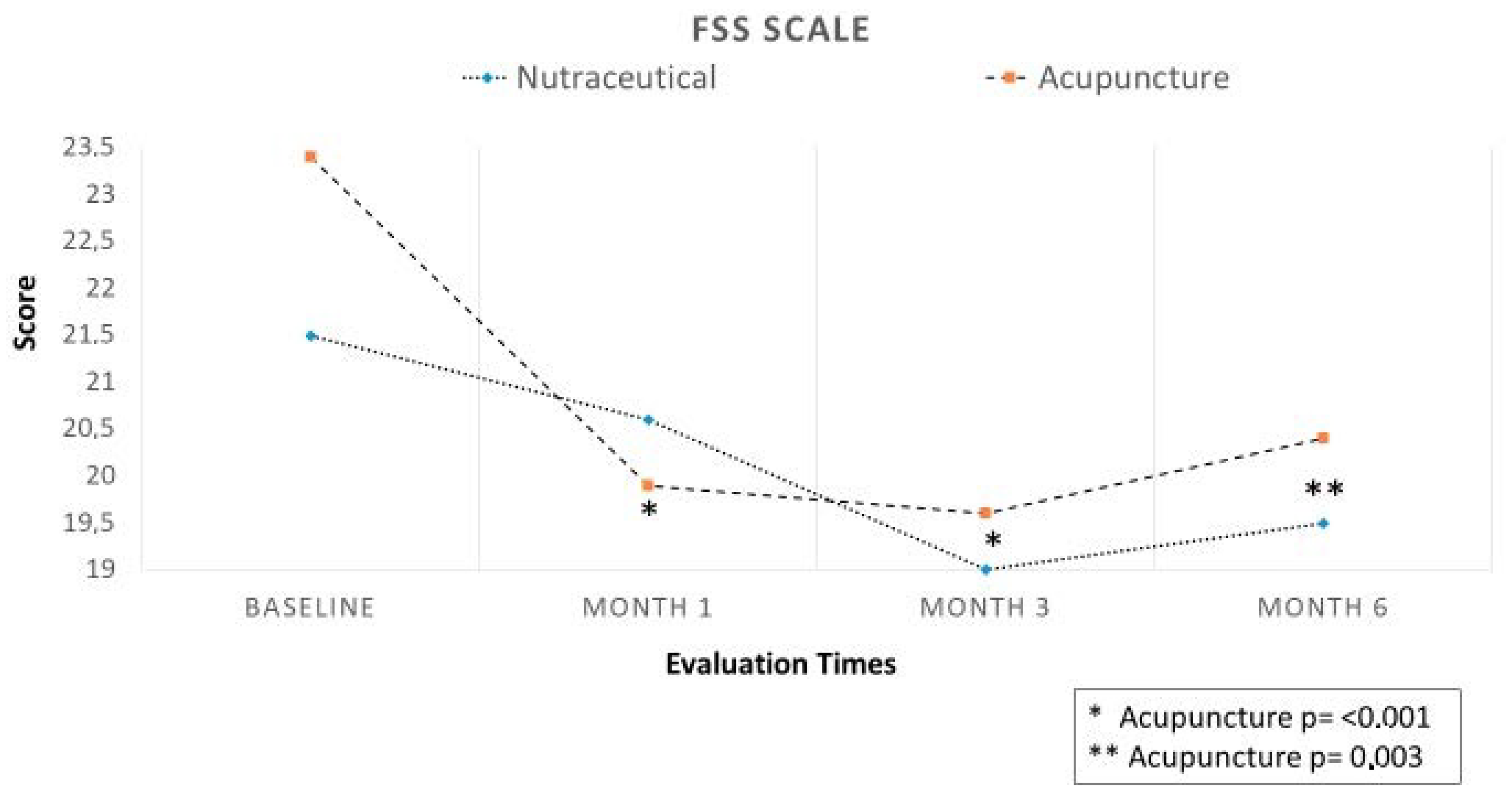
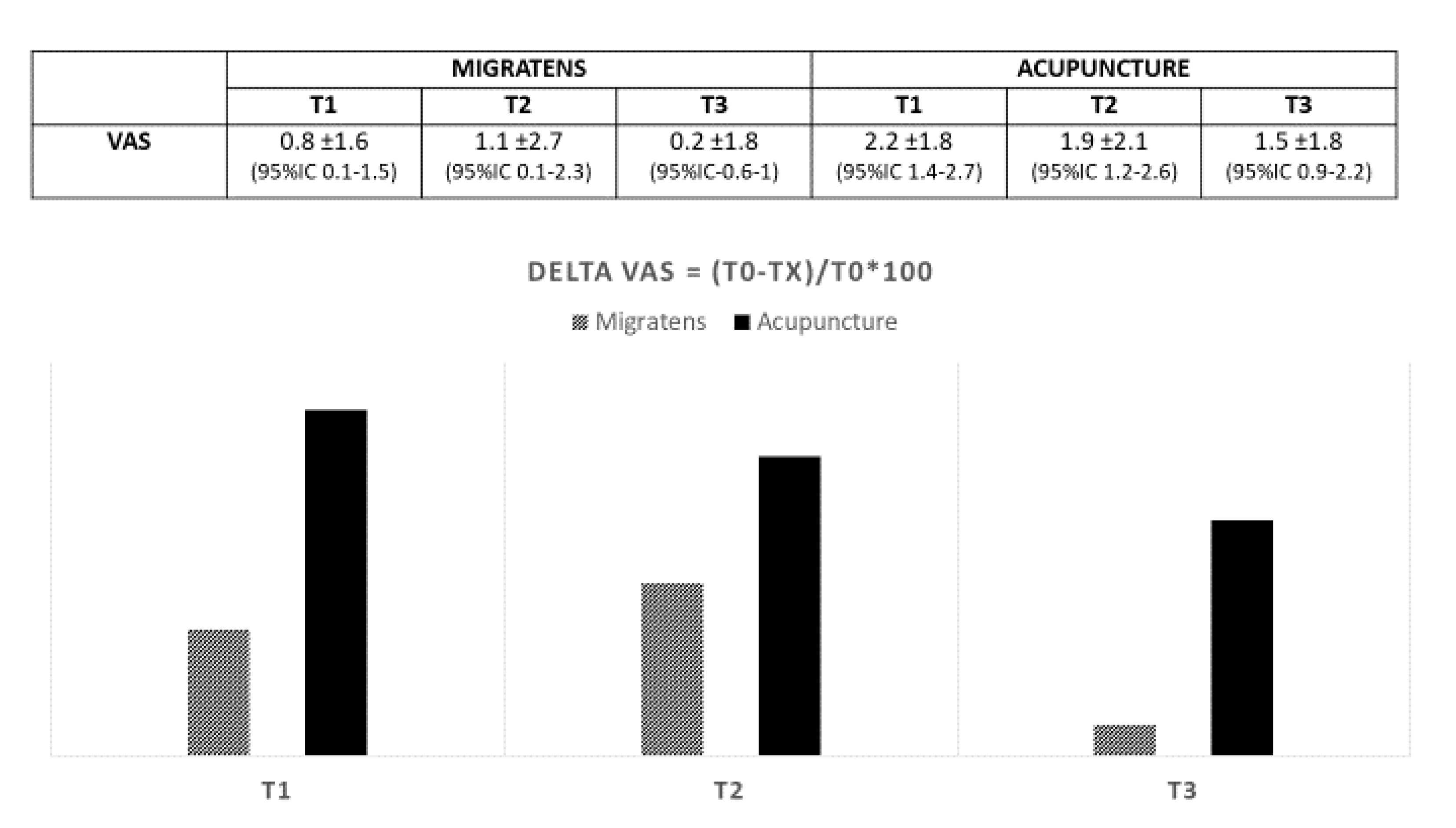
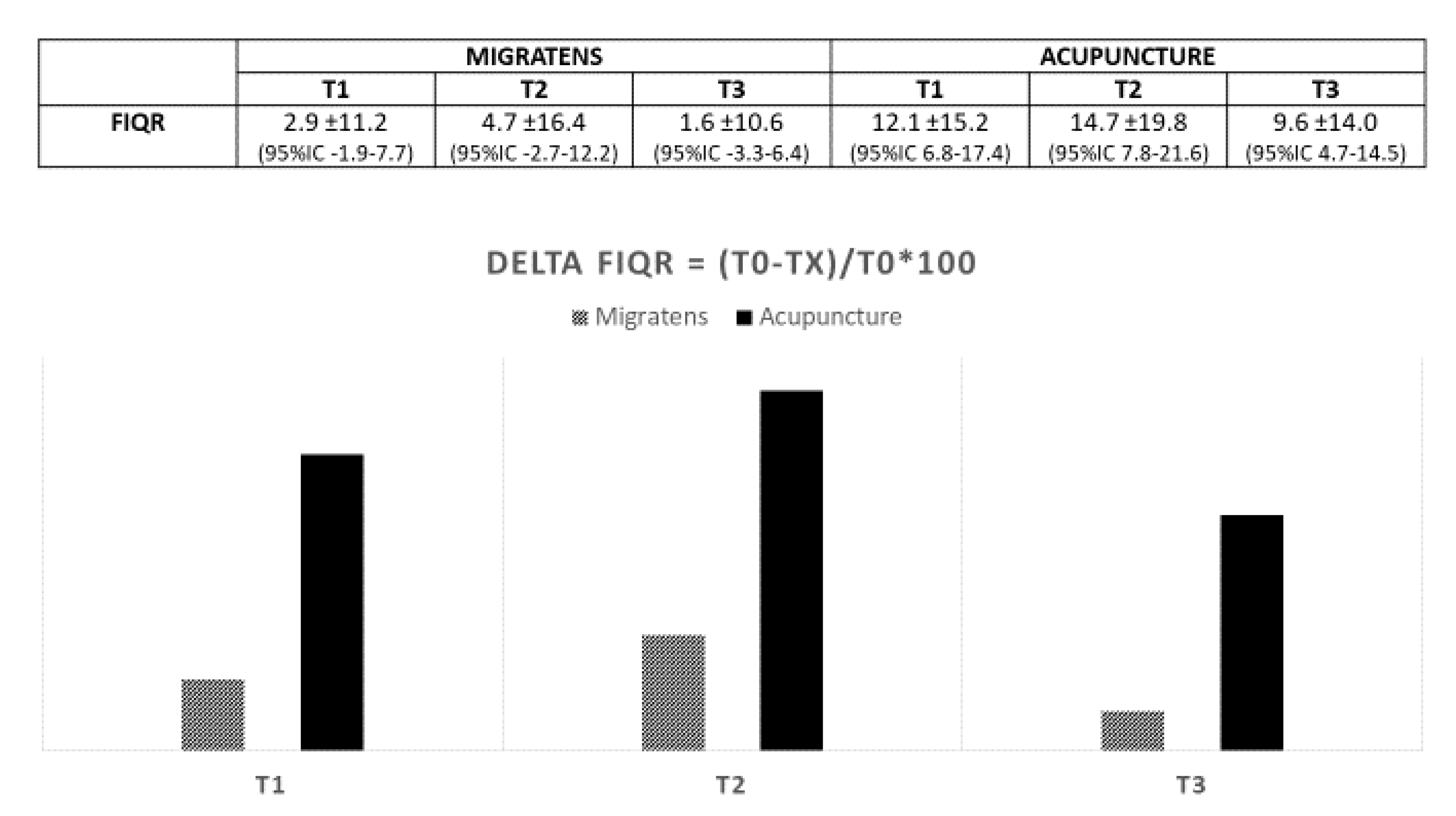
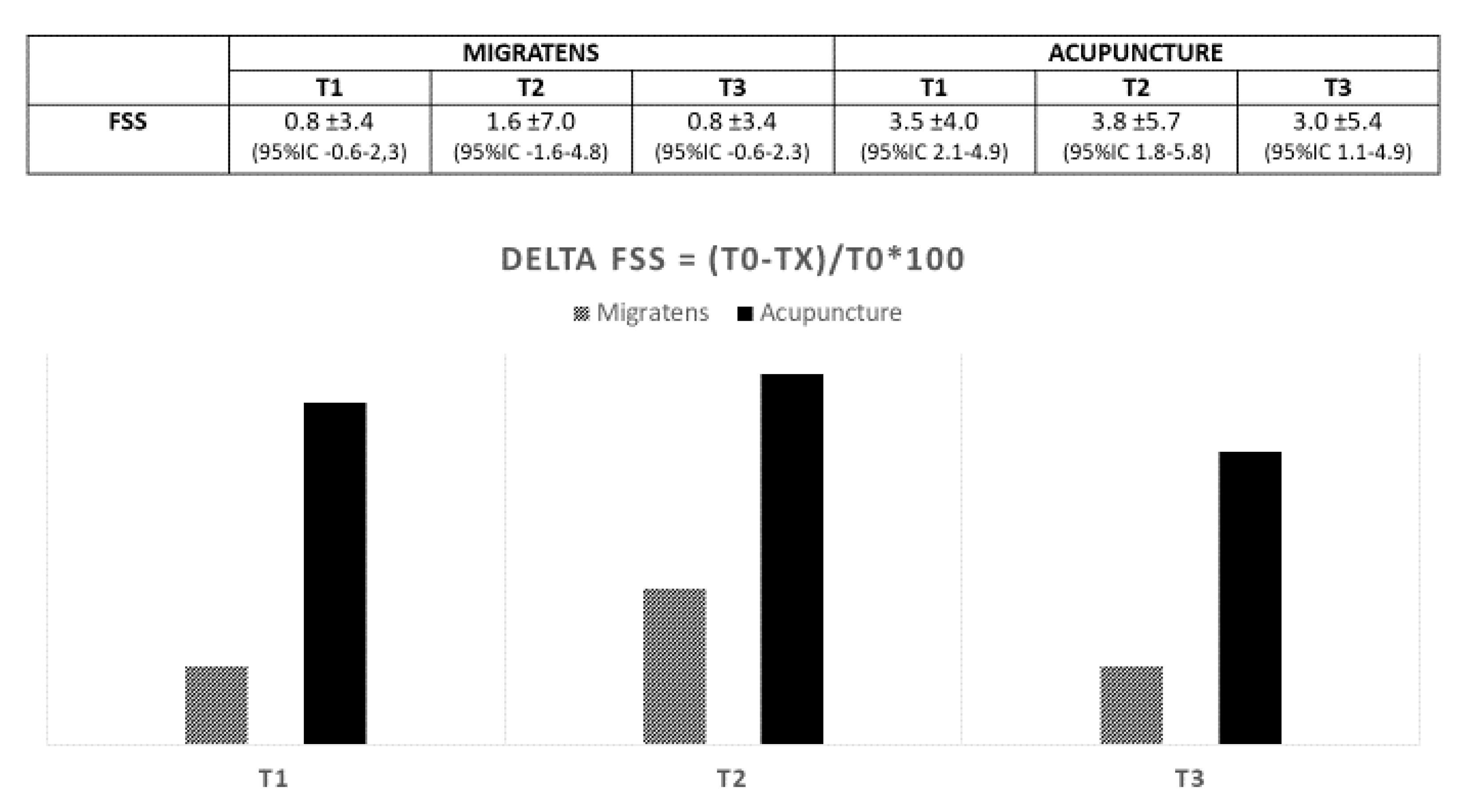
3.2. Efficacy
3.3. Treatments Discontinuation
3.4. Safety
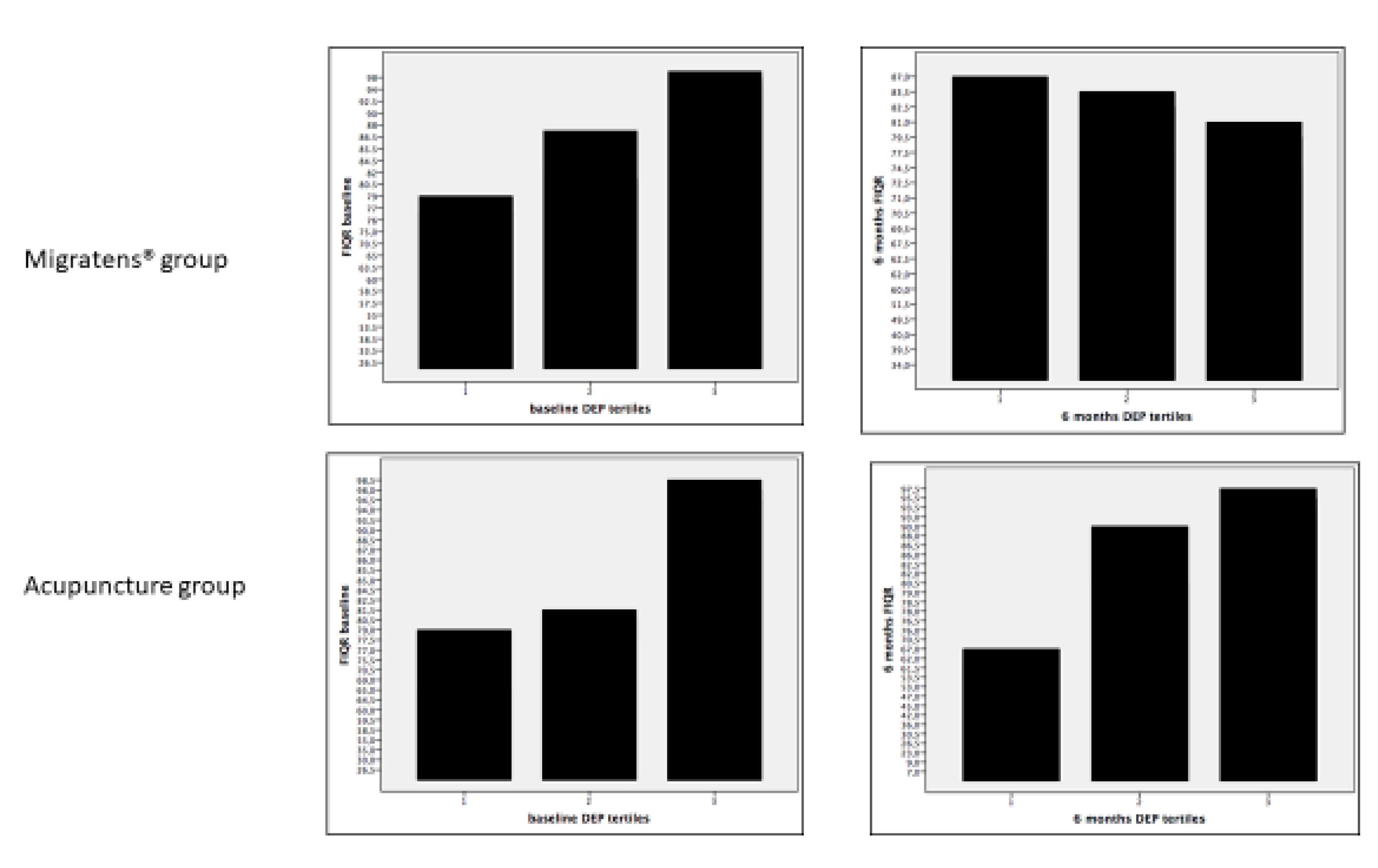
3.5. Correlations
4. Discussion
4.1. Summary of Current Findings
4.2. Nutrient Considerations
4.3. Acupuncture Considerations
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Clauw, D.J. Fibromyalgia: A clinical review. JAMA 2014, 311, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Martini, A.; Schweiger, V.; Del Balzo, G.; Sartori, G.; Parolini, M.; Chinellato, E.; Sarzi-Puttini, P.; Polati, E. Epidemiological, pharmacological and sociodemographic characterisation of a population of 386 fibromyalgic patients referred to a tertiary pain centre in Italy. Clin. Exp. Rheumatol. 2019, 37 (Suppl. 116), 27–38. [Google Scholar] [PubMed]
- Marques, A.P.; Santo, A.; Berssaneti, A.A.; Matsutani, L.A.; Yuan, S.L.K. Prevalence of fibromyalgia: Literature review update. Rev. Bras. Reumatol. Engl. Ed. 2017, 57, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.L.; Dukes, E.M. The health status burden of people with fibromyalgia: A review of studies that assessed health status with the SF-36 or the SF-12. Int. J. Clin. Pract. 2008, 62, 115–126. [Google Scholar] [CrossRef]
- Schweiger, V.; Del Balzo, G.; Raniero, D.; De Leo, D.; Martini, A.; Sarzi-Puttini, P.; Polati, E. Current trends in disability claims due to fibromyalgia syndrome. Clin. Exp. Rheumatol. 2017, 35, S119–S126. [Google Scholar]
- Thieme, K.; Mathys, M.; Turk, D.C. Evidenced-Based Guidelines on the Treatment of Fibromyalgia Patients: Are They Consistent and If Not, Why Not? Have Effective Psychological Treatments Been Overlooked? J. Pain 2017, 18, 747–756. [Google Scholar] [CrossRef]
- Bjorklund, G.; Dadar, M.; Chirumbolo, S.; Aaseth, J. Fibromyalgia and nutrition: Therapeutic possibilities? Biomed. Pharmacother. 2018, 103, 531–538. [Google Scholar] [CrossRef]
- Calandre, E.P.; Rico-Villademoros, F.; Slim, M. An update on pharmacotherapy for the treatment of fibromyalgia. Expert Opin. Pharmacother. 2015, 16, 1347–1368. [Google Scholar] [CrossRef]
- Prabhakar, A.; Kaiser, J.M.; Novitch, M.B.; Cornett, E.M.; Urman, R.D.; Kaye, A.D. The Role of Complementary and Alternative Medicine Treatments in Fibromyalgia: A Comprehensive Review. Curr. Rheumatol. Rep. 2019, 21, 14. [Google Scholar] [CrossRef]
- Holton, K. The role of diet in the treatment of fibromyalgia. Pain Manag. 2016, 6, 317–320. [Google Scholar] [CrossRef]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.A.; Goldenberg, D.L.; Häuser, W.; Katz, R.L.; Mease, P.J.; Russell, A.S.; Russell, I.J.; Walitt, B. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin. Arthritis Rheum. 2016, 46, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Makrani, A.H.; Afshari, M.; Ghajar, M.; Forooghi, Z.; Moosazadeh, M. Vitamin D and fibromyalgia: A meta-analysis. Korean J. Pain 2017, 30, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Garrido-Maraver, J.; Cordero, M.D.; Oropesa-Avila, M.; Vega, A.F.; de la Mata, M.; Pavon, A.D.; Alcocer-Gomez, E.; Calero, C.P.; Paz, M.V.; Alanis, M.; et al. Clinical applications of coenzyme Q10. Front. Biosci. (Landmark Ed.) 2014, 19, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Bazzichi, L.; Palego, L.; Giannaccini, G.; Rossi, A.; De Feo, F.; Giacomelli, C.; Betti, L.; Giusti, L.; Mascia, G.; Bombardieri, S.; et al. Altered amino acid homeostasis in subjects affected by fibromyalgia. Clin. Biochem. 2009, 42, 1064–1070. [Google Scholar] [CrossRef]
- Bagis, S.; Karabiber, M.; As, I.; Tamer, L.; Erdogan, C.; Atalay, A. Is magnesium citrate treatment effective on pain, clinical parameters and functional status in patients with fibromyalgia? Rheumatol. Int. 2013, 33, 167–172. [Google Scholar] [CrossRef]
- Laborest srl, I. Migratens® Data Sheet; Fast-Slow Delivery Technology: Assago, Italy, 2016. [Google Scholar]
- Deare, J.C.; Zheng, Z.; Xue, C.C.L.; Liu, J.P.; Shang, J.; Scott, S.W.; Littlejohn, G. Acupuncture for treating fibromyalgia. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Bennett, R.M.; Friend, R.; Jones, K.D.; Ward, R.; Han, B.K.; Ross, R.L. The Revised Fibromyalgia Impact Questionnaire (FIQR): Validation and psychometric properties. Arthritis Res. Ther. 2009, 11, R120. [Google Scholar] [CrossRef]
- Salaffi, F.; Franchignoni, F.; Giordano, A.; Ciapetti, A.; Sarzi-Puttini, P.; Ottonello, M. Psychometric characteristics of the Italian version of the revised Fibromyalgia Impact Questionnaire using classical test theory and Rasch analysis. Clin. Exp. Rheumatol. 2013, 31 (Suppl. 79), S41–S49. [Google Scholar]
- Wolfe, F.; Hauser, W. Fibromyalgia diagnosis and diagnostic criteria. Ann. Med. 2011, 43, 495–502. [Google Scholar] [CrossRef]
- Winkelmann, A. Is Aerobic Exercise Training Beneficial for Adults With Fibromyalgia? A Cochrane Review Summary with Commentary. Am. J. Phys. Med. Rehab. 2019, 98, 169–170. [Google Scholar] [CrossRef]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Ugurlu, F.G.; Sezer, N.; Aktekin, L.; Fidan, F.; Tok, F.; Akkus, S. The effects of acupuncture versus sham acupuncture in the treatment of fibromyalgia: A randomized controlled clinical trial. Acta Reumatol. Port. 2017, 42, 32–37. [Google Scholar] [PubMed]
- Hauser, W.; Walitt, B.; Fitzcharles, M.A.; Sommer, C. Review of pharmacological therapies in fibromyalgia syndrome. Arthritis Res. Ther. 2014, 16, 201. [Google Scholar] [CrossRef] [PubMed]
- Kia, S.; Choy, E. Update on Treatment Guideline in Fibromyalgia Syndrome with Focus on Pharmacology. Biomedicines 2017, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.J.; Steer, M.; Ashe, S.C.; Furness, P.J.; Haywood-Small, S.; Lawson, K. Patients’ perspective of the effectiveness and acceptability of pharmacological and non-pharmacological treatments of fibromyalgia. Scand. J. Pain 2019, 19, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Di Lollo, A.C.; Guzzo, M.P.; Giacomelli, C.; Atzeni, F.; Bazzichi, L.; Di Franco, M. Fibromyalgia and nutrition: What news? Clin. Exp. Rheumatol. 2015, 33, S117–S125. [Google Scholar]
- Bourre, J.M. Effects of nutrients (in food) on the structure and function of the nervous system: Update on dietary requirements for brain. Part 2: Macronutrients. J. Nutr. Health Aging 2006, 10, 386–399. [Google Scholar]
- Schwarz, M.J.; Offenbaecher, M.; Neumeister, A.; Ewert, T.; Willeit, M.; Praschak-Rieder, N.; Zach, J.; Zacherl, M.; Lossau, K.; Weisser, R.; et al. Evidence for an altered tryptophan metabolism in fibromyalgia. Neurobiol. Dis. 2002, 11, 434–442. [Google Scholar] [CrossRef]
- Juhl, J.H. Fibromyalgia and the serotonin pathway. Altern. Med. Rev. 1998, 3, 367–375. [Google Scholar]
- Cuciureanu, M.D.; Vink, R. Magnesium and stress. In Magnesium in the Central Nervous System; Vink, R., Nechifor, M., Eds.; AU: Adelaide, Australia, 2011. [Google Scholar]
- Sakarya, S.T.; Akyol, Y.; Bedir, A.; Canturk, F. The relationship between serum antioxidant vitamins, magnesium levels, and clinical parameters in patients with primary fibromyalgia syndrome. Clin. Rheumatol. 2011, 30, 1039–1043. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.R.; Lee, H.; Nam, D.H. Comparing Verum and Sham Acupuncture in Fibromyalgia Syndrome: A Systematic Review and Meta-Analysis. Evid.-Based Complement. Alternat. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, K.M.; Lee, D.J.; Kim, B.T.; Park, S.B.; Cho, D.Y.; Suh, C.H.; Kim, H.A.; Park, R.W.; Joo, N.S. Women with Fibromyalgia Have Lower Levels of Calcium, Magnesium, Iron and Manganese in Hair Mineral Analysis. J. Korean Med. Sci. 2011, 26, 1253–1257. [Google Scholar] [CrossRef] [PubMed]
- Andretta, A.; Dias Batista, E.; Madalozzo Schieferdecker, M.E.; Rasmussen Petterle, R.; Boguszewski, C.L.; Dos Santos Paiva, E. Relation between magnesium and calcium and parameters of pain, quality of life and depression in women with fibromyalgia. Adv. Rheumatol. 2019, 59, 55. [Google Scholar] [CrossRef] [PubMed]
- Tibullo, D.; Li Volti, G.; Giallongo, C.; Grasso, S.; Tomassoni, D.; Anfuso, C.D.; Lupo, G.; Amenta, F.; Avola, R.; Bramanti, V. Biochemical and clinical relevance of alpha lipoic acid: Antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflamm. Res. 2017, 66, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Gilron, I.; Tu, D.; Holden, R.; Towheed, T.; Vandenkerkhof, E.; Milev, R. Combination Analgesic Development for Enhanced Clinical Efficacy (CADENCE Trial): Study Protocol for a Double-Blind, Randomized, Placebo-Controlled Crossover Trial of an Alpha-Lipoic Acid—Pregabalin Combination for the Treatment of Fibromyalgia Pain. JMIR Res. Protoc. 2017, 6, e154. [Google Scholar] [CrossRef] [PubMed]
- Gilron, I.; Tu, D.; Holden, R.; Towheed, T.; Ziegler, D.; Wang, L.; Milev, R.; Gray, C. Innovations in the Management of Musculoskeletal Pain with Alpha-Lipoic Acid (IMPALA Trial): Study protocol for a Double-Blind, Randomized, Placebo-Controlled Crossover Trial of Alpha-Lipoic Acid for the Treatment of Fibromyalgia Pain. JMIR Res. Protoc. 2017, 6, e41. [Google Scholar] [CrossRef]
- Hernandez-Camacho, J.D.; Bernier, M.; Lopez-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef]
- Cordero, M.D.; de Miguel, M.; Carmona-Lopez, I.; Bonal, P.; Campa, F.; Moreno-Fernandez, A.M. Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuro Endocrinol. Lett. 2010, 31, 169–173. [Google Scholar]
- Alcocer-Gomez, E.; Cano-Garcia, F.J.; Cordero, M.D. Effect of coenzyme Q10 evaluated by 1990 and 2010 ACR Diagnostic Criteria for Fibromyalgia and SCL-90-R: Four case reports and literature review. Nutrition 2013, 29, 1422–1425. [Google Scholar] [CrossRef]
- Cordero, M.D.; Alcocer-Gomez, E.; Culic, O.; Carrión, A.M.; de Miguel, M.; Díaz-Parrado, E.; Pérez-Villegas, E.M.; Bullón, P.; Battino, M.; Sánchez-Alcazar, J.A. NLRP3 inflammasome is activated in fibromyalgia: The effect of coenzyme Q10. Antioxid. Redox Signal. 2014, 20, 1169–1180. [Google Scholar] [CrossRef]
- Dalle Carbonare, L.; Valenti, M.T.; del Forno, F.; Caneva, E.; Pietrobelli, A. Vitamin D: Daily vs. Monthly Use in Children and Elderly-What Is Going On? Nutrients 2017, 9, 652. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Valenti, M.T.; del Forno, F.; Piacentini, G.; Pietrobelli, A. Vitamin D Daily versus Monthly Administration: Bone Turnover and Adipose Tissue Influences. Nutrients 2018, 10, 1934. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.Q.; Malihi, Z.; Stewart, A.W.; Lawes, C.M.M.; Scragg, R. The association between vitamin D concentration and pain: A systematic review and meta-analysis. Public Health Nutr. 2018, 21, 2022–2037. [Google Scholar] [CrossRef] [PubMed]
- Anglin, R.E.S.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, D.L.; Hirotsu, C.; Tufik, S.; Andersen, M.L. The interfaces between vitamin D, sleep and pain. J. Endocrinol. 2017, 234, R23–R36. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, R.; Salli, A.; Cingoz, H.T.; Kucuksen, S.; Ugurlu, H. Efficacy of vitamin D replacement therapy on patients with chronic nonspecific widespread musculoskeletal pain with vitamin D deficiency. Int. J. Rheum. Dis. 2016, 19, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Ellis, S.D.; Kelly, S.T.; Shurlock, J.H.; Hepburn, A.L.N. The role of vitamin D testing and replacement in fibromyalgia: A systematic literature review. BMC Rheumatol. 2018, 2, 28. [Google Scholar] [CrossRef]
- Martins, Y.A.; Cardinali, C.; Ravanelli, M.I.; Brunaldi, K. Is hypovitaminosis D associated with fibromyalgia? A systematic review. Nutr. Rev. 2020, 78, 115–133. [Google Scholar] [CrossRef]
- Gasperi, V.; Sibilano, M.; Savini, I.; Catani, M.V. Niacin in the Central Nervous System: An Update of Biological Aspects and Clinical Applications. Int. J. Mol. Sci. 2019, 20, 974. [Google Scholar] [CrossRef]
- Deiana, M.; Malerba, G.; Dalle Carbonare, L.; Cheri, S.; Patuzzo, C.; Tsenov, G.; Moron Dalla Tor, L.; Mori, A.; Saviola, G.; Zipeto, D.; et al. Physical Activity Prevents Cartilage Degradation: A Metabolomics Study Pinpoints the Involvement of Vitamin B6. Cells 2019, 8, 1374. [Google Scholar] [CrossRef]
- Zhang, X.C.; Chen, H.; Xu, W.T.; Song, Y.Y.; Gu, Y.H.; Ni, G.X. Acupuncture therapy for fibromyalgia: A systematic review and meta-analysis of randomized controlled trials. J. Pain Res. 2019, 12, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Iannuccelli, C.; Guzzo, M.P.; Atzeni, F.; Mannocci, F.; Alessandri, C.; Gerardi, M.C.; Valesini, G.; Di Franco, M. Pain modulation in patients with fibromyalgia undergoing acupuncture treatment is associated with fluctuations in serum neuropeptide Y levels. Clin. Exp. Rheumatol. 2017, 35, S81–S85. [Google Scholar]
- Karatay, S.; Okur, S.C.; Uzkeser, H.; Yildirim, K.; Akcay, F. Effects of Acupuncture Treatment on Fibromyalgia Symptoms, Serotonin, and Substance P Levels: A Randomized Sham and Placebo-Controlled Clinical Trial. Pain Med. 2018, 19, 615–628. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.; Leach, V.; Davies, P.; Penfold, C.; Ness, A.; Churchill, R. An overview of systematic reviews of complementary and alternative therapies for fibromyalgia using both AMSTAR and ROBIS as quality assessment tools. Syst. Rev. 2017, 6, 97. [Google Scholar] [CrossRef]
| Included in the Study | 60 Patients | p | |
|---|---|---|---|
| Treatment Groups | Group A (26) | Group B (34) | |
| Age, yr., mean (SD) | 48.2 ± 7.4 | 52.9 ± 8.5 | 0.04 |
| Pain (VAS 0–10) | 7.7 ± 1.7 | 8.5 ± 1.4 | 0.04 |
| FIQ-R (0–100) | 69 ± 15.9 | 74.2 ± 18.2 | ns |
| FSS (0–31) | 21.5 ± 5.2 | 23.4 ± 4 | ns |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schweiger, V.; Secchettin, E.; Castellani, C.; Martini, A.; Mazzocchi, E.; Picelli, A.; Polati, E.; Donadello, K.; Valenti, M.T.; Dalle Carbonare, L. Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial. Nutrients 2020, 12, 821. https://doi.org/10.3390/nu12030821
Schweiger V, Secchettin E, Castellani C, Martini A, Mazzocchi E, Picelli A, Polati E, Donadello K, Valenti MT, Dalle Carbonare L. Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial. Nutrients. 2020; 12(3):821. https://doi.org/10.3390/nu12030821
Chicago/Turabian StyleSchweiger, Vittorio, Erica Secchettin, Cinzia Castellani, Alvise Martini, Elena Mazzocchi, Alessandro Picelli, Enrico Polati, Katia Donadello, Maria Teresa Valenti, and Luca Dalle Carbonare. 2020. "Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial" Nutrients 12, no. 3: 821. https://doi.org/10.3390/nu12030821
APA StyleSchweiger, V., Secchettin, E., Castellani, C., Martini, A., Mazzocchi, E., Picelli, A., Polati, E., Donadello, K., Valenti, M. T., & Dalle Carbonare, L. (2020). Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial. Nutrients, 12(3), 821. https://doi.org/10.3390/nu12030821








