Dietary Patterns and Health Outcomes among African American Maintenance Hemodialysis Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Collection of Clinical Information
2.3. Blood Sampling and Lipid Measurement
2.4. Assessment of Kidney Disease Quality of Life (KDQOL)
2.5. Assessment of Dietary Intake
2.6. Statistical Analysis
3. Results
3.1. Clusters Identified for Dietary Patterns
3.2. Baseline Characteristics
3.3. Characteristics of Patients According to Dietary Cluster
3.4. Nutrient Intake According to Dietary Cluster
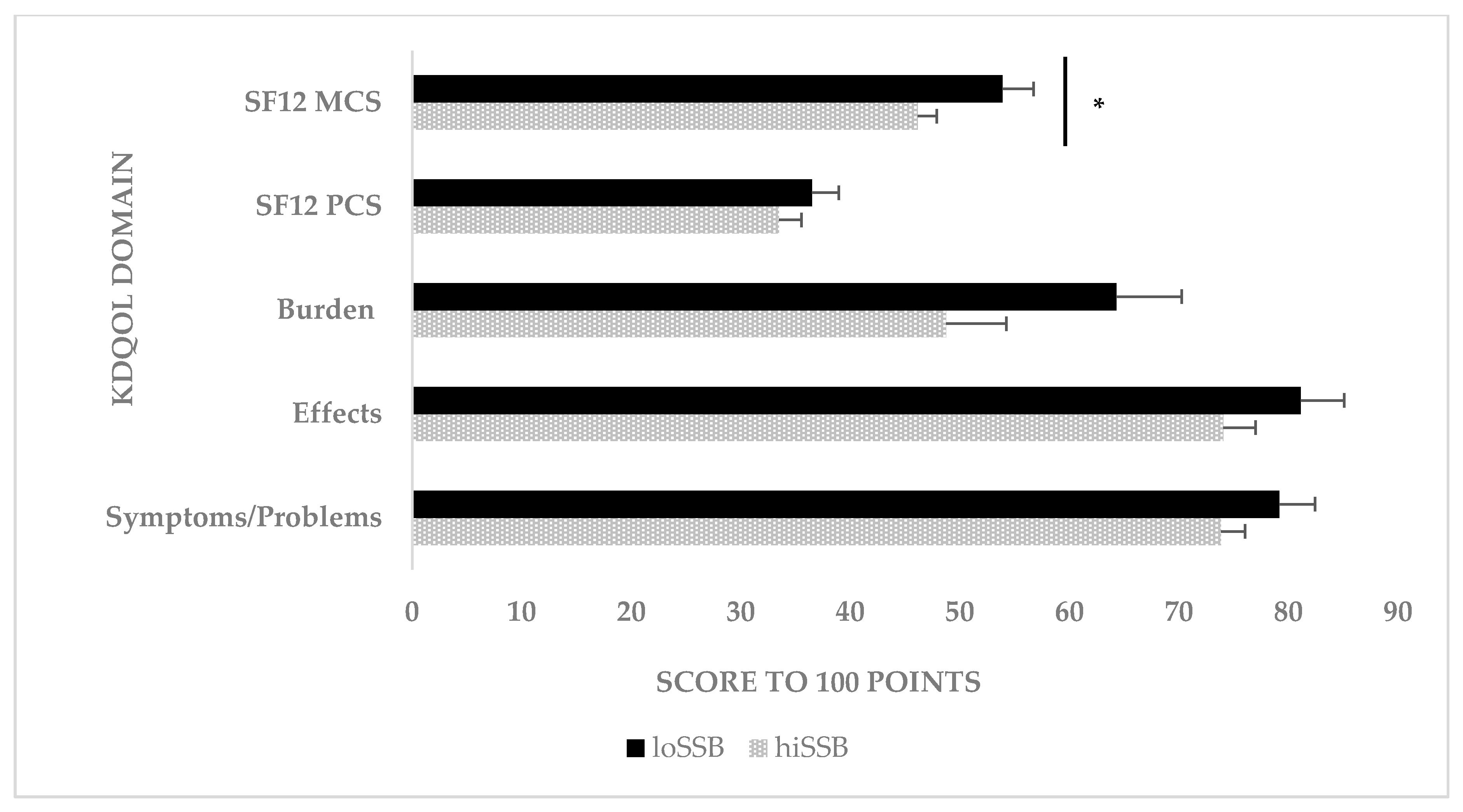
3.5. KDQOL among MHD Patients According to Diet Clusters among MHD Patients
4. Discussion
5. Conclusions
6. Practical Implication
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef] [PubMed]
- Wang, V.; Vilme, H.; Maciejewski, M.L.; Boulware, L.E. The Economic Burden of Chronic Kidney Disease and End-Stage Renal Disease. Semin. Nephrol. 2016, 36, 319–330. [Google Scholar] [CrossRef] [PubMed]
- The United States Renal Data System. Chapter 1: Incidence, Prevalence, Patient Characteristics, and Treatment Modalities. Am. J. Kidney Dis. 2017, 69, S261–S300. [Google Scholar] [CrossRef]
- NKF. African Americans and Kidney Disease. Available online: https://www.kidney.org/news/newsroom/factsheets/African-Americans-and-CKD (accessed on 9 January 2020).
- Harding, K.; Mersha, T.B.; Webb, F.A.; Vassalotti, J.A.; Nicholas, S.B. Current State and Future Trends to Optimize the Care of African Americans with End-Stage Renal Disease. Am. J. Nephrol. 2017, 46, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Asghari, G.; Momenan, M.; Yuzbashian, E.; Mirmiran, P.; Azizi, F. Dietary pattern and incidence of chronic kidney disease among adults: A population-based study. Nutr. Metab. 2018, 15, 88. [Google Scholar] [CrossRef]
- Ajjarapu, A.S.; Hinkle, S.N.; Li, M.; Francis, E.C.; Zhang, C. Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies. Nutrients 2019, 11, 1877. [Google Scholar] [CrossRef]
- Bach, K.E.; Kelly, J.T.; Palmer, S.C.; Khalesi, S.; Strippoli, G.F.M.; Campbell, K.L. Healthy Dietary Patterns and Incidence of CKD: A Meta-Analysis of Cohort Studies. Clin. J. Am. Soc. Nephrol. 2019. [Google Scholar] [CrossRef]
- Biruete, A.; Jeong, J.H.; Barnes, J.L.; Wilund, K.R. Modified Nutritional Recommendations to Improve Dietary Patterns and Outcomes in Hemodialysis Patients. J. Ren. Nutr. 2016. [Google Scholar] [CrossRef]
- Kelly, J.T.; Palmer, S.C.; Wai, S.N.; Ruospo, M.; Carrero, J.J.; Campbell, K.L.; Strippoli, G.F. Healthy Dietary Patterns and Risk of Mortality and ESRD in CKD: A Meta-Analysis of Cohort Studies. Clin. J. Am. Soc. Nephrol. 2017, 12, 272–279. [Google Scholar] [CrossRef]
- Ocke, M.C. Evaluation of methodologies for assessing the overall diet: Dietary quality scores and dietary pattern analysis. Proc. Nutr. Soc. 2013, 72, 191–199. [Google Scholar] [CrossRef]
- Devlin, U.M.; McNulty, B.A.; Nugent, A.P.; Gibney, M.J. The use of cluster analysis to derive dietary patterns: Methodological considerations, reproducibility, validity and the effect of energy mis-reporting. Proc. Nutr. Soc. 2012, 71, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Sauvageot, N.; Schritz, A.; Leite, S.; Alkerwi, A.; Stranges, S.; Zannad, F.; Streel, S.; Hoge, A.; Donneau, A.F.; Albert, A.; et al. Stability-based validation of dietary patterns obtained by cluster analysis. Nutr. J. 2017, 16, 4. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Tucker, K.L. Empirically derived eating patterns using factor or cluster analysis: A review. Nutr. Rev. 2004, 62, 177–203. [Google Scholar] [CrossRef] [PubMed]
- Garrow, J.S.; Webster, J. Quetelet’s index (W/H2) as a measure of fatness. Int. J. Obes. 1985, 9, 147–153. [Google Scholar] [PubMed]
- Peipert, J.D.; Hays, R.D. Using Patient-Reported Measures in Dialysis Clinics. Clin. J. Am. Soc. Nephrol. 2017, 12, 1889–1891. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hays, R.D.; Kallich, J.D.; Mapes, D.L.; Coons, S.J.; Carter, W.B. Development of the kidney disease quality of life (KDQOL) instrument. Qual. Life Res. 1994, 3, 329–338. [Google Scholar] [CrossRef]
- Hays, R.D.; Kallich, J.D.; Mapes, D.L. Kidney Disease Quality of Life Short Form (KDQOL-SF™), Version 1.3: A Manual for Use and Scoring; RAND: Santa Monica, CA, USA, 1997. [Google Scholar]
- Kidney Disease Quality of Life Instrument (KDQOL): Scoring the KDQOL-36. Available online: https://www.rand.org/health/surveys_tools/kdqol.html (accessed on 31 August 2019).
- De Keyzer, W.; Huybrechts, I.; De Vriendt, V.; Vandevijvere, S.; Slimani, N.; Van Oyen, H.; De Henauw, S. Repeated 24-hour recalls versus dietary records for estimating nutrient intakes in a national food consumption survey. Food Nutr. Res. 2011, 55. [Google Scholar] [CrossRef]
- Bailey, R.L.; Gutschall, M.D.; Mitchell, D.C.; Miller, C.K.; Lawrence, F.R.; Smiciklas-Wright, H. Comparative strategies for using cluster analysis to assess dietary patterns. J. Am. Diet. Assoc. 2006, 106, 1194–1200. [Google Scholar] [CrossRef]
- Raper, N.; Perloff, B.; Ingwersen, L.; Steinfeldt, L.; Anand, J. An overview of USDA’s Dietary Intake Data System. J. Food Compos. Anal. 2004, 17, 545–555. [Google Scholar] [CrossRef]
- Bailey, R.L.; Mitchell, D.C.; Miller, C.; Smiciklas-Wright, H. Assessing the effect of underreporting energy intake on dietary patterns and weight status. J. Am. Diet. Assoc. 2007, 107, 64–71. [Google Scholar] [CrossRef]
- McCrory, M.A.; McCrory, M.A.; Hajduk, C.L.; Roberts, S.B. Procedures for screening out inaccurate reports of dietary energy intake. Public Health Nutr. 2002, 5, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D. National kidney foundation K/DOQI clinical practice guidelines for nutrition in chronic renal failure. Am. J. Kidney Dis. 2001, 37, S66–S70. [Google Scholar] [CrossRef] [PubMed]
- Hearty, A.P.; Gibney, M.J. Comparison of cluster and principal component analysis techniques to derive dietary patterns in Irish adults. Br. J. Nutr. 2009, 101, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.L.; Harris, T.B.; Houston, D.K.; Tylavsky, F.A.; Lee, J.S.; Sellmeyer, D.E.; Sahyoun, N.R. Relationships of dietary patterns with body composition in older adults differ by gender and PPAR-gamma Pro12Ala genotype. Eur. J. Nutr. 2010, 49, 385–394. [Google Scholar] [CrossRef]
- Foundation, N.K. KDOQI Clinical Practice Guidelines for Managing Dyslipidemias in CKD. Am. J. Kidney Dis. 2003, 41, S1–S91. [Google Scholar]
- Choose My Plate. Available online: https://www.choosemyplate.gov/ (accessed on 11 October 2019).
- Kopple, J.; Wolfson, M. KDOQI Nutrition in Chronic Renal Failure. AJKD 2000, 25, S1–S139. [Google Scholar] [CrossRef]
- United States Department of Agriculture. 2015–2020 Dietary Guidelines; USDA: Washington, DC, USA, 2015. [Google Scholar]
- Nutrient Recommendations: Dietary Reference Intakes (DRI). Available online: https://ods.od.nih.gov/Health_Information/Dietary_Reference_Intakes.aspx (accessed on 11 October 2019).
- Duncan, M.J.; Kline, C.E.; Vandelanotte, C.; Sargent, C.; Rogers, N.L.; Di Milia, L. Cross-sectional associations between multiple lifestyle behaviors and health-related quality of life in the 10,000 Steps cohort. PLoS ONE 2014, 9, e94184. [Google Scholar] [CrossRef]
- Godos, J.; Castellano, S.; Marranzano, M. Adherence to a Mediterranean Dietary Pattern Is Associated with Higher Quality of Life in a Cohort of Italian Adults. Nutrients 2019, 11, 981. [Google Scholar] [CrossRef]
- Bonaccio, M.; Di Castelnuovo, A.; Bonanni, A.; Costanzo, S.; De Lucia, F.; Pounis, G.; Zito, F.; Donati, M.B.; de Gaetano, G.; Iacoviello, L.; et al. Adherence to a Mediterranean diet is associated with a better health-related quality of life: A possible role of high dietary antioxidant content. BMJ Open 2013, 3. [Google Scholar] [CrossRef]
- Ribeiro, S.M.F.; Braga, C.B.M.; Peria, F.M.; Martinez, E.Z.; Rocha, J.; Cunha, S.F.C. Effects of zinc supplementation on fatigue and quality of life in patients with colorectal cancer. Einstein (Sao Paulo) 2017, 15, 24–28. [Google Scholar] [CrossRef]
- Markiewicz-Zukowska, R.; Gutowska, A.; Borawska, M.H. Serum zinc concentrations correlate with mental and physical status of nursing home residents. PLoS ONE 2015, 10, e0117257. [Google Scholar] [CrossRef] [PubMed]
- Manley, K.J. Saliva composition and upper gastrointestinal symptoms in chronic kidney disease. J. Ren. Care 2014, 40, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.J.; Ma, F.; Wang, Q.Y.; He, S.L. The effects of oral nutritional supplements in patients with maintenance dialysis therapy: A systematic review and meta-analysis of randomized clinical trials. PLoS ONE 2018, 13, e0203706. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, C.; Wiese, G.; Moorthi, R.N.; Moe, S.M.; Hill Gallant, K.; Running, C.A. Characterizing Dysgeusia in Hemodialysis Patients. Chem. Senses 2019, 44, 165–171. [Google Scholar] [CrossRef] [PubMed]
- González, S.; Huerta, J.M.; Fernández, S.; Patterson, A.M.; Lasheras, C. Life-quality indicators in elderly people are influenced by selenium status. Aging Clin. Exp. Res. 2007, 19, 10–15. [Google Scholar] [CrossRef]
- Lobo, J.C.; Stockler-Pinto, M.B.; Farage, N.E.; Faulin Tdo, E.; Abdalla, D.S.; Torres, J.P.; Velarde, L.G.; Mafra, D. Reduced plasma zinc levels, lipid peroxidation, and inflammation biomarkers levels in hemodialysis patients: Implications to cardiovascular mortality. Ren. Fail. 2013, 35, 680–685. [Google Scholar] [CrossRef]
- Hsu, C.W.; Weng, C.H.; Lee, C.C.; Yen, T.H.; Huang, W.H. Association of serum chromium levels with malnutrition in hemodialysis patients. BMC Nephrol. 2019, 20, 302. [Google Scholar] [CrossRef]
- Ngala, R.A.; Awe, M.A.; Nsiah, P. The effects of plasma chromium on lipid profile, glucose metabolism and cardiovascular risk in type 2 diabetes mellitus. A case—Control study. PLoS ONE 2018, 13, e0197977. [Google Scholar] [CrossRef]
- Bai, J.; Xun, P.; Morris, S.; Jacobs, D.R., Jr.; Liu, K.; He, K. Chromium exposure and incidence of metabolic syndrome among American young adults over a 23-year follow-up: The CARDIA Trace Element Study. Sci. Rep. 2015, 5, 15606. [Google Scholar] [CrossRef]
- Tonelli, M.; Wiebe, N.; Bello, A.; Field, C.J.; Gill, J.S.; Hemmelgarn, B.R.; Holmes, D.T.; Jindal, K.; Klarenbach, S.W.; Manns, B.J.; et al. Concentrations of Trace Elements and Clinical Outcomes in Hemodialysis Patients: A Prospective Cohort Study. Clin. J. Am. Soc. Nephrol. 2018, 13, 907–915. [Google Scholar] [CrossRef]
- Maes, M.; Smith, R.; Christophe, A.; Vandoolaeghe, E.; Van Gastel, A.; Neels, H.; Demedts, P.; Wauters, A.; Meltzer, H.Y. Lower serum high-density lipoprotein cholesterol (HDL-C) in major depression and in depressed men with serious suicidal attempts: Relationship with immune-inflammatory markers. Acta Psychiatr. Scand. 1997, 95, 212–221. [Google Scholar] [CrossRef]
- Parekh, A.; Smeeth, D.; Milner, Y.; Thure, S. The Role of Lipid Biomarkers in Major Depression. Healthcare (Basel) 2017, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Brites, F.; Martin, M.; Guillas, I.; Kontush, A. Antioxidative activity of high-density lipoprotein (HDL): Mechanistic insights into potential clinical benefit. BBA Clin. 2017, 8, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Soran, H.; Schofield, J.D.; Durrington, P.N. Antioxidant properties of HDL. Front. Pharmacol. 2015, 6, 222. [Google Scholar] [CrossRef] [PubMed]
- Santos-Gallego, C.G.; Badimon, J.J.; Rosenson, R.S. Beginning to understand high-density lipoproteins. Endocrinol. Metab. Clin. N. Am. 2014, 43, 913–947. [Google Scholar] [CrossRef]
- Palmer, S.C.; Hanson, C.S.; Craig, J.C.; Strippoli, G.F.; Ruospo, M.; Campbell, K.; Johnson, D.W.; Tong, A. Dietary and fluid restrictions in CKD: A thematic synthesis of patient views from qualitative studies. Am. J. Kidney Dis. 2015, 65, 559–573. [Google Scholar] [CrossRef]
- Stevenson, J.; Tong, A.; Gutman, T.; Campbell, K.L.; Craig, J.C.; Brown, M.A.; Lee, V.W. Experiences and Perspectives of Dietary Management Among Patients on Hemodialysis: An Interview Study. J. Ren. Nutr. 2018, 28, 411–421. [Google Scholar] [CrossRef]
- Govindaraju, T.; Sahle, B.W.; McCaffrey, T.A.; McNeil, J.J.; Owen, A.J. Dietary Patterns and Quality of Life in Older Adults: A Systematic Review. Nutrients 2018, 10, 971. [Google Scholar] [CrossRef]
- Kim, N.H.; Song, S.; Jung, S.Y.; Lee, E.; Kim, Z.; Moon, H.G.; Noh, D.Y.; Lee, J.E. Dietary pattern and health-related quality of life among breast cancer survivors. BMC Women’s Health 2018, 18, 65. [Google Scholar] [CrossRef]
- Porter, A.; Fischer, M.J.; Wang, X.; Brooks, D.; Bruce, M.; Charleston, J.; Cleveland, W.H.; Dowie, D.; Faulkner, M.; Gassman, J.; et al. Quality of life and outcomes in African Americans with CKD. J. Am. Soc. Nephrol. 2014, 25, 1849–1855. [Google Scholar] [CrossRef]
- Fischer, M.J.; Kimmel, P.L.; Greene, T.; Gassman, J.J.; Wang, X.; Brooks, D.H.; Charleston, J.; Dowie, D.; Thornley-Brown, D.; Cooper, L.A.; et al. Elevated depressive affect is associated with adverse cardiovascular outcomes among African Americans with chronic kidney disease. Kidney Int. 2011, 80, 670–678. [Google Scholar] [CrossRef][Green Version]
- Kalantar-Zadeh, K.; Block, G.; McAllister, C. Appetite and inflammation, nutrition, anemia, and clinical outcome in hemodialysis patients. Am. J. Clin. Nutr. 2004, 80, 299–307. [Google Scholar] [CrossRef]
- Feroze, U.; Noori, N.; Kovesdy, C.P.; Molnar, M.Z.; Martin, D.J.; Reina-Patton, A.; Benner, D.; Bross, R.; Norris, K.C.; Kopple, J.D.; et al. Quality-of-life and mortality in hemodialysis patients: Roles of race and nutritional status. Clin. J. Am. Soc. Nephrol. 2011, 6, 1100–1111. [Google Scholar] [CrossRef]
- Gabbay, E.; Meyer, K.B.; Griffith, J.L.; Richardson, M.M.; Miskulin, D.C. Temporal trends in health-related quality of life among hemodialysis patients in the United States. Clin. J. Am. Soc. Nephrol. 2010, 5, 261–267. [Google Scholar] [CrossRef][Green Version]
- Nissenson, A.R. Improving outcomes for ESRD patients: Shifting the quality paradigm. Clin. J. Am. Soc. Nephrol. 2014, 9, 430–434. [Google Scholar] [CrossRef]
- Sualeheen, A.; Khor, B.H.; Balasubramanian, G.V.; Sahathevan, S.; Ali, M.S.M.; Narayanan, S.S.; Chinna, K.; Daud, Z.A.M.; Khosla, P.; Gafor, A.H.A.; et al. Habitual Dietary Patterns of Patients on Hemodialysis Indicate Nutritional Risk. J. Ren. Nutr. 2019. [Google Scholar] [CrossRef]
| Pattern 1 (hiSSB) | Pattern 2 (loSSB) | ||
|---|---|---|---|
| Food Groups | Mean (SD) %TE Food | p | |
| Sugar sweetened beverages | 27.97 ± 9.27 | 9.45 ± 5.65 | <0.001 |
| Unprocessed red meat | 0.95 ± 1.49 | 2.17 ± 3.49 | 0.022 |
| Poultry | 4.67 ± 5.99 | 2.51 ± 2.47 | 0.024 |
| Fish and shellfish | 0.49 ± 0.93 | 1.42 ± 2.87 | 0.028 |
| Puddings, ice cream, cheesecake | 0.24 ± 0.78 | 0.76 ± 1.70 | 0.049 |
| Processed and cured meats (bacon, sausage, hot dogs) | 2.67 ± 2.47 | 3.62 ± 3.39 | 0.106 |
| Dairy, low-fat and 2% | 1.75 ± 4.61 | 0.59 ± 2.30 | 0.122 |
| Egg and egg dishes | 2.43 ± 2.60 | 3.29 ± 3.08 | 0.132 |
| Vegetables, canned, fresh and frozen | 3.25 ± 4.20 | 4.45 ± 4.11 | 0.151 |
| Fast foods, frozen and convenience entrees | 4.46 ± 6.67 | 2.92 ± 4.87 | 0.194 |
| Pizza, pasta and lasagna | 2.77 ± 4.95 | 4.25 ± 6.58 | 0.200 |
| Butter, margarine, animal fats | 0.25 ± 0.41 | 0.41± 0.95 | 0.256 |
| Potatoes, mashed and salad | 0.87 ± 2.16 | 1.39 ± 2.55 | 0.270 |
| Beans and legumes | 0.43 ± 1.25 | 0.75 ± 1.92 | 0.315 |
| Potatoes, fried and hash browns | 1.39 ± 2.26 | 0.96 ± 2.18 | 0.330 |
| Fruit, canned, fresh and dried | 2.02 ± 4.61 | 1.28 ± 2.53 | 0.335 |
| Oils (vegetable, olive, canola) | 0.03 ± 0.09 | 0.05 ± 0.20 | 0.398 |
| Crackers, chips and popcorn | 2.12 ± 4.80 | 1.60 ± 2.57 | 0.509 |
| Candy | 0.47 ± 1.11 | 0.34 ± 1.01 | 0.542 |
| Sauce and condiments, savory | 0.83 ± 1.50 | 0.69 ± 0.92 | 0.597 |
| Nuts and seeds and nut butters | 0.24 ± 0.84 | 0.17 ± 0.86 | 0.665 |
| Dairy, full-fat and creamer | 1.00 ± 3.00 | 0.75 ± 3.18 | 0.691 |
| Cakes, cookies, pie, donuts, and rich dough | 1.71 ± 2.34 | 1.78 ± 2.70 | 0.885 |
| Grains | 6.38 ± 8.63 | 6.62 ± 8.26 | 0.888 |
| Pork | 0.71 ± 2.22 | 0.76 ± 1.65 | 0.889 |
| Sauces and condiments, sweet | 0.40 ± 0.62 | 0.39 ± 0.68 | 0.945 |
| All (n = 100 †) | hiSSB (n = 47) | loSSB (n = 53) | p Value between Groups | |
|---|---|---|---|---|
| Age, year | 60 (53–68) | 59 ± 12 | 60 ± 14 | 0.662 |
| Ethnicity | ||||
| African American, n (%) | 100 (100) | 47 (47) | 54 (53) | --- |
| Males, n (%) | 59 (59) | 29 (29) | 30 (30) | 0.605 |
| BMI, kg/m2 | 27.4 (23.3–31.4) | 29.7 ± 6.7 | 26.9 ± 6.0 | 0.029 |
| BMI Category, n(%) | ||||
| Underweight (BMI < 18.5) | 3 (3) | 1 (1) | 2(2) | 0.028 |
| Normal Weight (18.5–24.9) | 31 (31) | 8 (8) | 23 (23) | |
| Overweight (BMI 25–29.9) | 31 (31) | 19 (19) | 12 (12) | |
| Obese (BMI > 30) | 35 (35) | 19 (19) | 16 (16) | |
| Vintage (months) | 45 (19–88) | 62 ± 57 | 66 ± 68 | 0.770 |
| Cause of Kidney Failure | ||||
| Diabetes Mellitus, n (%) | 47 (47) | 24 (24) | 23 (23) | 0.663 |
| Hypertension, n (%) | 37 (37) | 18 (18) | 19 (19) | |
| Glomerulonephritis, n (%) | 5 (5) | 2 (2) | 3 (3) | |
| SLE, n (%) | 2 (2) | 0 (0) | 2 (2) | |
| HIV-Nephropathy, n (%) | 2 (2) | 0 (0) | 2 (2) | |
| Others, n (%) | 5 (5) | 2 (2) | 3 (3) | |
| Unknown, n (%) | 2 (2) | 1 (1) | 1 (1) | |
| Insulin Use, n (%) | 30 (30) | 20 (20) | 10 (10) | 0.010 |
| Oral Hypoglycemic Agent Use, n (%) | 8 (8) | 2 (2) | 6 (6) | 0.194 |
| Tobacco Use, n (%) | 29 (29) | 14 (14) | 15 (15) | 0.870 |
| Vascular Access | ||||
| Arteriovenous fistula, n (%) | 59 (59) | 28 (28) | 31 (31) | 0.993 |
| Arteriovenous graft, n (%) | 26 (26) | 12 (12) | 15 (14) | |
| Catheter, n (%) | 15 (15) | 7 (7) | 8 (8) | |
| Blood Pressure, mmHg (post-sitting) | ||||
| 1 Systolic | 138 (118–155) | 141 ± 24 | 139 ± 21 | 0.620 |
| 1 Diastolic | 77 (70–83) | 78 ± 10 | 80 ± 22 | 0.483 |
| 2 Kt/V | 1.5 (1.4–1.6) | 1.5 ± 0.2 | 1.6 ± 0.3 | 0.084 |
| Antidepressant use, % | 17 (17) | 12 (12) | 5 (5) | 0.032 |
| Renal vitamin * use, % | 56 (56) | 24 (24) | 32 (32) | 0.349 |
| Serum Potassium (mEq/L) | 4.6 (4.1–5.1) | 4.7 ± 0.6 | 4.6 ± 0.6 | 0.632 |
| Serum Phosphorus (mg/dL) | 5.0 (4.3–5.8) | 5.2 ± 1.5 | 5.0 ± 1.0 | 0.285 |
| 3 Serum Albumin (g/dL) | 3.8 (3.6–4.0) | 3.8 ± 0.4 | 3.8 ± 0.3 | 0.948 |
| 4 CRP (mg/L) | 6.1 (3.1–8.4) | 6.2 ± 4.8 | 6.0 ± 3.2 | 0.745 |
| 4 IL-6 (pg/mL) | 1.7 (0.01–5.3) | 10.9 ± 36.7 | 13.2 ± 54.4 | 0.799 |
| 5 IL-18 (pg/mL) | 238 (172–320) | 276 ± 146 | 259 ± 163 | 0.594 |
| 4 MCP-1 (pg/mL) | 113 (89–160) | 155 ± 127 | 134 ± 97 | 0.373 |
| Total Cholesterol (mg/dL) | 148 (113–188) | 147 ± 41 | 154 ± 47 | 0.471 |
| Triglycerides (mg/dL) | 81 (53–125) | 106 ± 52 | 87 ± 51 | 0.069 |
| LDL-C (mg/dL) | 76 (46–104) | 80 ± 34 | 80 ± 45 | 0.987 |
| HDL-C (mg/dL) | 47 (39–62) | 46 ± 16 | 56 ± 21 | 0.007 |
| Large HDL (mg/dL) | 17.0 (11.0–31.8) | 17.9 ± 12.7 | 27.2 ± 17.8 | 0.003 |
| Intermediate HDL (mg/dL) | 23.0 (18.3–26.0) | 21.7 ± 5.4 | 23.8 ± 6.1 | 0.081 |
| Small HDL (mg/dL) | 6.0 (4.0–8.0) | 6.7 ± 3.1 | 5.5 ± 3.4 | 0.076 |
| 6 Large LDL(mg/dL) | 20.5 (14.0–28.0) | 20.4 ± 9.2 | 23.2 ± 10.1 | 0.154 |
| 6 Intermediate LDL (mg/dL) | 11.0 (7.0–16.0) | 13.7 ± 7.8 | 11.8 ± 8.4 | 0.251 |
| 6 Small LDL (mg/dL) | 2.0 (0.0–5.3) | 4.9 ± 6.1 | 3.1 ± 4.8 | 0.103 |
| 6 Mean LDL Size (Å) | 270.0 (266.0–273.0) | 268.0 ± 4.6 | 270.3 ± 4.1 | 0.009 |
| LDL Pattern A, n (%) | 62 (61.4) | 22 (21.8) | 40 (39.6) | 0.050 |
| LDL Pattern B, n (%) | 16 (15.8) | 10 (9.9) | 6 (5.9) | 0.050 |
| Nutrient | All (n = 100 †) | hiSSB (n = 47) | loSSB (n = 53) | P Value between Groups | Nutrient Recommendations |
|---|---|---|---|---|---|
| Energy, kcals | 2027 ± 414 | 2123 ± 432 | 1941 ± 381 | 0.029 | Per renal Rx |
| Protein, g | 86 ± 26 | 83 ± 21 | 88 ± 29 | 0.311 | Per renal Rx |
| % kcals from protein | 17 ± 4 | 16 ± 4 | 18 ± 4 | 0.008 | 10–35 * |
| Fat, g | 90 ± 24 | 89 ± 25 | 91 ± 24 | 0.713 | |
| % kcals from fat | 40 ± 7 | 38 ± 6 | 42 ± 7 | 0.001 | 20–35 * |
| CHO, g | 218 ± 65 | 249 ± 68 | 191 ± 50 | <0.001 | |
| % kcals from CHO | 43 ± 8 | 47 ± 7 | 40 ± 7 | <0.001 | 45–65 * |
| Fiber, g | 12 ± 4 | 13.2 ± 3.9 | 11.8 ± 4.5 | 0.095 | 30 * |
| Sugars, g | 87 ± 45 | 115 ± 47 | 63 ± 23 | <0.001 | <50 ** |
| Phosphorus, mg | 833 ± 303 | 797 ± 260 | 864 ± 335 | 0.263 | Per renal Rx |
| Iron, g | 11.5 ± 4.5 | 11.3 ± 4.1 | 11.6 ± 4.9 | 0.800 | Per renal Rx |
| Magnesium, mg | 149 ± 64 | 150 ± 65 | 147 ± 64 | 0.852 | 420 * |
| Sodium, mg | 2996 ± 961 | 3036 ± 1013 | 2960 ± 921 | 0.695 | <2300 ** |
| Potassium, mg | 1500 ± 585 | 1487 ± 636 | 1512 ± 542 | 0.834 | Per renal Rx |
| Zinc, mg | 8.5 ± 4.1 | 7.4 ± 2.9 | 9.4 ± 4.8 | 0.017 | 11 * |
| Vitamin C, mg | 58 ± 46 | 69 ± 55 | 48 ± 33 | 0.023 | 90 * |
| Vitamin E, mg | 4.3 ± 3.1 | 4.4 ± 3.4 | 4.1 ± 2.9 | 0.628 | 15 * |
| Chromium (µg) | 4.4 ± 6.9 | 2.9 ± 4.5 | 5.7 ± 8.2 | 0.031 | 30 * |
| Selenium (µg) | 88.2 ± 37.8 | 79.6 ± 35.3 | 95.8 ± 38.7 | 0.023 | 55 * |
| Folic acid, mg | 225 ± 108 | 230 ± 112 | 221 ± 105 | 0.707 | 400 * |
| Cholesterol, mg | 412 ± 177 | 368 ± 153 | 452 ± 188 | 0.016 | Per renal Rx or (<200) [28] |
| USDA My plate recommendations (%) | |||||
| Grain | 76 ± 33 | 75 ± 37 | 77 ± 30 | 0.749 | Based on age and gender [29] |
| Vegetable | 33 ± 27 | 30 ± 25 | 35 ± 28 | 0.369 | |
| Fruit | 24 ± 33 | 32 ± 41 | 16 ± 21 | 0.023 | |
| Dairy | 13 ± 14 | 15 ± 14 | 12 ± 15 | 0.306 | |
| Protein | 148 ± 64 | 132 ± 59 | 162 ± 65 | 0.019 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tallman, D.A.; Latifi, E.; Kaur, D.; Sulaheen, A.; Ikizler, T.A.; Chinna, K.; Mat Daud, Z.A.; Karupaiah, T.; Khosla, P. Dietary Patterns and Health Outcomes among African American Maintenance Hemodialysis Patients. Nutrients 2020, 12, 797. https://doi.org/10.3390/nu12030797
Tallman DA, Latifi E, Kaur D, Sulaheen A, Ikizler TA, Chinna K, Mat Daud ZA, Karupaiah T, Khosla P. Dietary Patterns and Health Outcomes among African American Maintenance Hemodialysis Patients. Nutrients. 2020; 12(3):797. https://doi.org/10.3390/nu12030797
Chicago/Turabian StyleTallman, Dina A., Eno Latifi, Deepinder Kaur, Ayesha Sulaheen, T. Alp Ikizler, Karuthan Chinna, Zulfitri Azuan Mat Daud, Tilakavati Karupaiah, and Pramod Khosla. 2020. "Dietary Patterns and Health Outcomes among African American Maintenance Hemodialysis Patients" Nutrients 12, no. 3: 797. https://doi.org/10.3390/nu12030797
APA StyleTallman, D. A., Latifi, E., Kaur, D., Sulaheen, A., Ikizler, T. A., Chinna, K., Mat Daud, Z. A., Karupaiah, T., & Khosla, P. (2020). Dietary Patterns and Health Outcomes among African American Maintenance Hemodialysis Patients. Nutrients, 12(3), 797. https://doi.org/10.3390/nu12030797







