Micronutrient Deficiencies Following Minimally Invasive Esophagectomy for Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Search of the Literature
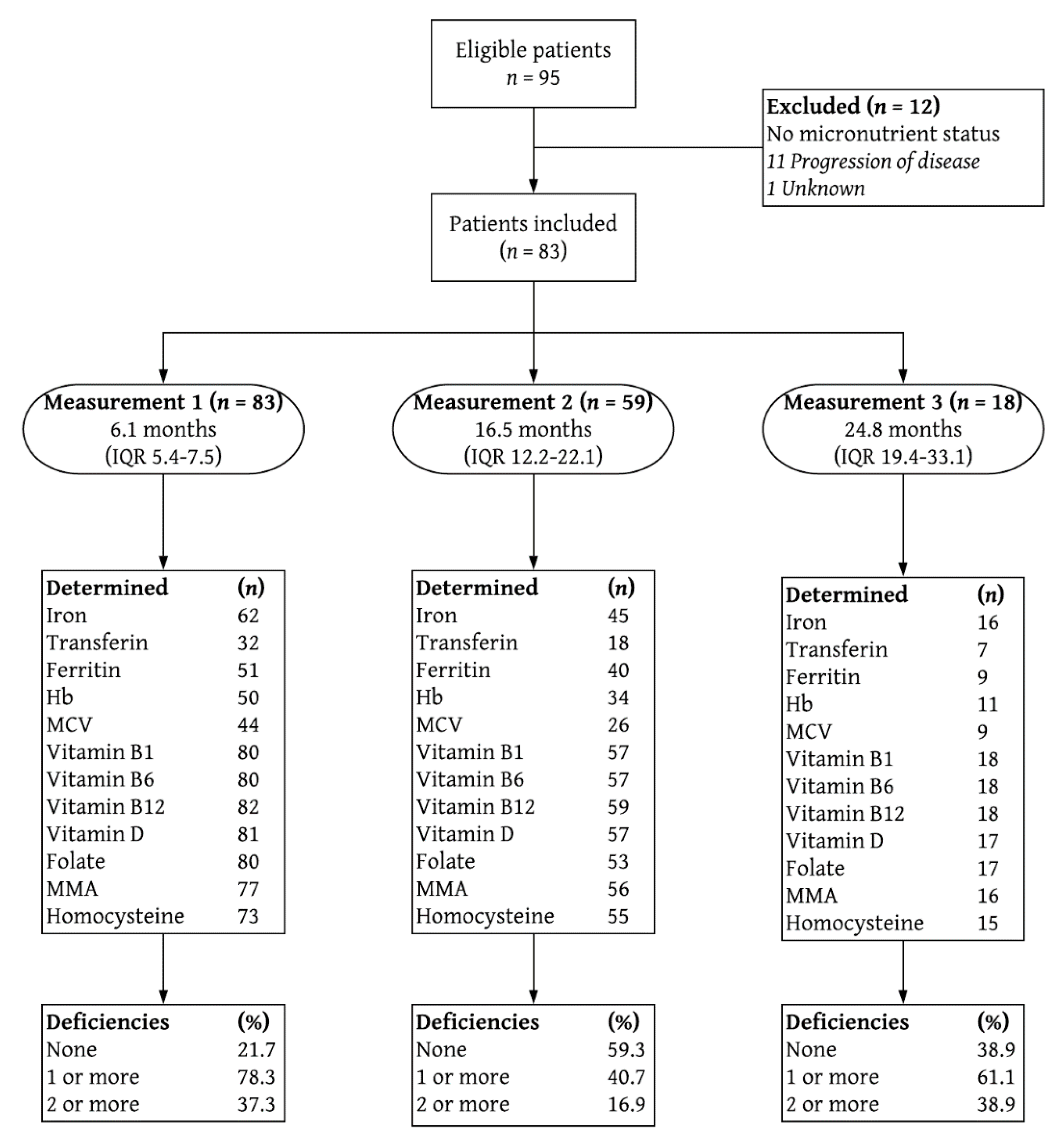
2.2. Study Design and Cohort
2.3. Definitions
2.4. Statistical Analysis
3. Results
3.1. Descriptive Literature Review
3.2. Characteristics of the Study Group
3.3. Trends of Micronutrient Values Following an Esophagectomy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Faiz, Z.; Lemmens, V.E.; Siersema, P.D.; Nieuwenhuijzen, G.A.; Wouters, M.W.; Rozema, T.; Coebergh, J.W.; Wijnhoven, B.P. Increased resection rates and survival among patients aged 75 years and older with esophageal cancer: A Dutch nationwide population-based study. World J. Surg. 2012, 36, 2872–2878. [Google Scholar] [CrossRef] [PubMed]
- Njei, B.; McCarty, T.R.; Birk, J.W. Trends in esophageal cancer survival in United States adults from 1973 to 2009: A SEER database analysis. J. Gastroenterol. Hepatol. 2016, 31, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, K.; Rouvelas, I.; Tsai, J.A.; Mariosa, D.; Lind, P.A.; Lindblad, M.; Ye, W.; Lundell, L.; Schuhmacher, C.; Mauer, M.; et al. Survival benefit and additional value of preoperative chemoradiotherapy in resectable gastric and gastro-oesophageal junction cancer: A direct and adjusted indirect comparison meta-analysis. Eur. J. Surg. Oncol. 2015, 41, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Straatman, J.; van der Wielen, N.; Cuesta, M.A.; Daams, F.; Roig Garcia, J.; Bonavina, L.; Rosman, C.; van Berge Henegouwen, M.I.; Gisbertz, S.S.; van der Peet, D.L. Minimally Invasive Versus Open Esophageal Resection: Three-year Follow-up of the Previously Reported Randomized Controlled Trial: The TIME Trial. Ann. Surg. 2017, 266, 232–236. [Google Scholar] [CrossRef]
- Luketich, J.D.; Pennathur, A.; Awais, O.; Levy, R.M.; Keeley, S.; Shende, M.; Christie, N.A.; Weksler, B.; Landreneau, R.J.; Abbas, G.; et al. Outcomes after minimally invasive esophagectomy: Review of over 1000 patients. Ann. Surg. 2012, 256, 95–103. [Google Scholar] [CrossRef]
- Treitl, D.; Hurtado, M.; Ben-David, K. Minimally Invasive Esophagectomy: A New Era of Surgical Resection. J. Laparoendosc. Adv. Surg. Tech. A 2016, 26, 276–280. [Google Scholar] [CrossRef]
- Berkelmans, G.H.; van Workum, F.; Weijs, T.J.; Nieuwenhuijzen, G.A.; Ruurda, J.P.; Kouwenhoven, E.A.; van Det, M.J.; Rosman, C.; van Hillegersberg, R.; Luyer, M.D. The feeding route after esophagectomy: A review of literature. J. Thorac. Dis. 2017, 9, S785–S791. [Google Scholar] [CrossRef]
- Findlay, J.M.; Gillies, R.S.; Millo, J.; Sgromo, B.; Marshall, R.E.; Maynard, N.D. Enhanced recovery for esophagectomy: A systematic review and evidence-based guidelines. Ann. Surg. 2014, 259, 413–431. [Google Scholar] [CrossRef]
- Greene, C.L.; DeMeester, S.R.; Worrell, S.G.; Oh, D.S.; Hagen, J.A.; DeMeester, T.R. Alimentary satisfaction, gastrointestinal symptoms, and quality of life 10 or more years after esophagectomy with gastric pull-up. J. Thorac. Cardiovasc. Surg. 2014, 147, 909–914. [Google Scholar] [CrossRef]
- Jacobs, M.; Macefield, R.C.; Elbers, R.G.; Sitnikova, K.; Korfage, I.J.; Smets, E.M.; Henselmans, I.; van Berge Henegouwen, M.I.; de Haes, J.C.; Blazeby, J.M.; et al. Meta-analysis shows clinically relevant and long-lasting deterioration in health-related quality of life after esophageal cancer surgery. Qual. Life Res. 2014, 23, 1097–1115. [Google Scholar] [CrossRef]
- Djarv, T.; Lagergren, P. Quality of life after esophagectomy for cancer. Expert Rev. Gastroenterol. Hepatol. 2012, 6, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Djarv, T.; Lagergren, J.; Blazeby, J.M.; Lagergren, P. Long-term health-related quality of life following surgery for oesophageal cancer. Br. J. Surg. 2008, 95, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.; Stier, C.; Raab, H.; Weiner, R. Review article: The nutritional and pharmacological consequences of obesity surgery. Aliment. Pharmacol. Ther. 2014, 40, 582–609. [Google Scholar] [CrossRef] [PubMed]
- Gudzune, K.A.; Huizinga, M.M.; Chang, H.Y.; Asamoah, V.; Gadgil, M.; Clark, J.M. Screening and diagnosis of micronutrient deficiencies before and after bariatric surgery. Obes. Surg. 2013, 23, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Gehrer, S.; Kern, B.; Peters, T.; Christoffel-Courtin, C.; Peterli, R. Fewer nutrient deficiencies after laparoscopic sleeve gastrectomy (LSG) than after laparoscopic Roux-Y-gastric bypass (LRYGB)-a prospective study. Obes. Surg. 2010, 20, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Levinson, R.; Silverman, J.B.; Catella, J.G.; Rybak, I.; Jolin, H.; Isom, K. Pharmacotherapy prevention and management of nutritional deficiencies post Roux-en-Y gastric bypass. Obes. Surg. 2013, 23, 992–1000. [Google Scholar] [CrossRef]
- Brzozowska, M.M.; Sainsbury, A.; Eisman, J.A.; Baldock, P.A.; Center, J.R. Bariatric surgery, bone loss, obesity and possible mechanisms. Obes. Rev. 2013, 14, 52–67. [Google Scholar] [CrossRef]
- Kim, J.H.; Bae, Y.J.; Jun, K.H.; Chin, H.M. Long-Term Trends in Hematological and Nutritional Status after Gastrectomy for Gastric Cancer. J. Gastrointest. Surg. 2017, 21, 1212–1219. [Google Scholar] [CrossRef]
- Lim, C.H.; Kim, S.W.; Kim, W.C.; Kim, J.S.; Cho, Y.K.; Park, J.M.; Lee, I.S.; Choi, M.G.; Song, K.Y.; Jeon, H.M.; et al. Anemia after gastrectomy for early gastric cancer: Long-term follow-up observational study. World J. Gastroenterol. 2012, 18, 6114–6119. [Google Scholar] [CrossRef]
- Hu, Y.; Kim, H.I.; Hyung, W.J.; Song, K.J.; Lee, J.H.; Kim, Y.M.; Noh, S.H. Vitamin B(12) deficiency after gastrectomy for gastric cancer: An analysis of clinical patterns and risk factors. Ann. Surg. 2013, 258, 970–975. [Google Scholar] [CrossRef]
- Rino, Y.; Oshima, T.; Yoshikawa, T. Changes in fat-soluble vitamin levels after gastrectomy for gastric cancer. Surg. Today 2017, 47, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Berkelmans, G.H.K.; Fransen, L.F.C.; Dolmans-Zwartjes, A.C.P.; Kouwenhoven, E.A.; van Det, M.J.; Nilsson, M.; Nieuwenhuijzen, G.A.P.; Luyer, M.D.P. Direct Oral Feeding Following Minimally Invasive Esophagectomy (NUTRIENT II trial): An International, Multicenter, Open-label Randomized Controlled Trial. Ann. Surg. 2019. [Google Scholar] [CrossRef]
- Snow, C.F. Laboratory diagnosis of vitamin B12 and folate deficiency: A guide for the primary care physician. Arch. Intern. Med. 1999, 159, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Hankey, G.J.; Eikelboom, J.W. Homocysteine and vascular disease. Lancet 1999, 354, 407–413. [Google Scholar] [CrossRef]
- Van Hagen, P.; de Jonge, R.; van Berge Henegouwen, M.I.; Hotte, G.J.; van der Stok, E.P.; Lindemans, J.; van Lanschot, J.J.B.; Wijnhoven, B.P.L. Vitamin B12 deficiency after esophagectomy with gastric tube reconstruction for esophageal cancer. Dis. Esophagus 2017, 30, 1–8. [Google Scholar] [CrossRef]
- Heneghan, H.M.; Zaborowski, A.; Fanning, M.; McHugh, A.; Doyle, S.; Moore, J.; Ravi, N.; Reynolds, J.V. Prospective Study of Malabsorption and Malnutrition After Esophageal and Gastric Cancer Surgery. Ann. Surg. 2015, 262, 803–807. [Google Scholar] [CrossRef]
- Elliott, J.A.; Casey, S.; Murphy, C.F.; Docherty, N.G.; Ravi, N.; Beddy, P.; Reynolds, J.V.; le Roux, C.W. Risk factors for loss of bone mineral density after curative esophagectomy. Arch. Osteoporos. 2019, 14, 6. [Google Scholar] [CrossRef]
- Allen, L.H. Causes of vitamin B12 and folate deficiency. Food Nutr. Bull. 2008, 29, S20–S34. [Google Scholar] [CrossRef]
- Palacios, C.; Gonzalez, L. Is vitamin D deficiency a major global public health problem? J. Steroid Biochem. Mol. Biol. 2014, 144, 138–145. [Google Scholar] [CrossRef]
- Lopez, A.; Cacoub, P.; Macdougall, I.C.; Peyrin-Biroulet, L. Iron deficiency anaemia. Lancet 2016, 387, 907–916. [Google Scholar] [CrossRef]
| Author | Country | Year | Study Period | Study Design | Study population | Type of Surgery | Follow-up Period | Micronutrient Assessment | Prevalence of Deficiencies after Surgery | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Size | Age | Gender | |||||||||||||
| n | Years | (SD) or IQR | Male | % | Esophagectomy | Gastrectomy | Months | [IQR] | |||||||
| van Hagen et al. | Netherlands | 2017 | August 2010–July 2012 | Group A: single center, cross-sectional cohort study | 99 | 62 | 38–79 | 73 | 74 | 99 | – | 19.4 | [13.5–30.1] | Vitamin B12 | 11% |
| Group B: double center, prospective cohort study | 88 | 63 | 19–79 | 63 | 72 | 88 | – | 6.4 | [1.6–10.8] | Vitamin B12 | 10.2% | ||||
| Heneghan et al. | Ireland | 2015 | January 2013–July 2013 | Single center, prospective cohort study | 45 | 63.3 | (8.9) | 30 | 68 | 30 | 15 | 23.1 | [18,19,20,21,22,23,24,25,26,27,28] | Vitamin A Vitamin B12 Vitamin D Vitamin E Iron | 81.5% missing 44.4% 61.5% 46–49% |
| Elliot et al. | Ireland | 2019 | 2000–2014 | Single center, retrospective cohort study | 75 | 60.6 | (9.6) | 58 | 77 | 75 | – | 43.4 | Missing | Vitamin A Vitamin D Vitamin E | missing 53%–70% † missing |
| Clinical Characteristics | (n = 83) | |
|---|---|---|
| Sex (male) | 66 | (79.5) |
| Age at inclusion (SD) | 65 | (7.7) |
| ASA score | ||
| I | 7 | (8.4) |
| II | 63 | (75.9) |
| III | 13 | (15.7) |
| Preoperative BMI | 25.3 | (23.6–28.7) |
| Alcohol use (at inclusion) | ||
| No | 18 | (21.7) |
| Daily | 46 | (55.4) |
| Weekly | 5 | (6.0) |
| Monthly | 14 | (16.9) |
| Any comorbidity | 54 | (65.1) |
| pTNM stage | ||
| Stage 0 | 29 | (34.9) |
| Stage I | 20 | (24.1) |
| Stage II | 19 | (22.9) |
| Stage III | 15 | (18.1) |
| Histology type | ||
| Adenocarcinoma | 68 | (81.9) |
| Squamous-cell carcinoma | 15 | (18.1) |
| Tumor localization | ||
| Mid esophagus | 4 | (4.8) |
| Distal esophagus | 51 | (61.4) |
| Esophagogastric junction | 28 | (33.7) |
| Neo-adjuvant treatment | 76 | (91.6) |
| NUTRIENT II treatment allocation | ||
| Direct oral feeding | 42 | (50.6) |
| Standard of care | 41 | (49.4) |
| Any 30-day postoperative complication | 63 | (75.9) |
| BMI at discharge | 25.0 | (23.5–28.7) |
| BMI 3 months postoperatively | 23.3 | (22.0–26.9) |
| Measurement 1 | Measurement 2 | Measurement 3 | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 6.1 Months (5.4–7.5) | 16.5 Months (12.2–22.1) | 24.8 Months (19.4–33.1) | |||||||||||||||||
| Micronutrient | Normal Value | Total | Abnormal Value | Total | Abnormal Value | Total | Abnormal Value | ||||||||||||
| Standard | (n) | Value | (%) | Value | (n) | Value | (%) | Value | (n) | Value | (%) | Value | |||||||
| Vitamin B1 | 70–200 nmol/L | 80 | 132 | (31) | – | – | – | 57 | 137 | (37) | – | – | – | 18 | 136 | (31) | 5.6 | 68 | – |
| Vitamin B6 | 35–110 nmol/L | 80 | 75 | (58–95) | – | – | – | 57 | 81 | (64–110) | – | – | – | 18 | 67 | (54–90) | – | – | – |
| Vitamin B12 | 140–700 pmol/L | 82 | 240 | (200–323) | 6.1 | 110 | (89–130) | 59 | 310 | (220–470) | 1.7 | 130 | – | 18 | 240 | (210–410) | – | – | – |
| Vitamin D | >50 nmol/L | 81 | 48 | (36–68) | 50.6 | 36 | (29–42) | 57 | 63 | (55–74) | 19.3 | 34 | (33–41) | 17 | 63 | (55–77) | 11.8 | 47 | (45–49) |
| Folate | >10 nmol/L | 80 | 14 | (10–22) | 28.8 | 7 | (6–9) | 53 | 20 | (13–35) | 20.8 | 8 | (7–9) | 17 | 14 | (10–24) | 23.5 | 7 | (5–8) |
| MMA (↑) | 0–430 nmol/L | 77 | 230 | (186–302) | 14.3 | 569 | (482–692) | 56 | 195 | (168–246) | 5.4 | 475 | (470–611) | 16 | 220 | (173–403) | 12.5 | 721 | (667–775) |
| Homocysteine (↑) | <15 μmol/L | 73 | 13 | (11–16) | 35.6 | 18 | (16–20) | 55 | 12 | (10–14) | 21.8 | 18 | (16–20) | 15 | 13 | (10–20) | 33.3 | 21 | (18–24) |
| On indication | |||||||||||||||||||
| Hb | (♂) 8.5–11.0 mmol/L | 50 | 8.6 | (8.0–8.9) | 32 | 7.7 | (7.2–8.3) | 34 | 8.6 | (8.1–8.9) | 26.5 | 7.9 | (7.7–8.3) | 11 | 8.7 | (7.7–9.3) | 18.2 | 7.6 | (7.2–7.9) |
| (♀) 7.5–10.0 mmol/L | 8.4 | (8.0–8.6) | – | – | – | 8.7 | (7.9–9.1) | – | – | – | 8.4 | (7.5–8.7) | 9.1 | 6.7 | – | ||||
| MCV (↑) | 80–100 fL | 44 | 91 | (87–93) | 4.5 | 80 | (79–80) | 26 | 93 | (89–96) | 11.5 | 103 | (102–108) | 9 | 90 | (88–91) | 11.1 | 68 | – |
| MCV (↓) | 4.5 | 101 | (100–101) | – | – | – | 11.1 | 101 | – | ||||||||||
| Iron | 14–35 μmol/L | 59 | 14 | (12–17) | 35.5 | 10 | (8–13) | 45 | 16 | (14–20) | 17.8 | 9 | (7–12) | 16 | 16 | (12–19) | 25 | 10 | (5–12) |
| Transferrin | 2.2–3.6 g/L | 32 | 2.6 | (2.2–2.9) | 18.8 | 2.1 | (2.0–2.2) | 18 | 2.5 | (2.1–2.8) | 33.3 | 2.0 | (1.6–2.1) | 7 | 2.8 | (2.0–3.3) | 28.6 | 2.0 | (1.9–2.0) |
| Ferritin | 30–400 μg/L | 51 | 76 | (32–180) | 19.6 | 16 | (14–23) | 40 | 94 | (33–169) | 20 | 14 | (11–24) | 9 | 32 | (25–100) | 22.2 | 19 | (13–25) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janssen, H.J.B.; Fransen, L.F.C.; Ponten, J.E.H.; Nieuwenhuijzen, G.A.P.; Luyer, M.D.P. Micronutrient Deficiencies Following Minimally Invasive Esophagectomy for Cancer. Nutrients 2020, 12, 778. https://doi.org/10.3390/nu12030778
Janssen HJB, Fransen LFC, Ponten JEH, Nieuwenhuijzen GAP, Luyer MDP. Micronutrient Deficiencies Following Minimally Invasive Esophagectomy for Cancer. Nutrients. 2020; 12(3):778. https://doi.org/10.3390/nu12030778
Chicago/Turabian StyleJanssen, Henricus J.B., Laura F.C. Fransen, Jeroen E.H. Ponten, Grard A.P. Nieuwenhuijzen, and Misha D.P. Luyer. 2020. "Micronutrient Deficiencies Following Minimally Invasive Esophagectomy for Cancer" Nutrients 12, no. 3: 778. https://doi.org/10.3390/nu12030778
APA StyleJanssen, H. J. B., Fransen, L. F. C., Ponten, J. E. H., Nieuwenhuijzen, G. A. P., & Luyer, M. D. P. (2020). Micronutrient Deficiencies Following Minimally Invasive Esophagectomy for Cancer. Nutrients, 12(3), 778. https://doi.org/10.3390/nu12030778






