The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review
Abstract
1. Introduction
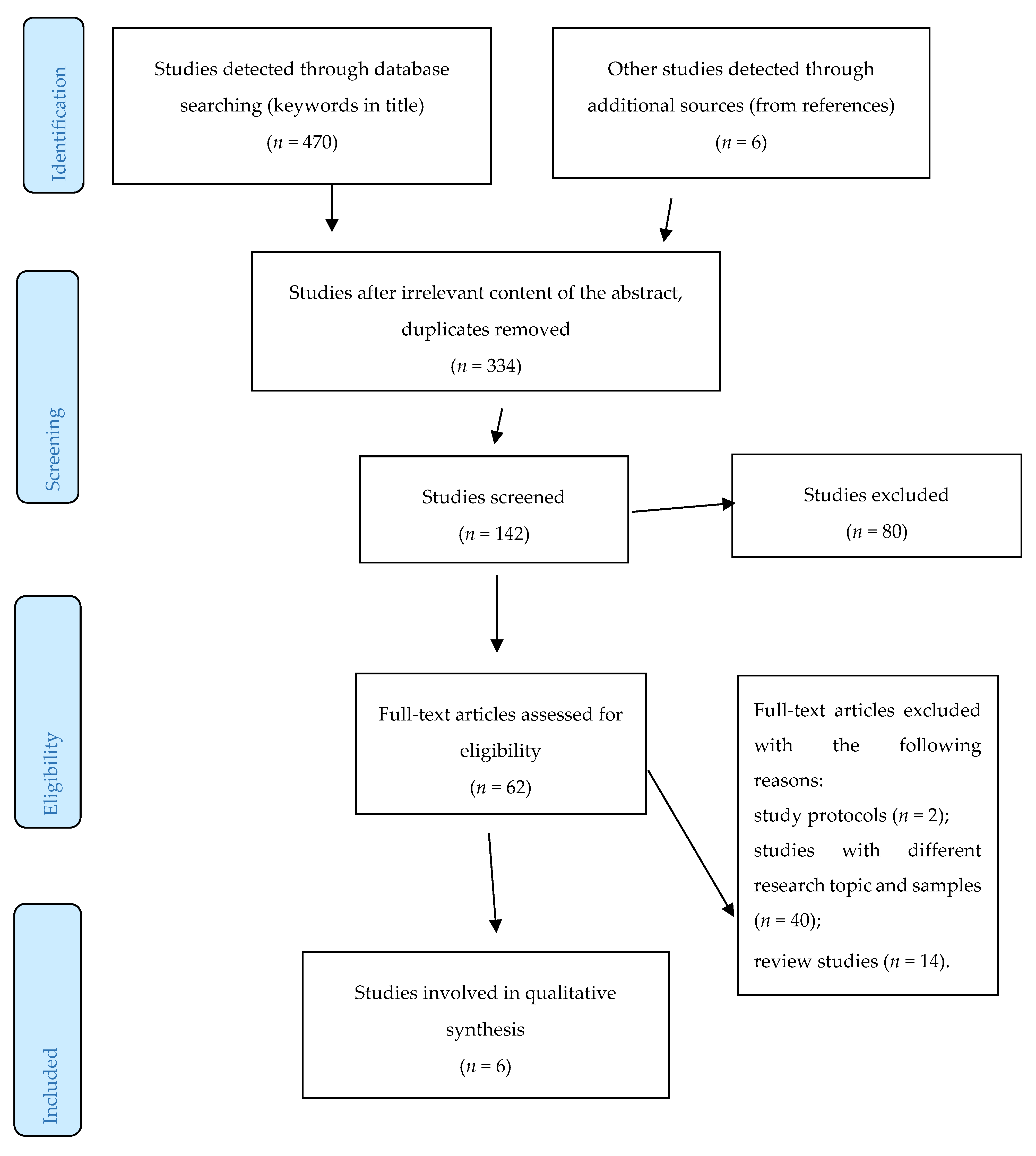
2. Methods
- to explore the impact of nutrition and intestinal microbiome on elderly depression;
- to discuss possible associations between nutrition, microbiome, and serotonin molecules and its tryptophan precursor.
3. Results
- The time span of the issuing the article was limited by January 1, 2013 up to December 31, 2018.
- Only peer-reviewed English-written full-text journal articles were involved.
- Only randomized controlled trials and experimental/cross-sectional studies were included.
- The primary outcome focused on the association of nutrition and depressive symptoms.
- The subjects had to be at the age 50+ with or without depression.
- The study protocols, e.g., [27], the studies focusing on other aging diseases in which depression was comorbidity, e.g., [28,29], the studies which contain subjects of all ages, e.g., [30,31], the studies which were aimed at dietary counselling as an intervention, e.g., [32], behavioral studies, e.g., [33], the studies with the reverse hypothesis, e.g., [34], and the review studies, e.g., [23,24,25], were also excluded.
| Author | Objective | Intervention Period, Type(s) of Nutrition | Number of Subjects | Main Outcome Measures | Findings | Limitations |
|---|---|---|---|---|---|---|
| Duffy et al. [35] RCT (Australia) | To explore the impact of ω-3 FA supplementation on in vivo GSH concentration in elderly people at risk for depression. | 12 weeks; four 1000-mg ω-3 fatty acid (FA) supplements daily (intervention g.), placebo (control). | 51 subjects, mean age: 72.2 years. | Magnetic resonance spectroscopy, medical, neuropsychological, and self-report assessments, Patient Health Questionnaire. | ω-3 FA supplementation can decrease oxidative stress mechanisms. | Not reported. |
| Grønning et al. [37] Cross-sectional study (Norway) | To examine the connections between psychological distress and diet patterns. | Irrelevant for this type of study. | 11,621 participants at the age of 65+ years. | Hospital Anxiety and Depression Scale, multivariable regression analyses. | Applying a healthy diet is connected with less anxiety and stress in older adults. | Questionnaires are all based on self-report, and the study has no objective measures of food intake or biological data. |
| Hussin et al. [36] RCT (Malaysia) | To explore dietary effectiveness in enhancing mood states and depression status in older adults. | Three months; reduction of 300–500 kcal/day. | 31 healthy males (Mean±SD), aged 59.7±6.3 years. | Profile of Mood States, Beck Depression Inventory-II, Geriatric Depression Scale, statistical analysis. | Fasting and calorie restriction dietary regime is efficient in enhancing mood states and nutritional status among older men (p<0.05). | Small sample size, short duration. |
| Miskulin et al. [38] Cross-sectional study (Croatia) | To determine the frequency of vitamin B12 deficiency among older adults and to assess whether there is an association between this deficiency and depressive symptoms in these people. | Three months; irrelevant for this type of study. | 140 subjects, mean age 71.0±6.7 years. | Questionnaire, competitive immunoassay vitamin B12 kit. | Depressive symptoms appeared in 100.0% (10/10) people with the vitamin B12 deficiency. | Not a randomized controlled trial (no assessment of causality), socio-economic status was not measured. |
| Nguyen et al. [39] Cross-sectional study (Japan) | To explore the association between vitamin consumption and depressive symptoms in Japanese adults. | Irrelevant for this type of study. | 1634 subjects at the age of 65+ years. | Self-administered questionnaires, interviews, comprehensive health examination, Geriatric Depression Scale, statistical analysis. | There are connections between vitamin deficiencies and depressive symptoms in women and overweight older adults. | Not an RCT (no assessment of causality), self-reported assessments might be bias. |
| Park et al. [40] Cross-sectional study (South Korea) | To examine the dominant and envisaging factors of depression in older Korean people. | Irrelevant for this type of study. | 258 subjects at the age of 65+ years. | Questionnaires, Geriatric Depression Scale, statistical analysis. | In men—deficient protein consumption, suffering from more chronic diseases; in women—deficient vitamin B6 consumption, lower cognitive functions, and higher social isolation. | The same as above + limited representativeness of the sample. |
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Mental Health of Older Adults. Available online: https://www.who.int/news-room/fact-sheets/detail/mental-health-of-older-adults (accessed on 23 January 2019).
- Maresova, P.; Klimova, B. Non-pharmacological approaches in the depression treatment—Strengths and weaknesses of mobile applications use. In Pervasive Computing Paradigms for Mental Health; Oliver, N., Serino, S., Matic, A., Cipresso, P., Filipovic, N., Gavrilovska, L., Eds.; MindCare 2016, FABULOUS 2016, IIOT 2015. Lecture Notes of the Institute for Computer Sciences, Social Informatics and Telecommunications Engineering 207; Springer: Cham, Switzerland, 2018. [Google Scholar]
- World Health Organization. Depression: Definition. Available online: http://www.euro.who.int/en/health-topics/noncommunicable-diseases/pages/news/news/2012/10/depression-in-europe/depession-definition (accessed on 23 January 2019).
- WebMD—Better Information. Better Health. Depression in Special Situations. Available online: https://www.webmd.com/depression/guide/depression-in-special-situations#1 (accessed on 23 January 2019).
- Rodda, J.; Carter, J. Depression in older adults. BMJ 2011, 343, d5219. [Google Scholar] [CrossRef] [PubMed]
- Klimova, B.; Maresova, P.; Kuca, K. Non-pharmacological approaches to the prevention and treatment of Alzheimer´s disease with respect to the rising treatment costs. Curr. Alzheimer Res. 2016, 13, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Yang, J.; Luo, A.; Hashimoto, K. Molecular and cellular mechanisms underlying the antidepressant effects of ketamine enantiomers and its metabolites. Transl. Psychiatry 2019, 9, 280. [Google Scholar] [CrossRef] [PubMed]
- Al-Rasheed, R.; Alrasheedi, R.; Al Johani, R.; AlRashidi, H.; Almaimany, B.; Alshalawi, B.; Kelantan, A.; Banjar, G.; Alzaher, A.; Alqadheb, A. Malnutrition in elderly and its relation to depression. Int. J. Community Med. Public Heal. 2018, 5, 2156–2160. [Google Scholar] [CrossRef]
- Cabout, M.; Brouwe, I.A.; Visser, M. The MooDFOOD project: Prevention of depression through nutritional strategies. Nutr. Bull. 2017, 42, 94–103. [Google Scholar] [CrossRef]
- Guligowska, A.; Pigłowska, M.; Fife, E.; Kostka, J.; Sołtysik, B.; Łukasz, K.; Kostka, T. Inappropriate nutrients intake is associated with lower functional status and inferior quality of life in older adults with depression. Clin. Interv. Aging 2016, 11, 1505–1517. [Google Scholar] [CrossRef]
- Sathyanarayana Rao, T.S.; Asha, M.R.; Ramesh, B.N.; Jagannatha, Rao, K.S. Understanding nutrition, depression and mental illnesses. Indian J. Psychiatry 2008, 50, 77–82. [Google Scholar] [CrossRef]
- Akbaraly, T.N.; Brunner, E.J.; Ferrie, J.E.; Marmot, M.G.; Kivimaki, M.; Singh-Manoux, A. Dietary pattern and depressive symptoms in middle age. Br. J. Psychiatry 2009, 195, 408–413. [Google Scholar] [CrossRef]
- Cheung, S.G.; Goldenthal, A.R.; Uhlemann, A.C.; Mann, J.J.; Miller, J.M.; Sublette, M.E. Systematic review of gut microbiota and major depression. Front. Psychiatry 2019, 10, 34. [Google Scholar] [CrossRef]
- Stower, H. Depression linked to the microbiome. Nat. Med. 2019, 25, 358. [Google Scholar] [CrossRef]
- Novotny, M.; Klimova, B.; Valis, M. Microbiome and cognitive impairment: Can any diets influence learning processes in a positive way? Front. Aging Neurosci. 2019, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. Regulation of the stress response by the gut microbiota: Implications for psychoneuroendocrinology. Psychoneuroendocrinology 2012, 37, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Marques, T.M.; Cryan, J.F.; Shanahan, F.; Fitzgerald, G.F.; Ross, R.P.; Dinan, T.G.; Stanton, C. Gut microbiota modulation and implications for host health: Dietary strategies to influence the gut–brain axis. Innov. Food Sci. Emerg. Technol. 2014, 22, 239–247. [Google Scholar] [CrossRef]
- Tsai, Y.-L.; Lin, T.-L.; Chang, C.-J.; Wu, T.-R.; Lai, W.-F.; Lu, C.-C.; Lai, H.-C. Probiotics, prebiotics and amelioration of diseases. J. Biomed. Sci. 2019, 26, 3. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Lepaslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.D.R.; Tap, J.; Bruls, T.; Batto, J.-M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Costea, P.I.; Hildebrand, F.; Arumugam, M.; Bäckhed, F.; Blaser, M.; Bushman, F.D.; De Vos, W.M.; Ehrlich, S.D.; Fraser, C.M.; Hattori, M.; et al. Enterotypes in the landscape of gut microbial community composition. Nat. Microbiol. 2018, 3, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kasper, L.H. The role of microbiome in central nervous system disorders. Brain Behav. Immun. 2014, 38, 1–12. [Google Scholar] [CrossRef]
- Foster, J.A.; McVey Neufeld, K.A. Gut-brain axis: How the microbiome influences anxiety and depression. Trends Neurosci. 2013, 36, 305–312. [Google Scholar] [CrossRef]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef]
- Phillips, R.M. Nutrition and depression in the community-based oldest-old. Home Healthc. Now 2012, 30, 462–471. [Google Scholar] [CrossRef]
- Tarleton, E. Depression and nutrition in the elderly. Digest 2014, 49, 1–9. [Google Scholar]
- Williamson, C. Dietary factors and depression in older people. Br. J. Community Nurs. 2009, 14, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Roca, M.; Kohls, E.; Gili, M.; Watkins, E.; Owens, M.; Hegerl, U.; Van Grootheest, G.; Bot, M.; Cabout, M.; Brouwer, I.; et al. Prevention of depression through nutritional strategies in high-risk persons: Rationale and design of the MooDFOOD prevention trial. BMC Psychiatry 2016, 16, 192. [Google Scholar] [CrossRef] [PubMed]
- Bitarafan, S.; Saboor-Yaraghi, A.; Sahraian, M.; Soltani, D.; Nafissi, S.; Togha, M. Effect of vitamin A supplementation on fatigue and depression in Multiple Sclerosis patients: A Double-Blind Placebo-Controlled Clinical Trial. Iran J. Allergy Asthma Immunol. 2016, 15, 13–19. [Google Scholar] [PubMed]
- De Koning, E.J.; Van Der Zwaluw, N.L.; Van Wijngaarden, J.P.; Sohl, E.; Brouwer-Brolsma, E.M.; Van Marwijk, H.; Enneman, A.W.; Swart, K.M.A.; Van Dijk, S.C.; Ham, A.C.; et al. Effects of two-year vitamin B12 and folic acid supplementation on depressive symptoms and quality of life in older adults with elevated homocysteine concentrations: Additional results from the B-PROOF study, an RCT. Nutrients 2016, 8, 748. [Google Scholar] [CrossRef] [PubMed]
- Kaner, G.; Soylu, M.; Yüksel, N.; Inanç, N.; Ongan, D.; Başmısırlı, E. Evaluation of nutritional status of patients with depression. Biomed. Res. Int. 2015, 2015, 521481. [Google Scholar] [CrossRef]
- Muss, C.; Mosgoeller, W.; Endler, T. Mood improving potential of a vitamin trace element composition—A randomized, double blind, placebo controlled clinical study with healthy volunteers. Neuro Endocrinol. Lett. 2016, 37, 18–28. [Google Scholar]
- acka, F.N.; O’Neil, A.; Opie, R.; Itsiopoulos, C.; Cotton, S.; Mohebbi, M.; Castle, D.; Dash, S.; Mihalopoulos, C.; Chatterton, M.L.; et al. A randomised controlled trial of dietary improvement for adults with major depression (the ‘SMILES’ trial). BMC Med. 2017, 15, 23. [Google Scholar]
- Tani, Y.; Sasaki, Y.; Haseda, M.; Kondo, K.; Kondo, N. Eating alone and depression in older men and women by cohabitation status: The JAGES longitudinal survey. Age Ageing 2015, 44, 1019–1026. [Google Scholar] [CrossRef]
- Gougeon, L.; Payette, H.; Morais, J.A.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K. A prospective evaluation of the depression-nutrient intake reverse causality hypothesis in a cohort of community-dwelling older Canadians. Br. J. Nutr. 2017, 117, 1032–1041. [Google Scholar] [CrossRef]
- Duffy, S.L.; Lagopoulos, J.; Cockayne, N.; Lewis, S.J.; Hickie, I.B.; Hermens, D.; Naismith, S. The effect of 12-wk ω-3 fatty acid supplementation on in vivo thalamus glutathione concentration in patients “at risk” for major depression. Nutrition 2015, 31, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Hussin, N.M.; Shahar, S.; Teng, N.I.; Ngah, W.Z.; Das, S.K. Efficacy of fasting and calorie restriction (FCR) on mood and depression among ageing men. J. Nutr. Health Aging 2013, 17, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Grønning, K.; Espnes, G.; Nguyen, C.; Rodrigues, A.M.; Gregório, M.J.; Sousa, R.; Canhão, H.; Andre, B. Psychological distress in elderly people is associated with diet, wellbeing, health status, social support and physical functioning- a HUNT3 study. BMC Geriatr. 2018, 18, 205. [Google Scholar] [CrossRef] [PubMed]
- Miškulin, M.; Kristić, M.; Vlahović, J. Vitamin B12 deficiency and depression in elderly: Cross-sectional study in Eastern Croatia. J. Health Sci. 2014, 4, 143–148. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Tsujiguchi, H.; Kambayashi, Y.; Hara, A.; Miyagi, S.; Yamada, Y.; Nakamura, H.; Shimizu, Y.; Hori, D.; Suzuki, F.; et al. Relationship between vitamin intake and depressive symptoms in elderly Japanese individuals: Differences with gender and body mass index. Nutrients 2017, 9, 1319. [Google Scholar] [CrossRef]
- Park, Y.H.; Choi-Kwon, S.; Park, K.A.; Suh, M.; Jung, Y.S. Nutrient deficiencies and depression in older adults according to sex: A cross sectional study. Nurs. Health Sci. 2017, 19, 88–94. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Apostolopoulos, V. The effects of vitamin B in depression. Curr. Med. Chem. 2016, 23, 4317–4337. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The effects of vitamin B on the immune/cytokine network and their involvement in depression. Maturitas 2017, 96, 58–71. [Google Scholar] [CrossRef]
- Black, C.N.; Bot, M.; Scheffer, P.G.; Cuijpers, P.; Penninx, B.W. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 2015, 51, 164–175. [Google Scholar] [CrossRef]
- Hashimoto, K. Role of Soluble Epoxide Hydrolase in Metabolism of PUFAs in Psychiatric and Neurological Disorders. Front. Pharm. 2019, 10, 36. [Google Scholar] [CrossRef]
- Ren, Q.; Ma, M.; Ishima, T.; Morisseau, C.; Yang, J.; Wagner, K.M.; Zhang, J.; Yang, C.; Yao, W.; Dong, C.; et al. Gene deficiency and pharmacological inhibition of soluble epoxide hydrolase confers resilience to repeated social defeat stress. Proc. Natl. Acad. Sci. USA 2016, 113, E1944–E1952. [Google Scholar] [CrossRef] [PubMed]
- Atone, J.; Wagner, K.; Hashimoto, K.; Hammock, B.D. Cytochrome P450 derived epoxidized fatty acids as a therapeutic tool against neuroinflammatory diseases. Prostaglandins Other Lipid Mediat. 2019, 147, 106385. [Google Scholar] [CrossRef] [PubMed]
- Stockmeier, C.A. Neurobiology of serotonin in depression and suicide. Ann. N. Y. Acad. Sci. 1997, 836, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.M. Depression, suicide and the metabolism of serotonin in the brain. J. Affect. Disord. 1983, 4, 275–290. [Google Scholar] [CrossRef]
- Gregório, M.J.; Rodrigues, A.M.; Eusébio, M.; De Sousa, R.D.; Dias, S.; André, B.; Grønning, K.; Coelho, P.S.; Mendes, J.M.; Graça, P.; et al. Dietary patterns characterized by high meat consumption are associated with other unhealthy life styles and depression symptoms. Front. Nutr. 2017, 4, 25. [Google Scholar] [CrossRef]
- WebMD—Better Information. Better Health. Depression in the Elderly. Available online: https://www.webmd.com/depression/guide/depression-elderly#1 (accessed on 23 January 2019).
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota regulation of tryptophan metabolism in health and disease. Cell Host Microbe 2018, 23, 716–724. [Google Scholar] [CrossRef]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef]
- Banskota, S.; Ghia, J.E.; Khan, W.I. Serotonin in the gut: Blessing or a curse. Biochimie 2019, 161, 56–64. [Google Scholar] [CrossRef]
- Mawe, G.M.; Hoffman, J.M. Serotonin signalling in the gut functions, dysfunctions and therapeutic targets. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 473–486. [Google Scholar] [CrossRef]
- Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of tryptophan and serotonin on mood and cognition with a possible role of the gut-brain axis. Nutrients 2016, 8, 56. [Google Scholar] [CrossRef]
- Xie, R.; Jiang, P.; Lin, L.; Jiang, J.; Yu, B.; Rao, J.; Liu, H.; Wei, W.; Qiao, Y. Oral treatment with Lactobacillus reuteri attenuates depressive-like behaviors and serotonin metabolism alterations induced by chronic social defeat stress. J. Psychiatr. Res. 2020, 122, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.; Myint, A.M.; Schwarz, M.J. The impact of neuroimmune dysregulation on neuroprotection and neurotoxicity in psychiatric disorders—Relation to drug treatment. Dialogues Clin. Neurosci. 2009, 11, 319–332. [Google Scholar] [PubMed]
- Kelly, J.R.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain-gut-microbiota axis: Challenges for translation in psychiatry. Ann. Epidemiol. 2016, 26, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Julio-Pieper, M.; Forsythe, P.; Kunze, W.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Communication between gastrointestinal bacteria and the nervous system. Curr. Opin. Pharm. 2012, 12, 667–672. [Google Scholar] [CrossRef]
- Wang, S.; Qu, Y.; Chang, L.; Pu, Y.; Zhang, K.; Hashimoto, K. Antibiotic-induced microbiome depletion is associated with resilience in mice after chronic social defeat stress. J. Affect. Disord. 2020, 260, 448–457. [Google Scholar] [CrossRef]
- Zhang, K.; Fujita, Y.; Chang, L.; Qu, Y.; Pu, Y.; Wang, S.; Shirayama, Y.; Hashimoto, K. Abnormal composition of gut microbiota is associated with resilience versus susceptibility to inescapable electric stress. Transl. Psychiatry 2019, 9, 231. [Google Scholar] [CrossRef]
- Collins, S.M.; Bercik, P. The relationship between intestinal microbiota and the central nervous system in normal gastrointestinal function and disease. Gastroenterology 2009, 136, 2003–2014. [Google Scholar] [CrossRef]
- Luna, R.A.; Foster, J.A. Gut brain axis: Diet microbiota interactions and implications for modulation of anxiety and depression. Curr. Opin. Biotechnol. 2015, 32, 35–41. [Google Scholar] [CrossRef]
- Maes, M.; Kubera, M.; Leunis, J.C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuro Endocrinol. Lett. 2008, 29, 117–124. [Google Scholar]
- Ekwaru, J.P.; Ohinmaa, A.; Veugelers, P.J. The effectiveness of a preventive health program and vitamin D status in improving health-related quality of life of older Canadians. Qual. Life Res. 2016, 25, 661–668. [Google Scholar] [CrossRef]
- Forsyth, A.; Deane, F.P.; Williams, P. A lifestyle intervention for primary care patients with depression and anxiety: A randomised controlled trial. Psychiatry Res. 2015, 230, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Cheong, C.Y.; Nyunt, M.S.Z.; Feng, L.; Niti, M.; Tan, B.Y.; Chan, G.; Khoo, S.A.; Chan, S.M.; Yap, P.; Yap, K.B. Multi-domains lifestyle interventions reduces depressive symptoms among frail and pre-frail older persons: Randomized Controlled Trial. J. Nutr. Health Aging 2017, 21, 918–926. [Google Scholar]
- Toman, J.; Klímová, B.; Vališ, M. Multidomain lifestyle intervention strategies for the delay of cognitive impairment in healthy aging. Nutrients 2018, 10, 1560. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klimova, B.; Novotny, M.; Valis, M. The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review. Nutrients 2020, 12, 710. https://doi.org/10.3390/nu12030710
Klimova B, Novotny M, Valis M. The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review. Nutrients. 2020; 12(3):710. https://doi.org/10.3390/nu12030710
Chicago/Turabian StyleKlimova, Blanka, Michal Novotny, and Martin Valis. 2020. "The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review" Nutrients 12, no. 3: 710. https://doi.org/10.3390/nu12030710
APA StyleKlimova, B., Novotny, M., & Valis, M. (2020). The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review. Nutrients, 12(3), 710. https://doi.org/10.3390/nu12030710






