Effects of Lutein and Astaxanthin Intake on the Improvement of Cognitive Functions among Healthy Adults: A Systematic Review of Randomized Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. The SR Protocol and Registration
2.2. Search Strategy
2.3. Detail of Included Studies
2.3.1. Types of Study
2.3.2. Participants/Population
2.3.3. Intervention and Control
2.4. Main Outcomes
2.5. Data Extraction (Selection and Coding)
2.6. Risk of Bias Assessment
2.7. Data Synthesis
3. Results
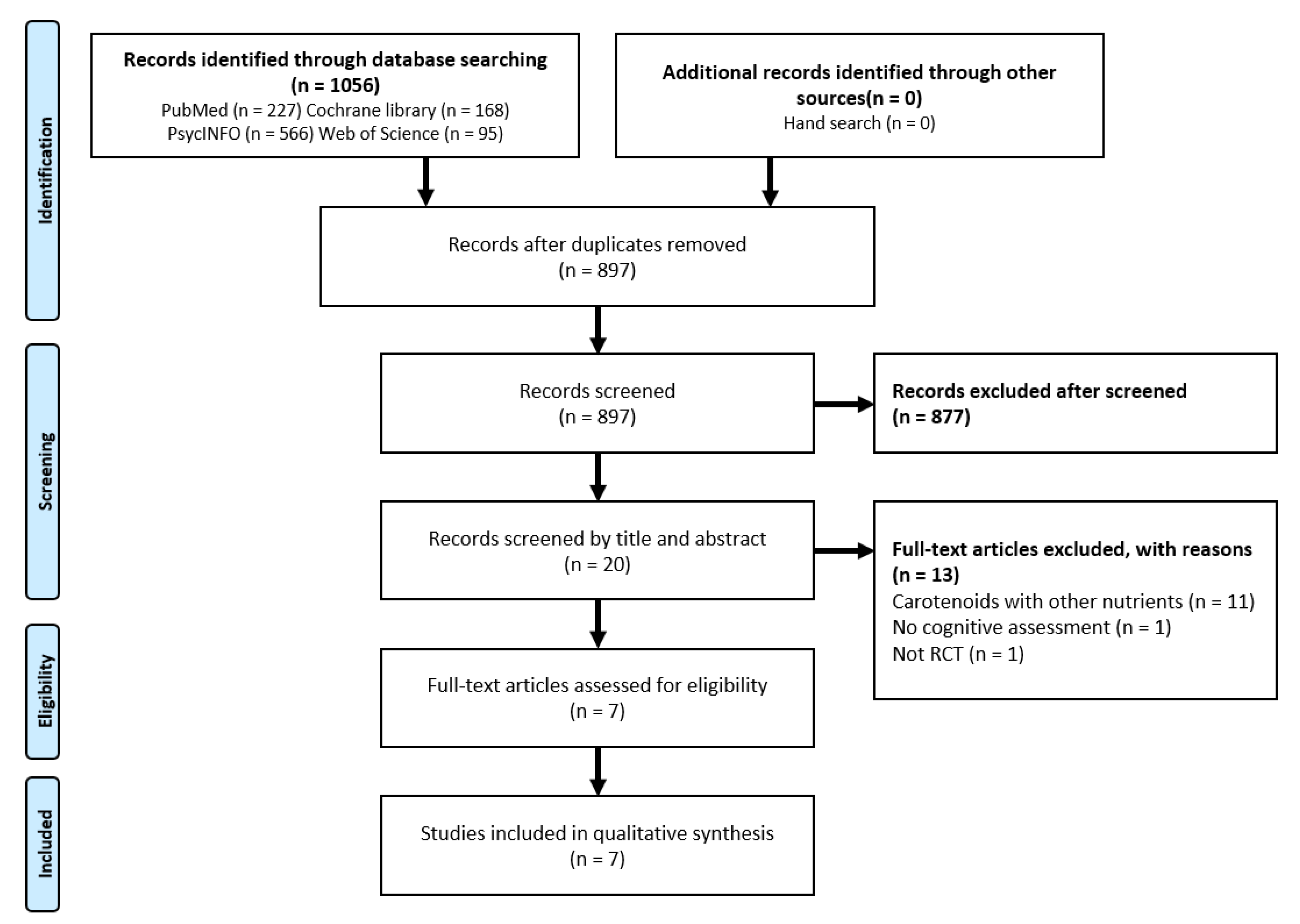
3.1. Results of the Search
3.2. Included Studies
3.3. Participants
3.4. Intervention
3.5. Outcome
3.6. Lutein (and its isomers) Intervention
3.6.1. Macular Pigment (MP)
3.6.2. Serum Lutein (SL)
3.6.3. Episodic Memory (Visual Stimuli)
3.6.4. Episodic Memory (verbal stimuli)
3.6.5. Short-term Memory/Working Memory
3.6.6. Reasoning
3.6.7. Attention
3.6.8. Inhibition
3.6.9. Shifting
3.6.10. Processing Speed
3.7. Adverse Events
3.8. Astaxanthin Intervention
3.8.1. Episodic Memory (visual stimuli)
3.8.2. Episodic Memory (Verbal Stimuli)
3.8.3. Short-term Memory/Working Memory
3.8.4. Inhibition
3.8.5. Attention
3.8.6. Shifting
3.8.7. Processing Speed
3.9. Adverse Events
3.10. Quality Assessment
3.10.1. Methodological Quality in Included Studies
3.10.2. Other Potential Sources of Bias
3.11. Excluded Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Park, D.C.; Lautenschlager, G.; Hedden, T.; Davidson, N.S.; Smith, A.D.; Smith, P.K. Models of visuospatial and verbal memory across the adult life span. Psychol. Aging 2002, 17, 299–320. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.J.; Korosi, A.; Layé, S.; Shukitt-Hale, B.; Barrientos, R.M. Food for thought: How nutrition impacts cognition and emotion. NPJ Sci. Food 2017, 1, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Taki, Y.; Hashizume, H.; Sassa, Y.; Takeuchi, H.; Asano, M.; Asano, K.; Kawashima, R. Breakfast staple types affect brain gray matter volume and cognitive function in healthy children. PLoS ONE 2010, 5, e15213. [Google Scholar] [CrossRef]
- Shimizu, K.; Ihira, H.; Mizumoto, A.; Makino, K.; Ishida, T.; Shimada, H.; Furuna, T. Relationship between dietary habits and cognitive function among community-dwelling elderly adults. Physiotherapy 2015, 101, e1390. [Google Scholar] [CrossRef][Green Version]
- Petersson, S.D.; Philippou, E. Mediterranean Diet, Cognitive Function, and Dementia: A Systematic Review of the Evidence. Adv. Nutr. 2016, 7, 889–904. [Google Scholar] [CrossRef]
- Péneau, S.; Galan, P.; Jeandel, C.; Ferry, M.; Andreeva, V.; Hercberg, S.; Kesse-Guyot, E.; Vogt, L.; Escande, M.; Sérot, J.M.; et al. Fruit and vegetable intake and cognitive function in the SU.VI.MAX 2 prospective study. Am. J. Clin. Nutr. 2011, 94, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Mottaghi, T.; Amirabdollahian, F.; Haghighatdoost, F. Fruit and Vegetable Intake and Cognitive Impairment: A Systematic Review and Meta-Analysis of Observational Studies; Nature Publishing Group: Berlin, Germany, 2018; Volume 72, pp. 1336–1344. [Google Scholar]
- Beydoun, M.A.; Fanelli-Kuczmarski, M.T.; Kitner-Triolo, M.H.; Beydoun, H.A.; Kaufman, J.S.; Mason, M.A.; Evans, M.K.; Zonderman, A.B. Dietary antioxidant intake and its association with cognitive function in an ethnically diverse sample of US adults. Psychosom. Med. 2015, 77, 68–82. [Google Scholar] [CrossRef]
- Devore, E.E.; Grodstein, F.; van Rooij, F.J.A.; Hofman, A.; Stampfer, M.J.; Witteman, J.C.M.; Breteler, M.M.B. Dietary Antioxidants and Long-term Risk of Dementia. Arch. Neurol. 2010, 67, 819–825. [Google Scholar] [CrossRef]
- Alharbi, M.H.; Lamport, D.J.; Dodd, G.F.; Saunders, C.; Harkness, L.; Butler, L.T.; Spencer, J.P.E. Flavonoid-rich orange juice is associated with acute improvements in cognitive function in healthy middle-aged males. Eur. J. Nutr. 2016, 55, 2021–2029. [Google Scholar] [CrossRef]
- Ceravolo, S.A.; Hammond, B.R.; Oliver, W.; Clementz, B.; Miller, L.S.; Renzi-Hammond, L.M. Dietary Carotenoids Lutein and Zeaxanthin Change Brain Activation in Older Adult Participants: A Randomized, Double-Masked, Placebo-Controlled Trial. Mol. Nutr. Food Res. 2019, 63, 1801051. [Google Scholar] [CrossRef]
- Lamport, D.J.; Dye, L.; Wightman, J.L.D.; Lawton, C.L. The effects of flavonoid and other polyphenol consumption on cognitive performance: A systematic research review of human experimental and epidemiological studies. Nutr. Aging 2012, 1, 5–25. [Google Scholar] [CrossRef]
- Ma, Q.P.; Huang, C.; Cui, Q.Y.; Yang, D.J.; Sun, K.; Chen, X.; Li, X.H. Meta-Analysis of the Association between Tea Intake and the Risk of Cognitive Disorders. PLoS ONE 2016, 11, e0165861. [Google Scholar] [CrossRef] [PubMed]
- Yabuzaki, J. Carotenoids Database: Structures, chemical fingerprints and distribution among organisms. Database 2017, 2017, bax004. [Google Scholar] [CrossRef] [PubMed]
- Vershinin, A. Biological functions of carotenoids--diversity and evolution. Biofactors 1999, 10, 99–104. [Google Scholar] [CrossRef]
- Yeum, K.J.; Aldini, G.; Russell, R.M.; Krinsky, N.I. Antioxidant/pro-oxidant actions of carotenoids. In Carotenoids; Britton, G., Liaaen-Jensen, S., Pfander, H., Eds.; Birkhäuser Verlag: Basel, Switzerland, 2009; pp. 235–268. ISBN 978-3-7643-7500-3. [Google Scholar]
- Khachik, F. Distribution and metabolism of dietary carotenoids in humans as a criterion for development of nutritional supplements. Pure Appl. Chem. 2006, 78, 1551–1557. [Google Scholar] [CrossRef]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Johnson, E.J. Role of lutein and zeaxanthin in visual and cognitive function throughout the lifespan. Nutr. Rev. 2014, 72, 605–612. [Google Scholar] [CrossRef]
- Power, R.; Coen, R.F.; Beatty, S.; Mulcahy, R.; Moran, R.; Stack, J.; Howard, A.N.; Nolan, J.M. Supplemental Retinal Carotenoids Enhance Memory in Healthy Individuals with Low Levels of Macular Pigment in A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Alzheimer’s Dis. 2018, 61, 947–961. [Google Scholar] [CrossRef]
- Hammond, B.R.; Stephen Miller, L.; Bello, M.O.; Lindbergh, C.A.; Mewborn, C.; Renzi-Hammond, L.M. Effects of lutein/zeaxanthin supplementation on the cognitive function of community dwelling older adults: A randomized, double-masked, placebo-controlled trial. Front. Aging Neurosci. 2017, 9, 1–9. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Nagao, A. Absorption and metabolism of xanthophylls. Mar. Drugs 2011, 9, 1024–1037. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, C.; Glanville, J.; Briscoe, S.; Littlewood, A.; Marshall, C.; Metzendorf, M.I.; Noel-Storr, A.; Rader, T.; Shokraneh, F.; Thomas, J.; et al. No Title. In Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Eds.; Cochrane: London, UK, 2019. [Google Scholar]
- Nouchi, R.; Kawashima, R. Improving Cognitive Function from Children to Old Age: A Systematic Review of Recent Smart Ageing Intervention Studies. Adv. Neurosci. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Verhagen, A.P.; De Vet, H.C.W.; De Bie, R.A.; Kessels, A.G.H.; Boers, M.; Bouter, L.M.; Knipschild, P.G. The Delphi List: A Criteria List for Quality Assessment of Randomized Clinical Trials for Conducting Systematic Reviews Developed by Delphi Consensus. J. Clin. Epidemiol. 1998, 51, 1235–1241. [Google Scholar] [CrossRef]
- Antes, G. The new CONSORT statement. BMJ 2010, 340, 666. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Renzi-Hammond, L.M.; Bovier, E.R.; Fletcher, L.M.; Miller, L.S.; Mewborn, C.M.; Lindbergh, C.A.; Baxter, J.H.; Hammond, B.R. Effects of a lutein and zeaxanthin intervention on cognitive function: A randomized, double-masked, placebo-controlled trial of younger healthy adults. Nutrients 2017, 9, 1246. [Google Scholar] [CrossRef]
- Katagiri, M.; Satoh, A.; Tsuji, S.; Shirasawa, T. Effects of astaxanthin-rich Haematococcus pluvialis extract on cognitive function: A randomised, double-blind, placebo-controlled study. J. Clin. Biochem. Nutr. 2012, 51, 102–107. [Google Scholar] [CrossRef]
- Hayashi, M.; Ishibashi, T.; Maoka, T. Effect of astaxanthinnrich extract derived from Paracoccus carotinifaciens on cognitive function in middleeaged and older individuals. J. Clin. Biochem. Nutr. 2018, 62, 195–205. [Google Scholar] [CrossRef]
- Lindbergh, C.A.; Renzi-Hammond, L.M.; Hammond, B.R.; Terry, D.P.; Mewborn, C.M.; Puente, A.N.; Miller, L.S. Lutein and Zeaxanthin Influence Brain Function in Older Adults: A Randomized Controlled Trial. J. Int. Neuropsychol. Soc. 2018, 24, 77–90. [Google Scholar] [CrossRef]
- Ajana, S.; Weber, D.; Helmer, C.; Merle, B.M.; Stuetz, W.; Dartigues, J.F.; Rougier, M.B.; Korobelnik, J.F.; Grune, T.; Delcourt, C.; et al. Plasma concentrations of lutein and zeaxanthin, macular pigment optical density, and their associations with cognitive performances among older adults. Investig. Ophthalmol. Vis. Sci. 2018, 59, 1828–1835. [Google Scholar] [CrossRef]
- Feeney, J.; O’Leary, N.; Moran, R.; O’Halloran, A.M.; Nolan, J.M.; Beatty, S.; Young, I.S.; Kenny, R.A. Plasma Lutein and Zeaxanthin Are Associated With Better Cognitive Function Across Multiple Domains in a Large Population-Based Sample of Older Adults: Findings from The Irish Longitudinal Study on Aging. J. Gerontol. Ser. A 2017, 72, 1431–1436. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Rühl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600685. [Google Scholar] [CrossRef] [PubMed]
- Giordano, E.; Quadro, L. Lutein, zeaxanthin and mammalian development: Metabolism, functions and implications for health. Arch. Biochem. Biophys. 2018, 647, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Green-Gomez, M.; Bernstein, P.S.; Curcio, C.A.; Moran, R.; Roche, W.; Nolan, J.M. Standardizing the assessment of macular pigment using a dual-wavelength autofluorescence technique. Transl. Vis. Sci. Technol. 2019, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Zygouris, S.; Tsolaki, M. Computerized cognitive testing for older adults: A review. Am. J. Alzheimers. Dis. Other Demen. 2015, 30, 13–28. [Google Scholar] [CrossRef] [PubMed]
- GUALTIERI, C.; JOHNSON, L. Reliability and validity of a computerized neurocognitive test battery, CNS Vital Signs. Arch. Clin. Neuropsychol. 2006, 21, 623–643. [Google Scholar] [CrossRef] [PubMed]
- Johansson, B. The MIR–Memory-in-Reality Test; Psykologiforlaget AB: Stockholm, Sweden, 1989. [Google Scholar]
- Wechsler. The Wechsler Memory Scale, 4th ed.; Pearson Assessments: San Antonio, TX, USA, 2009. [Google Scholar]
- McCarthy, M.; Ferris, S.H.; Clark, E.; Crook, T. Acquistion and retention of categorized material in normal aging and senile dementia. Exp. Aging Res. 1981, 7, 127–135. [Google Scholar] [CrossRef]
- Moms, J.C.; Heyman, A.; Mohs, R.C.; Hughes, J.P.; van Belle, G.; Fillenbaum, G.; Mellits, E.D.; Clark, C. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part I. Clinical and neuropsychological assesment of Alzheimer’s disease. Neurology 1989, 39, 1159. [Google Scholar] [CrossRef]
- Wechsler DA Manual for the Wechsler Adult Intelligence Scale-Revised; Psychological Corporation: New York, NY, USA, 1981.
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643–662. [Google Scholar] [CrossRef]
- Shao, Z.; Janse, E.; Visser, K.; Meyer, A.S. What do verbal fluency tasks measure? Predictors of verbal fluency performance in older adults. Front. Psychol. 2014, 5, 772. [Google Scholar] [CrossRef]
- Borkowski, J.G.; Benton, A.L.; Spreen, O. Word fluency and brain damage. Neuropsychologia 1967, 5, 135–140. [Google Scholar] [CrossRef]
- Hakoda, Y.; Watanabe, M. Manual for New Stroop Test II; Toyo Physical: Fukuoka, Japan, 2004. [Google Scholar]
- Matsumoto, A.; Hakoda, Y.; Watanabe, M. Life-span development of Stroop and reverse-Stroop interference measured using matching responses. Jpn. J. Psychol. 2012, 83, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Houx, P.J.; Jolles, J.; Vreeling, F.W. Stroop interference: Aging effects assessed with the stroop color-word test. Exp. Aging Res. 1993, 19, 209–224. [Google Scholar] [CrossRef] [PubMed]
- Macleod, C.M. Half a Century of Research on the Stroop Effect-An Integrative Review the Production Effect View project. Psychol. Bull. 1991, 109, 163. [Google Scholar] [CrossRef]
- Scott, T.M.; Rasmussen, H.M.; Chen, O.; Johnson, E.J. Avocado Consumption Increases Macular Pigment Density in Older Adults: A Randomized, Controlled Trial. Nutrients 2017, 9, 919. [Google Scholar] [CrossRef]
- Ito, N.; Saito, H.; Seki, S.; Ueda, F.; Asada, T. Effects of Composite Supplement Containing Astaxanthin and Sesamin on Cognitive Functions in People with Mild Cognitive Impairment: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Alzheimer’s Dis. 2018, 62, 1767–1775. [Google Scholar] [CrossRef]
- Smith, A.P.; Clark, R.E.; Nutt, D.J.; Haller, J.; Hayward, S.G.; Perry, K. Vitamin C, Mood and Cognitive Functioning in the Elderly. Nutr. Neurosci. 1999, 2, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, K.; Clemons, T.E.; McBee, W.L.; Lindblad, A.S. Impact of antioxidants, zinc, and copper on cognition in the elderly: A randomized, controlled trial. Neurology 2004, 63, 1705–1707. [Google Scholar]
- Wolters, M.; Hickstein, M.; Flintermann, A.; Tewes, U.; Hahn, A. Cognitive performance in relation to vitamin status in healthy elderly German women-The effect of 6-month multivitamin supplementation. Prev. Med. Balt. 2005, 41, 253–259. [Google Scholar] [CrossRef]
- Grodstein, F.; Kang, J.H.; Glynn, R.J.; Cook, N.R.; Gaziano, J.M. A randomized trial of beta carotene supplementation and cognitive function in men: The physicians’ health study II. Arch. Intern. Med. 2007, 167, 2184–2190. [Google Scholar] [CrossRef]
- Kang, J.H.; Cook, N.R.; Manson, J.E.; Buring, J.E.; Albert, C.M.; Grodstein, F. Vitamin E, Vitamin C, Beta carotene, and cognitive function among women with or at risk of cardiovascular disease: The women’s antioxidant and cardiovascular study. Circulation 2009, 119, 2772–2780. [Google Scholar] [CrossRef] [PubMed]
- Yagi, A.; Fujimoto, K.; Michihiro, K.; Goh, B.; Tsi, D.; Nagai, H. The effect of lutein supplementation on visual fatigue: A psychophysiological analysis. Appl. Erg. 2009, 40, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Fezeu, L.; Jeandel, C.; Ferry, M.; Andreeva, V.; Amieva, H.; Hercberg, S.; Galan, P. French adults’ cognitive performance after daily supplementation with antioxidant vitamins and minerals at nutritional doses: A post hoc analysis of the supplementation in vitamins and mineral antioxidants (SU.VI.MAX) trial. Am. J. Clin. Nutr. 2011, 94, 892–899. [Google Scholar] [CrossRef]
- Bovier, E.R.; Hammond, B.R. A randomized placebo-controlled study on the effects of lutein and zeaxanthin on visual processing speed in young healthy subjects. Arch. Biochem. Biophys. 2015, 572, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Bovier, E.R.; Renzi, L.M.; Hammond, B.R. A Double-Blind, Placebo-Controlled Study on the Effects of Lutein and Zeaxanthin on Neural Processing Speed and Efficiency. PLoS ONE 2014, 9, e108178. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Coen, R.F.; Akuffo, K.O.; Beatty, S.; Dennison, J.; Moran, R.; Stack, J.; Howard, A.N.; Mulcahy, R.; Nolan, J.M. Cognitive function and its relationship with macular pigment optical density and serum concentrations of its constituent carotenoids. J. Alzheimer’s Dis. 2015, 48, 261–277. [Google Scholar] [CrossRef]
- Feeney, J.; O’Sullivan, M.; Kenny, R.A.; Robertson, I.H. Change in perceived stress and 2-year change in cognitive function among older adults: The Irish Longitudinal Study on Ageing. Stress Heal. 2018, 34, 403–410. [Google Scholar] [CrossRef]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef]
- Ozawa, Y.; Sasaki, M.; Takahashi, N.; Kamoshita, M.; Miyake, S.; Tsubota, K. Neuroprotective Effects of Lutein in the Retina. Curr. Pharm. Des. 2012, 18, 51–56. [Google Scholar] [CrossRef]
- Stringham, N.T.; Holmes, P.V.; Stringham, J.M. Effects of macular xanthophyll supplementation on brain-derived neurotrophic factor, pro-inflammatory cytokines, and cognitive performance. Physiol. Behav. 2019, 211, 112650. [Google Scholar] [CrossRef]
- Calabrese, F.; Rossetti, A.C.; Racagni, G.; Gass, P.; Riva, M.A.; Molteni, R. Brain-derived neurotrophic factor: A bridge between inflammation and neuroplasticity. Front. Cell. Neurosci. 2014, 8, 430. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Johnson, E.J.; Hammond, B.R. Lutein across the Lifespan: From Childhood Cognitive Performance to the Aging Eye and Brain. Curr. Dev. Nutr. 2019, 3, nzz066. [Google Scholar] [CrossRef] [PubMed]
- Craft, N.E.; Haitema, T.B.; Garnett, K.M.; Fitch, K.A.; Dorey, C.K. Carotenoid, tocopherol, and retinol concentrations in elderly human brain. J. Nutr. Heal. Aging 2004, 8, 156–162. [Google Scholar]
- Vishwanathan, R.; Kuchan, M.J.; Sen, S.; Johnson, E.J. Lutein and preterm infants with decreased concentrations of brain carotenoids. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 659–665. [Google Scholar] [CrossRef]
- Zamroziewicz, M.K.; Paul, E.J.; Zwilling, C.E.; Johnson, E.J.; Kuchan, M.J.; Cohen, N.J.; Barbey, A.K. Parahippocampal Cortex Mediates the Relationship between Lutein and Crystallized Intelligence in Healthy, Older Adults. Front. Aging Neurosci. 2016, 8, 297. [Google Scholar] [CrossRef]
- Lindbergh, C.A.; Mewborn, C.M.; Hammond, B.R.; Renzi-Hammond, L.M.; Curran-Celentano, J.M.; Miller, L.S. Relationship of Lutein and Zeaxanthin Levels to Neurocognitive Functioning: An fMRI Study of Older Adults. J. Int. Neuropsychol. Soc. 2017, 23, 11–22. [Google Scholar] [CrossRef]
- Laird, A.R.; McMillan, K.M.; Lancaster, J.L.; Kochunov, P.; Turkeltaub, P.E.; Pardo, J.V.; Fox, P.T. A comparison of label-based review and ALE meta-analysis in the Stroop task. Hum. Brain Mapp. 2005, 25, 6–21. [Google Scholar] [CrossRef]
- Simmonds, D.J.; Pekar, J.J.; Mostofsky, S.H. Meta-analysis of Go/No-go tasks demonstrating that fMRI activation associated with response inhibition is task-dependent. Neuropsychologia 2008, 46, 224–232. [Google Scholar] [CrossRef]
- Jeong, W.; Chung, C.K.; Kim, J.S. Episodic Memory in Aspects of Large-Scale Brain Networks; Frontiers Media SA: Lausanne, Switzerland, 2015; Volume 9, p. 454. [Google Scholar]
- Squire, L.R.; Wixted, J.T. The Cognitive Neuroscience of Human Memory since H.M. Annu. Rev. Neurosci. 2011, 34, 259–288. [Google Scholar] [CrossRef]
- Alvarado-Ramos, K.E.; De Leon, L.; Fontes, F.; Rios-Castillo, I. Dietary Consumption of Lutein and Zeaxanthin in Panama: A Cross-Sectional Study. Curr. Dev. Nutr. 2018, 2, nzy064. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000; ISBN 978-0-309-06935-9. [Google Scholar]
- Aizawa, K.; Inakuma, T. Quantitation of Carotenoids in Commonly Consumed Vegetables in Japan. Food Sci. Technol. Res. 2007, 13, 247–252. [Google Scholar] [CrossRef][Green Version]
- Bjerkeng, B.; Peisker, M.; von Schwartzenberg, K.; Ytrestøyl, T.; Åsgård, T. Digestibility and muscle retention of astaxanthin in Atlantic salmon, Salmo salar, fed diets with the red yeast Phaffia rhodozyma in comparison with synthetic formulated astaxanthin. Aquaculture 2007, 269, 476–489. [Google Scholar] [CrossRef]
- Turujman, S.A.; Wamer, W.G.; Wei, R.R.; Albert, R.H. Rapid Liquid Chromatographic Method to Distinguish Wild Salmon from Aquacultured Salmon Fed Synthetic Astaxanthin. J. Aoac Int. 1997, 80, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Mortality in Randomized Trials of Antioxidant Supplements for Primary and Secondary Prevention. JAMA 2007, 297, 842. [Google Scholar] [CrossRef] [PubMed]
| Lead Author; Year; Country | Study Design, Duration | Sample size (Female) | Age (mean ± SD) | Health Status | Cognitive Status | Intervention (Timing or Method) | Control (Contents) |
|---|---|---|---|---|---|---|---|
| Power; 2018; Ireland | A parallel-group, double-blind, placebo-controlled, block-randomized clinical trial, 12 months | 91 (48%) P: 46 A: 45 | P: 46.43 ± 13.21 A: 44.38 ± 11.57 | Low MP volume without the retinal disease, no consumption of L and/or Z and/or MZ supplement | No impairment | L: 10 mg/d MZ: 10 mg/d Z: 2 mg/d (with a meal) | Placebo (capsule containing sunflower oil) |
| Lindberg; 2017; USA | A single-site, double-blind RCT, 12 months | 44 (59%) P: 14 A: 30 | P: 70.43 ± 5.43 A: 72.43 ± 6.48 | Community-dwelling older adults, good overall health, no consumption of xanthophyll supplement | n/R | L: 10 mg/d Z: 2 mg/d (n/R) | Placebo (n/R) |
| Renzi-Hammond; 2017; USA | A randomized, double-masked, placebo-controlled trial, 12 months | 51 (43%) P: 14 A: 37 | P: 20.5 ± 1.91 A: 21.5 ± 2.69 | Healthy young college students, no consumption of the supplement | No impairment | L: 10 mg/d Z: 2 mg/d (with the highest fat meal) | Placebo (n/R) |
| Hammond; 2017; USA | The double-masked, randomized, placebo-controlled trial, 12 months | 51 (59%) P: 15 A: 36 | P: 70.93 ± 5.70 A: 72.51 ± 6.24 | Healthy community-dwelling older adults, no consumption of L&Z supplement | No impairment | L: 10 mg/d Z: 2 mg/d (with the highest fat meal) | Placebo (n/R) |
| Johnson; 2008; USA | Randomized, double-blind, intervention trial, 4 months | 49 (100%) P: 10 D: 14 L: 11 D+L: 14 | P: 68.0 ± 1.2 D: 68.5 ± 1.3 L: 66.7 ± 1.9 D+L: 68.6 ± 1.3 | Healthy, non-smoking older women, no consumption of carotenoids supplement | No impairment | L: 12 mg/d (with nutritional energy drink) | Placebo (n/R) |
| Katagiri; 2012; Japan | The randomized double-blind placebo-controlled study, 12 weeks | 89 (n/R) HAx: 29 LAx: 29 P: 31 | HAx: 51.5 ± 5.7 LAx: 51.1 ± 5.9 P: 51.6 ± 5.3 | Healthy men and women | Complaints of age-related forgetfulness | HAx: A 12 mg/d LAx: A 6 mg/d (after breakfast or lunch or supper) | Placebo (a pill containing corn oil) |
| Hayashi; 2018; Japan | Randomized, double-blind, placebo-controlled, parallel inter-group comparison 8 weeks | 54 (54%) P: 26 Ax: 28 | P: 54.4 ± 6.0 Ax: 56.0 ± 5.2 | Healthy men and women | No impairment | Ax 8 mg/d (after breakfast and dinner) | Placebo (jelly without Ax) |
| Lead author; Year; Country | Test Battery | Tasks for Calculating the Index Score | Domain of Cognitive Functions | Measurement Indices | Statistical Analysis Methods | Results | |
|---|---|---|---|---|---|---|---|
| Analytical Objects | Intervention Effects | ||||||
| Power; 2018; Ireland | CANTAB | MOT | Processing speed | Mean latency | rANOVA (Only time effects and time-group interaction effects are shown) | Value | n/R |
| Mean total correct | Value | n/R | |||||
| Mean total errors | Value | n/R | |||||
| AST | Inhibition | AST correct latency | Value | 0 | |||
| AST percent correct | Value | 0 | |||||
| AST congruency cost | Value | + | |||||
| VRM | Episodic memory (verbal stimuli) | VRM Trial 1 immediate free recall | Value | n/R | |||
| VRM Trial 2 immediate free recall | Value | n/R | |||||
| VRM Trial 3 immediate free recall | Value | n/R | |||||
| VRM Total immediate free recall | Value | n/R | |||||
| VRM Trial 1 intrusion errors | Value | n/R | |||||
| VRM Trial 2 intrusion errors | Value | n/R | |||||
| VRM Trial 3 intrusion errors | Value | n/R | |||||
| VRM Learning slope | Value | n/R | |||||
| VRM delayed free recall | Value | n/R | |||||
| VRM delayed intrusion errors | Value | + | |||||
| VRM delayed recognition total | Value | n/R | |||||
| VRM recognition false positives | Value | n/R | |||||
| PAL | Episodic memory (visual stimuli) | PAL memory score | Value | + | |||
| PAL total errors | Value | n/R | |||||
| PAL total errors stage 6 | Value | + | |||||
| “FAS” and “Animal” test | FAS test | Shifting | Sum of the number of words beginning with the letters F, A, and S generated with a 1-min time limit per letter | Value | 0 | ||
| Animal test | Shifting | The number of animal names generated with a 1-min time limit | Value | n/R | |||
| Lindberg; 2018; USA | WTAR | WTAR | Global cognition | Number of words correctly pronounced | 2 × 2 mixed-design ANOVA | Value | n/R |
| fMRI task | Wechsler memory scale paired associates learning test | Episodic memory (verbal stimuli) | Number of cued recall | t-test | Value | 0 | |
| Renzi-Hammond; 2017; USA | CNSVS | VBM | Episodic memory (verbal stimuli) | Verbal memory score (VBM correct hits (immediate) + VBM correct – passes (immediate) + VBM correct hits (delay) + VBM correct − passes (delay)) | RCI | Delta | n/R |
| VIM | Episodic memory (visual stimuli) | Visual memory score (VIM correct hits (immediate) + VIM correct − passes (immediate) + VIM correct hits (delay) + VIM correct − passes (delay)) | Delta | + | |||
| NVRT | Reasoning | Reasoning score (NVRT correct responses − NVRT errors) | Delta | +*(“MPOD increaser”) | |||
| SAT | Shifting | Executive function score (SAT correct responses − SAT errors) | Delta | n/R | |||
| FTT, SDC | Processing speed | Psychomotor speed score (FTT right response + FTT left response + SDC corrects) | Delta | n/R | |||
| ST, SAT, CPT | Attention | Complex Attention score (ST errors + SAT errors + CPT errors + CPT correct − passes) | Delta | +*(“MPOD increaser”) | |||
| ST, SAT | Inhibition | Cognitive Flexibility score (SAT correct responses − SAT error − ST error) | Delta | n/R | |||
| Hammond; 2017; USA | CNSVS | VBM | Episodic memory (verbal stimuli) | Verbal memory score (VBM correct hits (immediate) + VBM correct − passes (immediate) + VBM correct hits (delay) + VBM correct − passes (delay)) | RCI | Delta | n/R |
| VIM | Episodic memory (visual stimuli) | Visual memory score (VIM correct hits (immediate) + VIM correct − passes (immediate) + VIM correct hits (delay) + VIM correct − passes (delay)) | Delta | n/R | |||
| NVRT | Reasoning | Reasoning score (NVRT correct responses − NVRT errors) | Delta | n/R | |||
| SAT | Shifting | Executive function score (SAT correct responses − SAT errors) | Delta | +‡ | |||
| FTT, SDC | Processing speed | Psychomotor speed score (FTT right response + FTT left response + SDC corrects) | Delta | n/R | |||
| ST, SAT, CPT | Attention | Complex Attention score (ST errors + SAT errors + CPT errors + CPT correct-passes) | Delta | + | |||
| ST, SAT | Inhibition | Cognitive Flexibility score (SAT correct responses − SAT error − ST error) | Delta | + | |||
| VBM, VIM, FTT, SDC, ST, SAT, CPT | Global cognition | Neurocognitive Index (NCI) (Average of following five scores; Composite Memory, Psychomotor Speed, Reaction Time, Complex Attention, and Cognitive Flexibility) | Delta | n/R | |||
| Johnson; 2008; USA | Original test battery | Verbal Fluency | Shifting | Number of words from a category during a 1-min | Student’s paired t-test | Value | +†(L) |
| Digit Span Forward and Backward | Short-term memory/working memory | Forward digit span length | Value | 0 | |||
| Forward digit span total | Value | 0 | |||||
| Backward digit span length | Value | 0 | |||||
| Backward digit span total | Value | 0 | |||||
| Shopping List Task | Episodic memory (verbal stimuli) | Trial 1 items recalled (max. 10) | Value | 0 | |||
| Trials to learn list (max. 4) | Value | 0 | |||||
| Delayed recall (max. 10) | Value | 0 | |||||
| Word List Memory Test (computer version) | Episodic memory (verbal stimuli) | Trial 1 items recalled (max. 10) | Value | 0 | |||
| Trials to learn list (max. 6) | Value | 0 | |||||
| Delayed recall (max. 10) | Value | 0 | |||||
| Memory in Reality (MIR) Apartment Test | Episodic memory (visual stimuli) | Delayed recall (max. 10) | Value | 0 | |||
| Location recall (max. 10) | Value | 0 | |||||
| NES2 Pattern Comparison Test | Processing speed | Number correct (max.15) | Value | 0 | |||
| Mean response time-correct (s) | Value | - | |||||
| Stroop Test | Processing speed | Mean RT, read words-black (ms) | Value | 0 | |||
| Processing speed | Mean RT, read words-color (ms) | Value | 0 | ||||
| Processing speed | Mean RT, name colors (ms) | Value | 0 | ||||
| Inhibition | Mean RT, name color-words (ms) | Value | 0 | ||||
| Inhibition | Total RT, interference (NC-C) (s) | Value | 0 | ||||
| Katagiri; 2012; Japan | CogHealth | Simple reaction test | Processing speed | Response time (ms) | Two-way factorial ANOVA, adjusted for age and sex (between-group) One-way repeated measure ANOVA, adjusted for age and sex (inter-group) Bonferroni correction (multiple comparisons) | Value | 0 |
| Choice reaction test | Response time (ms) | Value | 0 | ||||
| Working memory test | Short-term memory/working memory | Response time (ms) | Value | +†(HAx) | |||
| Accuracy (%) | Value | 0 | |||||
| Delayed recall test | Episodic memory (visual stimuli) | Response time (ms) | Value | 0 | |||
| Accuracy (%) | Value | +†(HAx) | |||||
| Divided attention test | Attention | Response time (ms) | Value | 0 | |||
| GMLT | GMLT test | Short-term memory/working memory | Total duration (s) | Value | +†(HAx, LAx, P) | ||
| Total errors | Value | +†(HAx, LAx, P) | |||||
| Hayashi; 2018; Japan | Original test battery | Word memory test | Short-term memory/working memory | Immediate recall | Two-way repeated measures ANOVA | Delta | 0 |
| Immediate recall + cued recall | Delta | 0 | |||||
| Episodic memory (verbal stimuli) | Recall after 5 min | Delta | +*(age < 55) | ||||
| Recall after 5 min + cued recall | Delta | 0 | |||||
| Verbal fluency test | Shifting | Names of vegetables | Delta | 0 | |||
| Words that begin with “a” | Delta | 0 | |||||
| Animal words | Delta | 0 | |||||
| Stroop test | Processing speed | Stroop test Step 1 | Delta | 0 | |||
| Inhibition | Stroop test Step 2 | Delta | 0 | ||||
| Processing speed | Stroop test Step 3 | Delta | 0 | ||||
| Inhibition | Stroop test Step 4 | Delta | 0 | ||||
| Cognitive Function | Lead Author; Year; Country | Measurement Indices (Task Name) | Correlation with Task (p-Value) | Task Score, Mean ± SD (Pre) [Post]{Change} | Observed Effect | SMD [95%CI] (Bigger Number Shows Active Favors) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| SL | MP | Active | Placebo | Statistical Method (p-Value) | P pre vs. A pre (p-Value) | P Post vs. A Post (p-Value) | P Change vs. A Change (p-Value) | |||
| Episodic memory (visual stimuli) | Power; 2018; Ireland | PAL memory score (PAL, CANTAB) | r = 0.159 (p = 0.226) | r = 0.219 (p = 0.078) | (18.91 ± 4.96) [20.77 ± 4.57] {1.86 ± 4.72} | (21.26 ± 3.52) [20.32 ± 4.57] {−0.94 ± 3.48} | rANOVA (time effect p = 0.376, time × group effect p = 0.009) | 0.55 [0.05, 1.04] (p = 0.03) | 0.10 [−0.39, 0.58] (p = 0.69) | 0.66 [1.16, 0.16] (p = 0.009) |
| PAL total errors (PAL, CANTAB) | n/R | n/R | (21.50 ± 28.20) [n/R] {n/R} | (17.22 ± 16.93) [n/R] {n/R} | rANOVA (n.S.) | −0.18 [−0.59, 0.23] (p = 0.38) | - | - | ||
| PAL errors adjusted for stage 6 (PAL, CANTAB) | r = −0.346 (p = 0.006) | r = −0.342 (p = 0.005) | (6.78 ± 7.10) [3.17 ± 4.52] {−3.61 ± 7.01} | (4.19 ± 3.82) [4.48 ± 4.89] {0.29 ± 5.79} | rANOVA (time effect p = 0.040, time × group effect p = 0.017) | −0.44 [−0.93, 0.05] (p = 0.08) | 0.28 [−0.21, 0.76] (p = 0.28) | 0.60 [0.10, 1.09] (p = 0.02) | ||
| Renzi-Hammond; 2017; USA | Visual memory (VIM, CNSVS) | n/R | n/R | (n/R) [n/R] {9.43 ± n/R} | (n/R) [n/R] {4.93 ± n/R} | RCI (RCI active = 6.77 vs. RCI placebo = 1.88, p < 0.04) | - | - | - | |
| Hammond; 2017; USA | Visual memory (VIM, CNSVS) | n/R | r = 0.24 (p = 0.09, in A only) | (41.03 ± 6.68) [n/R] {n/R} | (43.87 ± 5.18) [n/R] {n/R} | RCI (n.S.) | - | - | - | |
| Johnson; 2008; USA | Delayed recall (MIR apartment test) | −0.16 (n.S.) | n/R | (8.3 ± 1.6) [8.6 ± 2.1] {n/R} | (9.3 ± 0.8) [9.4 ± 0.7] {n/R} | Student’s paired t-test (n.S.) | 0.75 [−0.15, 1.64] (p = 0.10) | 0.48 [−0.39, 1.35] (p = 0.28) | - | |
| Location recall (MIR apartment test) | n/R | n/R | (9.5 ± 1.0) [9.5 ± 0.8] {n/R} | (9.7 ± 0.7) [9.7 ± 0.7] {n/R} | 0.22 [−0.64, 1.08] (p = 0.62) | 0.25 [−0.61, 1.12] (p = 0.56) | - | |||
| Episodic memory (verbal stimuli) | Power; 2018; Ireland | VRM Trial 1 immediate free recall (VRM, CANTAB) | n/R | n/R | (8.17 ± 1.75) [8.69 ± 1.67] {0.52 ± 1.68} | (8.57 ± 1.59) [8.73 ± 1.84] {0.17 ± 1.58} | rANOVA (n.S.) | −0.24 [−0.72, 0.25] (p = 0.34) | −0.02 [−0.51, 0.46] (p = 0.93) | 0.21 [−0.27, 0.70] (p = 0.39) |
| VRM Trial 2 immediate free recall (VRM, CANTAB) | n/R | n/R | (9.83 ± 1.48) [10.11 ± 2.30] {0.28 ± 1.67} | (10.10 ± 1.24) [10.60 ± 1.57] {0.50 ± 1.80} | −0.19 [−0.68, 0.29] (p = 0.43) | −0.24 [−0.73, 0.24] (p = 0.33) | −0.13 [−0.61, 0.36] (p = 0.61) | |||
| VRM Trial 3 immediate free recall (VRM, CANTAB) | n/R | n/R | (10.71 ± 1.72) [10.51 ± 2.13] {−0.20 ± 1.35} | (10.80 ± 1.10) [11.03 ± 1.13] {0.23 ± 1.17} | −0.06 [−0.55, 0.43] (p = 0.81) | −0.29 [−0.79, 0.20] (p = 0.24) | −0.33 [−0.83, 0.16] (p = 0.18) | |||
| VRM Total immediate free recall (VRM, CANTAB) | n/R | n/R | (28.67 ± 4.51) [29.03 ± 5.52] {0.36 ± 3.22} | (29.47 ± 3.17) [30.37 ± 3.77] {0.90 ± 3.25} | −0.2 [−0.69, 0.29] (p = 0.42) | −0.28 [−0.76, 0.21] (p = 0.27) | −0.17 [−0.65, 0.32] (p = 0.51) | |||
| VRM Trial 1 intrusion errors (VRM, CANTAB) | n/R | n/R | (0.11 ± 0.32) [0.11 ± 0.32] {0 ± 0.48} | (0.17 ± 0.46) [0.20 ± 0.55] {0.03 ± 0.49} | −0.15 [−0.64, 0.33] (p = 0.54) | −0.20 [−0.69, 0.28] (p = 0.54) | −0.06 [−0.55, 0.42] (p = 0.80) | |||
| VRM Trial 2 intrusion errors (VRM, CANTAB) | n/R | n/R | (0.08 ± 0.28) [0.06 ± 0.23] {−0.02 ± 0.38} | (0.03 ± 0.18) [0.20 ± 0.48] {0.17 ± 0.46} | 0.21 [−0.28, 0.69] (p = 0.41) | −0.38 [−0.87, 0.11] (p = 0.13) | −0.45 [−0.94, 0.04] (p = 0.07) | |||
| VRM Trial 3 intrusion errors (VRM, CANTAB) | n/R | n/R | (0.06 ± 0.23) [0 ± 0] {−0.06 ± 0.23} | (0.07 ± 0.36) [0.13 ± 0.50] {0.06 ± 0.63} | −0.03 [−0.52, 0.45] (p = 0.89) | - | −0.26 [−0.75, 0.23] (p = 0.30) | |||
| VRM Learning slope (VRM, CANTAB) | n/R | n/R | (2.54 ± 2.09) [1.89 ± 1.84] {−0.65 ± 1.89} | (2.20 ± 1.32) [2.30 ± 1.64] {0.10 ± 1.63} | 0.20 [−0.29, 0.68] (p = 0.43) | −0.23 [−0.72, 0.26] (p = 0.35) | −0.42 [−0.92, 0.07] (p = 0.09) | |||
| VRM delayed free recall (VRM, CANTAB) | n/R | n/R | (9.07 ± 3.25) [9.92 ± 1.72] {0.85 ± 3.22} | (10.04 ± 1.43) [10.08 ± 2.73] {0.04 ± 2.18} | −0.38 [−0.94, 0.19] (p = 0.19) | −0.07 [−0.62, 0.49] (p = 0.81) | 0.29 [−0.27, 0.85] (p = 0.31) | |||
| VRM delayed intrusion errors (VRM, CANTAB) | r = −0.189 (p = 0.220) | r = −0.306 (p = 0.033) | (0.12 ± 0.33) [0.04 ± 0.20] {−0.08 ± 0.39} | (0.00 ± 0.00) [0.21 ± 0.51] {0.21 ± 0.51} | rANOVA (time effect p = 0.309, time × group effect p = 0.030) | −0.5 [−1.06, 0.07] (p = 0.08) | 0.44 [−0.12, 1.00] (p = 0.13) | 0.63 [0.06, 1.20] (p = 0.03) | ||
| VRM delayed recognition total (VRM, CANTAB) | n/R | n/R | (23.85 ± 0.36) [23.82 ± 0.52] {−0.03 ± 0.52} | (23.83 ± 0.38) [23.87 ± 0.43] {0.04 ± 0.49} | rANOVA (n.S.) | 0.05 [−0.44, 0.54] (p = 0.83) | −0.1 [−0.59, 0.39] (p = 0.68) | −0.14 [−0.63, 0.35] (p = 0.59) | ||
| VRM recognition false positives (VRM, CANTAB) | n/R | n/R | (0.03 ± 0.17) [0.09 ± 0.38] {0.06 ± 0.34} | (0.10 ± 0.31) [0.07 ± 0.37] {−0.03 ± 0.32} | 0.27 [−0.22, 0.76] (p = 0.29) | 0.05 [−0.44, 0.54] (p = 0.83) | −0.28 [−0.78, 0.21] (p = 0.26) | |||
| Lindberg; 2018; USA | Wechsler memory scale paired associates learning test | n/R | n/R | (8.87 ± 1.50) [8.80 ± 2.16] {n/R} | (9.36 ± 0.75) [8.21 ± 2.29] {n/R} | t-test, 2 × 2 mixed design ANOVA (n.S.) | −0.37 [−1.01, 0.27] (p = 0.26) | 0.26 [−0.37,0.90] (p = 0.42) | - | |
| Renzi-Hammond; 2017; USA | Verbal memory (VEM, CNSVS) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (n.S.) | - | - | - | |
| Hammond; 2017; USA | Verbal memory (VEM, CNSVS) | n/R | r = 0.31 (p = 0.07, “MPOD increaser”) | (49.91 ± 5.66) [n/R] {n/R} | (52.67 ± 5.29) [n/R] {n/R} | RCI (n.S.) | −0.49 [−1.10, 0.12] (p = 0.12) | - | - | |
| Johnson; 2008; USA | Trial 1 items recalled (max. 10) (Shopping List Task) | n/R | n/R | (6.9 ± 1.8) [6.5 ± 2.1] {n/R} | (6.5 ± 1.2) [7.7 ± 1.5] {n/R} | Student’s paired t-test (n.S.) | 0.25 [−0.61, 1.11] (p = 0.57) | −0.63 [−1.51, 0.26] (p = 0.16) | - | |
| Trials to learn list (max. 6) (Shopping List Task) | 0.30 (p < 0.05) | n/R | (4.2 ± 1.5) [3.9 ± 1.4] {n/R} | (3.0 ± 0.8) [2.8 ± 0.9] {n/R} | −0.94 [−1.86, −0.03] (p = 0.04) | −0.89 [−1.79, 0.02] (p = 0.06) | - | |||
| Delayed recall (max. 10) (Shopping List Task) | n/R | n/R | (8.3 ± 1.9) [7.6 ± 3.0] {n/R} | (9.5 ± 0.9) [9.5 ± 0.7] {n/R} | 0.25 [−0.61, 1.11] (p = 0.09) | −0.82 [−1.72, 0.08] (p = 0.07) | - | |||
| Trial 1 items recalled (max. 10) (Word List Memory Test) | n/R | n/R | (5.8 ± 1.8) [5.8 ± 1.8] {n/R} | (6.2 ± 1.3) [6.6 ± 1.8] {n/R} | −0.24 [−1.10, 0.62] (p = 0.58) | −0.43 [−1.29, 0.44] (p = 0.34) | - | |||
| Trials to learn list (max. 4) (Word List Memory Test) | −0.04 (n.S.) | n/R | (3.4 ± 0.7) [3.5 ± 0.8] {n/R} | (3.1 ± 0.9) [2.8 ± 0.9] {n/R} | −0.36 [−1.22, 0.51] (p = 0.42) | −0.79 [−1.69, 0.10] (p = 0.08) | - | |||
| Delayed recall (max. 10) (Word List Memory Test) | n/R | n/R | (6.8 ± 2.9) [7.6 ± 2.4] {n/R} | (8.1 ± 1.1) [8.3 ± 1.8] {n/R} | −0.56 [−1.43, 0.32] (p = 0.21) | −0.31 [−1.18, 0.55] (p = 0.48) | - | |||
| Short-term memory/working memory | Johnson; 2008; USA | Forward digit span length | n/R | n/R | (6.6 ± 1.2) [7.0 ± 1.5] {n/R} | (7.2 ± 1.2) [7.2 ± 1.4] {n/R} | Student’s paired t-test (n.S.) | −0.48 [−1.35, 0.39] (p = 0.28) | −0.13 [−0.99, 0.73] (p = 0.76) | - |
| Forward digit span total | n/R | n/R | (8.1 ± 2.3) [8.7 ± 2.5] {n/R} | (9.7 ± 2.5) [9.0 ± 2.4] {n/R} | −0.64 [−1.52, 0.24] (p = 0.28) | −0.12 [−0.97, 0.74] (p = 0.79) | - | |||
| Backward digit span length | n/R | n/R | (5.1 ± 1.6) [4.7 ± 1.4] {n/R} | (5.9 ± 1.4) [5.8 ± 1.7] {n/R} | −0.51 [−1.38, 0.36] (p = 0.25) | −0.68 [−1.57, 0.20] (p = 0.13) | - | |||
| Backward digit span total | n/R | n/R | (7.5 ± 3.1) [6.9 ± 2.7] {n/R} | (8.2 ± 2.7) [8.4 ± 3.3] {n/R} | −0.23 [−1.09, 0.63] (p = 0.60) | −0.48 [−1.35, 0.39] (p = 0.28) | - | |||
| Reasoning | Renzi-Hammond; 2017; USA | Reasoning (NVRT, CNSVS) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (RCI “MPOD increase” = 1.94 vs. RCI “no change” = 0.18, p < 0.05*) | - | - | - |
| Hammond; 2017; USA | Reasoning (NVRT, CNSVS) | n/R | r = 0.45 (p = 0.04) | (2.97 ± 3.95) [n/R] {n/R} | (2.47 ± 3.83) [n/R] {n/R} | RCI (n.S.) | 0.13 [−0.48, 0.73] (p = 0.68) | - | - | |
| Attention | Renzi-Hammond; 2017; USA | Complex attention (ST & SAT & CPT, CNSVS) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (RCI “MPOD increase” = 2.02 vs. RCI “no change” = 0.00, p < 0.04) | - | - | - |
| Hammond; 2017; USA | Complex attention (ST & SAT & CPT, CNSVS) | n/R | r = −0.31 (p = 0.04, in A only) | (13.12 ± 11.12) [n/R] {n/R} | (10.21 ± 7.21) [n/R] {n/R} | RCI (RCI active = 3.71 vs. RCI placebo = 0.34, p < 0.02) | 0.28 [−0.32, 0.89] (p = 0.36) | - | - | |
| Inhibition | Power; 2018; Ireland | AST correct latency (AST, CANTAB) | n/R | n/R | (832 ± 191.86) [751.63 ± 191.70] {−80.37 ± 132.98} | (841.41 ± 158.95) [775.38 ± 217.58] {−66.03 ± 167.79} | rANOVA (time effect p < 0.001, time × group effect p = 0.695) | 0.05 [−0.42,0.53] (p = 0.83) | 0.12 [−0.36,0.59] (p = 0.83) | −0.09 [−0.57, 0.38] (p = 0.70) |
| AST percent correct (AST, CANTAB) | n/R | n/R | (93.71 ± 8.14) [96.84 ± 3.49] {3.13 ± 6.94} | (93.89 ± 7.57) [95.02 ± 7.61] {1.13 ± 4.10} | rANOVA (time effect p = 0.004, time × group effect p = 0.164) | −0.02 [−0.50, 0.45] (p = 0.93) | 0.31 [−0.17, 0.79] (p = 0.20) | 0.34 [−0.14, 0.82] (p = 0.17) | ||
| AST congruency cost (AST, CANTAB) | n/R | n/R | (98.46 ± 103.1) [94.43 ± 70.01] {−4.03 ± 93.04} | (128.1 ± 99.07) [72.74 ± 86.65] {−55.36 ± 110.56} | rANOVA (time effect p = 0.019, time × group effect p = 0.041) | −0.29 [−0.77, 0.19] (p = 0.24) | 0.27 [−0.20, 0.75] (p = 0.26) | 0.5 [0.02, 0.99] (p = 0.104) | ||
| Renzi-Hammond; 2017; USA | Cognitive flexibility (ST & SAT, CNSVS) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (n.S.) | - | - | - | |
| Hammond; 2017; USA | Cognitive flexibility (ST & SAT, CNSVS) | n/R | r = 0.20 (p = 0.10) | (30.32 ± 19.09) [n/R] {n/R} | (35.5 ± 16.01) [n/R] {n/R} | RCI (RCI active = 6.31 vs. RCI placebo = 0.84, p < 0.04) | −0.28 [−0.88, 0.33] (p = 0.37) | - | - | |
| Johnson; 2008; USA | Mean RT, name colors – words (Stroop test) | n/R | n/R | (1492 ± 329) [1462 ± 221] {n/R} | (1419 ± 308) [1413 ± 508] {n/R} | Student’s paired t-test (n.S.) | −0.22 [−1.08, 0.64] (p = 0.62) | −0.12 [−0.98, 0.74] (p = 0.78) | - | |
| Total RT, interference (NC-C) (Stroop test) | n/R | n/R | (24.2 ± 10.9) [22.4 ± 7.1] {n/R} | (25.0 ± 14.8) [23.1 ± 22.0] {n/R} | 0.06 [−0.80, 0.92] (p = 0.89) | 0.04 [−0.81, 0.90] (p = 0.92) | - | |||
| Shifting | Power; 2018; Ireland | Phonemic (letter) fluency (FAS test) | n/R | n/R | (44.44 ± 15.49) [50.11 ± 15.55] {5.67 ± 7.85} | (40.23 ± 11.84) [45.93 ± 10.91] {5.70 ± 7.88} | rANOVA (time effect p < 0.001, time × group effect p = 0.986) | 0.30 [−0.19, 0.79] (p = 0.23) | 0.30 [−0.18, 0.79] (p = 0.22) | 0.00 [−0.49, 0.48] (p = 0.99) |
| Semantic (category) fluency (Animal test) | n/R | n/R | (23.72 ± 6.96) [24.25 ± 6.97] {0.53 ± 5.20} | (19.87 ± 3.86) [21.30 ± 3.66] {1.43 ± 3.57} | rANOVA (n.S.) | 0.7 [0.28, 1.13] (p = 0.001) | 0.51 [0.02, 1.00] (p = 0.04) | −0.2 [−0.68, 0.29] (p = 0.43) | ||
| Renzi-Hammond; 2017; USA | Executive function (SAT, CNSVS) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (n.S.) | - | - | - | |
| Hammond; 2017; USA | Executive function (SAT, CNSVS) | n/R | n/R | (32.33 ± 18.33) [n/R] {n/R} | (35.87 ± 15.90) [n/R] {n/R} | RCI (RCI active = 5.64 vs. RCI placebo = 1.27, p = 0.07) | −0.2 [−0.80, 0.41] (p = 0.52) | - | - | |
| Johnson; 2008; USA | Verbal Fluency (Verbal Fluency) | 0.03 (n.S.) | n/R | (11.3 ± 5.1) [15.5 ± 5.5] {n/R} | (12.9 ± 6.2) [13.8 ± 3.5] {n/R} | Student’s paired t-test (p < 0.05) | −0.27 [−1.13, 0.59] (p = 0.54) | 0.35 [−0.51,1.21] (p = 0.43) | - | |
| Processing speed | Power; 2018; Ireland | Mean latency (CANTAB, MOT) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | rANOVA (n.S.) | - | - | - |
| Mean total correct (CANTAB, MOT) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | - | - | - | |||
| Mean total errors (CANTAB, MOT) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | - | - | - | |||
| Renzi-Hammond; 2017; USA | Psychomotor speed score (CNSVS, FTT+SDC) | n/R | n/R | (n/R) [n/R] {n/R} | (n/R) [n/R] {n/R} | RCI (n.S.) | - | - | - | |
| Hammond; 2017; USA | Psychomotor speed score (CNSVS, FTT+SDC) | n/R | n/R | (145.5 ± 19.37) [n/R] {n/R} | (140.2 ± 20.11) [n/R] {n/R} | RCI (n.S.) | 0.27 [−0.34, 0.87] (p = 0.39) | - | - | |
| Johnson; 2008; USA | Number correct (max. 15) (NES2 Pattern Comparison Test) | n/R | n/R | (14.5 ± 0.9) [14.3 ± 1.8] {n/R} | (14.5 ± 0.7) [14.9 ± 0.3] {n/R} | Student’s paired t-test (n.S.) | 0.00 [−0.86, 0.86] (p = 1.00) | −0.44 [−1.30, 0.43] (p = 0.33) | - | |
| Mean response time-correct (s) (NES2 Pattern Comparison Test) | n/R | n/R | (6.1 ± 2.3) [6.4 ± 2.3] {n/R} | (6.8 ± 3.0) [5.9 ± 2.3] {n/R} | −0.25 [−1.11, 0.61] (p = 0.33) | 0.21 [−0.65, 1.07] (p = 0.63) | - | |||
| Stroop test Mean RT, read words-black (ms) | n/R | n/R | (844 ± 239) [945 ± 185] {n/R} | (1040 ± 380) [891 ± 222] {n/R} | −0.6 [−1.48, 0.28] (p = 0.33) | 0.25 [−0.61, 1.12] (p = 0.33) | - | |||
| Stroop test Mean RT, read words-color (ms) | n/R | n/R | (753 ± 210) [883 ± 213] {n/R} | (788 ± 200) [804 ± 202] {n/R} | −0.16 [−1.02, 0.69] (p = 0.33) | 0.36 [−0.50, 1.23] (p = 0.33) | - | |||
| Stroop test Mean RT, name colors (ms) | n/R | n/R | (1008 ± 217) [1014 ± 193] {n/R} | (919 ± 173) [951 ± 220] {n/R} | 0.43 [−0.44, 1.30] (p = 0.33) | 0.29 [−0.57, 1.16] (p = 0.33) | - | |||
| Lead Author; Year; Country | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Q9 | Q10 | Q11 | Q12 | Q13 | Q14 | Total score (Max. = 14) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Power; 2018; Ireland | Y | Y | N | Y | Y | Y | Y | Y | ? | Y | Y | Y | ? | N | 10 |
| Lindberg; 2018; USA | Y | Y | ? | Y | Y | Y | Y | ? | ? | Y | N | Y | Y | N | 9 |
| Renzi-Hammond; 2017; USA | Y | Y | ? | Y | Y | Y | Y | ? | N | Y | N | Y | Y | N | 9 |
| Hammond; 2017; USA | Y | Y | Y | Y | Y | Y | Y | ? | N | Y | N | Y | Y | N | 10 |
| Johnson; 2008; USA | Y | Y | Y | Y | Y | Y | Y | Y | ? | N | N | N | Y | N | 9 |
| Katagiri; 2012; Japan | Y | Y | Y | Y | Y | Y | Y | Y | N | N | Y | Y | Y | N | 11 |
| Hayashi; 2018; Japan | Y | ? | Y | Y | Y | Y | Y | Y | N | N | Y | Y | Y | N | 10 |
| Total score across studies | 7 | 6 | 4 | 7 | 7 | 7 | 7 | 4 | 0 | 4 | 3 | 6 | 6 | 0 | – |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nouchi, R.; Suiko, T.; Kimura, E.; Takenaka, H.; Murakoshi, M.; Uchiyama, A.; Aono, M.; Kawashima, R. Effects of Lutein and Astaxanthin Intake on the Improvement of Cognitive Functions among Healthy Adults: A Systematic Review of Randomized Controlled Trials. Nutrients 2020, 12, 617. https://doi.org/10.3390/nu12030617
Nouchi R, Suiko T, Kimura E, Takenaka H, Murakoshi M, Uchiyama A, Aono M, Kawashima R. Effects of Lutein and Astaxanthin Intake on the Improvement of Cognitive Functions among Healthy Adults: A Systematic Review of Randomized Controlled Trials. Nutrients. 2020; 12(3):617. https://doi.org/10.3390/nu12030617
Chicago/Turabian StyleNouchi, Rui, Takahiko Suiko, Eriko Kimura, Hiroki Takenaka, Michiaki Murakoshi, Akira Uchiyama, Megumi Aono, and Ryuta Kawashima. 2020. "Effects of Lutein and Astaxanthin Intake on the Improvement of Cognitive Functions among Healthy Adults: A Systematic Review of Randomized Controlled Trials" Nutrients 12, no. 3: 617. https://doi.org/10.3390/nu12030617
APA StyleNouchi, R., Suiko, T., Kimura, E., Takenaka, H., Murakoshi, M., Uchiyama, A., Aono, M., & Kawashima, R. (2020). Effects of Lutein and Astaxanthin Intake on the Improvement of Cognitive Functions among Healthy Adults: A Systematic Review of Randomized Controlled Trials. Nutrients, 12(3), 617. https://doi.org/10.3390/nu12030617





