Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease
Abstract
1. Introduction
2. Components of a Healthy Diet and Their Benefits
3. Common Health-Promoting Dietary Patterns
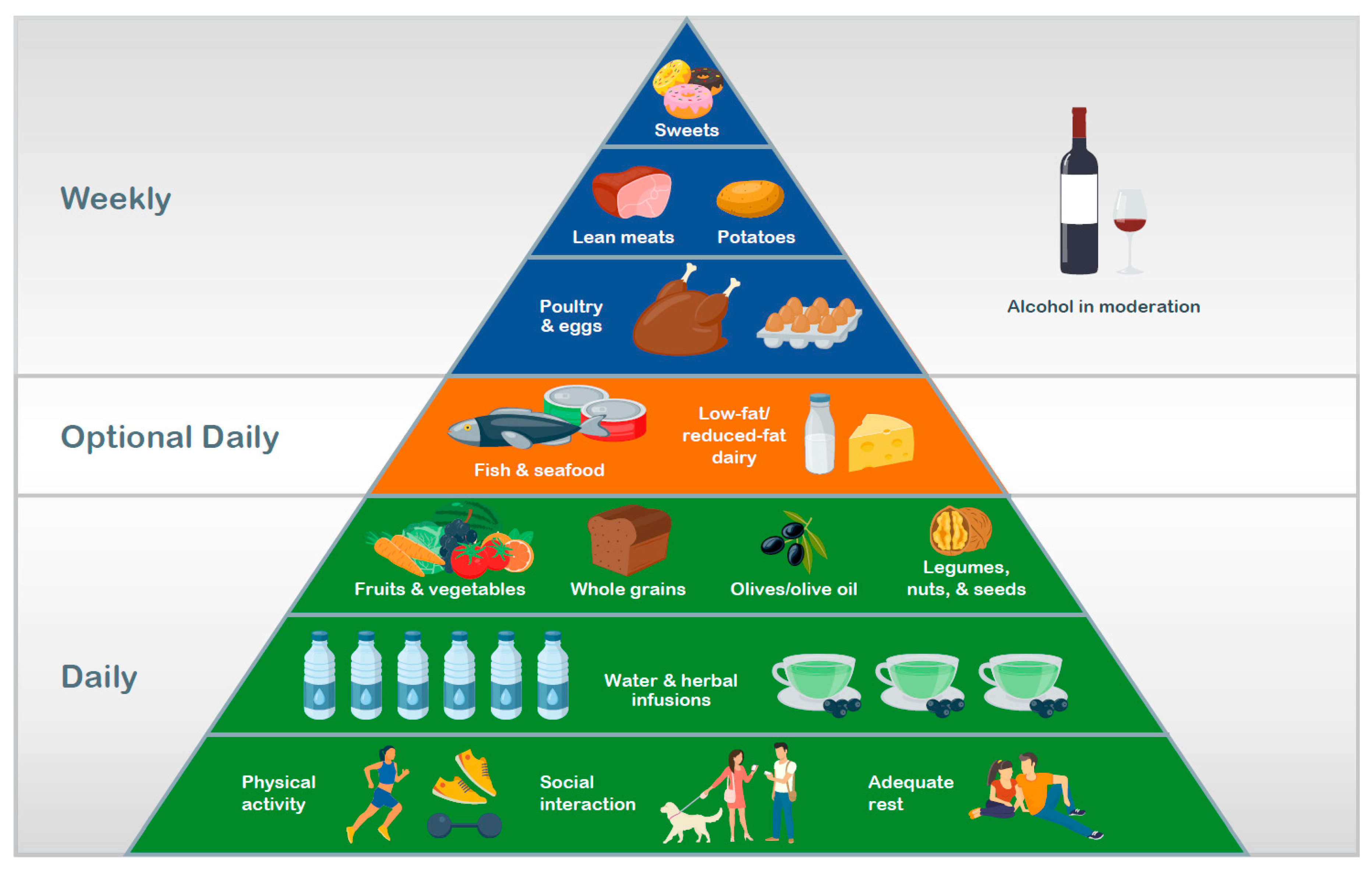
3.1. Mediterranean Diet
3.2. Dietary Approaches to Stop Hypertension (DASH)
3.3. Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND)
3.4. Nordic Diet
3.5. Traditional Asian Diets
4. Additional Factors
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020; World Health Organ: Geneva, Switzerland, 2013.
- Tamimi, R.M.; Spiegelman, D.; Smith-Warner, S.A.; Wang, M.; Pazaris, M.; Willett, W.C.; Eliassen, A.H.; Hunter, D.J. Population attributable risk of modifiable and nonmodifiable breast cancer risk factors in postmenopausal breast cancer. Am. J. Epidemiol. 2016, 184, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.; Rimm, E.; Qi, L.; Rexrode, K.; Albert, C.M.; Sun, Q.; Willett, W.C.; Hu, F.B.; Manson, J.E. Diet, lifestyle, biomarkers, genetic factors, and risk of cardiovascular disease in the Nurses’ Health Studies. Am. J. Public Health 2016, 106, 1616–1623. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 131. [Google Scholar] [CrossRef] [PubMed]
- Koene, R.J.; Prizment, A.E.; Blaes, A.; Konety, S.H. Shared risk factors in cardiovascular disease and cancer. Circulation 2016, 133, 1104–1114. [Google Scholar] [CrossRef]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef]
- Marcason, W. What are the components to the MIND diet? J. Acad. Nutr. Diet 2015, 115, 1744. [Google Scholar] [CrossRef]
- Stark, C. Guidelines for Food and Nutrient Intake. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 34–47. [Google Scholar]
- Stipanuk, M.H.; Caudill, M.A. Structure and Properties of the Macronutrients. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; p. 49. [Google Scholar]
- Stipanuk, M.H.; Caudill, M.A. The Vitamins. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 537–539. [Google Scholar]
- Stipanuk, M.H.; Caudill, M.A. The Minerals and Water. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 719–720. [Google Scholar]
- Slavin, J. Structure, Nomenclature, and Properties of Carbohydrates. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 50–68. [Google Scholar]
- Benisi-Kohansal, S.; Saneei, P.; Salehi-Marzijarani, M.; Larijani, B.; Esmaillzadeh, A. Whole-grain intake and mortality from all causes, cardiovascular disease, and cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Adv. Nutr. 2016, 7, 1052–1065. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef]
- Zong, G.; Gao, A.; Hu, F.B.; Sun, Q. Whole grain intake and mortality from all causes, cardiovascular disease, and cancer: A meta-analysis of prospective cohort studies. Circulation 2016, 133, 2370–2380. [Google Scholar] [CrossRef] [PubMed]
- McRorie, J.W., Jr.; McKeown, N.M. Understanding the physics of functional fibers in the gastrointestinal tract: An evidence-based approach to resolving enduring misconceptions about insoluble and soluble fiber. J. Acad. Nutr. Diet 2017, 117, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Probst, Y.C.; Guan, V.X.; Kent, K. Dietary phytochemical intake from foods and health outcomes: A systematic review protocol and preliminary scoping. BMJ Open 2017, 7, e013337. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomas-Barberan, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Li, F.; Wang, L.; Zhang, D. Fruit and vegetables consumption and risk of hypertension: A meta-analysis. J. Clin. Hypertens (Greenwich) 2016, 18, 468–476. [Google Scholar] [CrossRef]
- Gan, Y.; Tong, X.; Li, L.; Cao, S.; Yin, X.; Gao, C.; Herath, C.; Li, W.; Jin, Z.; Chen, Y.; et al. Consumption of fruit and vegetable and risk of coronary heart disease: A meta-analysis of prospective cohort studies. Int. J. Cardiol. 2015, 183, 129–137. [Google Scholar] [CrossRef]
- Zhan, J.; Liu, Y.J.; Cai, L.B.; Xu, F.R.; Xie, T.; He, Q.Q. Fruit and vegetable consumption and risk of cardiovascular disease: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 1650–1663. [Google Scholar] [CrossRef]
- Kaluza, J.; Larsson, S.C.; Orsini, N.; Linden, A.; Wolk, A. Fruit and vegetable consumption and risk of COPD: A prospective cohort study of men. Thorax 2017, 72, 500–509. [Google Scholar] [CrossRef]
- Wang, Y.; Li, F.; Wang, Z.; Qiu, T.; Shen, Y.; Wang, M. Fruit and vegetable consumption and risk of lung cancer: A dose-response meta-analysis of prospective cohort studies. Lung Cancer 2015, 88, 124–130. [Google Scholar] [CrossRef]
- Tian, Y.; Su, L.; Wang, J.; Duan, X.; Jiang, X. Fruit and vegetable consumption and risk of the metabolic syndrome: A meta-analysis. Public Health Nutr. 2018, 21, 756–765. [Google Scholar] [CrossRef]
- Lonnie, M.; Hooker, E.; Brunstrom, J.M.; Corfe, B.M.; Green, M.A.; Watson, A.W.; Williams, E.A.; Stevenson, E.J.; Penson, S.; Johnstone, A.M. Protein for life: Review of optimal protein intake, sustainable dietary sources and the effect on appetite in ageing adults. Nutrients 2018, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Demeyer, D.; Mertens, B.; De Smet, S.; Ulens, M. Mechanisms linking colorectal cancer to the consumption of (processed) red meat: A review. Crit. Rev. Food Sci. Nutr. 2016, 56, 2747–2766. [Google Scholar] [CrossRef] [PubMed]
- Meat, Fish and Dairy Products and the Risk of Cancer. Available online: https://www.wcrf.org/sites/default/files/Meat-Fish-and-Dairy-products.pdf (accessed on 28 May 2019).
- Della Guardia, L.; Roggi, C.; Cena, H. Diet-induced acidosis and alkali supplementation. Int. J. Food Sci. Nutr. 2016, 67, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Della Guardia, L.; Thomas, M.A.; Cena, H. Insulin sensitivity and glucose homeostasis can be influenced by metabolic acid load. Nutrients 2018, 10, 618. [Google Scholar] [CrossRef]
- Kim, J.E.; O’Connor, L.E.; Sands, L.P.; Slebodnik, M.B.; Campbell, W.W. Effects of dietary protein intake on body composition changes after weight loss in older adults: A systematic review and meta-analysis. Nutr. Rev. 2016, 74, 210–224. [Google Scholar] [CrossRef]
- Curneen, J.M.G.; Casey, M.; Laird, E. The relationship between protein quantity, BMD and fractures in older adults. Ir. J. Med. Sci. 2018, 187, 111–121. [Google Scholar] [CrossRef]
- Cheng, H.; Kong, J.; Underwood, C.; Petocz, P.; Hirani, V.; Dawson, B.; O’Leary, F. Systematic review and meta-analysis of the effect of protein and amino acid supplements in older adults with acute or chronic conditions. Br. J. Nutr. 2018, 119, 527–542. [Google Scholar] [CrossRef]
- Brenna, J.T.; Sacks, G.L. Lipid Structure, Nomenclature, and Chemical Properties. In Biochemistry, Physiology and Molecular Aspects of Human Nutrition, 3rd ed.; Stipanuk, M.H., Caudill, M.A., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 91–119. [Google Scholar]
- de Souza, R.J.; Mente, A.; Maroleanu, A.; Cozma, A.I.; Ha, V.; Kishibe, T.; Uleryk, E.; Budylowski, P.; Schunemann, H.; Beyene, J.; et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ 2015, 351, h3978. [Google Scholar] [CrossRef]
- Ricci, C.; Baumgartner, J.; Zec, M.; Kruger, H.S.; Smuts, C.M. Type of dietary fat intakes in relation to all-cause and cause-specific mortality in US adults: An iso-energetic substitution analysis from the American National Health and Nutrition Examination Survey linked to the US mortality registry. Br. J. Nutr. 2018, 119, 456–463. [Google Scholar] [CrossRef]
- Cederholm, T.; Salem, N., Jr.; Palmblad, J. Omega-3 fatty acids in the prevention of cognitive decline in humans. Adv. Nutr. 2013, 4, 672–676. [Google Scholar] [CrossRef]
- Manuelli, M.; Della Guardia, L.; Cena, H. Enriching diet with n-3 PUFAs to help prevent cardiovascular diseases in healthy adults: Results from clinical trials. Int. J. Mol. Sci. 2017, 18, 1552. [Google Scholar] [CrossRef]
- Buoite Stella, A.; Gortan Cappellari, G.; Barazzoni, R.; Zanetti, M. Update on the impact of omega 3 fatty acids on inflammation, insulin resistance and sarcopenia: A review. Int. J. Mol. Sci. 2018, 19, 218. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3: The good oil. Nutr. Bull. 2017, 42, 132–140. [Google Scholar] [CrossRef]
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018, 77, 52–72. [Google Scholar] [CrossRef]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef]
- Ames, B.N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA 2006, 103, 17589–17594. [Google Scholar] [CrossRef]
- Hohn, A.; Weber, D.; Jung, T.; Ott, C.; Hugo, M.; Kochlik, B.; Kehm, R.; Konig, J.; Grune, T.; Castro, J.P. Happily (n)ever after: Aging in the context of oxidative stress, proteostasis loss and cellular senescence. Redox Biol. 2017, 11, 482–501. [Google Scholar] [CrossRef]
- Popkin, B.M.; D’Anci, K.E.; Rosenberg, I.H. Water, hydration, and health. Nutr. Rev. 2010, 68, 439–458. [Google Scholar] [CrossRef]
- World Health Organization. Nutrients in Drinking Water; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Pistollato, F.; Iglesias, R.C.; Ruiz, R.; Aparicio, S.; Crespo, J.; Lopez, L.D.; Manna, P.P.; Giampieri, F.; Battino, M. Nutritional patterns associated with the maintenance of neurocognitive functions and the risk of dementia and Alzheimer’s disease: A focus on human studies. Pharmacol. Res. 2018, 131, 32–43. [Google Scholar] [CrossRef]
- Campbell, A.P. DASH eating plan: An eating pattern for diabetes management. Diabetes Spectr. 2017, 30, 76–81. [Google Scholar] [CrossRef]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Adamsson, V.; Reumark, A.; Cederholm, T.; Vessby, B.; Riserus, U.; Johansson, G. What is a healthy Nordic diet? Foods and nutrients in the NORDIET study. Food Nutr. Res. 2012, 56. [Google Scholar] [CrossRef] [PubMed]
- Asian Heritage Diet. Available online: https://oldwayspt.org/traditional-diets/asian-heritage-diet (accessed on 10 July 2019).
- Altomare, R.; Cacciabaudo, F.; Damiano, G.; Palumbo, V.D.; Gioviale, M.C.; Bellavia, M.; Tomasello, G.; Lo Monte, A.I. The mediterranean diet: A history of health. Iran. J. Public Health 2013, 42, 449–457. [Google Scholar]
- Rosato, V.; Temple, N.J.; La Vecchia, C.; Castellan, G.; Tavani, A.; Guercio, V. Mediterranean diet and cardiovascular disease: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2019, 58, 173–191. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: An updated systematic review and meta-analysis. Nutrition 2017, 9, 63. [Google Scholar] [CrossRef]
- Esposito, K.; Maiorino, M.I.; Bellastella, G.; Chiodini, P.; Panagiotakos, D.; Giugliano, D. A journey into a Mediterranean diet and type 2 diabetes: A systematic review with meta-analyses. BMJ Open 2015, 5, e008222. [Google Scholar] [CrossRef]
- Petersson, S.D.; Philippou, E. Mediterranean diet, cognitive function, and dementia: A systematic review of the evidence. Adv. Nutr. 2016, 7, 889–904. [Google Scholar] [CrossRef]
- Loughrey, D.G.; Lavecchia, S.; Brennan, S.; Lawlor, B.A.; Kelly, M.E. The impact of the Mediterranean diet on the cognitive functioning of healthy older adults: A systematic review and meta-analysis. Adv. Nutr. 2017, 8, 571–586. [Google Scholar]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Medina-Remon, A.; Tresserra-Rimbau, A.; Pons, A.; Tur, J.A.; Martorell, M.; Ros, E.; Buil-Cosiales, P.; Sacanella, E.; Covas, M.I.; Corella, D.; et al. Effects of total dietary polyphenols on plasma nitric oxide and blood pressure in a high cardiovascular risk cohort. The PREDIMED randomized trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 60–67. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Romero-Mamani, E.S.; Gilabert, R.; Nunez, I.; de la Torre, R.; Corella, D.; Ruiz-Gutierrez, V.; Lopez-Sabater, M.C.; Pinto, X.; Rekondo, J.; et al. Changes in ultrasound-assessed carotid intima-media thickness and plaque with a Mediterranean diet: A substudy of the PREDIMED trial. Arter. Thromb Vasc. Biol. 2014, 34, 439–445. [Google Scholar] [CrossRef]
- Medina-Remon, A.; Zamora-Ros, R.; Rotches-Ribalta, M.; Andres-Lacueva, C.; Martinez-Gonzalez, M.A.; Covas, M.I.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; et al. Total polyphenol excretion and blood pressure in subjects at high cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 323–331. [Google Scholar] [CrossRef]
- Medina-Remon, A.; Casas, R.; Tressserra-Rimbau, A.; Ros, E.; Martinez-Gonzalez, M.A.; Fito, M.; Corella, D.; Salas-Salvado, J.; Lamuela-Raventos, R.M.; Estruch, R. Polyphenol intake from a Mediterranean diet decreases inflammatory biomarkers related to atherosclerosis: A substudy of the PREDIMED trial. Br. J. Clin. Pharmacol. 2017, 83, 114–128. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Chowdhury, S.; Ashor, A.; Oggioni, C.; Mathers, J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 1–15. [Google Scholar] [CrossRef]
- Soltani, S.; Shirani, F.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) diet on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Obes. Rev. 2016, 17, 442–454. [Google Scholar] [CrossRef]
- Shirani, F.; Salehi-Abargouei, A.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH) diet on some risk for developing type 2 diabetes: A systematic review and meta-analysis on controlled clinical trials. Nutrients 2013, 29, 939–947. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Blanco Mejia, S.; Rahelic, D.; Kahleova, H.; Salas-Salvado, J.; Kendall, C.W.; Sievenpiper, J.L. DASH dietary pattern and cardiometabolic outcomes: An umbrella review of systematic reviews and meta-analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef]
- Salehi-Abargouei, A.; Maghsoudi, Z.; Shirani, F.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases--incidence: A systematic review and meta-analysis on observational prospective studies. Nutrients 2013, 29, 611–618. [Google Scholar] [CrossRef]
- Asemi, Z.; Samimi, M.; Tabassi, Z.; Esmaillzadeh, A. The effect of DASH diet on pregnancy outcomes in gestational diabetes: A randomized controlled clinical trial. Eur. J. Clin. Nutr. 2014, 68, 490–495. [Google Scholar] [CrossRef]
- Asghari, G.; Yuzbashian, E.; Mirmiran, P.; Hooshmand, F.; Najafi, R.; Azizi, F. Dietary Approaches to Stop Hypertension (DASH) dietary pattern is associated with reduced incidence of metabolic syndrome in children and adolescents. J. Pediatr. 2016, 174, 178–184.e1. [Google Scholar] [CrossRef]
- Han, S.; Middleton, P.; Shepherd, E.; Van Ryswyk, E.; Crowther, C.A. Different types of dietary advice for women with gestational diabetes mellitus. Cochrane Database Syst. Rev. 2017, 2, Cd009275. [Google Scholar] [CrossRef]
- van de Rest, O.; Berendsen, A.A.; Haveman-Nies, A.; de Groot, L.C. Dietary patterns, cognitive decline, and dementia: A systematic review. Adv. Nutr. 2015, 6, 154–168. [Google Scholar] [CrossRef]
- Chen, X.; Maguire, B.; Brodaty, H.; O’Leary, F. Dietary patterns and cognitive health in older adults: A systematic review. J. Alzheimers Dis. 2019, 67, 583–619. [Google Scholar] [CrossRef]
- Wengreen, H.; Munger, R.G.; Cutler, A.; Quach, A.; Bowles, A.; Corcoran, C.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K.A. Prospective study of Dietary Approaches to Stop Hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: The Cache County Study on Memory, Health and Aging. Am. J. Clin. Nutr. 2013, 98, 1263–1271. [Google Scholar] [CrossRef]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Barnes, L.L.; Bennett, D.A.; Aggarwal, N.T. MIND diet slows cognitive decline with aging. Alzheimers Dement 2015, 11, 1015–1022. [Google Scholar] [CrossRef]
- Adamsson, V.; Reumark, A.; Fredriksson, I.B.; Hammarstrom, E.; Vessby, B.; Johansson, G.; Riserus, U. Effects of a healthy Nordic diet on cardiovascular risk factors in hypercholesterolaemic subjects: A randomized controlled trial (NORDIET). J. Intern. Med. 2011, 269, 150–159. [Google Scholar] [CrossRef]
- Jensen, J.D.; Poulsen, S.K. The new Nordic diet–consumer expenditures and economic incentives estimated from a controlled intervention. BMC Public Health 2013, 13, 1114. [Google Scholar] [CrossRef]
- Mithril, C.; Dragsted, L.O.; Meyer, C.; Blauert, E.; Holt, M.K.; Astrup, A. Guidelines for the New Nordic Diet. Public Health Nutr. 2012, 15, 1941–1947. [Google Scholar] [CrossRef]
- Nordic Nutrition Recommendations 2012: Integral Nutrition and Physical Activity; Nordic Council of Ministers: Copenhagen, Denmark, 2014.
- Uusitupa, M.; Hermansen, K.; Savolainen, M.J.; Schwab, U.; Kolehmainen, M.; Brader, L.; Mortensen, L.S.; Cloetens, L.; Johansson-Persson, A.; Onning, G.; et al. Effects of an isocaloric healthy Nordic diet on insulin sensitivity, lipid profile and inflammation markers in metabolic syndrome—A randomized study (SYSDIET). J. Intern. Med. 2013, 274, 52–66. [Google Scholar] [CrossRef]
- Brader, L.; Uusitupa, M.; Dragsted, L.O.; Hermansen, K. Effects of an isocaloric healthy Nordic diet on ambulatory blood pressure in metabolic syndrome: A randomized SYSDIET sub-study. Eur. J. Clin. Nutr. 2014, 68, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.K.; Due, A.; Jordy, A.B.; Kiens, B.; Stark, K.D.; Stender, S.; Holst, C.; Astrup, A.; Larsen, T.M. Health effect of the New Nordic Diet in adults with increased waist circumference: A 6-mo randomized controlled trial. Am. J. Clin. Nutr. 2014, 99, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Marklund, M.; Magnusdottir, O.K.; Rosqvist, F.; Cloetens, L.; Landberg, R.; Kolehmainen, M.; Brader, L.; Hermansen, K.; Poutanen, K.S.; Herzig, K.H.; et al. A dietary biomarker approach captures compliance and cardiometabolic effects of a healthy Nordic diet in individuals with metabolic syndrome. J. Nutr. 2014, 144, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, L.B.; Damsgaard, C.T.; Dalskov, S.M.; Petersen, R.A.; Egelund, N.; Dyssegaard, C.B.; Stark, K.D.; Andersen, R.; Tetens, I.; Astrup, A.; et al. Diet-induced changes in iron and n-3 fatty acid status and associations with cognitive performance in 8–11-year-old Danish children: Secondary analyses of the Optimal Well-Being, Development and Health for Danish Children through a Healthy New Nordic Diet School Meal Study. Br. J. Nutr. 2015, 114, 1623–1637. [Google Scholar]
- Akesson, A.; Andersen, L.F.; Kristjansdottir, A.G.; Roos, E.; Trolle, E.; Voutilainen, E.; Wirfalt, E. Health effects associated with foods characteristic of the Nordic diet: A systematic literature review. Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef]
- Kim, S.; Kim, M.; Lee, M.; Park, Y.; Lee, H.; Kang, S.; Lee, H.; Lee, K.; Yang, H.; Kim, M.; et al. Korean diet: Characteristics and historical background. J. Ethn. Foods 2016, 3, 26–31. [Google Scholar] [CrossRef]
- Lee, K.W.; Cho, M.S. The traditional Korean dietary pattern is associated with decreased risk of metabolic syndrome: Findings from the Korean National Health and Nutrition Examination Survey, 1998–2009. J. Med. Food 2014, 17, 43–56. [Google Scholar] [CrossRef]
- Jung, S.J.; Park, S.H.; Choi, E.K.; Cha, Y.S.; Cho, B.H.; Kim, Y.G.; Kim, M.G.; Song, W.O.; Park, T.S.; Ko, J.K.; et al. Beneficial effects of Korean traditional diets in hypertensive and type 2 diabetic patients. J. Med. Food 2014, 17, 161–171. [Google Scholar] [CrossRef]
- Ma, G. Food, eating behavior and culture in Chinese society. J. Ethn. Foods 2015, 2, 195–199. [Google Scholar] [CrossRef]
- Introducing the Updated Asian Diet Pyramid. Available online: https://oldwayspt.org/blog/introducing-updated-asian-diet-pyramid (accessed on 29 May 2019).
- Wan, Y.; Wang, F.; Yuan, J.; Li, J.; Jiang, D.; Zhang, J.; Huang, T.; Zheng, J.; Mann, J.; Li, D. Effects of macronutrient distribution on weight and related cardiometabolic profile in healthy non-obese Chinese: A 6-month, randomized controlled-feeding trial. EBioMedicine 2017, 22, 200–207. [Google Scholar] [CrossRef]
- Leonetti, F.; Liguori, A.; Petti, F.; Rughini, S.; Silli, L.; Liguori, S.; Bangrazi, S. Effects of basic traditional Chinese diet on body mass index, lean body mass, and eating and hunger behaviours in overweight or obese individuals. J. Tradit Chin. Med. 2016, 36, 456–463. [Google Scholar] [CrossRef]
- Liguori, A.; Petti, F.; Rughini, S.; Silli, L.; Asprino, R.; Maglio, C.; Leonetti, F. Effect of a basic Chinese traditional diet in overweight patients. J. Tradit Chin. Med. 2013, 33, 322–324. [Google Scholar] [CrossRef]
- Gabriel, A.S.; Ninomiya, K.; Uneyama, H. The role of the Japanese traditional diet in healthy and sustainable dietary patterns around the world. Nutrients 2018, 10, 173. [Google Scholar] [CrossRef] [PubMed]
- Niu, K.; Momma, H.; Kobayashi, Y.; Guan, L.; Chujo, M.; Otomo, A.; Ouchi, E.; Nagatomi, R. The traditional Japanese dietary pattern and longitudinal changes in cardiovascular disease risk factors in apparently healthy Japanese adults. Eur. J. Nutr. 2016, 55, 267–279. [Google Scholar] [CrossRef]
- Htun, N.C.; Suga, H.; Imai, S.; Shimizu, W.; Takimoto, H. Food intake patterns and cardiovascular risk factors in Japanese adults: Analyses from the 2012 National Health and nutrition survey, Japan. Nutr. J. 2017, 16, 61. [Google Scholar] [CrossRef]
- Turconi, G.T.; Nucera, A.; Roggi, C.; Cena, H. Food consumption and diet cost: A northern Italian survey. Italy J. Food Sci. 2010, 22, 352–358. [Google Scholar]
- Bourassa, M.W.; Osendarp, S.J.M.; Adu-Afarwuah, S.; Ahmed, S.; Ajello, C.; Bergeron, G.; Black, R.; Christian, P.; Cousens, S.; de Pee, S.; et al. Review of the evidence regarding the use of antenatal multiple micronutrient supplementation in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2019, 1444, 6–21. [Google Scholar] [CrossRef] [PubMed]
- Swed-Tobia, R.; Haj, A.; Militianu, D.; Eshach, O.; Ravid, S.; Weiss, R.; Aviel, Y.B. Highly selective eating in autism spectrum disorder leading to scurvy: A series of three patients. Pediatr. Neurol. 2019, 94, 61–63. [Google Scholar] [CrossRef]
- National Institute of Health. Calcium Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/ (accessed on 12 February 2019).
- African Heritage Diet. Available online: https://oldwayspt.org/traditional-diets/african-heritage-diet (accessed on 6 January 2020).
| Dietary Component | Recommended Servings | ||||
|---|---|---|---|---|---|
| Mediterranean [7] | DASH [49] a | MIND [9,50] | Healthy Nordic [51] | Traditional Asian [52] | |
| Fruits | 1–2/meal | 4–5 servings/day | Berries: ≥ 2 servings/week | Fruits, berries, vegetables, and potatoes: ≥ 500 g/day | Daily |
| Vegetables | ≥ 2 servings/meal | 4–5 servings/day | Green leafy: ≥ 6 servings/week Other: ≥ 1 serving/day | Daily | |
| Whole grains | 1–2 servings/meal | 7–8 servings/day | ≥ 3 servings/day | Bread: 4–6 slices/day Cereal: 1.5 servings/day Pasta: 3 servings/week β-glucan-rich foods: 3 g/d | Daily |
| Dairy | Low-fat: 2 servings/day | Low- or non-fat: 2–3 servings/day | Cheese: < 1 serving/week Butter: < 1 Tbsp/day | Low-fat milk: ≤ 5 dL/day Cheese: for cooking b | Yogurt: daily to weekly |
| Nuts, seeds, and legumes | Olives/nuts/seeds: 1–2 servings/day Legumes: ≥ 2 servings/week | 4–5 servings/week | Nuts: ≥ 5 servings/week Beans: > 3 servings/week | Nuts (mostly almonds): 15 g/day | Daily |
| Beef, pork, ham, lamb, veal, poultry | Red meat: < 2 servings/week Processed meat: ≤ 1 servings/week White meat: 2 servings/week | Lean protein: ≤ 2 servings/day | Red meat: < 4 servings/week | Meat: ≤ 500 g/week | Red meat: infrequent |
| Poultry: ≥ 2 servings/week | Poultry: ≤ 300 g/week | Poultry: Daily to weekly | |||
| Fish/seafood | ≥ 2 servings/week | ≥ 1 serving/week | 3–5 servings/week | 2 servings/week | |
| Fats, oils, and salad dressing | Olive oil: 1–2 servings/meal | 2–3 servings /day | Olive oil as primary oil | 5 g/bread slice 0.5 dL/day as dressing | Healthy cooking oils: daily to weekly |
| Sweets | ≤ 2 servings/week | ≤ 5 servings/week | Pastries & sweets: < 5 servings/week | On weekends | Infrequent |
| Other | Eggs: 2–4 servings/week Potatoes: ≤ 3 servings//week | Sodium < 2,300 mg/day | Fried or fast food: < 1 serving/week | Eggs: Stay within daily recommended cholesterol intake Fruit/vegetable juice: 4 dL/week | Eggs: daily to weekly |
| Alcohol | Wine: in moderation | Women: ≤ 1 drink/day Men: ≤ 2 drinks/day | 1 glass/day | Habitual amount | In moderation |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cena, H.; Calder, P.C. Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease. Nutrients 2020, 12, 334. https://doi.org/10.3390/nu12020334
Cena H, Calder PC. Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease. Nutrients. 2020; 12(2):334. https://doi.org/10.3390/nu12020334
Chicago/Turabian StyleCena, Hellas, and Philip C. Calder. 2020. "Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease" Nutrients 12, no. 2: 334. https://doi.org/10.3390/nu12020334
APA StyleCena, H., & Calder, P. C. (2020). Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease. Nutrients, 12(2), 334. https://doi.org/10.3390/nu12020334