The Emerging Role of Vitamin C as a Treatment for Sepsis
Abstract
1. Introduction
2. Pharmacology and Physiology of Vitamin C
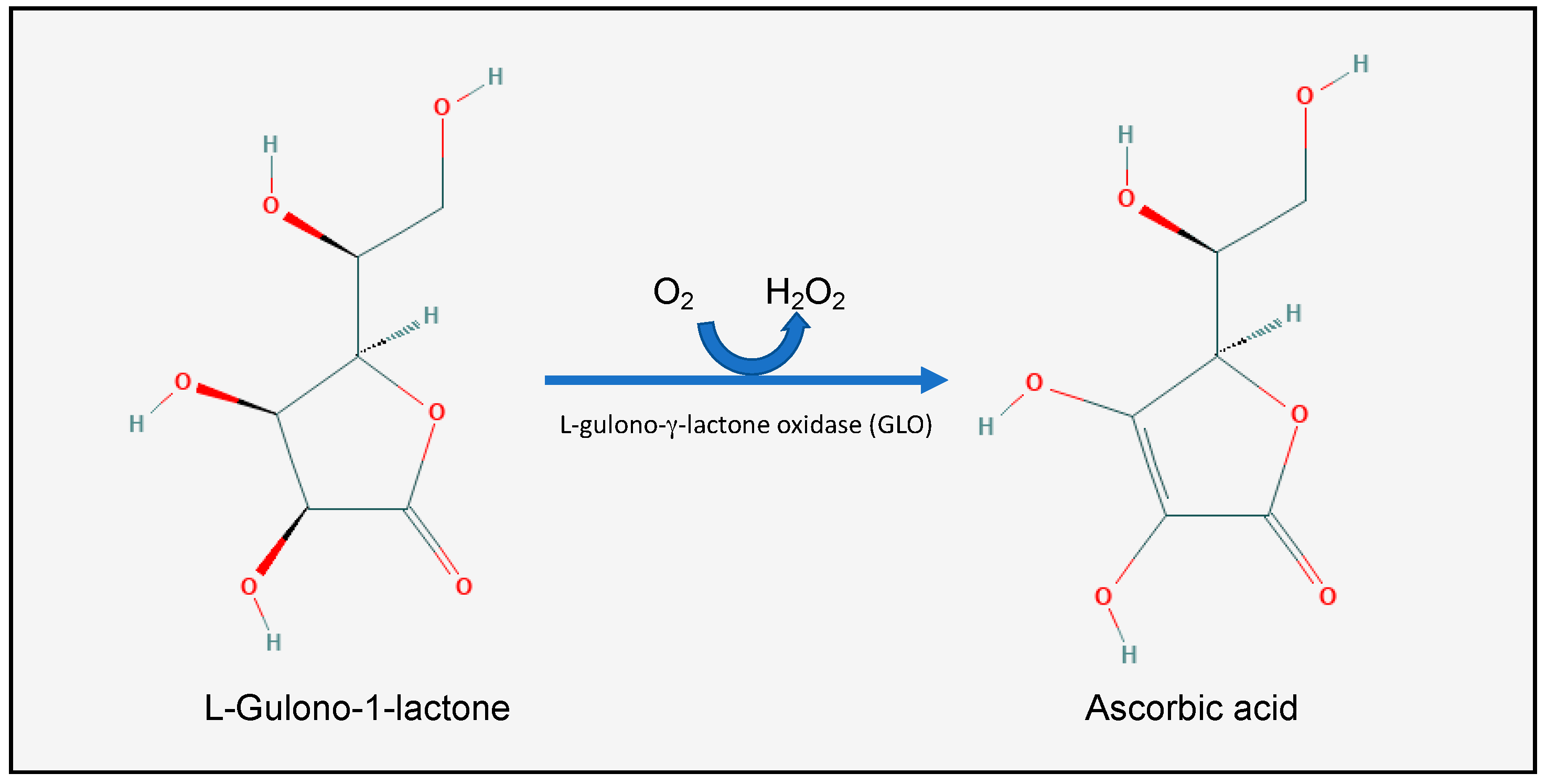
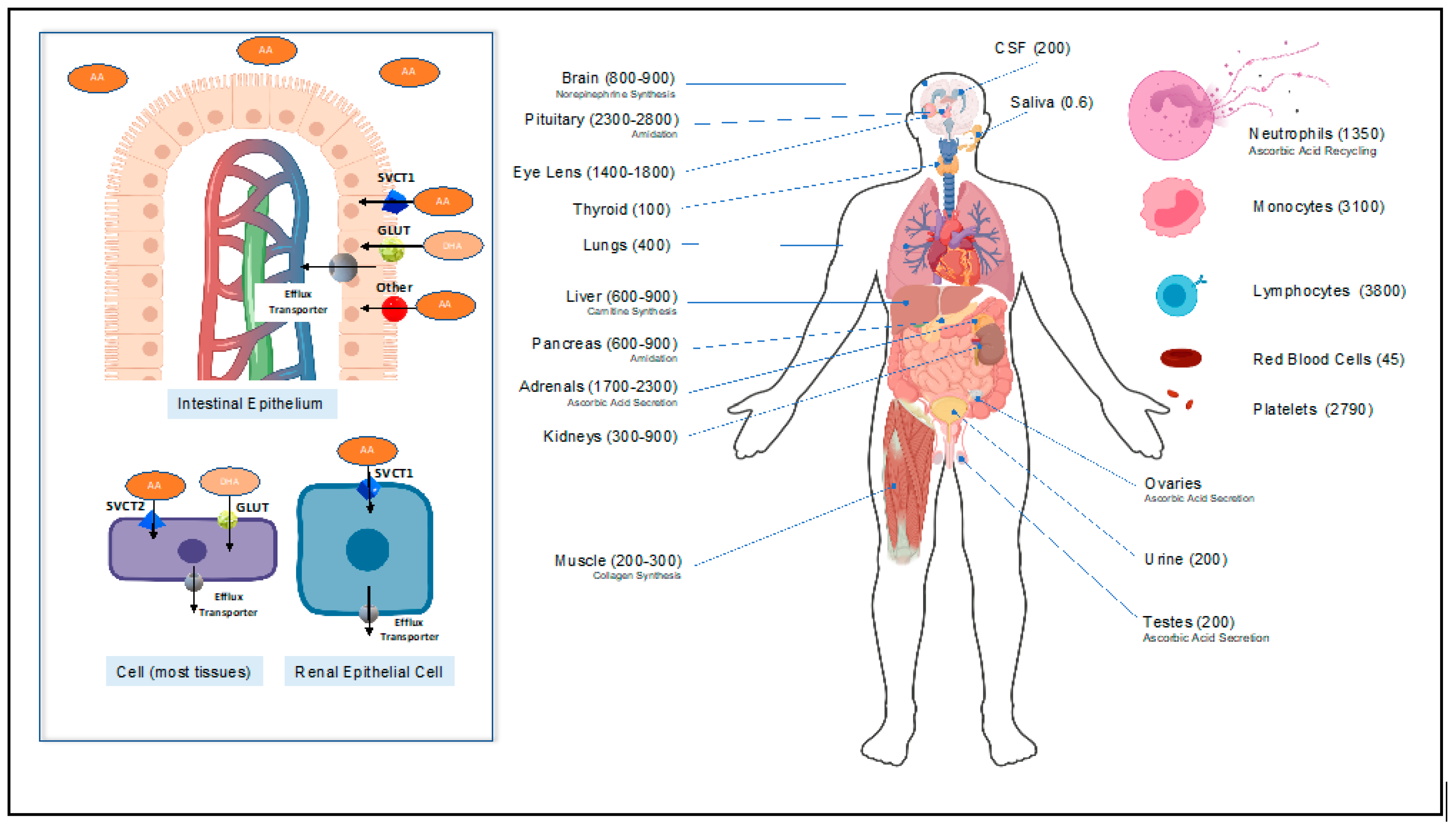
2.1. Vitamin C Homeostasis
2.2. Pleiotropic Physiologic Functions of Vitamin C
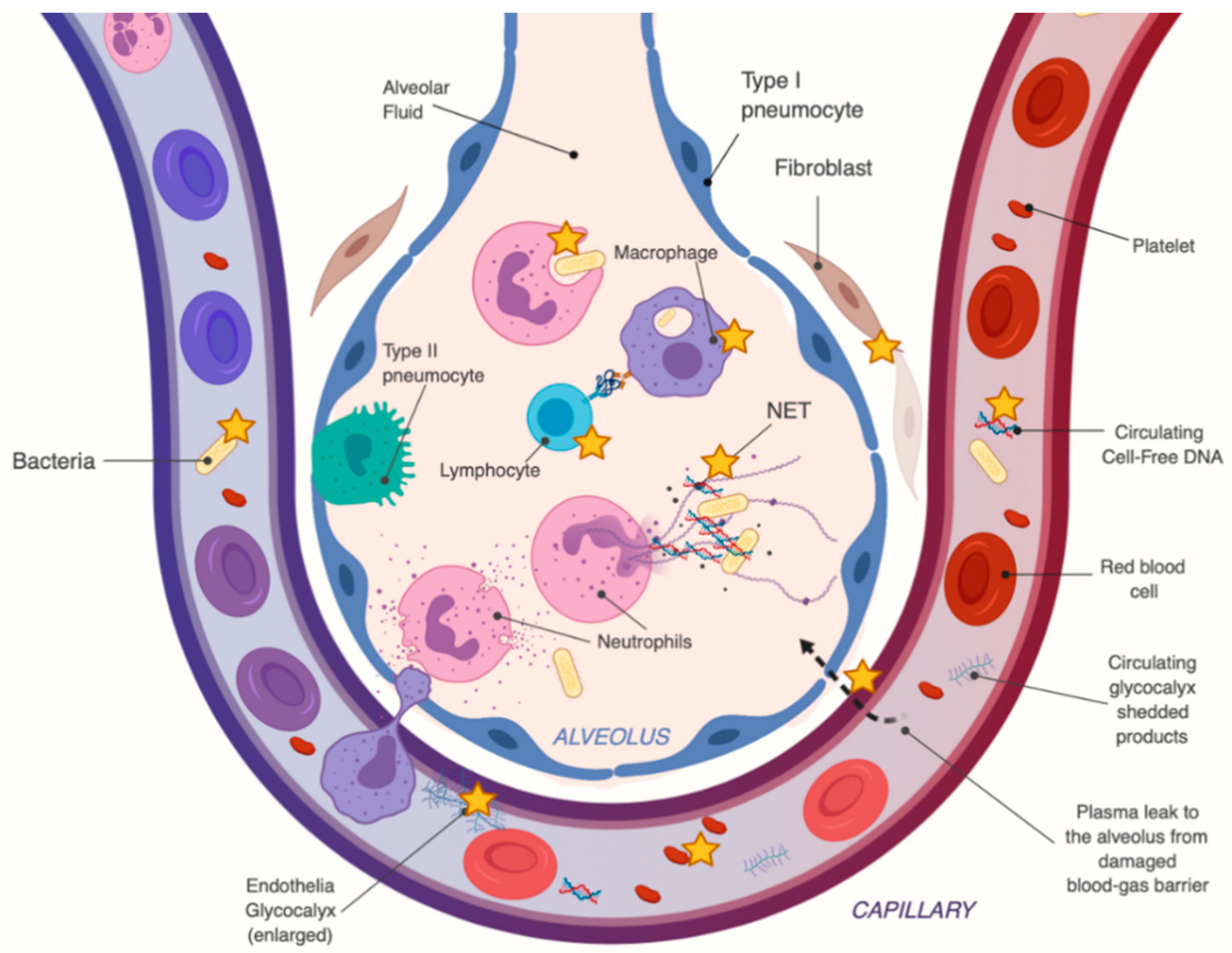
2.3. Vitamin C’s Mechanism of Action in Sepsis and ARDS
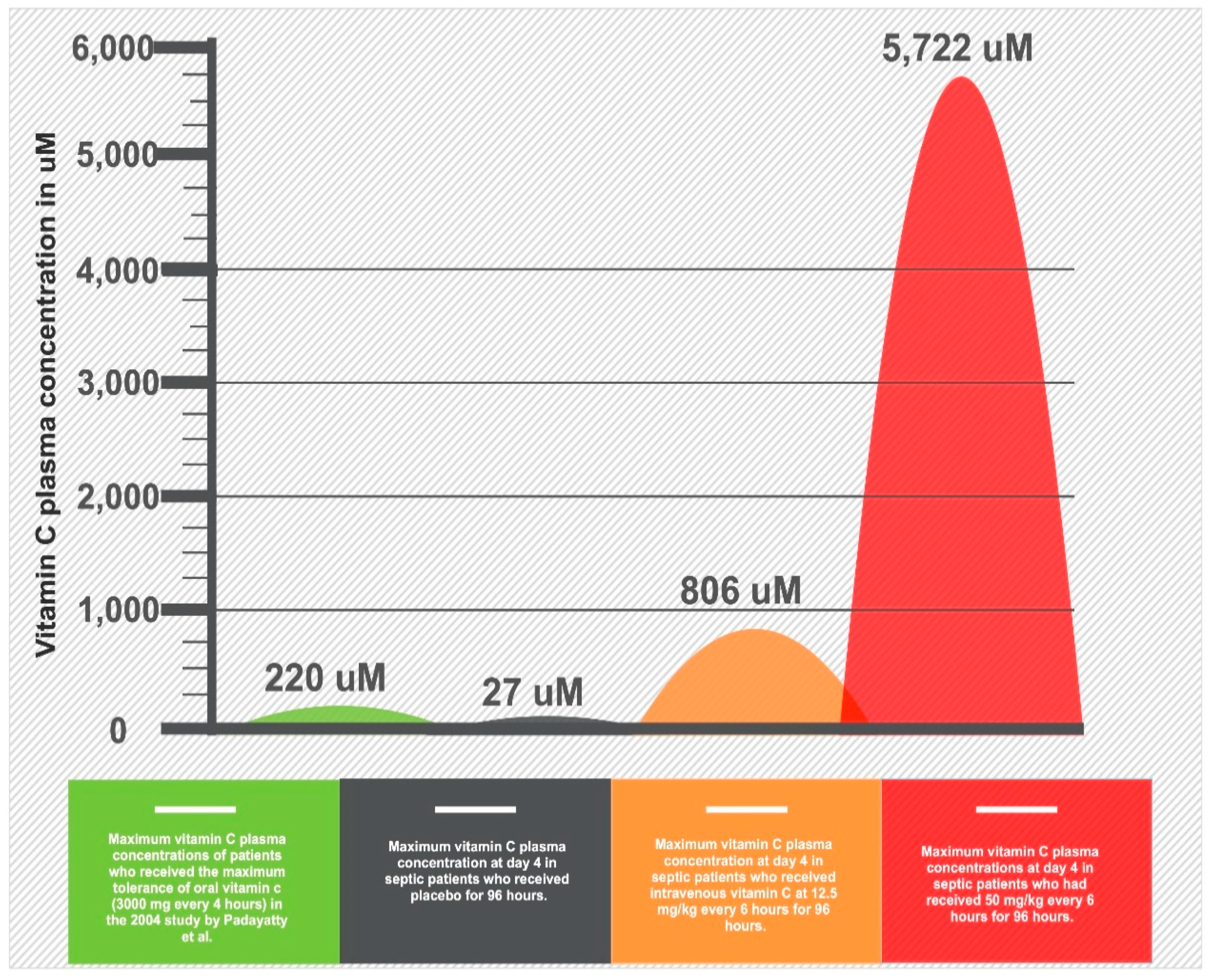
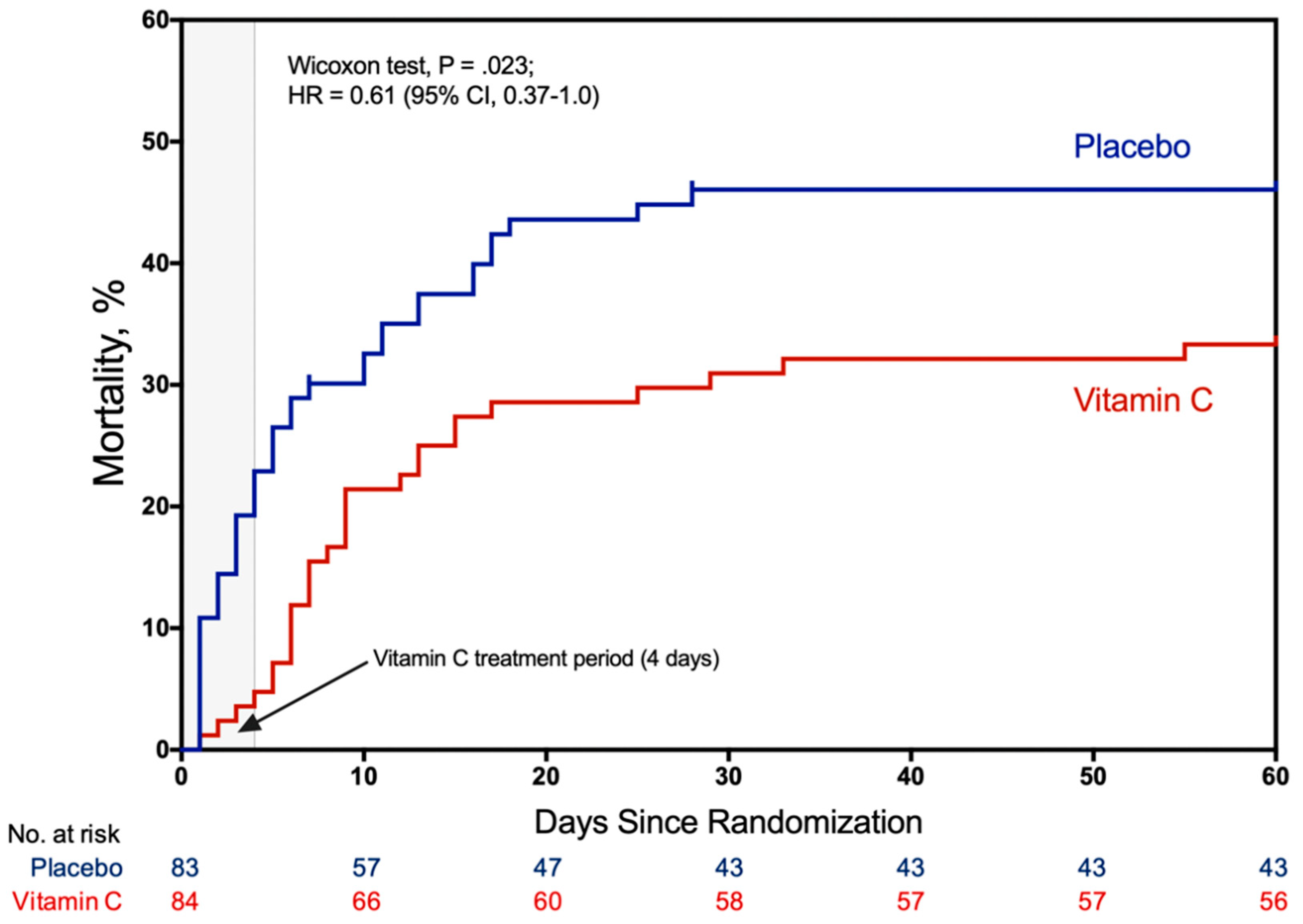
3. Clinical Trials
4. Adverse Effects of Vitamin C Therapy
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Fleischmann, C.; Scherag, A.; Adhikari, N.K.J.; Hartog, C.S.; Tsaganos, T.; Schlattmann, P.; Angus, D.C.; Reinhart, K. International Forum of Acute Care Trialists Assessment of Global Incidence and Mortality of Hospital-treated Sepsis. Current Estimates and Limitations. Am. J. Respir. Crit. Care Med. 2016, 193, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Walkey, A.J.; Lagu, T.; Lindenauer, P.K. Trends in sepsis and infection sources in the United States. A population-based study. Ann. Am. Thorac. Soc. 2015, 12, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Andreu Ballester, J.C.; Ballester, F.; González Sánchez, A.; Almela Quilis, A.; Colomer Rubio, E.; Peñarroja Otero, C. Epidemiology of sepsis in the Valencian Community (Spain), 1995–2004. Infect. Control Hosp. Epidemiol. 2008, 29, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Vincent, J.-L.; Adhikari, N.K.J.; Machado, F.R.; Angus, D.C.; Calandra, T.; Jaton, K.; Giulieri, S.; Delaloye, J.; Opal, S.; et al. Sepsis: A roadmap for future research. Lancet Infect. Dis. 2015, 15, 581–614. [Google Scholar] [CrossRef]
- Sheu, C.-C.; Gong, M.N.; Zhai, R.; Chen, F.; Bajwa, E.K.; Clardy, P.F.; Gallagher, D.C.; Thompson, B.T.; Christiani, D.C. Clinical characteristics and outcomes of sepsis-related vs non-sepsis-related ARDS. Chest 2010, 138, 559–567. [Google Scholar] [CrossRef]
- Kaukonen, K.-M.; Bailey, M.; Suzuki, S.; Pilcher, D.; Bellomo, R. Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000–2012. JAMA 2014, 311, 1308–1316. [Google Scholar] [CrossRef]
- Rhee, C.; Jones, T.M.; Hamad, Y.; Pande, A.; Varon, J.; O’Brien, C.; Anderson, D.J.; Warren, D.K.; Dantes, R.B.; Epstein, L.; et al. Centers for Disease Control and Prevention (CDC) Prevention Epicenters Program Prevalence, Underlying Causes, and Preventability of Sepsis-Associated Mortality in US Acute Care Hospitals. JAMA Netw. Open 2019, 2, e187571. [Google Scholar] [CrossRef]
- Salvemini, D.; Cuzzocrea, S. Oxidative stress in septic shock and disseminated intravascular coagulation. Free Radic. Biol. Med. 2002, 33, 1173–1185. [Google Scholar] [CrossRef]
- Marshall, J.C. Clinical trials of mediator-directed therapy in sepsis: What have we learned? Intensive Care Med. 2000, 26 (Suppl. 1), S75–S83. [Google Scholar] [CrossRef]
- Kruger, P.; Bailey, M.; Bellomo, R.; Cooper, D.J.; Harward, M.; Higgins, A.; Howe, B.; Jones, D.; Joyce, C.; Kostner, K.; et al. ANZ-STATInS Investigators–ANZICS Clinical Trials Group A multicenter randomized trial of atorvastatin therapy in intensive care patients with severe sepsis. Am. J. Respir. Crit. Care Med. 2013, 187, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Truwit, J.D.; Bernard, G.R.; Steingrub, J.; Matthay, M.A.; Liu, K.D.; Albertson, T.E.; Brower, R.G.; Shanholtz, C.; Rock, P.; Douglas, I.S.; et al. Rosuvastatin for sepsis-associated acute respiratory distress syndrome. N. Engl. J. Med. 2014, 370, 2191–2200. [Google Scholar] [PubMed]
- Ranieri, V.M.; Thompson, B.T.; Barie, P.S.; Dhainaut, J.-F.; Douglas, I.S.; Finfer, S.; Gårdlund, B.; Marshall, J.C.; Rhodes, A.; Artigas, A.; et al. PROWESS-SHOCK Study Group Drotrecogin alfa (activated) in adults with septic shock. N. Engl. J. Med. 2012, 366, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, E.J.; Fisher, C.J.; Sprung, C.L.; Straube, R.C.; Sadoff, J.C.; Foulke, G.E.; Wortel, C.H.; Fink, M.P.; Dellinger, R.P.; Teng, N.N. Treatment of gram-negative bacteremia and septic shock with HA-1A human monoclonal antibody against endotoxin. A randomized, double-blind, placebo-controlled trial. The HA-1A Sepsis Study Group. N. Engl. J. Med. 1991, 324, 429–436. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute Ascorbic Acid (Code C285). Available online: https://ncit.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCI_Thesaurus&ns=NCI_Thesaurus&code=C285 (accessed on 11 September 2019).
- Drouin, G.; Godin, J.-R.; Pagé, B. The genetics of vitamin C loss in vertebrates. Curr. Genom. 2011, 12, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Lachapelle, M.Y.; Drouin, G. Inactivation dates of the human and guinea pig vitamin C genes. Genetica 2011, 139, 199–207. [Google Scholar] [CrossRef]
- Pauling, L. Evolution and the need for ascorbic acid. Proc. Natl. Acad. Sci. USA 1970, 67, 1643–1648. [Google Scholar] [CrossRef]
- Granger, M.; Eck, P. Dietary vitamin C in human health. Adv. Food Nutr. Res. 2018, 83, 281–310. [Google Scholar]
- Hodges, R.E.; Hood, J.; Canham, J.E.; Sauberlich, H.E.; Baker, E.M. Clinical manifestations of ascorbic acid deficiency in man. Am. J. Clin. Nutr. 1971, 24, 432–443. [Google Scholar] [CrossRef]
- Tsukaguchi, H.; Tokui, T.; Mackenzie, B.; Berger, U.V.; Chen, X.Z.; Wang, Y.; Brubaker, R.F.; Hediger, M.A. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature 1999, 399, 70–75. [Google Scholar] [CrossRef]
- Daruwala, R.; Song, J.; Koh, W.S.; Rumsey, S.C.; Levine, M. Cloning and functional characterization of the human sodium-dependent vitamin C transporters hSVCT1 and hSVCT2. FEBS Lett. 1999, 460, 480–484. [Google Scholar] [CrossRef]
- Corpe, C.P.; Eck, P.; Wang, J.; Al-Hasani, H.; Levine, M. Intestinal dehydroascorbic acid (DHA) transport mediated by the facilitative sugar transporters, GLUT2 and GLUT8. J. Biol. Chem. 2013, 288, 9092–9101. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, V.; Diederich, L.; Keller, T.C.S.; Kramer, C.M.; Lückstädt, W.; Panknin, C.; Suvorava, T.; Isakson, B.E.; Kelm, M.; Cortese-Krott, M.M.; et al. Red blood cell function and dysfunction: Redox regulation, nitric oxide metabolism, anemia. Antioxid. Redox Signal. 2017, 26, 718–742. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef]
- Schorah, C.J.; Downing, C.; Piripitsi, A.; Gallivan, L.; Al-Hazaa, A.H.; Sanderson, M.J.; Bodenham, A. Total vitamin C, ascorbic acid, and dehydroascorbic acid concentrations in plasma of critically ill patients. Am. J. Clin. Nutr. 1996, 63, 760–765. [Google Scholar] [CrossRef]
- Long, C.L.; Maull, K.I.; Krishnan, R.S.; Laws, H.L.; Geiger, J.W.; Borghesi, L.; Franks, W.; Lawson, T.C.; E Sauberlich, H. Ascorbic acid dynamics in the seriously ill and injured. J. Surg. Res. 2003, 109, 144–148. [Google Scholar] [CrossRef]
- Metnitz, P.G.; Bartens, C.; Fischer, M.; Fridrich, P.; Steltzer, H.; Druml, W. Antioxidant status in patients with acute respiratory distress syndrome. Intensive Care Med. 1999, 25, 180–185. [Google Scholar] [CrossRef]
- Fulzele, S.; Chothe, P.; Sangani, R.; Chutkan, N.; Hamrick, M.; Bhattacharyya, M.; Prasad, P.B.; Zakhary, I.; Bowser, M.; Ganapathy, V.; et al. Sodium-dependent vitamin C transporter SVCT2: Expression and function in bone marrow stromal cells and in osteogenesis. Stem Cell Res. 2013, 10, 36–47. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, S.M.; Katz, A.; Wesley, R.A.; Levine, M. Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef]
- Wilson, J.X. Mechanism of action of vitamin C in sepsis: Ascorbate modulates redox signaling in endothelium. Biofactors 2009, 35, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.X. Evaluation of vitamin C for adjuvant sepsis therapy. Antioxid. Redox Signal. 2013, 19, 2129–2140. [Google Scholar] [CrossRef]
- Frei, B.; Stocker, R.; England, L.; Ames, B.N. Ascorbate: The most effective antioxidant in human blood plasma. Adv. Exp. Med. Biol. 1990, 264, 155–163. [Google Scholar] [PubMed]
- Koekkoek, W.A.C.K.; van Zanten, A.R.H. Antioxidant vitamins and trace elements in critical illness. Nutr. Clin. Pract. 2016, 31, 457–474. [Google Scholar] [CrossRef] [PubMed]
- Nathens, A.B.; Neff, M.J.; Jurkovich, G.J.; Klotz, P.; Farver, K.; Ruzinski, J.T.; Radella, F.; Garcia, I.; Maier, R.V. Randomized, prospective trial of antioxidant supplementation in critically ill surgical patients. Ann. Surg. 2002, 236, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.T.; Lin, H.I.; Yeh, D.Y.; Chou, T.Y.; Chen, C.F.; Leu, F.C.; Wang, D.; Hu, R.T. Effects of the antioxidants lycium barbarum and ascorbic acid on reperfusion liver injury in rats. Transpl. Proc. 2009, 41, 4110–4113. [Google Scholar] [CrossRef]
- Lee, W.-Y.; Lee, J.-S.; Lee, S.-M. Protective effects of combined ischemic preconditioning and ascorbic acid on mitochondrial injury in hepatic ischemia/reperfusion. J. Surg. Res. 2007, 142, 45–52. [Google Scholar] [CrossRef]
- Berger, M.M.; Soguel, L.; Shenkin, A.; Revelly, J.-P.; Pinget, C.; Baines, M.; Chioléro, R.L. Influence of early antioxidant supplements on clinical evolution and organ function in critically ill cardiac surgery, major trauma, and subarachnoid hemorrhage patients. Crit. Care 2008, 12, R101. [Google Scholar] [CrossRef]
- Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, 418. [Google Scholar] [CrossRef]
- Kagan, V.E.; Serbinova, E.A.; Forte, T.; Scita, G.; Packer, L. Recycling of vitamin E in human low density lipoproteins. J. Lipid Res. 1992, 33, 385–397. [Google Scholar] [PubMed]
- Noguchi, N.; Iwaki, Y.; Takahashi, M.; Komuro, E.; Kato, Y.; Tamura, K.; Cynshi, O.; Kodama, T.; Niki, E. 2,3-Dihydro-5-hydroxy-2,2-dipentyl-4,6-di-tert-butylbenzofuran: Design and evaluation as a novel radical-scavenging antioxidant against lipid peroxidation. Arch. Biochem. Biophys. 1997, 342, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Eipper, B.A.; Mains, R.E. The role of ascorbate in the biosynthesis of neuroendocrine peptides. Am. J. Clin. Nutr. 1991, 54, 1153S–1156S. [Google Scholar] [CrossRef]
- Arrigoni, O.; De Tullio, M.C. The role of ascorbic acid in cell metabolism: Between gene-directed functions and unpredictable chemical reactions. J. Plant Physiol. 2000, 157, 481–488. [Google Scholar] [CrossRef]
- Gorres, K.L.; Raines, R.T. Prolyl 4-hydroxylase. Crit. Rev. Biochem. Mol. Biol. 2010, 45, 106–124. [Google Scholar] [CrossRef]
- Kivirikko, K.I.; Myllylä, R. Post-translational processing of procollagens. Ann. N. Y. Acad. Sci. 1985, 460, 187–201. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Saito, N.; Kurita, K.; Shimokado, K.; Maruyama, N.; Ishigami, A. Ascorbic acid enhances the expression of type 1 and type 4 collagen and SVCT2 in cultured human skin fibroblasts. Biochem. Biophys. Res. Commun. 2013, 430, 579–584. [Google Scholar] [CrossRef]
- Nytko, K.J.; Maeda, N.; Schläfli, P.; Spielmann, P.; Wenger, R.H.; Stiehl, D.P. Vitamin C is dispensable for oxygen sensing in vivo. Blood 2011, 117, 5485–5493. [Google Scholar] [CrossRef]
- Bruick, R.K.; McKnight, S.L. A conserved family of prolyl-4-hydroxylases that modify HIF. Science 2001, 294, 1337–1340. [Google Scholar] [CrossRef]
- Grano, A.; De Tullio, M.C. Ascorbic acid as a sensor of oxidative stress and a regulator of gene expression: The Yin and Yang of vitamin C. Med. Hypotheses 2007, 69, 953–954. [Google Scholar] [CrossRef]
- Ke, Q.; Costa, M. Hypoxia-inducible factor-1 (HIF-1). Mol. Pharm. 2006, 70, 1469–1480. [Google Scholar] [CrossRef] [PubMed]
- Nelson, P.J.; Pruitt, R.E.; Henderson, L.L.; Jenness, R.; Henderson, L.M. Effect of ascorbic acid deficiency on the in vivo synthesis of carnitine. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1981, 672, 123–127. [Google Scholar] [CrossRef]
- Rebouche, C.J. Ascorbic acid and carnitine biosynthesis. Am. J. Clin. Nutr. 1991, 54, 1147S–1152S. [Google Scholar] [CrossRef] [PubMed]
- Winter, B.K.; Fiskum, G.; Gallo, L.L. Effects of L-carnitine on serum triglyceride and cytokine levels in rat models of cachexia and septic shock. Br. J. Cancer 1995, 72, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Foroozanfar, N.; Lucas, C.F.; Joss, D.V.; Hugh-Jones, K.; Hobbs, J.R. Ascorbate (1g/day) does not help the phagocyte killing defect of X-linked chronic granulomatous disease. Clin. Exp. Immunol. 1983, 51, 99–102. [Google Scholar]
- Goldschmidt, M.C. Reduced bactericidal activity in neutrophils from scorbutic animals and the effect of ascorbic acid on these target bacteria in vivo and in vitro. Am. J. Clin. Nutr. 1991, 54, 1214S–1220S. [Google Scholar] [CrossRef]
- Goldschmidt, M.C.; Masin, W.J.; Brown, L.R.; Wyde, P.R. The effect of ascorbic acid deficiency on leukocyte phagocytosis and killing of actinomyces viscosus. Int. J. Vitam. Nutr. Res. 1988, 58, 326–334. [Google Scholar]
- Rebora, A.; Dallegri, F.; Patrone, F. Neutrophil dysfunction and repeated infections: Influence of levamisole and ascorbic acid. Br. J. Dermatol. 1980, 102, 49–56. [Google Scholar] [CrossRef]
- Shilotri, P.G. Glycolytic, hexose monophosphate shunt and bactericidal activities of leukocytes in ascorbic acid deficient guinea pigs. J. Nutr. 1977, 107, 1507–1512. [Google Scholar] [CrossRef]
- Johnston, C.S.; Huang, S.N. Effect of ascorbic acid nutriture on blood histamine and neutrophil chemotaxis in guinea pigs. J. Nutr. 1991, 121, 126–130. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Washko, P.W.; Wang, Y.; Levine, M. Ascorbic acid recycling in human neutrophils. J. Biol. Chem. 1993, 268, 15531–15535. [Google Scholar] [PubMed]
- Chen, Y.; Luo, G.; Yuan, J.; Wang, Y.; Yang, X.; Wang, X.; Li, G.; Liu, Z.; Zhong, N. Vitamin C mitigates oxidative stress and tumor necrosis factor-alpha in severe community-acquired pneumonia and LPS-induced macrophages. Mediat. Inflamm. 2014, 2014, 426740. [Google Scholar] [CrossRef]
- Cárcamo, J.M.; Pedraza, A.; Bórquez-Ojeda, O.; Zhang, B.; Sanchez, R.; Golde, D.W. Vitamin C is a kinase inhibitor: Dehydroascorbic acid inhibits IkappaBalpha kinase beta. Mol. Cell. Biol. 2004, 24, 6645–6652. [Google Scholar] [CrossRef]
- Peng, Y.; Kwok, K.H.H.; Yang, P.-H.; Ng, S.S.M.; Liu, J.; Wong, O.G.; He, M.-L.; Kung, H.-F.; Lin, M.C.M. Ascorbic acid inhibits ROS production, NF-kappa B activation and prevents ethanol-induced growth retardation and microencephaly. Neuropharmacology 2005, 48, 426–434. [Google Scholar] [CrossRef]
- Ferrón-Celma, I.; Mansilla, A.; Hassan, L.; Garcia-Navarro, A.; Comino, A.-M.; Bueno, P.; Ferrón, J.-A. Effect of vitamin C administration on neutrophil apoptosis in septic patients after abdominal surgery. J. Surg. Res. 2009, 153, 224–230. [Google Scholar] [CrossRef]
- Hampton, M.B.; Fadeel, B.; Orrenius, S. Redox regulation of the caspases during apoptosis. Ann. N. Y. Acad. Sci. 1998, 854, 328–335. [Google Scholar] [CrossRef]
- Mikacenic, C.; Moore, R.; Dmyterko, V.; West, T.E.; Altemeier, W.A.; Liles, W.C.; Lood, C. Neutrophil extracellular traps (NETs) are increased in the alveolar spaces of patients with ventilator-associated pneumonia. Crit. Care 2018, 22, 358. [Google Scholar] [CrossRef]
- Zawrotniak, M.; Rapala-Kozik, M. Neutrophil extracellular traps (NETs)-formation and implications. Acta Biochim. Pol. 2013, 60, 277–284. [Google Scholar] [CrossRef]
- Lefrançais, E.; Mallavia, B.; Zhuo, H.; Calfee, C.S.; Looney, M.R. Maladaptive role of neutrophil extracellular traps in pathogen-induced lung injury. JCI Insight 2018, 3, 3. [Google Scholar] [CrossRef]
- Yang, S.; Qi, H.; Kan, K.; Chen, J.; Xie, H.; Guo, X.; Zhang, L. Neutrophil extracellular traps promote hypercoagulability in patients with sepsis. Shock 2017, 47, 132–139. [Google Scholar] [CrossRef]
- Liu, S.; Su, X.; Pan, P.; Zhang, L.; Hu, Y.; Tan, H.; Wu, D.; Liu, B.; Li, H.; Li, H.; et al. Neutrophil extracellular traps are indirectly triggered by lipopolysaccharide and contribute to acute lung injury. Sci. Rep. 2016, 6, 37252. [Google Scholar] [CrossRef]
- Czaikoski, P.G.; Mota, J.M.S.C.; Nascimento, D.C.; Sônego, F.; Castanheira, F.V.E.S.; Melo, P.H.; Scortegagna, G.T.; Silva, R.L.; Barroso-Sousa, R.; Souto, F.O.; et al. Neutrophil Extracellular Traps Induce Organ Damage during Experimental and Clinical Sepsis. PLoS ONE 2016, 11, e0148142. [Google Scholar] [CrossRef]
- Saffarzadeh, M.; Juenemann, C.; Queisser, M.A.; Lochnit, G.; Barreto, G.; Galuska, S.P.; Lohmeyer, J.; Preissner, K.T. Neutrophil extracellular traps directly induce epithelial and endothelial cell death: A predominant role of histones. PLoS ONE 2012, 7, e32366. [Google Scholar] [CrossRef]
- Hirose, T.; Hamaguchi, S.; Matsumoto, N.; Irisawa, T.; Seki, M.; Tasaki, O.; Hosotsubo, H.; Yamamoto, N.; Yamamoto, K.; Akeda, Y.; et al. Presence of neutrophil extracellular traps and citrullinated histone H3 in the bloodstream of critically ill patients. PLoS ONE 2014, 9, e111755. [Google Scholar] [CrossRef]
- Kaplan, M.J.; Radic, M. Neutrophil extracellular traps: Double-edged swords of innate immunity. J. Immunol. 2012, 189, 2689–2695. [Google Scholar] [CrossRef]
- Sørensen, O.E.; Borregaard, N. Neutrophil extracellular traps - the dark side of neutrophils. J. Clin. Investig. 2016, 126, 1612–1620. [Google Scholar] [CrossRef]
- Maruchi, Y.; Tsuda, M.; Mori, H.; Takenaka, N.; Gocho, T.; Huq, M.A.; Takeyama, N. Plasma myeloperoxidase-conjugated DNA level predicts outcomes and organ dysfunction in patients with septic shock. Crit. Care 2018, 22, 176. [Google Scholar] [CrossRef]
- Manda, A.; Pruchniak, M.P.; Araźna, M.; Demkow, U.A. Neutrophil extracellular traps in physiology and pathology. Cent. Eur. J. Immunol. 2014, 39, 116–121. [Google Scholar] [CrossRef]
- O’Brien, X.M.; Biron, B.M.; Reichner, J.S. Consequences of extracellular trap formation in sepsis. Curr. Opin. Hematol. 2017, 24, 66–71. [Google Scholar] [CrossRef]
- Mohammed, B.M.; Fisher, B.J.; Kraskauskas, D.; Farkas, D.; Brophy, D.F.; Fowler, A.A.; Natarajan, R. Vitamin C: A novel regulator of neutrophil extracellular trap formation. Nutrients 2013, 5, 3131–3151. [Google Scholar] [CrossRef] [PubMed]
- Hamaguchi, S.; Akeda, Y.; Yamamoto, N.; Seki, M.; Yamamoto, K.; Oishi, K.; Tomono, K. Origin of circulating free DNA in sepsis: Analysis of the CLP mouse model. Mediat. Inflamm. 2015, 2015, 614518. [Google Scholar] [CrossRef] [PubMed]
- Nakahira, K.; Kyung, S.-Y.; Rogers, A.J.; Gazourian, L.; Youn, S.; Massaro, A.F.; Quintana, C.; Osorio, J.C.; Wang, Z.; Zhao, Y.; et al. Circulating mitochondrial DNA in patients in the ICU as a marker of mortality: Derivation and validation. PLoS Med. 2013, 10, e1001577. [Google Scholar] [CrossRef] [PubMed]
- Manning, J.; Mitchell, B.; Appadurai, D.A.; Shakya, A.; Pierce, L.J.; Wang, H.; Nganga, V.; Swanson, P.C.; May, J.M.; Tantin, D.; et al. Vitamin C promotes maturation of T-cells. Antioxid. Redox Signal. 2013, 19, 2054–2067. [Google Scholar] [CrossRef]
- Huijskens, M.J.A.J.; Walczak, M.; Koller, N.; Briedé, J.J.; Senden-Gijsbers, B.L.M.G.; Schnijderberg, M.C.; Bos, G.M.J.; Germeraad, W.T.V. Technical advance: Ascorbic acid induces development of double-positive T cells from human hematopoietic stem cells in the absence of stromal cells. J. Leukoc. Biol. 2014, 96, 1165–1175. [Google Scholar] [CrossRef]
- Agathocleous, M.; Meacham, C.E.; Burgess, R.J.; Piskounova, E.; Zhao, Z.; Crane, G.M.; Cowin, B.L.; Bruner, E.; Murphy, M.M.; Chen, W.; et al. Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. Nature 2017, 549, 476–481. [Google Scholar] [CrossRef]
- Lee Chong, T.; Ahearn, E.L.; Cimmino, L. Reprogramming the epigenome with vitamin C. Front. Cell Dev. Biol. 2019, 7, 128. [Google Scholar] [CrossRef]
- Young, J.I.; Züchner, S.; Wang, G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef]
- Kallio, J.; Jaakkola, M.; Mäki, M.; Kilpeläinen, P.; Virtanen, V. Vitamin C inhibits staphylococcus aureus growth and enhances the inhibitory effect of quercetin on growth of Escherichia coli in vitro. Planta Med. 2012, 78, 1824–1830. [Google Scholar] [CrossRef]
- Rawal, B.D. Bactericidal action of ascorbic acid on Psuedomonas aeruginosa: Alteration of cell surface as a possible mechanism. Chemotherapy 1978, 24, 166–171. [Google Scholar] [CrossRef]
- Mikirova, N.; Riordan, N.; Casciari, J. Modulation of cytokines in cancer patients by intravenous ascorbate therapy. Med. Sci. Monit. 2016, 22, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, E.; Roux-Lombard, P.; Grau, G.E.; Girardin, E.; Ricou, B.; Dayer, J.; Suter, P.M. Plasma concentrations of cytokines, their soluble receptors, and antioxidant vitamins can predict the development of multiple organ failure in patients at risk. Crit. Care Med. 1996, 24, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Hamacher, J.; Hadizamani, Y.; Borgmann, M.; Mohaupt, M.; Männel, D.N.; Moehrlen, U.; Lucas, R.; Stammberger, U. Cytokine-Ion Channel Interactions in Pulmonary Inflammation. Front. Immunol. 2017, 8, 1644. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Fu, G.; Yao, Q.; Cheng, G. Relation of thrombomodulin, TFPI and plasma antioxidants in healthy individuals and patients with coronary heart disease. Acta Cardiol. 2008, 63, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Puskarich, M.A.; Cornelius, D.C.; Tharp, J.; Nandi, U.; Jones, A.E. Plasma syndecan-1 levels identify a cohort of patients with severe sepsis at high risk for intubation after large-volume intravenous fluid resuscitation. J. Crit. Care 2016, 36, 125–129. [Google Scholar] [CrossRef]
- Hayashida, K.; Parks, W.C.; Park, P.W. Syndecan-1 shedding facilitates the resolution of neutrophilic inflammation by removing sequestered CXC chemokines. Blood 2009, 114, 3033–3043. [Google Scholar] [CrossRef]
- Smart, L.; Bosio, E.; Macdonald, S.P.J.; Dull, R.; Fatovich, D.M.; Neil, C.; Arendts, G. Glycocalyx biomarker syndecan-1 is a stronger predictor of respiratory failure in patients with sepsis due to pneumonia, compared to endocan. J. Crit. Care 2018, 47, 93–98. [Google Scholar] [CrossRef]
- Wu, F.; Peng, Z.; Park, P.W.; Kozar, R.A. Loss of Syndecan-1 Abrogates the Pulmonary Protective Phenotype Induced by Plasma After Hemorrhagic Shock. Shock 2017, 48, 340–345. [Google Scholar] [CrossRef]
- Palaiologou, M.; Delladetsima, I.; Tiniakos, D. CD138 (syndecan-1) expression in health and disease. Histol. Histopathol. 2014, 29, 177–189. [Google Scholar]
- Sallisalmi, M.; Tenhunen, J.; Yang, R.; Oksala, N.; Pettilä, V. Vascular adhesion protein-1 and syndecan-1 in septic shock. Acta Anaesthesiol. Scand. 2012, 56, 316–322. [Google Scholar] [CrossRef]
- Wei, S.; Gonzalez Rodriguez, E.; Chang, R.; Holcomb, J.B.; Kao, L.S.; Wade, C.E. PROPPR Study Group Elevated Syndecan-1 after Trauma and Risk of Sepsis: A Secondary Analysis of Patients from the Pragmatic, Randomized Optimal Platelet and Plasma Ratios (PROPPR) Trial. J. Am. Coll. Surg. 2018, 227, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.; Johansson, J.; Tydén, J.; Bodelsson, M. Circulating syndecans during critical illness. APMIS 2017, 125, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, J.; Bollmann, M. Soluble syndecans: Biomarkers for diseases and therapeutic options. Br. J. Pharmacol. 2019, 176, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Murphy, L.S.; Wickersham, N.; McNeil, J.B.; Shaver, C.M.; May, A.K.; Bastarache, J.A.; Ware, L.B. Endothelial glycocalyx degradation is more severe in patients with non-pulmonary sepsis compared to pulmonary sepsis and associates with risk of ARDS and other organ dysfunction. Ann. Intensive Care 2017, 7, 102. [Google Scholar] [CrossRef]
- Nelson, A.; Berkestedt, I.; Schmidtchen, A.; Ljunggren, L.; Bodelsson, M. Increased levels of glycosaminoglycans during septic shock: Relation to mortality and the antibacterial actions of plasma. Shock 2008, 30, 623–627. [Google Scholar] [CrossRef]
- Kashiouris, M.G.; Qiao, X.; Fisher, B.J.; Truwit, J.D.; Hite, R.D.; Morris, P.E.; Martin, G.S.; Fowler, A.A. A Multicenter Parsimonious Biomarker Mortality Prediction Model for Sepsis-Induced ARDS. In A95. Critical Care: Alice in Wonderland—Biomarkers in Critical Illness: Clues to be Unravelled; American Thoracic Society: New York, NY, USA, 2019; p. A2381. [Google Scholar]
- Qiao, X.; Fisher, B.; Kashiouris, M.G.; Truwit, J.D.; Hite, R.D.; Morris, P.E.; Martin, G.S.; Fowler, A.A. Effects of High Dose Intravenous Vitamin C (IVC) on Plasma Cell-Free DNA Levels in Patients with Sepsis-Associated ARDS. In A60. Lung Injury, Sepsis, and Ards; American Thoracic Society: New York, NY, USA, 2019; p. A2100. [Google Scholar]
- Pignatelli, P.; Sanguigni, V.; Paola, S.G.; Lo Coco, E.; Lenti, L.; Violi, F. Vitamin C inhibits platelet expression of CD40 ligand. Free Radic. Biol. Med. 2005, 38, 1662–1666. [Google Scholar] [CrossRef]
- Horrobin, D.F. Ascorbic acid and prostaglandin synthesis. Subcell. Biochem. 1996, 25, 109–115. [Google Scholar]
- Mohammed, B.M.; Sanford, K.W.; Fisher, B.J.; Martin, E.J.; Contaifer, D.; Warncke, U.O.; Wijesinghe, D.S.; Chalfant, C.E.; Brophy, D.F.; Fowler Iii, A.A.; et al. Impact of high dose vitamin C on platelet function. World J. Crit. Care Med. 2017, 6, 37–47. [Google Scholar] [CrossRef]
- Cudjoe, E.K.; Hassan, Z.H.; Kang, L.; Reynolds, P.S.; Fisher, B.J.; McCarter, J.; Sweeney, C.; Martin, E.J.; Middleton, P.; Ellenberg, M.; et al. Temporal map of the pig polytrauma plasma proteome with fluid resuscitation and intravenous vitamin C treatment. J. Thromb. Haemost. 2019, 17, 1827–1837. [Google Scholar] [CrossRef]
- Galley, H.F.; Davies, M.J.; Webster, N.R. Ascorbyl radical formation in patients with sepsis: Effect of ascorbate loading. Free Radic. Biol. Med. 1996, 20, 139–143. [Google Scholar] [CrossRef]
- Fowler, A.A.; Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; De Wilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; et al. Medical Respiratory Intensive Care Unit Nursing, Fisher BJ, Natarajan R. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar] [CrossRef]
- Seno, T.; Inoue, N.; Matsui, K.; Ejiri, J.; Hirata, K.-I.; Kawashima, S.; Yokoyama, M. Functional expression of sodium-dependent vitamin C transporter 2 in human endothelial cells. J. Vasc. Res. 2004, 41, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Oudemans-van Straaten, H.M.; Elbers, P.W.G.; Spoelstra-de Man, A.M.E. How to give vitamin C a cautious but fair chance in severe sepsis. Chest 2017, 151, 1199–1200. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zabet, M.H.; Mohammadi, M.; Ramezani, M.; Khalili, H. Effect of high-dose Ascorbic acid on vasopressor’s requirement in septic shock. J. Res. Pharm. Pract. 2016, 5, 94–100. [Google Scholar] [PubMed]
- Bürzle, M.; Hediger, M.A. Functional and physiological role of vitamin C transporters. Curr. Top. Membr. 2012, 70, 357–375. [Google Scholar]
- Abrams, S.T.; Morton, B.; Alhamdi, Y.; Alsabani, M.; Lane, S.; Welters, I.D.; Wang, G.; Toh, C.-H. A novel assay for neutrophil extracellular trap formation independently predicts disseminated intravascular coagulation and mortality in critically ill patients. Am. J. Respir. Crit. Care Med. 2019, 200, 869–880. [Google Scholar] [CrossRef]
- Gupta, A.K.; Joshi, M.B.; Philippova, M.; Erne, P.; Hasler, P.; Hahn, S.; Resink, T.J. Activated endothelial cells induce neutrophil extracellular traps and are susceptible to NETosis-mediated cell death. FEBS Lett. 2010, 584, 3193–3197. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Bozonet, S.M.; Carr, A.C. The Role of Physiological Vitamin C Concentrations on Key Functions of Neutrophils Isolated from Healthy Individuals. Nutrients 2019, 11, 1363. [Google Scholar] [CrossRef]
- May, J.M.; Harrison, F.E. Role of vitamin C in the function of the vascular endothelium. Antioxid. Redox Signal. 2013, 19, 2068–2083. [Google Scholar] [CrossRef]
- Fisher, B.J.; Seropian, I.M.; Kraskauskas, D.; Thakkar, J.N.; Voelkel, N.F.; Fowler, A.A.; Natarajan, R. Ascorbic acid attenuates lipopolysaccharide-induced acute lung injury. Crit. Care Med. 2011, 39, 1454–1460. [Google Scholar] [CrossRef] [PubMed]
- Burchfield, J.S.; Li, Q.; Wang, H.Y.; Wang, R.-F. JMJD3 as an epigenetic regulator in development and disease. Int. J. Biochem. Cell Biol. 2015, 67, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Brogdon, J.L.; Xu, Y.; Szabo, S.J.; An, S.; Buxton, F.; Cohen, D.; Huang, Q. Histone deacetylase activities are required for innate immune cell control of Th1 but not Th2 effector cell function. Blood 2007, 109, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.D.; Le, T.; Fan, G. DNA Methylation and Its Basic Function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef]
- Intravenous Vitamin C Employed as Adjunctive Therapy in Recurrent Acute Respiratory Distress Syndrome (ARDS): A Case Report A54. Critical Care Case Reports: Acute Hypoxemic Respiratory Failure/Ards. Available online: https://www.atsjournals.org/doi/abs/10.1164/ajrccm-conference.2016.193.1_MeetingAbstracts.A1892 (accessed on 24 September 2019).
- Fisher, B.J.; Kraskauskas, D.; Martin, E.J.; Farkas, D.; Wegelin, J.A.; Brophy, D.; Ward, K.R.; Voelkel, N.F.; Fowler, A.A.; Natarajan, R. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L20–L32. [Google Scholar] [CrossRef]
- Sawyer, M.A.J.; Mike, J.J.; Chavin, K.; Marino, P.L. Antioxidant therapy and survival in ARDS. Crit. Care Med. 1989, 17, S153. [Google Scholar]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; De Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef]
- Marik, P.E.; Khangoora, V.; Rivera, R.; Hooper, M.H.; Catravas, J. Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest 2017, 151, 1229–1238. [Google Scholar] [CrossRef]
- Li, J. Evidence is stronger than you think: A meta-analysis of vitamin C use in patients with sepsis. Crit. Care 2018, 22, 258. [Google Scholar] [CrossRef]
- Fowler, A.A.; Truwit, J.D.; Hite, R.D.; Morris, P.E.; DeWilde, C.; Priday, A.; Fisher, B.; Thacker, L.R.; Natarajan, R.; Brophy, D.F.; et al. Effect of Vitamin C Infusion on Organ Failure and Biomarkers of Inflammation and Vascular Injury in Patients With Sepsis and Severe Acute Respiratory Failure: The CITRIS-ALI Randomized Clinical Trial. JAMA 2019, 322, 1261–1270. [Google Scholar] [CrossRef]
- Tanaka, H.; Matsuda, T.; Miyagantani, Y.; Yukioka, T.; Matsuda, H.; Shimazaki, S. Reduction of resuscitation fluid volumes in severely burned patients using ascorbic acid administration: A randomized, prospective study. Arch. Surg. 2000, 135, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Hager, D.N.; Martin, G.S.; Sevransky, J.E.; Hooper, M.H. Glucometry when using vitamin C in sepsis: A note of caution. Chest 2018, 154, 228–229. [Google Scholar] [CrossRef] [PubMed]
- Sartor, Z.; Kesey, J.; Dissanaike, S. The effects of intravenous vitamin C on point-of-care glucose monitoring. J. Burn Care Res. 2015, 36, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.E.; Brown, C.S.; Manning, B.M.; May, T.; Riker, R.R.; Lerwick, P.A.; Hayes, T.L.; Fraser, G.L. Accuracy of Point-of-Care Blood Glucose Level Measurements in Critically Ill Patients with Sepsis Receiving High-Dose Intravenous Vitamin, C. Pharmacotherapy 2018, 38, 1155–1161. [Google Scholar] [CrossRef]
| The key antioxidant of the body | Reverses the oxidation of lipids by the neutrophil reactive oxygen species (ROS) [32,33]. Reduces depletion of other antioxidants (e.g., vitamin E and glutathione) to prevent oxidation of lipids, proteins and DNA [34,35,36,37,38,39,40]. |
| Norepinephrine biosynthesis | Cofactor for Dopamine ß-Hydroxylase, catalyzing the formation of norepinephrine from dopamine. Enhances adrenergic receptor activity [41]. |
| Dopamine biosynthesis | Facilitates recycling of the enzyme cofactor tetrahydrobiopterin (BH4); a required and rate-limiting step in the hydroxylation of l-tyrosine to form l-DOPA [42,43]. |
| Vasopressin biosynthesis | Cofactor for peptidylglycine α-amidating monooxygenase (PAM), involved in vasopressin biosynthesis [44]. |
| Connective tissue maintenance | Vital in wound healing; cofactor for Propyl 3-hydroxylase, prolyl 4-hydroxylase, and lysyl hydroxylase which catalyze the formation of procollagen and elastin biosynthesis [45,46]. Catalyzes the hydroxylation of procollagen to form the collagen triple-helix [47]. Induces fibroblast collagen gene expression, stimulating the production of new collagen [48]. |
| Regulation of cellular gene expression in response to hypoxia and stress | Needed for the hydroxylation (thus downregulation) of Hypoxia Induced Factor 1α (HIF-1α) by propyl and lysyl hydroxylases and FIF-1 (asparaginyl hydroxylase or factor inhibiting HIF-1) [49,50,51]. HIF-1α is a protein-transcription factor that regulates hundreds of genes in response to hypoxia and cellular stress, and is a marker of cellular hypoxia with increased expression in states of shock [52]. |
| Carnitine biosynthesis | Cofactor for γ-butyrobetaine hydroxylase, a dioxygenase involved in carnitine synthesis, which transports fatty acids into the mitochondria [53,54]. L-Carnitine can down-modulate tumor necrosis factor (TNF-α) by endotoxins, affect lipid metabolism, and reduce septic shock severity [55]. |
| Phagocytic cell function | Severe vitamin C deficiency (scorbutic) results in impaired neutrophilic phagocytosis and ROS generation [56,57,58,59,60]. In situations of impaired neutrophilic ROS production, vitamin C enhances the hexose monophosphate shunt (HMPS) and antibody dependent cell cytotoxicity (ADCC) resulting in increased bacterial killing [60]. Improves chemotaxis [61]. Accumulation in neutrophils may protect them from neutrophil dependent oxidative bursts [62,63]. Reduces inflammation and ROS via attenuation of NF-κB activation [64,65,66]. |
| Inflammation: Immune cell clearance | Promotes neutrophil apoptosis, instead of necrosis via activation of caspase-3 proteins [67,68]. High-dose intravenous vitamin C (HDIVC) treatment has been shown to decrease circulating plasma cell-free DNA (resulting from neutrophil extracellular trap (NET) formations, or NETosis), and have been implicated in sepsis-induced end-organ failure [69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84]. |
| Lymphocytic function | May promote lymphocytic proliferation, differentiation, and maturation [85,86]. |
| Epigenetic modulation | Cofactor for ten-eleven translocation (TET) enzymes and Jumonji-C domain-containing histone demethylases (JHDMs); vitamin C increasing enzymatic activity of both, resulting in increased DNA demethylation and histone demethylation, respectively, which controls gene transcription and gene activation or repression [87,88,89]. |
| Direct antimicrobial activity | High concentrations directly inhibit bacterial growth and exhibits bactericidal activity in vitro [90,91]. |
| Inflammatory mediators | Modulates cytokine production and can decrease circulating histamine levels [61,92,93,94]. |
| Endothelial function | HDIVC decreases circulating thrombomodulin, an endothelial membrane protein receptor for thrombin that converts thrombin to an anticoagulant capable of activating protein C [95]. Decreases plasma Syndecan-1 levels, a by-product of endothelial glycocalyx shedding [96,97,98,99,100,101,102,103,104,105,106,107,108]. |
| Platelet function and Thrombosis | Alters platelet oxidative states by inhibiting CD40 ligand expression on platelet surfaces [109]. Prolonged platelet exposure to HDIVC increases Thromboxane-B2 and Prostagladin-E2 levels [110,111]. HDIVC stabilizes ADAMTS13 levels and its von-Willebrand factor cleavage activity [112]. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kashiouris, M.G.; L’Heureux, M.; Cable, C.A.; Fisher, B.J.; Leichtle, S.W.; Fowler, A.A. The Emerging Role of Vitamin C as a Treatment for Sepsis. Nutrients 2020, 12, 292. https://doi.org/10.3390/nu12020292
Kashiouris MG, L’Heureux M, Cable CA, Fisher BJ, Leichtle SW, Fowler AA. The Emerging Role of Vitamin C as a Treatment for Sepsis. Nutrients. 2020; 12(2):292. https://doi.org/10.3390/nu12020292
Chicago/Turabian StyleKashiouris, Markos G., Michael L’Heureux, Casey A. Cable, Bernard J. Fisher, Stefan W. Leichtle, and Alpha A. Fowler. 2020. "The Emerging Role of Vitamin C as a Treatment for Sepsis" Nutrients 12, no. 2: 292. https://doi.org/10.3390/nu12020292
APA StyleKashiouris, M. G., L’Heureux, M., Cable, C. A., Fisher, B. J., Leichtle, S. W., & Fowler, A. A. (2020). The Emerging Role of Vitamin C as a Treatment for Sepsis. Nutrients, 12(2), 292. https://doi.org/10.3390/nu12020292





