Epigenetic Patterns in Five-Year-Old Children Exposed to a Low Glycemic Index Dietary Intervention during Pregnancy: Results from the ROLO Kids Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Dietary Intervention During Pregnancy
2.3. Anthropometry and Body Composition
2.4. DNA Extraction and Genome-Wide Methylation Detection
2.5. Statistical Analysis
2.6. Pathway Analysis
3. Results
3.1. Cohort Characteristics and the Dietary Intervention
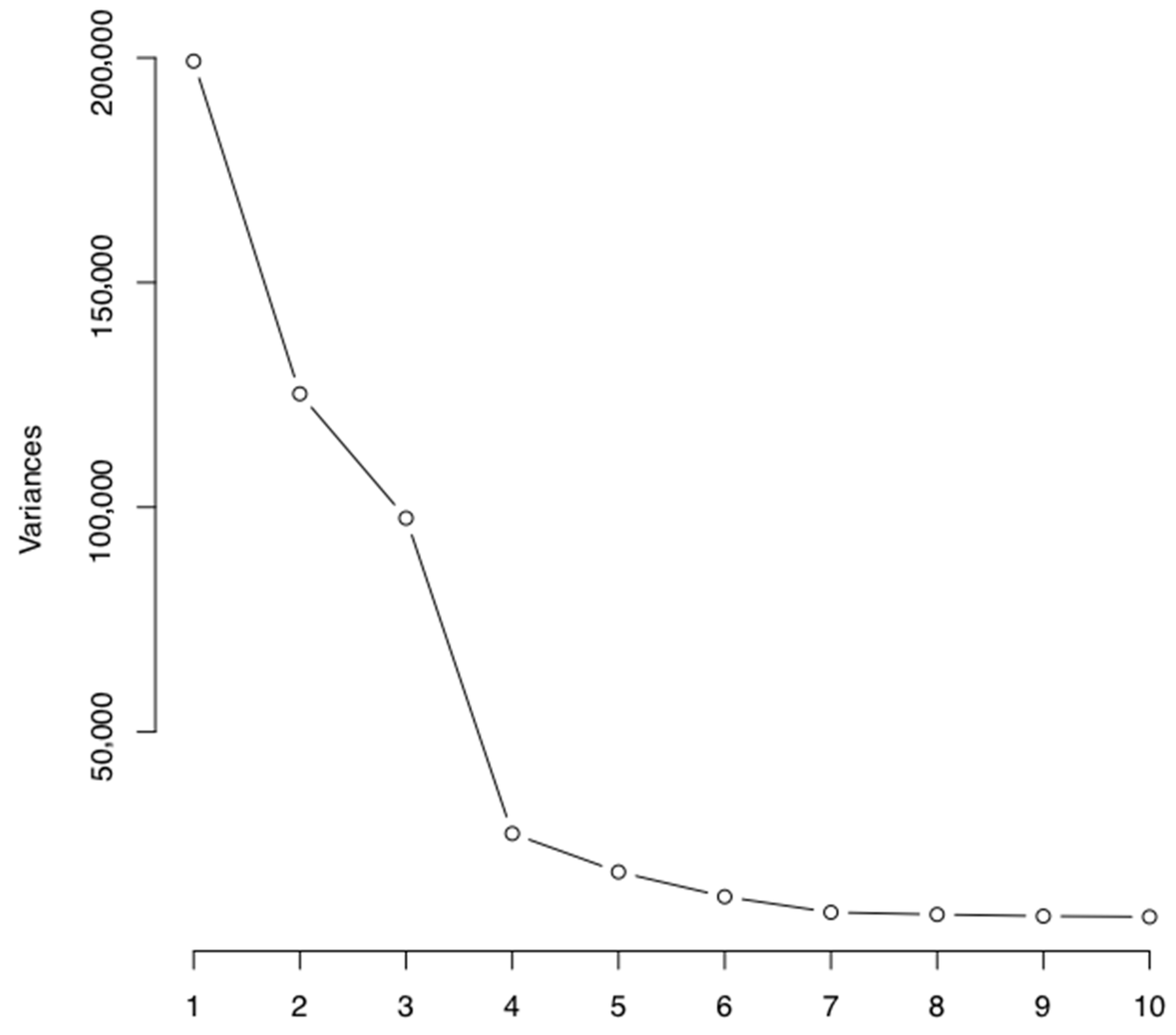
3.2. Principal Component Analysis
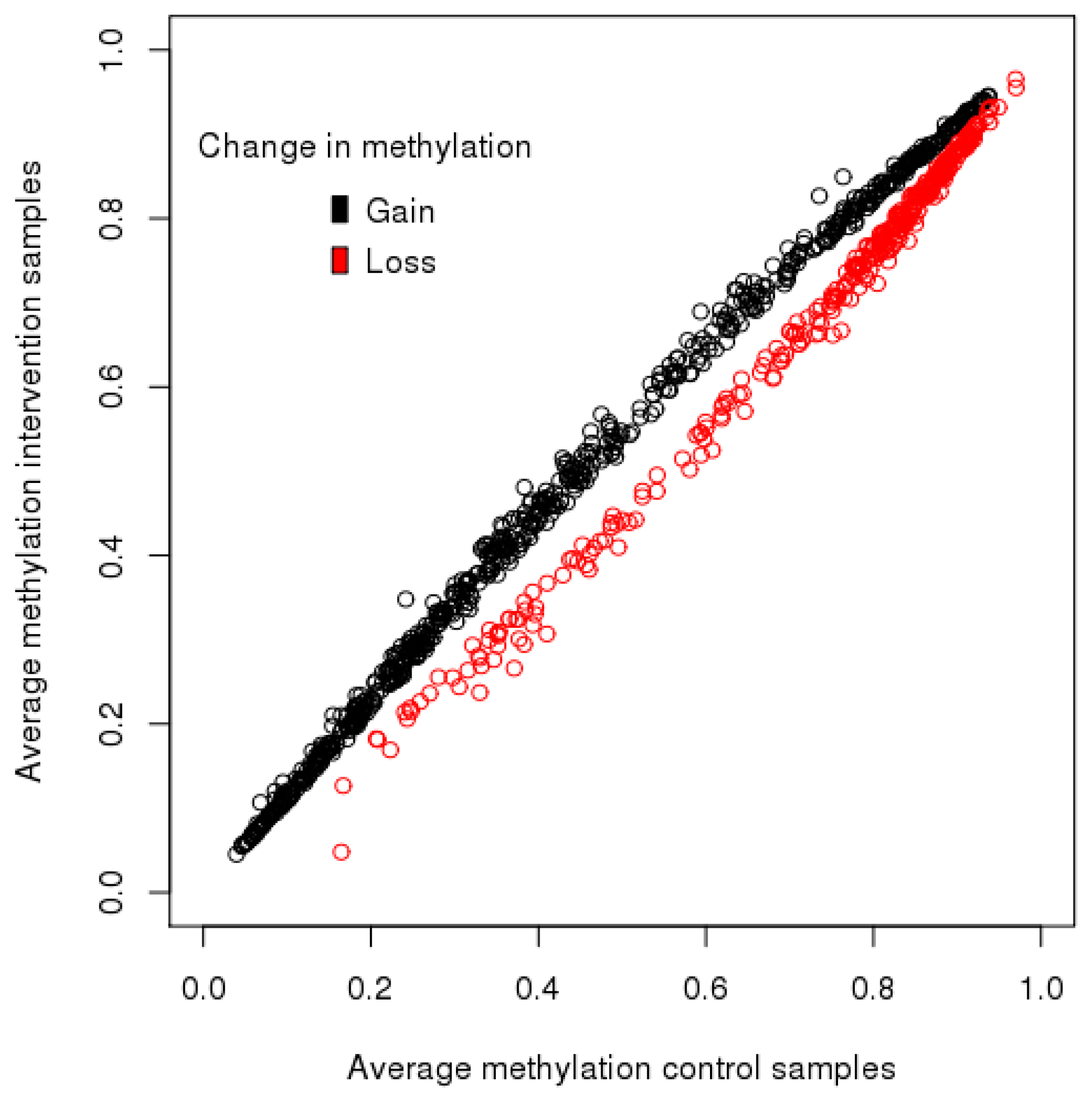
3.3. Low Glycemic Index Dietary Intervention
3.4. Pathway Analysis
3.5. Early Childhood Weight Status and Adiposity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mathers, J.C.; Strathdee, G.; Relton, C.L. Induction of Epigenetic Alterations by Dietary and Other Environmental Factors, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2010; Volume 71. [Google Scholar]
- McKay, J.; Mathers, J. Diet induced epigenetic changes and their implications for health. Acta Physiol. (Oxf.) 2011, 202, 103–118. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.A.; Gluckman, P.D. Developmental origins of health and disease: New insights. Basic Clin. Pharmacol. Toxicol. 2008, 102, 90–93. [Google Scholar] [CrossRef]
- Marsit, C.J. Influence of environmental exposure on human epigenetic regulation. J. Exp. Biol. 2015, 3, 71–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.S. Impact of maternal diet on the epigenome during in utero life and the developmental programming of diseases in childhood and adulthood. Nutrients 2015, 7, 9492–9507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, R.C.W.; Tutino, G.E.; Lillycrop, K.A.; Hanson, M.A.; Tam, W.H. Maternal diabetes, gestational diabetes and the role of epigenetics in their long term effects on offspring. Prog. Biophys. Mol. Biol. 2015, 118, 55–68. [Google Scholar] [CrossRef]
- Knopik, V.S.; Maccani, M.A.; Francazio, S.; McGeary, J.E. The Epigenetics of Maternal Cigarette Smoking During Pregnancy and Effects on Child Development. Dev. Psychopathol. 2012, 24, 1377–1390. [Google Scholar] [CrossRef] [Green Version]
- Rzehak, P.; Saffery, R.; Reischl, E.; Covic, M.; Wahl, S.; Grote, V.; Xhonneux, A.; Langhendries, J.P.; Ferre, N.; Closa-Monasterolo, R.; et al. Maternal smoking during pregnancy and DNA-methylation in children at age 5.5 years: Epigenome-wide-analysis in the European Childhood Obesity Project (CHOP)-study. PLoS ONE 2016, 11, e0155554. [Google Scholar] [CrossRef]
- Reichetzeder, C.; Dwi Putra, S.E.; Li, J.; Hocher, B. Developmental Origins of Disease—Crisis Precipitates Change. Cell. Physiol. Biochem. 2016, 39, 919–938. [Google Scholar] [CrossRef]
- Lumey, L.H.; Stein, A.D.; Kahn, H.S.; Van der Pal-de Bruin, K.M.; Blauw, G.J.; Zybert, P.A.; Susser, E.S. Cohort profile: The Dutch Hunger Winter families study. Int. J. Epidemiol. 2007, 36, 1196–1204. [Google Scholar] [CrossRef] [Green Version]
- Tobi, E.W.; Slagboom, P.E.; van Dongen, J.; Kremer, D.; Stein, A.D.; Putter, H.; Heijmans, B.T.; Lumey, L.H. Prenatal famine and genetic variation are independently and additively associated with DNA methylation at regulatory loci within IGF2/H19. PLoS ONE 2012, 7, e37933. [Google Scholar] [CrossRef] [Green Version]
- Tobi, E.W.; Slieker, R.C.; Stein, A.D.; Suchiman, H.E.D.; Slagboom, P.E.; Van Zwet, E.W.; Heijmans, B.T. Early Life Environment Early gestation as the critical time-window for changes in the prenatal environment to affect the adult human blood methylome. Int. J. Epidemiol. 2015, 1211–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Hajj, N.; Schneider, E.; Lehnen, H.; Haaf, T. Epigenetics and life-long consequences of an adverse nutritional and diabetic intrauterine environment. Reproduction 2014, 148, R111–R120. [Google Scholar] [CrossRef] [PubMed]
- Hjort, L.; Martino, D.; Grunnet, L.G.; Naeem, H.; Maksimovic, J.; Olsson, A.H.; Zhang, C.; Ling, C.; Olsen, S.F.; Saffery, R.; et al. Gestational diabetes and maternal obesity are associated with epigenome-wide methylation changes in children. JCI Insight 2018, 3, e122572. [Google Scholar] [CrossRef] [PubMed]
- Geraghty, A.A.; Sexton-Oates, A.; O’Brien, E.C.; Alberdi, G.; Fransquet, P.; Saffery, R.; McAuliffe, F.M. A low glycaemic index diet in pregnancy induces DNA methylation variation in blood of newborns: Results from the ROLO randomised controlled trial. Nutrients 2018, 10, 455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crume, T.L.; Ogden, L.; Daniels, S.; Hamman, R.F.; Norris, J.M.; Dabelea, D. The impact of in utero exposure to diabetes on childhood body mass index growth trajectories: The EPOCH study. J. Pediatr. 2011, 158, 941–946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef] [Green Version]
- Godfrey, K.M.; Sheppard, A.; Gluckman, P.D.; Lillycrop, K.A.; Burdge, G.C.; McLean, C.; Rodford, J.; Slater-Jefferies, J.L.; Garratt, E.; Crozier, S.R.; et al. Epigenetic gene promoter methylation at birth is associated with child’s later adiposity. Diabetes 2011, 60, 1528–1534. [Google Scholar] [CrossRef] [Green Version]
- Relton, C.L.; Groom, A.; St. Pourcain, B.; Sayers, A.E.; Swan, D.C.; Embleton, N.D.; Pearce, M.S.; Ring, S.M.; Northstone, K.; Tobias, J.H.; et al. DNA methylation patterns in cord blood DNA and body size in childhood. PLoS ONE 2012, 7, e31821. [Google Scholar] [CrossRef] [Green Version]
- Rounge, T.B.; Page, C.M.; Lepistö, M.; Ellonen, P.; Andreassen, B.K.; Weiderpass, E. Genome-wide DNA methylation in saliva and body size of adolescent girls. Epigenomics 2016, 8, 1495–1505. [Google Scholar] [CrossRef] [Green Version]
- Dick, K.J.; Nelson, C.P.; Tsaprouni, L.; Sandling, J.K.; Aïssi, D.; Wahl, S.; Meduri, E.; Morange, P.-E.; Gagnon, F.; Grallert, H.; et al. DNA methylation and body-mass index: A genome-wide analysis. Lancet 2014, 383, 1990–1998. [Google Scholar] [CrossRef] [Green Version]
- Walsh, J.; McGowan, C.; Mahony, R.; Foley, M.; McAuliffe, F. Low glycaemic index diet in pregnancy to prevent macrosomia (ROLO study): Randomised control trial. Br. Med. J. 2012, 345, e5605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donnelly, J.M.; Lindsay, K.L.; Walsh, J.M.; Horan, M.; Molloy, E.J.; McAuliffe, F.M. Fetal metabolic influences of neonatal anthropometry and adiposity. BMC Pediatr. 2015, 15, 175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Food Standards Agency. McCance and Widdowson’s The Composition of Foods, Sixth Summary Edition; Royal Society of Chemistry: Cambridge, UK, 2002.
- Levis, S.; McGowan, C.; McAuliffe, F. Methodology for adding and amending glycaemic index values to a nutrition analysis package. Br. J. Nutr. 2011, 105, 1117–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cole, T.; Freeman, J.; Preece, M. Body mass index reference curves for the UK, 1990. Arch. Dis. Child. 1995, 73, 25–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, H.; Cole, T. LMS Growth, a Microsoft Excel Add-In to Access Growth References Based on the LMS Method; Version 2.77; Medical Research Council the Institute of Child Health: London, UK, 2012. [Google Scholar]
- Maksimovic, J.; Gordon, L.; Oshlack, A. SWAN: Subset-quantile within array normalization for illumina infinium HumanMethylation450 BeadChips. Genome Biol. 2012, 13, R44. [Google Scholar] [CrossRef] [Green Version]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Benajmini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar]
- Huang, D.W.; Sherman, B.T.; Lempicki, R. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Sharp, G.C.; Lawlor, D.A.; Richmond, R.C.; Fraser, A.; Simpkin, A.; Suderman, M.; Shihab, H.A.; Lyttleton, O.; McArdle, W.; Ring, S.M.; et al. Maternal pre-pregnancy BMI and gestational weight gain, offspring DNA methylation and later offspring adiposity: Findings from the Avon Longitudinal Study of Parents and Children. Int. J. Epidemiol. 2015, 44, 1288–1304. [Google Scholar] [CrossRef] [Green Version]
- Richmond, R.C.; Timpson, N.J.; Felix, J.F.; Palmer, T.; Gaillard, R.; McMahon, G.; Davey Smith, G.; Jaddoe, V.W.; Lawlor, D.A. Using Genetic Variation to Explore the Causal Effect of Maternal Pregnancy Adiposity on Future Offspring Adiposity: A Mendelian Randomisation Study. PLoS Med. 2017, 14, e1002221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simpkin, A.J.; Suderman, M.; Gaunt, T.R.; Lyttleton, O.; McArdle, W.L.; Ring, S.M.; Tilling, K.; Davey Smith, G.; Relton, C.L. Longitudinal analysis of DNA methylation associated with birth weight and gestational age. Hum. Mol. Genet. 2015, 24, 3752–3763. [Google Scholar] [CrossRef] [Green Version]
- Danielsen, I.; Granström, C.; Haldorsson, T.; Rytter, D.; Hammer Bech, B.; Henriksen, T.B.; Vaag, A.A.; Olsen, S.F. Dietary Glycemic Index during Pregnancy Is Associated with Biomarkers of the Metabolic Syndrome in Offspring at Age 20 Years. PLoS ONE 2013, 8, e64887. [Google Scholar] [CrossRef]
- Mihaylova, M.; Shaw, R. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat. Cell Biol. 2011, 13, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Kizirian, N.V.; Kong, Y.; Muirhead, R.; Brodie, S.; Garnett, S.P.; Petocz, P.; Sim, K.A.; Celermajer, D.S.; Louie, J.C.Y.; Markovic, T.P.; et al. Effects of a low–glycemic index diet during pregnancy on offspring growth, body composition, and vascular health: A pilot randomized controlled trial 1. Am. J. Clin. Nutr. 2016, 103, 1073–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- High, F.A.; Epstein, J.A. The multifaceted role of Notch in cardiac development and disease. Nat. Rev. Genet. 2008, 9, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Azzi, S.; Sas, T.C.J.; Koudou, Y.; Le Bouc, Y.; Souberbielle, J.C.; Dargent-Molina, P.; Netchine, I.; Charles, M.A. Degree of methylation of ZAC1 (PLAGL1) is associated with prenatal and post-natal growth in healthy infants of the EDEN mother child cohort. Epigenetics 2014, 9, 338–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barker, D. Fetal origins of coronary heart disease. Br. Med. J. 1995, 311, 171–174. [Google Scholar] [CrossRef]
- Vickers, M.H. Early life nutrition, epigenetics and programming of later life disease. Nutrients 2014, 6, 2165–2178. [Google Scholar] [CrossRef]
- Lin, X.; Lim, I.Y.; Wu, Y.; Teh, A.L.; Chen, L.; Aris, I.M.; Soh, S.E.; Tint, M.T.; MacIsaac, J.L.; Morin, A.M.; et al. Developmental pathways to adiposity begin before birth and are influenced by genotype, prenatal environment and epigenome. BMC Med. 2017, 15, 50. [Google Scholar] [CrossRef] [Green Version]
| Total | Intervention | Control | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Maternal Characteristics | n | Mean/Median | SD/IQR | n | Mean/Median | SD/IQR | n | Mean/Median | SD/IQR | p |
| Maternal age at birth (mean, SD, years) | 63 | 33.07 | 3.66 | 31 | 32.88 | 3.99 | 32 | 31.79 | 7.91 | 0.697 |
| Gestational age at booking visit (mean, SD, weeks) | 63 | 13.00 | 2.34 | 31 | 13.23 | 2.45 | 32 | 12.78 | 2.24 | 0.454 |
| Maternal weight in early pregnancy (median, IQR, kg) | 63 | 68.5 | 8.8 | 31 | 69.6 | 10.7 | 32 | 66.2 | 9.38 | 0.375 |
| Maternal BMI in early pregnancy (mean, SD, cm) | 63 | 25.75 | 4.05 | 31 | 25.74 | 3.46 | 32 | 25.77 | 4.61 | 0.979 |
| Maternal smoking in pregnancy (n (%)) | 63 | 1 (1.6) | 31 | 1 (3.2) | 32 | 0 (0) | 0.987 | |||
| Maternal ethnicity: Caucasian (n (%)) | 63 | 61 (96.8) | 31 | 30 (96.8) | 32 | 31 (96.9) | 0.37 | |||
| Education higher level (n (%)) | 59 | 34 (57.6) | 29 | 18 (58.1) | 30 | 16 (53.3) | 0.678 | |||
| Trimester 1 GI (mean, SD) | 60 | 58.07 | 4.48 | 30 | 57.67 | 4.6 | 30 | 58.46 | 4.41 | 0.497 |
| Trimester 2 GI (mean, SD) | 60 | 56.57 | 3.38 | 30 | 55.58 | 3.05 | 30 | 57.56 | 3.46 | 0.022 * |
| Trimester 3 GI (mean, SD) | 60 | 56.92 | 4.27 | 30 | 55.57 | 3.88 | 30 | 58.26 | 4.27 | 0.013 * |
| Child Characteristics | Mean/Median | SD/IQR | Mean/Median | SD/IQR | Mean/Median | SD/IQR | p | |||
| Sex: male (n (%)) | 63 | 32 (50.8) | 31 | 16 (51.6) | 32 | 16 (50) | 1 | |||
| Birth weight (mean, SD, kg) | 63 | 4.06 | 0.47 | 31 | 4.14 | 0.52 | 32 | 3.98 | 0.41 | 0.173 |
| Birth weight centile (median, IQR) | 63 | 79.55 | 33.43 | 31 | 82.75 | 34.18 | 32 | 76.15 | 34.23 | 0.379 |
| Gestational age (median, IQR, weeks) | 63 | 40.57 | 1.57 | 31 | 40.43 | 1.57 | 32 | 40.714 | 2.18 | 0.94 |
| Breastfed (n (%)) | 39 | 27 (69.2) | 20 | 16 (80) | 19 | 11 (57.9) | 0.251 | |||
| Six Month Follow-Up | ||||||||||
| Weight (mean, SD, kg) | 50 | 8.57 | 1.27 | 23 | 9.03 | 1.49 | 27 | 8.19 | 0.91 | 0.024 * |
| Weight centile (median, IQR) | 50 | 72.5 | 39.25 | 23 | 84 | 46 | 27 | 67 | 54 | 0.078 |
| BMI centile (median, IQR) | 50 | 51 | 57.25 | 23 | 74 | 62 | 27 | 40 | 50 | 0.016 * |
| Two Year Follow-Up | ||||||||||
| Weight (mean, SD, kg) | 56 | 13.15 | 1.37 | 29 | 13.4 | 1.48 | 27 | 12.92 | 1.22 | 0.216 |
| Weight centile (median, IQR) | 56 | 67 | 44 | 29 | 77 | 48.5 | 27 | 63 | 41 | 0.298 |
| BMI centile (median, IQR) | 56 | 41.5 | 48.5 | 29 | 56 | 66 | 27 | 38 | 32 | 0.275 |
| Total | Intervention | Control | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Child Characteristics at the Five Year Follow-Up | n | Mean/Median | SD/IQR | n | Mean/Median | SD/IQR | n | Mean/Median | SD/IQR | p |
| Age (mean, SD, years) | 63 | 5.10 | 0.11 | 31 | 5.08 | 0.12 | 32 | 5.12 | 0.1 | 0.134 |
| Weight (mean, SD, kg) | 63 | 20.28 | 2.33 | 31 | 20.43 | 2.49 | 32 | 20.13 | 2.21 | 0.624 |
| Weight centile (median, IQR) | 63 | 71 | 45 | 31 | 70 | 48 | 32 | 74.5 | 20.5 | 0.625 |
| Change in weight centiles from birth to five years (mean, SD) | 63 | 8.9 | 30.95 | 31 | 9.94 | 30.53 | 32 | 7.9 | 31.79 | 0.799 |
| Height (mean, SD, cm) | 63 | 111.26 | 4.13 | 31 | 110.98 | 4.12 | 32 | 111.53 | 4.19 | 0.599 |
| Height centile (median, IQR) | 63 | 63 | 47 | 31 | 63 | 48 | 32 | 65 | 47.75 | 0.67 |
| BMI (mean, SD, kg/m2) | 63 | 16.35 | 1.32 | 31 | 16.55 | 1.46 | 32 | 16.16 | 1.15 | 0.237 |
| BMI centile (median, IQR) | 63 | 69 | 39 | 31 | 71 | 35 | 32 | 68.5 | 40.75 | 0.437 |
| Chest circumference (mean, SD, cm) | 63 | 56.44 | 2.62 | 31 | 57.05 | 2.61 | 32 | 55.84 | 2.53 | 0.066 |
| Abdomen circumference (mean, SD, cm) | 63 | 54.41 | 5.61 | 31 | 55.76 | 4.15 | 32 | 53.11 | 6.53 | 0.059 |
| Waist to height ratio (mean, SD) | 63 | 0.49 | 0.05 | 31 | 0.5 | 0.03 | 32 | 0.48 | 0.07 | 0.068 |
| Sum of skinfolds (median, IQR, mm) | 55 | 34.8 | 13.6 | 28 | 35.25 | 14.48 | 27 | 33.5 | 10 | 0.853 |
| Subscapular and Triceps skinfold (median, IQR, mm) | 55 | 15.28 | 3.89 | 27 | 15.89 | 4.6 | 28 | 14.69 | 3.15 | 0.256 |
| Subscular:triceps skinfold ratio (mean, SD) | 55 | 0.62 | 0.16 | 27 | 0.64 | 0.21 | 28 | 0.61 | 0.13 | 0.16 |
| Individual | Chip | Array Position | RCT Group | Maternal Age (years) | Maternal Weight (kg) | Maternal BMI | Smoking in Pregnancy | Maternal Ethnicity | Child Sex | Gestational Age (weeks) | Birth Weight (kg) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PC1 correlation | −0.056 | 0.131 | 0.392 * | 0.163 | −0.094 | 0.064 | 0.016 | −0.175 | 0.087 | −0.076 | −0.056 | −0.084 |
| PC1 p-value | 0.662 | 0.306 | 0.002 * | 0.202 | 0.465 | 0.620 | 0.904 | 0.171 | 0.497 | 0.556 | 0.665 | 0.512 |
| PC2 correlation | −0.047 | −0.002 | 0.232 | −0.194 | −0.069 | −0.103 | −0.084 | 0.015 | −0.011 | 0.040 | 0.241 | −0.116 |
| PC2 p-value | 0.713 | 0.985 | 0.067 | 0.128 | 0.590 | 0.421 | 0.515 | 0.905 | 0.929 | 0.757 | 0.058 | 0.364 |
| PC3 correlation | −0.110 | −0.137 | 0.685 * | −0.046 | −0.035 | 0.088 | 0.078 | 0.055 | 0.073 | 0.073 | −0.167 | −0.018 |
| PC3 p-value | 0.392 | 0.283 | 0.000 * | 0.720 | 0.783 | 0.495 | 0.543 | 0.671 | 0.571 | 0.568 | 0.190 | 0.887 |
| PC4 correlation | −0.101 | 0.234 | 0.083 | −0.154 | 0.003 | 0.064 | 0.111 | −0.053 | 0.069 | −0.002 | 0.041 | 0.152 |
| PC4 p-value | 0.430 | 0.065 | 0.519 | 0.228 | 0.982 | 0.620 | 0.385 | 0.681 | 0.589 | 0.989 | 0.750 | 0.234 |
| PC5 correlation | −0.296 * | 0.316 * | 0.151 | 0.012 | −0.016 | 0.018 | 0.007 | −0.099 | 0.010 | −0.042 | −0.148 | −0.035 |
| PC5 p-value | 0.019 * | 0.012 * | 0.237 | 0.923 | 0.903 | 0.890 | 0.957 | 0.438 | 0.935 | 0.746 | 0.247 | 0.786 |
| PC6 correlation | −0.132 | 0.109 | −0.147 | −0.192 | −0.191 | −0.044 | −0.011 | −0.013 | −0.068 | −0.303 * | −0.018 | 0.155 |
| PC6 p-value | 0.304 | 0.394 | 0.251 | 0.132 | 0.133 | 0.730 | 0.929 | 0.921 | 0.594 | 0.016 * | 0.886 | 0.225 |
| PC7 correlation | 0.048 | 0.179 | 0.051 | −0.078 | −0.039 | −0.125 | −0.152 | 0.011 | −0.029 | −0.362 * | −0.030 | 0.127 |
| PC7 p-value | 0.709 | 0.160 | 0.689 | 0.545 | 0.762 | 0.328 | 0.235 | 0.929 | 0.822 | 0.004 * | 0.816 | 0.323 |
| PC8 correlation | −0.117 | −0.210 | 0.065 | 0.009 | 0.039 | 0.018 | 0.047 | −0.013 | 0.070 | 0.198 | 0.246 | −0.088 |
| PC8 p-value | 0.360 | 0.099 | 0.612 | 0.944 | 0.761 | 0.890 | 0.713 | 0.918 | 0.583 | 0.120 | 0.052 | 0.494 |
| PC9 correlation | 0.035 | 0.155 | 0.000 | −0.057 | 0.146 | −0.176 | −0.167 | −0.011 | −0.009 | 0.009 | −0.058 | −0.064 |
| PC9 p-value | 0.787 | 0.225 | 1.000 | 0.658 | 0.255 | 0.169 | 0.192 | 0.931 | 0.944 | 0.944 | 0.652 | 0.621 |
| PC10 correlation | 0.055 | 0.142 | 0.067 | 0.236 | 0.013 | −0.168 | −0.155 | −0.012 | 0.023 | −0.223 | 0.035 | 0.139 |
| PC10 p-value | 0.668 | 0.268 | 0.602 | 0.063 | 0.921 | 0.188 | 0.225 | 0.924 | 0.856 | 0.080 | 0.787 | 0.278 |
| Individual | T1 GI | T2 GI | T3 GI | |
|---|---|---|---|---|
| PC1 correlation | −0.074 | −0.008 | −0.013 | 0.024 |
| PC1 p-value | 0.574 | 0.951 | 0.923 | 0.857 |
| PC2 correlation | 0.057 | 0.034 | 0.167 | 0.178 |
| PC2 p-value | 0.667 | 0.799 | 0.203 | 0.173 |
| PC3 correlation | 0.139 | −0.033 | −0.033 | 0.185 |
| PC3 p-value | 0.288 | 0.802 | 0.802 | 0.157 |
| PC4 correlation | −0.077 | 0.042 | −0.050 | −0.072 |
| PC4 p-value | 0.559 | 0.749 | 0.702 | 0.584 |
| PC5 correlation | −0.336 * | 0.158 | 0.208 | −0.016 |
| PC5 p-value | 0.009 * | 0.229 | 0.112 | 0.906 |
| PC6 correlation | −0.108 | 0.191 | −0.037 | 0.034 |
| PC6 p-value | 0.411 | 0.144 | 0.781 | 0.796 |
| PC7 correlation | −0.021 | 0.135 | 0.114 | 0.211 |
| PC7 p-value | 0.872 | 0.303 | 0.384 | 0.105 |
| PC8 correlation | 0.135 | −0.091 | −0.010 | −0.156 |
| PC8 p-value | 0.302 | 0.488 | 0.940 | 0.235 |
| PC9 correlation | −0.033 | −0.264 * | −0.105 | 0.030 |
| PC9 p-value | 0.803 | 0.041 * | 0.424 | 0.820 |
| PC10 correlation | −0.051 | −0.159 | −0.081 | −0.218 |
| PC10 p-value | 0.700 | 0.225 | 0.538 | 0.094 |
| Pathway Type | Function | Count | p | Benjamini | |
|---|---|---|---|---|---|
| Cluster 1: Insulin Functioning | |||||
| ES: 1.91 * | KEGG | Insulin resistance | 11 | 0.0023 | 0.17 |
| KEGG | AMPK signaling pathway | 11 | 0.0055 | 0.21 | |
| KEGG | Insulin signaling pathway | 11 | 0.013 | 0.25 | |
| KEGG | FoxO signaling pathway | 8 | 0.14 | 0.56 | |
| Cluster 2: NOTCH Functioning | |||||
| ES: 1.83 * | REACTOME | Constitutive signaling by NOTCH1 HD+PEST domain mutants | 8 | 0.0017 | 0.25 |
| REACTOME | Constitutive signaling by NOTCH1 PEST domain mutants | 8 | 0.0017 | 0.25 | |
| REACTOME | NOTCH1 intracellular domain regulates transcription | 6 | 0.014 | 0.65 | |
| KEGG | Notch signaling pathway | 6 | 0.018 | 0.29 | |
| REACTOME | Notch-HLH transcription pathway | 3 | 0.059 | 0.85 | |
| REACTOME | Pre-NOTCH transcription and translation | 3 | 0.22 | 0.98 | |
| Cluster 3: Cell Signal Functioning | |||||
| ES: 1.31 * | REACTOME | PIP3 activates AKT signaling | 8 | 0.012 | 0.71 |
| REACTOME | CD28 dependent PI3K/Akt signaling | 4 | 0.029 | 0.77 | |
| REACTOME | Constitutive signaling by AKT1 E17K in cancer | 4 | 0.04 | 0.8 | |
| REACTOME | VEGFR2 mediated vascular permeability | 3 | 0.22 | 0.98 | |
| Change in Weight Centiles | Weight Centile at Six Months (n = 50) | Weight Centile at Two Years (n = 56) | BMI Centile at Six Months (n = 50) | BMI Centile at Two Years (n = 56) | |
|---|---|---|---|---|---|
| PC1 correlation | −0.253 * | 0.119 | −0.020 | 0.099 | −0.098 |
| PC1 p-value | 0.047 * | 0.410 | 0.887 | 0.495 | 0.477 |
| PC2 correlation | −0.028 | 0.054 | −0.056 | −0.120 | −0.044 |
| PC2 p-value | 0.826 | 0.711 | 0.683 | 0.405 | 0.751 |
| PC3 correlation | −0.131 | −0.258 | 0.220 | −0.153 | 0.025 |
| PC3 p-value | 0.312 | 0.071 | 0.107 | 0.289 | 0.854 |
| PC4 correlation | 0.087 | −0.059 | −0.076 | −0.035 | −0.081 |
| PC4 p-value | 0.500 | 0.683 | 0.583 | 0.809 | 0.555 |
| PC5 correlation | −0.082 | 0.040 | 0.021 | 0.056 | −0.055 |
| PC5 p-value | 0.524 | 0.783 | 0.878 | 0.699 | 0.692 |
| PC6 correlation | 0.064 | −0.283 * | −0.132 | −0.386 * | −0.139 |
| PC6 p-value | 0.624 | 0.047 * | 0.336 | 0.006 * | 0.312 |
| PC7 correlation | −0.128 | −0.128 | 0.172 | −0.174 | 0.188 |
| PC7 p-value | 0.322 | 0.374 | 0.209 | 0.227 | 0.169 |
| PC8 correlation | 0.065 | −0.047 | −0.017 | −0.239 | 0.080 |
| PC8 p-value | 0.614 | 0.743 | 0.902 | 0.094 | 0.559 |
| PC9 correlation | 0.004 | −0.339 * | 0.131 | −0.216 | 0.022 |
| PC9 p-value | 0.978 | 0.016 * | 0.339 | 0.133 | 0.873 |
| PC10 correlation | −0.030 | −0.147 | −0.021 | −0.105 | 0.001 |
| PC10 p-value | 0.818 | 0.307 | 0.881 | 0.468 | 0.993 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geraghty, A.A.; Sexton-Oates, A.; O’Brien, E.C.; Saffery, R.; McAuliffe, F.M. Epigenetic Patterns in Five-Year-Old Children Exposed to a Low Glycemic Index Dietary Intervention during Pregnancy: Results from the ROLO Kids Study. Nutrients 2020, 12, 3602. https://doi.org/10.3390/nu12123602
Geraghty AA, Sexton-Oates A, O’Brien EC, Saffery R, McAuliffe FM. Epigenetic Patterns in Five-Year-Old Children Exposed to a Low Glycemic Index Dietary Intervention during Pregnancy: Results from the ROLO Kids Study. Nutrients. 2020; 12(12):3602. https://doi.org/10.3390/nu12123602
Chicago/Turabian StyleGeraghty, Aisling A., Alex Sexton-Oates, Eileen C. O’Brien, Richard Saffery, and Fionnuala M. McAuliffe. 2020. "Epigenetic Patterns in Five-Year-Old Children Exposed to a Low Glycemic Index Dietary Intervention during Pregnancy: Results from the ROLO Kids Study" Nutrients 12, no. 12: 3602. https://doi.org/10.3390/nu12123602
APA StyleGeraghty, A. A., Sexton-Oates, A., O’Brien, E. C., Saffery, R., & McAuliffe, F. M. (2020). Epigenetic Patterns in Five-Year-Old Children Exposed to a Low Glycemic Index Dietary Intervention during Pregnancy: Results from the ROLO Kids Study. Nutrients, 12(12), 3602. https://doi.org/10.3390/nu12123602





