Skeletal Muscle and the Maintenance of Vitamin D Status
Abstract
1. Characterization of Vitamin D Status
2. Evidence for Conservation of 25(OH)D
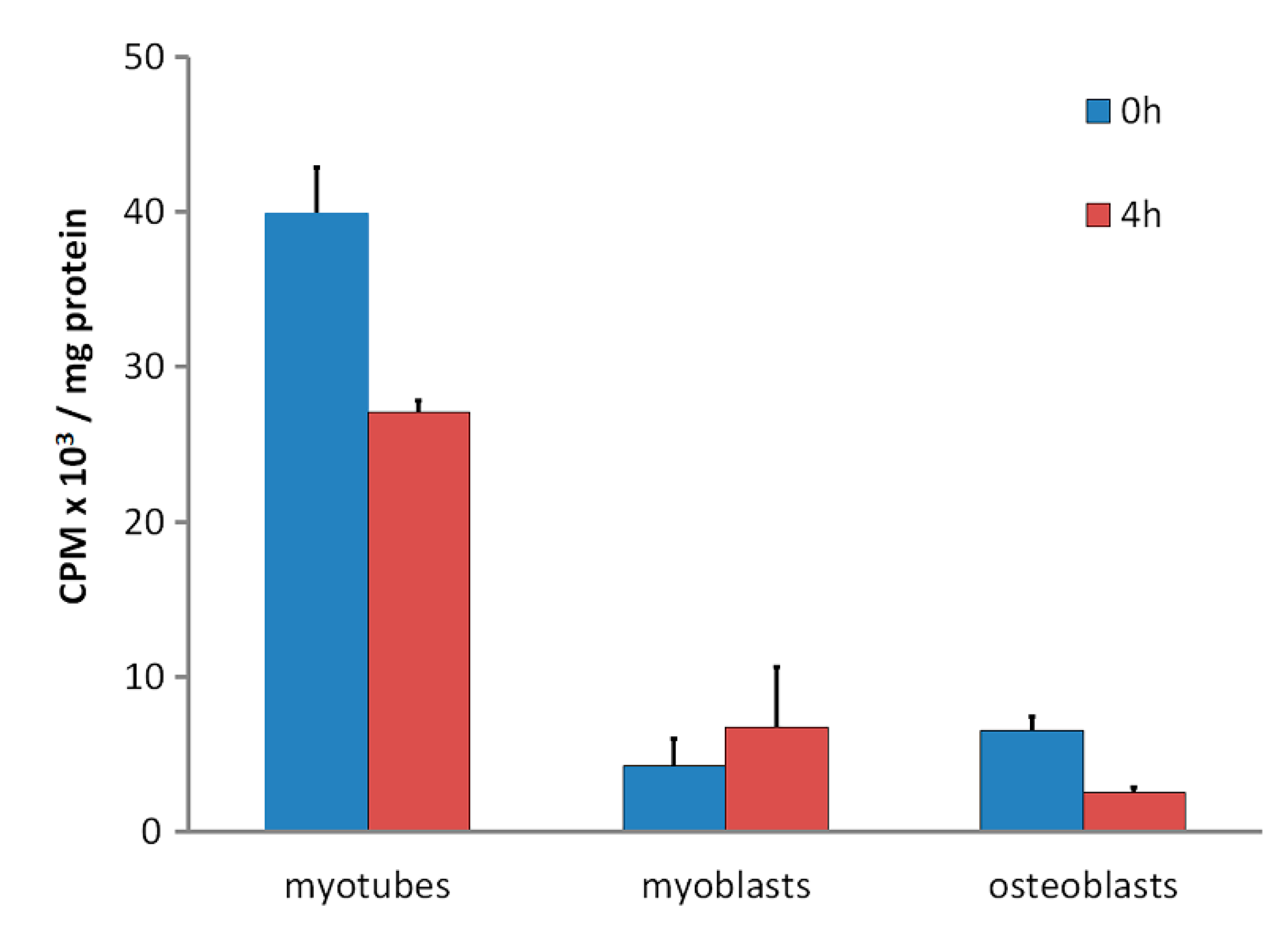
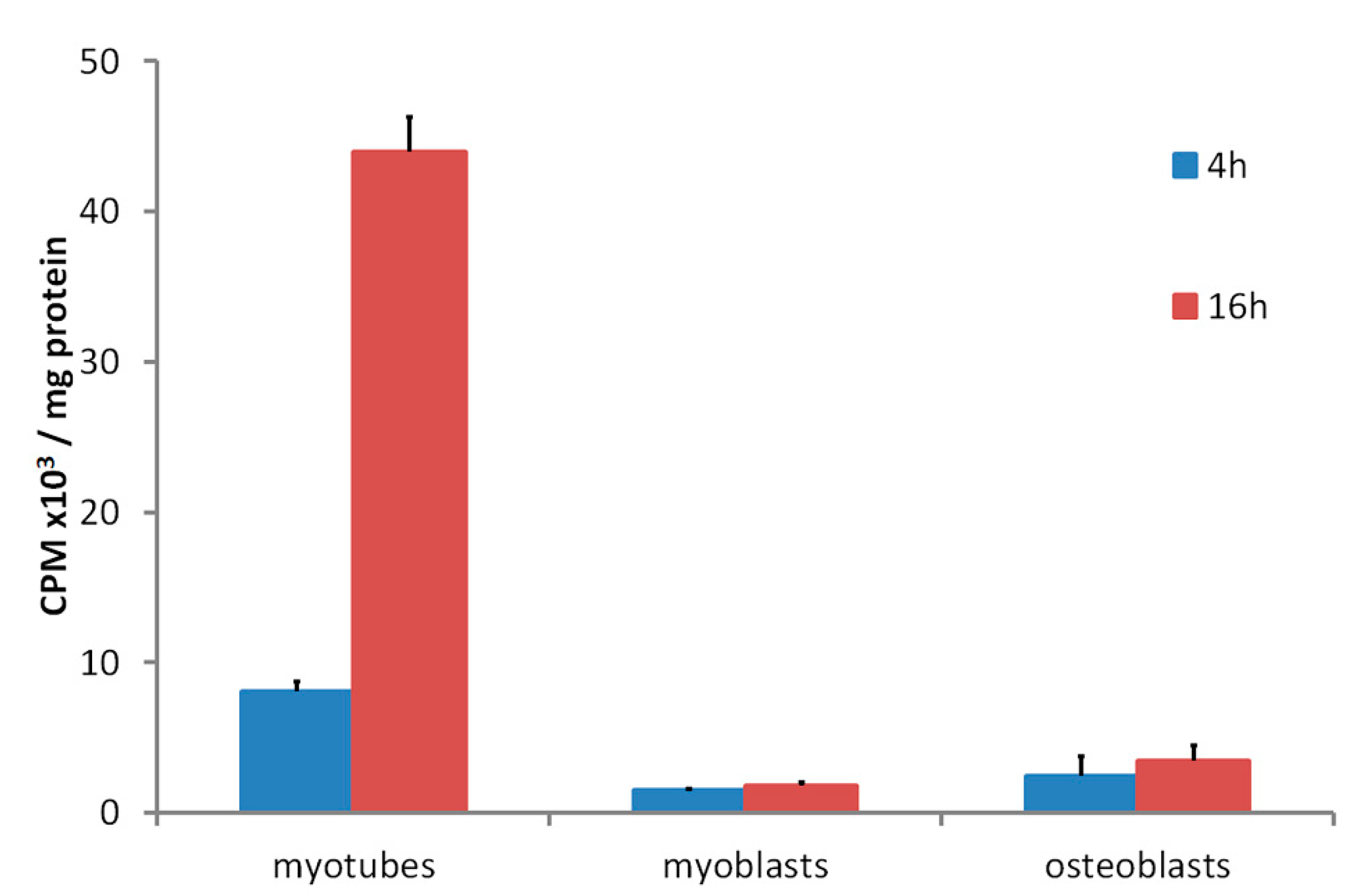
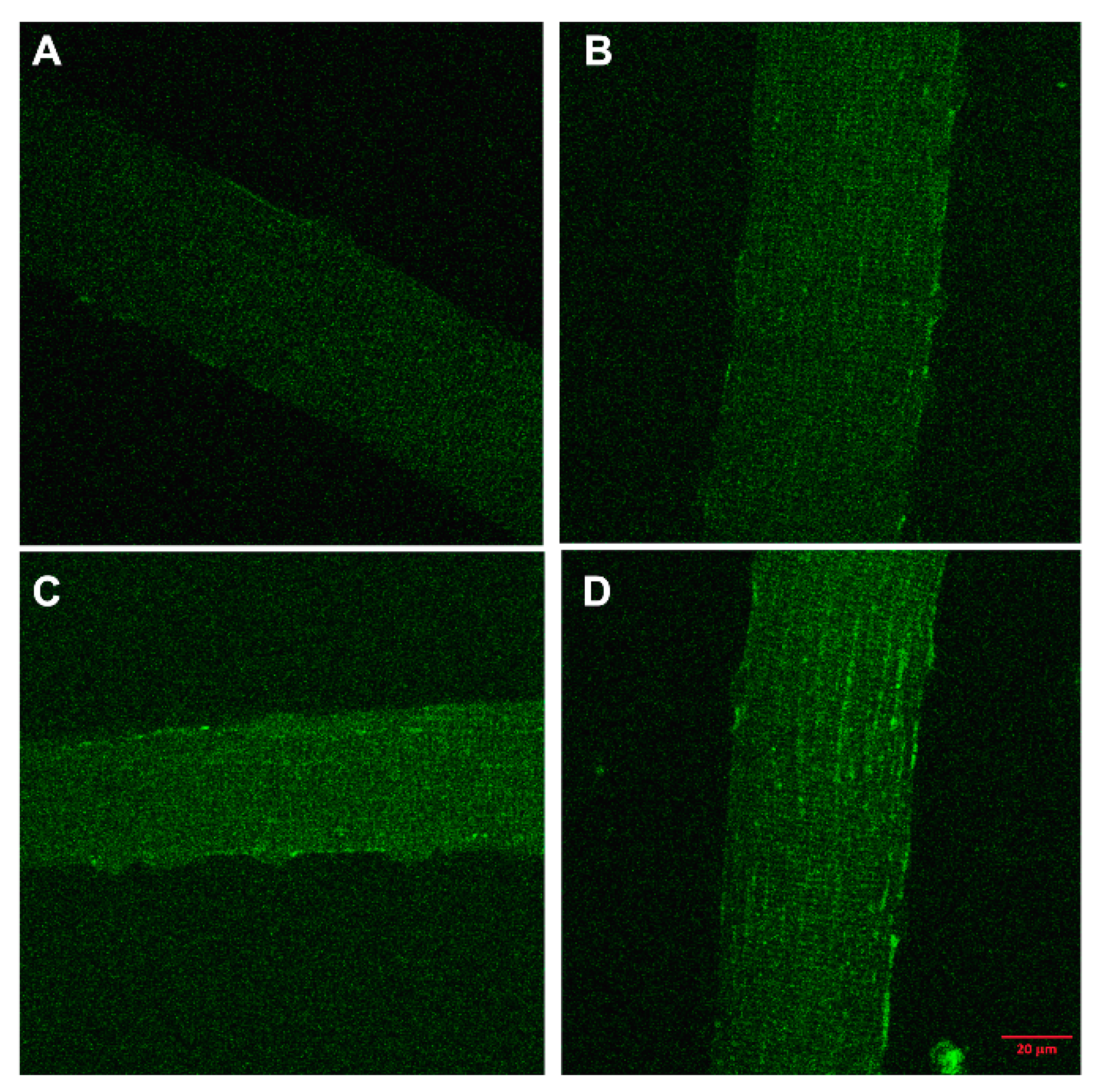
3. Skeletal Muscle Cell Uptake of DBP
4. Regulation of Conservation Mechanism for 25(OH)D
5. Failure of Conservation Mechanism for 25(OH)D
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Hassan Murad, M.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.J.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; Kovacs, C.S.; et al. IOM Committee Members Respond to Endocrine Society Vitamin D Guideline. J. Clin. Endocrinol. Metab. 2012, 97, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Vik, T.; Try, K.; Strømme, J.H. The vitamin D status of man at 70 degrees north. Scand. J. Clin. Lab. Investig. 1980, 40, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Parviainen, M.T.; Koskinen, T. Vitamin A, D and E status in a Finnish population—A multivitamin study. Hum. Nutr. Clin. Nutr. 1983, 37, 397–403. [Google Scholar] [PubMed]
- Nowson, C.A.; McGrath, J.J.; Ebeling, P.R.; Haikerwal, A.; Daly, R.M.; Sanders, K.M.; Seibel, M.J.; Mason, R.S.; Working Group of Australian and New Zealand Bone and Mineral Society; Endocrine Society of Australia and Osteoporosis Australia. Vitamin D and health in adults in Australia and New Zealand: A position statement. Med. J. Aust. 2012, 196, 686–687. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Didriksen, A.; Burild, A.; Jakobsen, J.; Fuskevåg, O.M.; Jorde, R. Vitamin D3 increases in abdominal subcutaneous fat tissue after supplementation with vitamin D3. Eur. J. Endocrinol. 2015, 172, 235–241. [Google Scholar] [CrossRef]
- Carrelli, A.; Bucovsky, M.; Horst, R.; Cremers, S.; Zhang, C.; Bessler, M.; Schrope, B.; Evanko, J.; Blanco, J.; Silverberg, S.J.; et al. Vitamin D Storage in Adipose Tissue of Obese and Normal Weight Women. J. Bone Miner. Res. 2017, 32, 237–242. [Google Scholar] [CrossRef]
- Connors, M.H.; Sheikholislam, B.M.; Iria, J. Vitamin D Toxicity after Dieting in Hypoparathyroidism. Pediatrics 1976, 57, 794–796. [Google Scholar]
- Ziaie, H.; Razmjou, S.; Jomhouri, R.; Jenabi, A. Vitamin D Toxicity; Stored and Released from Adipose Tissue? Arch. Iran Med. 2016, 19, 597–600. [Google Scholar] [PubMed]
- Perticone, M.; Maio, R.; Sciacqua, A.; Suraci, E.; Pinto, A.; Pujia, R.; Zito, R.; Gigliotti, S.; Sesti, G.; Perticone, F. Ketogenic Diet-Induced Weight Loss is Associated with an Increase in Vitamin D Levels in Obese Adults. Molecules 2019, 24, 2499. [Google Scholar] [CrossRef] [PubMed]
- Clements, M.R.; Davies, M.; Fraser, D.R.; Lumb, G.A.; Mawer, E.B.; Adams, P.H. Metabolic inactivation of vitamin D is enhanced in primary hyperparathyroidism. Clin. Sci. 1987, 73, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Datta, P.; Philipsen, P.A.; Olsen, P.; Bogh, M.K.; Johansen, P.; Schmedes, A.V.; Morling, N.; Wulfa, H.C. The half-life of 25(OH)D after UVB exposure depends on gender and vitamin D receptor polymorphism but mainly on the start level. Photochem. Photobiol. Sci. 2017, 16, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.S.; Lissner, D.; Posen, S.; Norman, A.W. Blood levels of dihydroxylated vitamin D metabolites after an oral dose. Brit. Med. J. 1980, 280, 449–450. [Google Scholar] [CrossRef][Green Version]
- Schapiro, S.; Percin, C.J.; Kotichas, F.J. Half-life of plasma corticosterone during development. Endocrinology 1971, 89, 284–286. [Google Scholar] [CrossRef]
- Haddad, J.G.; Fraser, D.R.; Lawson, D.E.M. Vitamin-D Plasma-Binding Protein—Turnover and Fate in the Rabbit. J. Clin. Investig. 1981, 67, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, M.; Blum, C.B.; Ramakrishnan, R.; Dell, R.B.; Goodman, D.S. Turnover of the plasma binding protein for vitamin D and its metabolites in normal human subjects. J. Clin. Endocrinol. Metab. 1981, 53, 1110–1116. [Google Scholar] [CrossRef]
- Abboud, M.; Rybchyn, M.S.; Rizk, R.; Fraser, D.R.; Mason, R.S. Sunlight exposure is just one of the factors which influence vitamin D status. Photochem. Photobiol. Sci. 2017, 16, 302–313. [Google Scholar] [CrossRef]
- Haddad, J.G. Purification, characterization and quantitation of the human serum binding protein for vitamin D and its metabolites. Methods Enzymol. 1980, 67, 449–459. [Google Scholar]
- Clements, M.R.; Fraser, D.R. Vitamin D supply to the rat fetus and neonate. J. Clin. Investig. 1988, 81, 1768–1773. [Google Scholar] [CrossRef]
- Liu, J.; Greenfield, H.; Strobel, N.; Fraser, D.R. The influence of latitude on the concentration of vitamin D3 and 25-hydroxy-vitamin D3 in Australian red meat. Food Chem. 2013, 140, 432–435. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Heymsfield, S.B.; Wang, Z.; Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 2000, 89, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Sawka, M.N.; Young, A.J.; Pandolf, K.B.; Dennis, R.C.; Valeri, C.R. Erythrocyte, plasma, and blood volume of healthy young men. Med. Sci. Sports 1992, 24, 447–453. [Google Scholar] [CrossRef]
- Foo, L.H.; Zhang, Q.; Zhu, K.; Ma, G.; Trube, A.; Greenfield, H.; Fraser, D.R. Relationship between vitamin D status, body composition and physical exercise of adolescent girls in Beijing. Osteoporos. Int. 2009, 20, 417–425. [Google Scholar] [CrossRef]
- Scragg, R.; Holdaway, I.; Jackson, R.; Lim, T. Plasma 25-hydroxyvitamin D3 and its relation to physical activity and other heart disease risk factors in the general population. Ann. Epidemiol. 1992, 2, 697–703. [Google Scholar] [CrossRef]
- Bell, N.H.; Godsen, R.N.; Henry, D.P.; Shary, J.; Epstein, S. The effects of muscle-building exercise on vitamin D and mineral metabolism. J. Bone Miner. Res. 1988, 3, 369–373. [Google Scholar] [CrossRef]
- Abboud, M.; Rybchyn, M.S.; Liu, J.; Gordon-Thomson, C.; Brennan-Speranza, T.C.; Cole, L.; Greenfield, H.; Fraser, D.R.; Mason, R.S. The effect of parathyroid hormone on the uptake and retention of 25-hydroxyvitamin D in skeletal muscle cells. J. Steroid Biochem. Mol. Biol. 2017, 173, 173–179. [Google Scholar] [CrossRef]
- Abboud, M.; Puglisi, D.A.; Davies, B.N.; Rybchyn, M.; Whitehead, N.P.; Brock, K.E.; Cole, L.; Gordon-Thomson, C.; Fraser, D.R.; Mason, R.S. Evidence for a specific uptake and retention mechanism for 25-hydroxyvitamin D (25OHD) in skeletal muscle cells. Endocrinology 2013, 154, 3022–3030. [Google Scholar] [CrossRef]
- Nykjaer, A.; Dragun, D.; Walther, D.; Vorum, H.; Jacobsen, C.; Herz, J.; Melsen, F.; Christensen, E.I.; Willnow, T.E. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell 1999, 96, 507–515. [Google Scholar] [CrossRef]
- Shiying, C.; Verroust, P.J.; Stiren, K.; Moestrup, S.K.; Christensen, E.I. Megalin/gp330 mediates uptake of albumin in renal proximal tubule. Am. J. Physiol. 1996, 271, F900–F907. [Google Scholar]
- Gressner, O.A.; Lahme, B.; Gressner, A.M. Gc-globulin (vitamin D binding protein) is synthesized and secreted by hepatocytes and internalized by hepatic stellate cells through Ca(2+)-dependent interaction with the megalin/gp330 receptor. Clin. Chim. Acta 2008, 390, 28–37. [Google Scholar] [CrossRef]
- Van Baelen, H.; Bouillon, R.; De Moor, P. Vitamin D binding protein (Gc-globulin) binds actin. J. Biol. Chem. 1980, 255, 2270–2272. [Google Scholar] [PubMed]
- Bouillon, R.; Schuit, F.; Antonio, L.; Rastinejad, F. Vitamin D Binding Protein: A Historic Overview. Front. Endocrinol. 2020, 10, 910. [Google Scholar] [CrossRef] [PubMed]
- Goldschmidt-Clermont, P.J.; Van Baelen, H.; Bouillon, R.; Shook, T.E.; Williams, M.H.; Nel, A.E.; Galbraith, R.M. Role of group-specific component (vitamin D binding protein) in clearance of actin from the circulation in the rabbit. J. Clin. Investig. 1988, 81, 1519–1527. [Google Scholar] [CrossRef] [PubMed]
- Otterbein, L.R.; Cosio, C.; Graceffa, P.; Dominguez, R. Crystal structures of the vitamin D-binding protein and its complex with actin: Structural basis of the actin-scavenger system. Proc. Natl. Acad. Sci. USA 2002, 99, 8003–8008. [Google Scholar] [CrossRef]
- Haddad, J.G. Human serum binding protein for vitamin D and its metabolites (DBP): Evidence that actin is the DBP binding compound in human skeletal muscle. Arch. Biochem. Biophys. 1982, 213, 538–544. [Google Scholar] [CrossRef]
- Brennan-Speranza, T.C.; Mor, D.; Mason, R.S.; Bartlett, J.R.; Duque, G.; Levinger, I.; Levinger, P. Skeletal muscle vitamin D in patients with end stage osteoarthritis of the knee. J. Steroid Biochem. Mol. Biol. 2017, 173, 180–184. [Google Scholar] [CrossRef]
- Reed, R.K. Albumin concentration and colloid osmotic pressure of interstitial fluid collected by wick technique from rat skeletal muscle. Evaluation of the method. Acta Physiol. Scand. 1981, 112, 1–5. [Google Scholar] [CrossRef]
- Haddad, J.G. Plasma vitamin D-binding protein (Gc globulin): Multiple tasks. J. Steroid Biochem. Mol. Biol. 1995, 53, 579–582. [Google Scholar] [CrossRef]
- Mason, R.S.; Rybchyn, M.S.; Abboud, M.; Brennan-Speranza, T.C.; Fraser, D.R. The role of skeletal muscle in maintaining vitamin D status in winter. Curr. Dev. Nutr. 2019, 3, nzz087. [Google Scholar] [CrossRef]
- Schiavon, C.R.; Zhang, T.; Zhao, B.; Moore, A.S.; Wales, P.; Andrade, L.R.; Wu, M.; Sung, T.-C.; Dayn, Y.; Feng, J.W.; et al. Actin chromobody imaging reveals sub-organellar actin dynamics. Nat. Methods 2020. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Pinckaers, P.J.M.; Smeets, J.S.J.; Betz, M.W.; Senden, J.M.; Goessens, J.P.B.; Gijsen, A.P.; Rollo, I.; Verdijk, L.B.; van Loon, L.J.C. Dose-response effects of dietary protein on muscle protein synthesis during recovery from endurance exercise in young men: A double-blind randomized trial. Am. J. Clin. Nutr. 2020, 112, 303–317. [Google Scholar] [CrossRef] [PubMed]
- McGlory, C.; Devries, M.C.; Phillips, S.M. Skeletal muscle and resistance exercise training; the role of protein synthesis in recovery and remodeling. J. Appl. Physiol. 2017, 122, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Dunnigan, M.G.; Henderson, J.B.; Hole, D.J.; Mawer, E.B.; Berry, J.L. Meat consumption reduces the risk of nutritional rickets and osteomalacia. Br. J. Nutr. 2005, 94, 983–991. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abboud, M.; Rybchyn, M.S.; Ning, Y.J.; Brennan-Speranza, T.C.; Girgis, C.M.; Gunton, J.E.; Fraser, D.R.; Mason, R.S. 1,25-Dihydroxycholecalciferol (calcitriol) modifies uptake and release of 25-hydroxycholecalciferol in skeletal muscle cells in culture. J. Steroid Biochem. Mol. Biol. 2018, 177, 109–115. [Google Scholar] [CrossRef]
- Girgis, C.M.; Mokbel, N.; Cha, K.M.; Houweling, P.J.; Abboud, M.; Fraser, D.R.; Mason, R.S.; Clifton-Bligh, R.J.; Gunton, J.E. The vitamin D receptor (VDR) is expressed in skeletal muscle of male mice and modulates 25-hydroxyvitamin D (25OHD) uptake in myofibers. Endocrinology 2014, 155, 3227–3237. [Google Scholar] [CrossRef]
- Das, G.; Taylor, P.N.; Javaid, H.; Tennant, B.P.; Green, J.; Aldridge, A.; Okosieme, O. Seasonal variation of vitamin D and serum thyrotropin levels and its relationship in a euthyroid Caucasian population. Endocr. Pract. 2018, 24, 53–59. [Google Scholar] [CrossRef]
- Yalla, Y.; Bobba, G.; Guo, G.; Stankiewicz, A.; Ostlund, R. Parathyroid hormone reference ranges in healthy individuals classified by vitamin D status. J. Endocrinol. Investig. 2019, 42, 1353–1360. [Google Scholar] [CrossRef]
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2014, 111, 23–45. [Google Scholar] [CrossRef]
- Alfredsson, L.; Armstrong, B.K.; Butterfield, D.A.; Chowdhury, R.; de Gruijl, F.R.; Feelisch, M.; Garland, C.F.; Hart, P.H.; Hoel, D.G.; Jacobsen, R.; et al. Insufficient Sun Exposure Has Become a Real Public Health Problem. Int. J. Environ. Res. Public Health 2020, 17, 5014. [Google Scholar] [CrossRef]
- McGee, M. Vitamin D: Insufficiency, Uncertainty and Achievability. Int. J. Vitam. Nutr. Res. 2020, 90, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Van Baelen, H.; Allewaert, K.; Bouillon, R. New Aspects of the plasma carrier protein for 25-hydroxycholecalciferol in vertebrates. Ann. N. Y. Acad. Sci. 1988, 538, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.W.M.; Watson, G. The plasma transport proteins of 25-hydroxycholeclciferol in fish, amphibia, reptiles and birds. Comp. Biochem. Physiol. 1976, 53B, 167–172. [Google Scholar]
- Pierens, S.L.; Fraser, D.R. The origin and metabolism of vitamin D in rainbow trout. J. Steroid Biochem. Mol. Biol. 2015, 145, 58–64. [Google Scholar] [CrossRef]
- Chick, H. Study of rickets in Vienna 1919–1922. Med. Hist. 1976, 20, 41–51. [Google Scholar] [CrossRef]
- McCance, R.A.; Widdowson, E.M. Studies of Undernutrition, Wuppertal 1946–49. 1. The German Background; Special Report Series; Medical Research Council: Great Britain, UK, 1951; Volume 275, pp. 1–20. [Google Scholar]
- Fraser, D.R. Vitamin D-deficiency in Asia. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 491–495. [Google Scholar] [CrossRef]
- Whitehead, R.G.; Alleyne, G.A. Pathophysiological factors of importance in protein-energy malnutrition. Br. Med. Bull. 1972, 28, 72–79. [Google Scholar] [CrossRef]
- Laing, C.J.; Fraser, D.R. Changes with malnutrition in the concentration of plasma vitamin D binding protein in growing rats. Br. J. Nutr. 2002, 88, 133–139. [Google Scholar] [CrossRef]
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
| Blood Plasma Volume (L) | Muscle Mass (kg) | |
|---|---|---|
| Total for 70 kg human | 2.7–4.3 L [23] | 21–26.6 kg ## [22] |
| 25(OH)D concentration | 20 µg/L ### | 1–3 µg/kg #### [21] |
| Total body 25(OH)D | 54–86 µg | 21–80 µg |
| Season | Plasma 25(OH)D3 | Muscle 25(OH)D |
|---|---|---|
| End of summer values (n = 5) | 10.67 ± 1.65 ng/mL | 0.84 ± 0.18 µg/100 g tissue |
| End of winter values (n = 5) | 5.36 ± 0.71 ng/mL * | 1.82 ± 0.39 µg/100 g tissue * |
| DBP Concentration | 0 | 1 nM | 10 nM | 100 nM |
|---|---|---|---|---|
| [3H]25(OH)D cpm/mg cell protein | 1284 ± 147 | 1233 ± 67 | 778 ± 76 * | 402 ± 20 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rybchyn, M.S.; Abboud, M.; Puglisi, D.A.; Gordon-Thomson, C.; Brennan-Speranza, T.C.; Mason, R.S.; Fraser, D.R. Skeletal Muscle and the Maintenance of Vitamin D Status. Nutrients 2020, 12, 3270. https://doi.org/10.3390/nu12113270
Rybchyn MS, Abboud M, Puglisi DA, Gordon-Thomson C, Brennan-Speranza TC, Mason RS, Fraser DR. Skeletal Muscle and the Maintenance of Vitamin D Status. Nutrients. 2020; 12(11):3270. https://doi.org/10.3390/nu12113270
Chicago/Turabian StyleRybchyn, Mark S., Myriam Abboud, David A. Puglisi, Clare Gordon-Thomson, Tara C. Brennan-Speranza, Rebecca S. Mason, and David R. Fraser. 2020. "Skeletal Muscle and the Maintenance of Vitamin D Status" Nutrients 12, no. 11: 3270. https://doi.org/10.3390/nu12113270
APA StyleRybchyn, M. S., Abboud, M., Puglisi, D. A., Gordon-Thomson, C., Brennan-Speranza, T. C., Mason, R. S., & Fraser, D. R. (2020). Skeletal Muscle and the Maintenance of Vitamin D Status. Nutrients, 12(11), 3270. https://doi.org/10.3390/nu12113270






