Age-Appropriate Feeding Practices in Cambodia and the Possible Influence on the Growth of the Children: A Longitudinal Study
Abstract
:1. Introduction
2. Materials and Methods
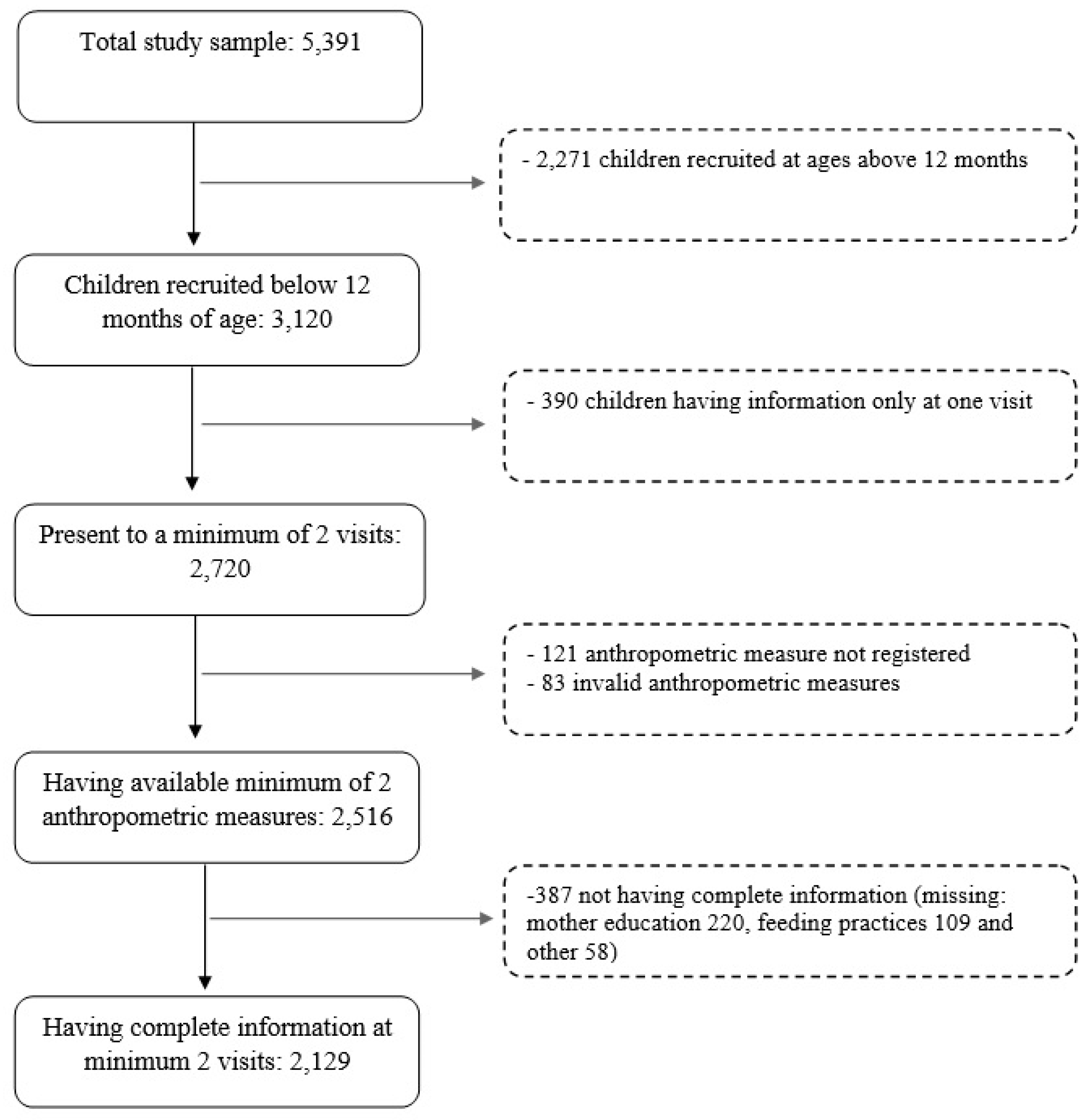
2.1. Study Design and Participants
2.2. Outcome Measures
2.3. Statistical Analysis
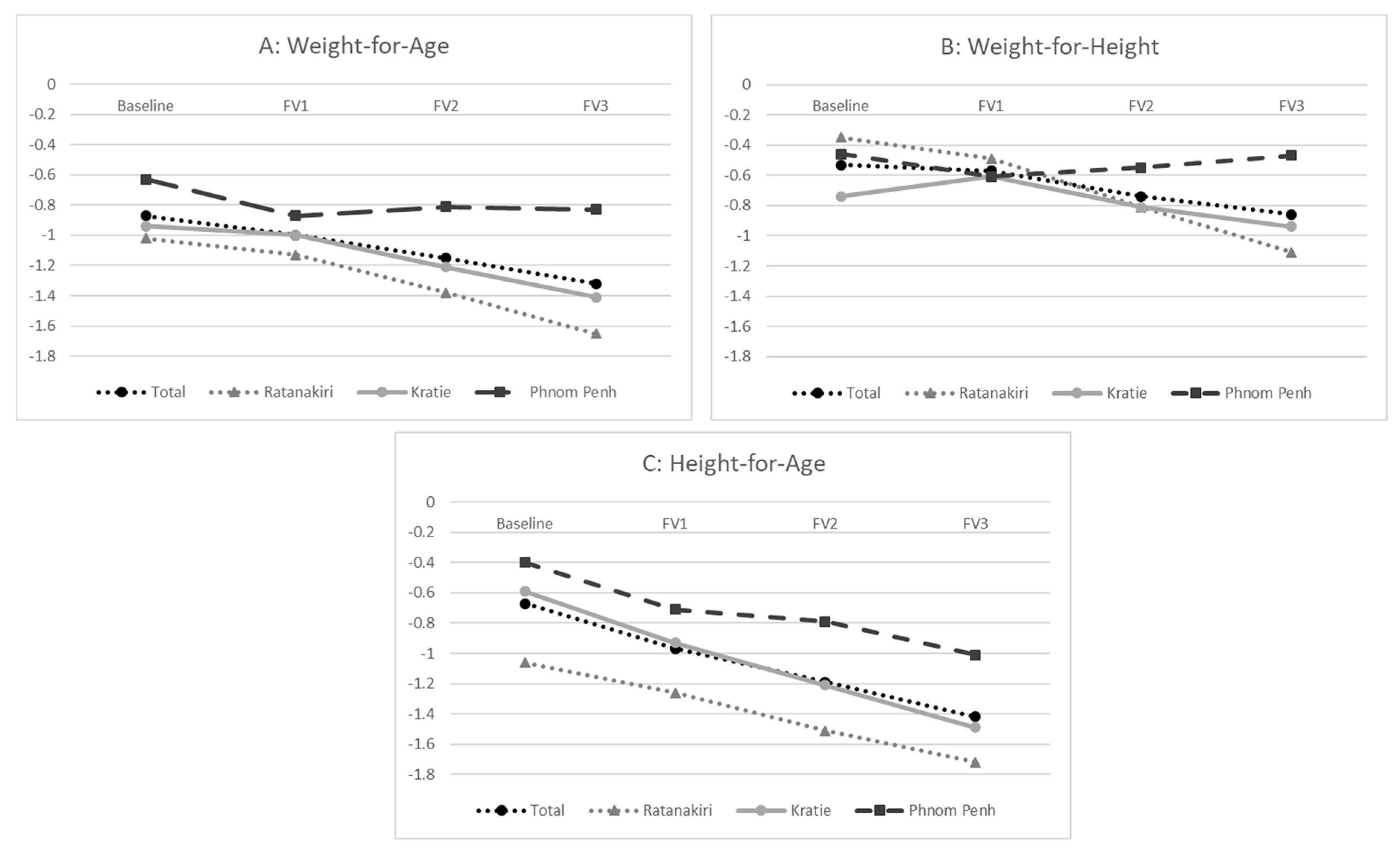
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, L.C.; Haddad, L. Reducing child undernutrition: Past drivers and priorities for the post-MDG era. World Dev. 2015, 68, 180–204. [Google Scholar] [CrossRef] [Green Version]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; De Onis, M.; Ezzati, M.; Grantham-Mcgregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Martins, V.J.B.; Toledo Florêncio, T.M.M.; Grillo, L.P.; Do Carmo, P.; Franco, M.; Martins, P.A.; Clemente, A.P.G.; Santos, C.D.L.; Vieira, M.D.F.A.; Sawaya, A.L. Long-lasting effects of undernutrition. Int. J. Environ. Res. Public Health 2011, 8, 1817–1846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bagriansky, J.; Champa, N.; Pak, K.; Whitney, S.; Laillou, A. The economic consequences of malnutrition in Cambodia, more than 400 million US dollar lost annually. Asia Pac. J. Clin. Nutr. 2014, 23, 524–531. [Google Scholar] [CrossRef]
- National Institute of Statistics; Directorate General for Health; ICF International. Cambodia Demographic and Health Survey. 2015. Available online: https://dhsprogram.com/pubs/pdf/fr312/fr312.pdf (accessed on 28 October 2019).
- Deki, P. Factors affecting early childhood growth and development: Golden 1000 days. Adv. Pract. Nurs. 2016, 1, 1–4. [Google Scholar] [CrossRef]
- United States Department of Agriculture. Infant Nutrition and Feeding: A guide for Use in the WIC and CSF Programs; United States Department of Agriculture: Washington, DC, USA, 2009; ISBN 978-8-57-811079-6.
- Jackson, A.A. Feeding the normal infant, child and adolescent. Medicine 2015, 43, 127–131. [Google Scholar] [CrossRef]
- Reinbott, A.; Jordan, I. Determinants of child malnutrition and infant and young child feeding approaches in Cambodia. World Rev. Nutr. Diet. 2016, 115, 61–67. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Indicators for Assessing Infant and Young Child Feeding Practices: Part II: Measurments; WHO: Geneva, Switzerland, 2009; ISBN 978-9-24-159929-0. Available online: https://www.who.int/maternal_child_adolescent/documents/9789241599290/en/ (accessed on 19 December 2019).
- World Health Organization. Indicators for Assessing Infant and Young Child Feeding Practices Part I: Definition; WHO: Geneva, Switzerland, 2008; ISBN 978-9-24-159666-4. Available online: https://www.who.int/maternal_child_adolescent/documents/9789241596664/en/ (accessed on 19 December 2019).
- Rakotomanana, H.; Gates, G.E.; Hildebrand, D.; Stoecker, B.J. Situation and determinants of the infant and young child feeding (IYCF) indicators in Madagascar: Analysis of the 2009 demographic and health survey. BMC Public Health 2017, 17, 812. [Google Scholar] [CrossRef] [Green Version]
- Ruel, M.T. Measuring infant and young child complementary feeding practices: Indicators, current practice, and research gaps. In Complementary Feeding: Building the Foundations for a Healthy Life: Nestlé Nutrition Institute Workshop Series; Nestec Ltd. Vevey/S. Karger AG Basel: Vevey, Switzerland, 2017; Volume 87, pp. 73–87. [Google Scholar] [CrossRef]
- Jones, A.D.; Ickes, S.B.; Smith, L.E.; Mbuya, M.N.N.; Chasekwa, B.; Heidkamp, R.A.; Menon, P.; Zongrone, A.A.; Stoltzfus, R.J. World Health Organization infant and young child feeding indicators and their associations with child anthropometry: A synthesis of recent findings. Matern. Child Nutr. 2014, 10, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Marriott, B.P.; White, A.; Hadden, L.; Davies, J.C.; Wallingford, J.C. World Health Organization (WHO) infant and young child feeding indicators: Associations with growth measures in 14 low-income countries. Matern. Child Nutr. 2012, 8, 354–370. [Google Scholar] [CrossRef]
- Mallard, S.R.; Houghton, L.A.; Filteau, S.; Mullen, A.; Nieuwelink, J.; Chisenga, M.; Siame, J.; Gibson, R.S. Dietary diversity at 6 months of age is associated with subsequent growth and mediates the effect of maternal education on infant growth in urban Zambia. J. Nutr. 2014, 144, 1818–1825. [Google Scholar] [CrossRef] [PubMed]
- Moursi, M.M.; Arimond, M.; Dewey, K.G.; Treche, S.; Ruel, M.T.; Delpeuch, F. Dietary diversity is a good predictor of the micronutrient density of the diet of 6- to 23-month-old children in Madagascar. J. Nutr. 2008, 138, 2448–2453. [Google Scholar] [CrossRef] [PubMed]
- Reinbott, A.; Jordan, I.; Herrmann, J.; Kuchenbecker, J.; Kevanna, O.; Krawinkel, M.B. Role of breastfeeding and complementary food on hemoglobin and ferritin levels in a cambodian cross-sectional sample of children aged 3 to 24 months. PLoS ONE 2016, 11, e0150750. [Google Scholar] [CrossRef] [PubMed]
- Lohia, N.; Udipi, S.A. Infant and child feeding index reflects feeding practices, nutritional status of urban slum children. BMC Pediatr. 2014, 14, 290. [Google Scholar] [CrossRef] [Green Version]
- Lourenço, B.H.; Cardoso, M.A. Infant feeding practices, childhood growth and obesity in adult life. Arq. Bras. Endocrinol. Metabol. 2009, 53, 528–539. [Google Scholar] [CrossRef] [Green Version]
- Irarrázaval, B.; Barja, S.; Bustos, E.; Doirsaint, R.; Senethmm, G.; Guzmán, M.P.; Uauy, R. Influence of feeding practices on malnutrition in haitian infants and young children. Nutrients 2018, 10, 382. [Google Scholar] [CrossRef] [Green Version]
- Lawan, U.; Sani, A.; Amole, G.; Jahum, M. Age-appropriate feeding practices and nutritional status of infants attending child welfare clinic at a Teaching Hospital in Nigeria. J. Fam. Community Med. 2014, 21, 6–12. [Google Scholar] [CrossRef] [Green Version]
- Semahegn, A.; Tesfaye, G.; Bogale, A. Complementary feeding practice of mothers and associated factors in Hiwot Fana specialized hospital, eastern Ethiopia. Pan Afr. Med. J. 2014, 18, 143. [Google Scholar] [CrossRef]
- Cambodian National Nutrition Program Nutrition Handbook for the Family—Cambodian National Guidelines. 2011. Available online: http://foodsecurity.gov.kh/sites/default/files/documents/Nutrition_hand_Book_Family_Eng_Version.pdf (accessed on 9 December 2019).
- WHO. WHO Child Growth Standards Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development Department of Nutrition for Health and Development; WHO: Geneva, Switzerland, 2006; ISBN 924154693X. [Google Scholar]
- Wieringa, F.; Gauthier, L.; Greffeuille, V.; Som, S.; Dijkhuizen, M.; Laillou, A.; Chamnan, C.; Berger, J.; Poirot, E. Identification of acute malnutrition in children in Cambodia requires both mid upper arm circumference and weight-for-height to offset gender bias of each indicator. Nutrients 2018, 10, 786. [Google Scholar] [CrossRef] [Green Version]
- Som, S.; Prak, S.; Laillou, A.; Gauthier, L.; Berger, J.; Poirot, E.; Wieringa, F. Diets and feeding practices during the first 1000 days window in the Phnom Penh and north eastern districts of Cambodia. Nutrients 2018, 10, 500. [Google Scholar] [CrossRef] [Green Version]
- Poirot, E.; Wieringa, F.T.; Treglown, S. Water quality for young children in Cambodia—High contamination at collection and consumption level. Matern. Child Nutr. 2019, e12744. Available online: https://www.unicef.org/cambodia/media/1716/file/Water%20quality%20for%20young%20children%20in%20Cambodia.pdf (accessed on 20 December 2019). [CrossRef]
- World Health Organization (WHO); The United Nations Children’s Fund (UNICEF); United States Agency for International Development (USAID); Academy for Educational Development(AED); University of California, Davis (UCDAVIS); International Food Policy Research Institute (IFPRI). Indicators for Assessing Infant and Young Child Feeding Practices: Conclusions of a Consensus Meeting; WHO: Geneva, Switzerland, 2008; Volume 2007, ISBN 978-9-24-159666-4. [Google Scholar]
- World Health Organization (WHO); The United Nations Children’s Fund (UNICEF). Core Questions on Drinking-Water and Sanitation for Household Surveys; WHO: Geneva, Switzerland, 2006; ISBN 9241563265. [Google Scholar]
- Johnson, W.; Balakrishna, N.; Griffiths, P.L. Modelling physical growth using mixed effects models. Am. J. Phys. Anthropol. 2014, 150, 612–625. [Google Scholar] [CrossRef] [Green Version]
- World Health Organisation. Anthro Software—Version 3; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Bates, D. Mixed Models in R Using the lme4 Package Part 2: Longitudinal Data, Modeling Interactions Longitudinal Data: Sleepstudy. 2011. Available online: http://lme4.r-forge.r-project.org/slides/2011-03-16-Amsterdam/2Longitudinal.pdf (accessed on 28 October 2019).
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 251–264. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. 2016. Available online: https://www.r-project.org/ (accessed on 12 May 2018).
- Champeny, M.; Pries, A.M.; Hou, K.; Adhikary, I.; Zehner, E.; Huffman, S.L. Predictors of breast milk substitute feeding among newborns in delivery facilities in urban Cambodia and Nepal. Matern. Child Nutr. 2019, 15 (Suppl. 4), e12754. [Google Scholar] [CrossRef] [Green Version]
- Kassa, T.; Meshesha, B.; Haji, Y.; Ebrahim, J. Appropriate complementary feeding practices and associated factors among mothers of children age 6–23 months in Southern Ethiopia, 2015. BMC Pediatr. 2016, 16, 131. [Google Scholar] [CrossRef] [Green Version]
- Krasevec, J.; An, X.; Kumapley, R.; Bégin, F.; Frongillo, E.A. Diet quality and risk of stunting among infants and young children in low- and middle-income countries. Matern. Child Nutr. 2017, 13, e12430. [Google Scholar] [CrossRef] [Green Version]
- Campbell, R.K.; Aguayo, V.; Kang, Y.; Dzed, L.; Joshi, V.; Waid, J.; Gupta, S.D.; Haselow, N.; West, K.P. Infant and young child feeding practices and nutritional status in Bhutan. Matern. Child Nutr. 2017, 14, e12580. [Google Scholar] [CrossRef]
- Ali, D.; Saha, K.K.; Nguyen, P.H.; Diressie, M.T.; Ruel, M.T.; Menon, P.; Rawat, R. Household food insecurity is associated with higher child undernutrition in Bangladesh, Ethiopia, and Vietnam, but the effect is not mediated by child dietary diversity. J. Nutr. 2013, 143, 2015–2021. [Google Scholar] [CrossRef] [Green Version]
- Ruel, M.; Levin, C.; Armar-Klemesu, M.; Maxwell, D.; Morris, S. Good Care Practices Can Mitigate the Negative Effects of Poverty and Low Maternal Schooling on Children’s Nutritional Status: Evidence from Accra. Am. Soc. Nutr. Sci. 2002, 27. [Google Scholar] [CrossRef]
- Saaka, M.; Galaa, S.Z. Relationships between wasting and stunting and their concurrent occurrence in Ghanaian preschool children. J. Nutr. Metab. 2016, 2016, 4654920. [Google Scholar] [CrossRef] [Green Version]
- Roba, K.T.; O’Connor, T.P.; Belachew, T.; O’Brien, N.M. Variations between post- and pre-harvest seasons in stunting, wasting, and Infant and Young Child Feeding (IYCF) practices among children 6–23 months of age in lowland and midland agro-ecological zones of rural Ethiopia. Pan Afr. Med. J. 2016, 24, 163. [Google Scholar] [CrossRef] [PubMed]
- Khara, T.; Mwangome, M.; Ngari, M.; Dolan, C. Children concurrently wasted and stunted: A meta-analysis of prevalence data of children 6–59 months from 84 countries. Matern. Child Nutr. 2018, 14, e12516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Briend, A.; Khara, T.; Dolan, C. Wasting and stunting-similarities and differences: Policy and programmatic implications. Food Nutr. Bull. 2015, 36, S15–S23. [Google Scholar] [CrossRef] [PubMed]
- Osgood-Zimmerman, A.; Millear, A.I.; Stubbs, R.W.; Shields, C.; Pickering, B.V.; Earl, L.; Graetz, N.; Kinyoki, D.K.; Ray, S.E.; Bhatt, S.; et al. Mapping child growth failure in Africa between 2000 and 2015. Nature 2018, 555, 41–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luke, S.G. Evaluating significance in linear mixed-effects models in R. Behav. Res. Methods 2017, 49, 1494–1502. [Google Scholar] [CrossRef] [PubMed]
| Age Group | Breast-Feeding | Complementary Feeding | |
|---|---|---|---|
| Frequency | Quantity 1 | ||
| 0–5.9 months | Predominant breast-feeding at least 6 times per day | No complementary feeding | |
| 6–8.9 months | Breast-feeding at least 6 times per day (complementary) | Maximum 3 meals per day | 2–3 tablespoons to half Chan Chang Koeh for each meal |
| 9–11.9 months | Breast-feeding at least 6 times per day (complementary) | Minimum of 3 meals per day | From half to a full Chan Chang Koeh for each meal |
| 12 months+ | Breast-feeding at least 3 times per day(complementary) | Minimum of 3 meals per day | Minimum half of a full Chan Chang Koeh for each meal |
| Variable | Total n = 2129 | Ratanakiri n = 682 | Kratie n = 799 | Phnom Penh n = 648 | p-Value |
|---|---|---|---|---|---|
| Age at recruitment | 6.19 (3.30) | 5.87 (3.33) | 6.22 (3.22) | 6.48 (3.33) | 0.005 |
| Age group | |||||
| 0–5.9 months | 48.4% (1030) | 53.1% (362) | 47.4% (379) | 44.6% (289) | |
| 6–8.9 months | 27.2% (580) | 24.4% (167) | 29.6% (236) | 27.3% (177) | |
| 9–11.9 months | 24.3% (519) | 22.4% (153) | 23.0% (184) | 28.1% (182) | |
| Sex: Male | 50.5% (1076) | 47.9% (327) | 50.6% (405) | 53.1% (344) | 0.08 |
| Breast-feeding status 1 | <0.001 | ||||
| Predominantly | 34.3% (731) | 43.2% (287) | 38.9% (303) | 24.2% (141) | |
| Partly | 51.9% (1105) | 51.5% (342) | 57.5% (448) | 54.0% (315) | |
| None | 8.9% (190) | 5.3% (35) | 3.6% (28) | 21.8% (127) | |
| NA’s | (103) | (18) | (20) | (65) | |
| ADF | 43.0% (845) | 47.7% (306) | 52.5% (399) | 25.0% (148) | <0.001 |
| 0–5.9 months | 55.9% (538) | 62.8% (221) | 65.4% (237) | 32.4% (80) | |
| 6–8.9 months | 45.6% (240) | 29.5% (60) | 59.2% (132) | 27.1% (48) | |
| 9–11.9 months | 14.1% (67) | 18.2% (25) | 16.7% (29) | 7.9% (13) | |
| NA’s | (166) | (40) | (40) | (85) | |
| Minimum Diet Diversity 2 | 19.1% (204) | 21.7% (68) | 17% (75) | 21.8% (74) | 0.03 |
| Nutritional status | <0.001 | ||||
| Not stunted nor wasted | 74.3% (1573) | 70.4% (477) | 71.5% (565) | 81.9% (531) | |
| Stunted | 13.8% (295) | 21.7% (147) | 11.6% (92) | 9.0% (56) | |
| Wasted | 14.8% (315) | 12.4% (84) | 20.4% (161) | 10.6% (69) | |
| NA’s | (14) | (5) | (9) | (0) | |
| Mother education | <0.001 | ||||
| No education | 23.0% (490) | 42.8% (292) | 16.8% (134) | 10.0% (65) | |
| Primary | 40.2% (855) | 32.3% (225) | 48.7% (389) | 37.4% (241) | |
| Secondary and more | 39.6% (780) | 24.2% (165) | 34.5% (276) | 52.6% (339) | |
| NA’s | (3) | (0) | (0) | (3) | |
| Number of children | 2.4 (1.37) | 2.59 (1.39) | 2.37(1.28) | 2.30 (1.31) | 0.08 |
| NA’s | (13) | (1) | (6) | (6) | |
| Safe drinking water | 93.4% (1931) | 97.5% (659) | 84.4% (666) | 99.3% (4) | 0.04 |
| NA’s | (63) | (10) | (12) | (41) | |
| Do not have latrine | 40.0% (836) | 52.9% (361) | 57.9% (463) | 2.0% (13) | <0.001 |
| NA’s | (40) | (0) | (0) | (40) |
| Variable (%) | 1st Visit n = 1898 | 2nd Visit n = 1332 | 3rd Visit n = 1212 | 4th Visit n = 1091 | p-Value |
|---|---|---|---|---|---|
| Age group | <0.001 | ||||
| 0–5.9 months | 49.0% (931) | 22.4% (299) | 0% (0) | 0% (0) | |
| 6–8.9 months | 26.7% (506) | 26.7% (355) | 12.9% (157) | 0% (0) | |
| 9–11.9 months | 24.3% (461) | 28.6% (381) | 27.3% (331) | 6.1% (67) | |
| 12 months+ | 0% (0) | 22.3% (297) | 59.7% (724) | 93.9%(1024) | |
| Province | 0.18 | ||||
| Ratanakiri | 33.0% (627) | 36.0% (480) | 34.9% (423) | 34.4% (276) | |
| Kratie | 38.5% (732) | 40.4% (538) | 38.5% (467) | 41.5% (453) | |
| Phnom Penh | 28.4% (539) | 23.6% (314) | 26.6% (322) | 24.0% (262) | |
| Breast-feeding status 1 | <0.001 | ||||
| Predominantly | 37.1% (705) | 16.7% (223) | 1.2% (15) | 1.6% (18) | |
| Partly | 53% (1006) | 77.9% (1037) | 88.2% (1059) | 72.9% (796) | |
| None | 9.9% (187) | 5.4% (72) | 10.6% (128) | 25.4% (277) | |
| ADF excluding quantity 2 | 50.6% (960) | 61.3% (816) | 61.5% (745) | 51.1% (558) | <0.001 |
| ADF | 42.9% (815) | 35.9% (479) | 17.7% (214) | 7.1% (70) | <0.001 |
| 0–5.9 months | 55.9% (520) | 56.5% (169) | 0% (0) | 0% (0) | |
| 6–8.9 months | 45.7% (231) | 48.2% (171) | 56% (88) | 0% (0) | |
| 9–11.9 months | 13.9% (64) | 25.7% (98) | 17.2% (57) | 14.9% (10) | |
| 12 months+ | 0% (0) | 13.8% (41) | 9.5% (69) | 5.9% (60) | |
| Minimum Diet Diversity 3 | 20.0% (194) | 26.9% (278) | 37.1% (449) | 34.9% (424) | <0.001 |
| Nutritional status | <0.001 | ||||
| Not stunted nor wasted | 74.9% (1423) | 71.8% (957) | 68.2% (826) | 61.6% (672) | |
| Stunting | 13.2% (250) | 18.9% (252) | 24.5% (297) | 32.4% (354) | |
| Wasting | 13.6% (259) | 14.7% (196) | 13.7% (166) | 15.4% (168) | |
| Mother education | 0.04 | ||||
| No education | 23.2% (440) | 23.2% (309) | 25.3% (307) | 26.2% (286) | |
| Primary | 39.8% (756) | 40.2% (550) | 40.4% (490) | 40.1% (437) | |
| Secondary and more | 36.9% (702) | 36.6% (473) | 34.2% (415) | 33.7% (368) | |
| Safe drinking water | 93.6% (1777) | 92.9% (1235) | 85.2% (1033) | 78.8% (860) | <0.001 |
| Do not have latrine | 39.9% (759) | 41.2% (559) | 41.2% (499) | 41.8% (456) | 0.06 |
| Model A | Model B | Model C | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Est. | S.E. | P. | Est. | S.E. | P. | Est. | S.E. | P. | |
| WAZ | 0.11 | 0.02 | <0.001 | 0.004 | 0.01 | 0.8 | 0.004 | 0.02 | 0.83 |
| HAZ | 0.17 | 0.02 | <0.001 | 0.01 | 0.02 | 0.91 | 0.01 | 0.02 | 0.90 |
| WHZ | 0.15 | 0.02 | <0.001 | 0.06 | 0.02 | 0.02 | 0.06 | 0.02 | 0.02 |
| Fully-Adjusted Model(C) | Ratanakiri | Kratie | Phnom Penh | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Est. | S.E. | P. | Est. | S.E. | P. | Est. | S.E. | P. | |
| WAZ | 0.05 | 0.03 | 0.12 | 0.001 | 0.03 | 0.88 | 0.02 | 0.04 | 0.19 |
| HAZ | 0.01 | 0.04 | 0.79 | 0.04 | 0.03 | 0.19 | −0.08 | 0.05 | 0.07 |
| WHZ | 0.13 | 0.04 | <0.001 | 0.01 | 0.04 | 0.81 | 0.02 | 0.05 | 0.72 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hondru, G.; Laillou, A.; Wieringa, F.T.; Poirot, E.; Berger, J.; Christensen, D.L.; Roos, N. Age-Appropriate Feeding Practices in Cambodia and the Possible Influence on the Growth of the Children: A Longitudinal Study. Nutrients 2020, 12, 12. https://doi.org/10.3390/nu12010012
Hondru G, Laillou A, Wieringa FT, Poirot E, Berger J, Christensen DL, Roos N. Age-Appropriate Feeding Practices in Cambodia and the Possible Influence on the Growth of the Children: A Longitudinal Study. Nutrients. 2020; 12(1):12. https://doi.org/10.3390/nu12010012
Chicago/Turabian StyleHondru, Gabriela, Arnaud Laillou, Frank T. Wieringa, Etienne Poirot, Jacques Berger, Dirk L. Christensen, and Nanna Roos. 2020. "Age-Appropriate Feeding Practices in Cambodia and the Possible Influence on the Growth of the Children: A Longitudinal Study" Nutrients 12, no. 1: 12. https://doi.org/10.3390/nu12010012
APA StyleHondru, G., Laillou, A., Wieringa, F. T., Poirot, E., Berger, J., Christensen, D. L., & Roos, N. (2020). Age-Appropriate Feeding Practices in Cambodia and the Possible Influence on the Growth of the Children: A Longitudinal Study. Nutrients, 12(1), 12. https://doi.org/10.3390/nu12010012





