A Calorie-Restricted DASH Diet Reduces Body Fat and Maintains Muscle Strength in Obese Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Study Design and Diet
2.3. Follow-Up Visits
2.4. Hemodynamic Measurements
2.5. Body Composition
2.6. Muscle Fitness and Function
2.7. Gait and Balance Analysis
2.8. Statistical Analysis
3. Results
3.1. Participant Characteristics and Baseline (wk 0) Measurements
3.2. Effects of the DASH Diet on Indicators of Body Composition
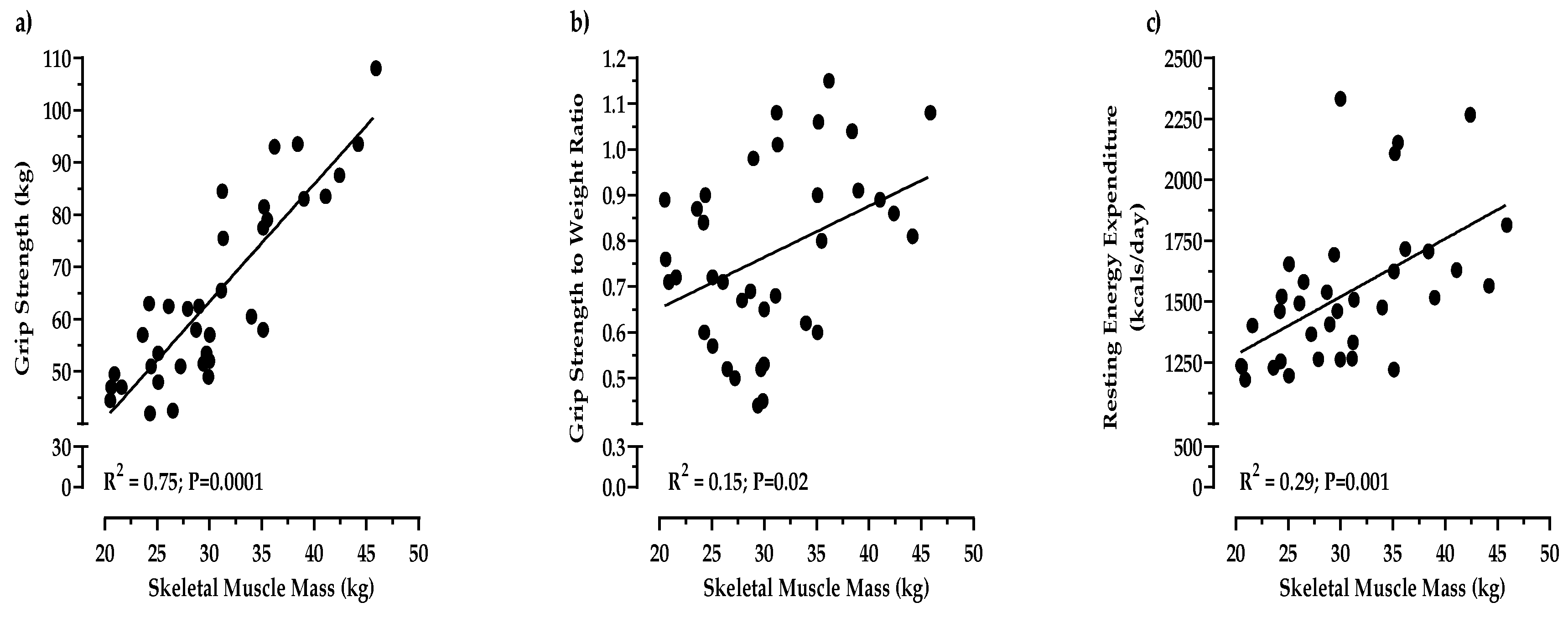
3.3. Effects of the DASH Diet on Measurements of Muscle Fitness
4. Discussion
- (1)
- Daily consumption of the study diet altered a comprehensive panel of body composition measurements in obese older adults.
- (2)
- Daily meat as a part of the DASH diet was associated with preserved handgrip strength, a significantly increased strength-to-weight ratio, and increased resting energy expenditure in adults 65 and older.
4.1. Body Composition Improved in Obese Older Adults Consuming the DASH Diet
4.2. Muscle Strength was Maintained in Obese Older Adults Consuming Meat as a Part of the DASH Diet
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AFM | Absolute Fat Mass |
| BMI | Body Mass Index |
| BP | Blood Pressure |
| DBP | Diastolic Blood Pressure |
| FFM | Fat Free Mass |
| LBM | Lean Body Mass |
| REE | Resting Energy Expenditure |
| SBP | Systolic Blood Pressure |
| SMM | Skeletal Muscle Mass |
| TBW | Total Body Water |
| WHR | Waist-to-Hip Ratio |
References
- Kim, J.; Lee, Y.; Kye, S.; Chung, Y.S.; Kim, J.H.; Chon, D.; Lee, K.E. Diet quality and osteosarcopenic obesity in community-dwelling adults 50 years and older. Maturitas 2017, 104, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Sieber, C.C. Malnutrition and Sarcopenia. Aging Exp. Res. 2019, 31, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate. Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; National Academies Press: Washington, DC, USA, 2005. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Leidy, H. Dietary protein and muscle in older persons. Curr. Opin. Clin. Nutr. Metab. Care 2014, 1, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Deer, R.R.; Volpi, E. Protein intake and muscle function in older adults. Curr. Opin. Clin. Nutr. Metab. Care 2015, 3, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.J.; Milan, A.M.; Mitchell, S.M.; Zeng, N.; Ramzan, F.; Sharma, P.; Knowles, S.; Roy, N.; Sjödin, A.; Wagner, K.; et al. The effects of dietary protein intake on appendicular lean mass and muscular function in elderly men: A 10-wk randomized controlled trial. Am. J. Clin. Nutr. 2017, 6, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Padden-Jones, D.; Campbell, W.W.; Jacques, P.F.; Kritchevsky, S.B.; Moore, L.L.; Rodriguiz, N.R.; van Loon, L.J. Protein and healthy aging. Am. J. Clin. Nutr. 2015, 6, 1339S–1345S. [Google Scholar] [CrossRef] [PubMed]
- Drehmer, M.; Odegaard, A.O.; Schmidt, M.I.; Duncan, B.B.; Cardoso, L.O.; Matos, S.M.A.; Molina, M.D.C.; Barreto, S.M.; Pereira, M.A. Brazilian dietary patterns and the dietary approaches to stop hypertension (DASH) diet-relationship with metabolic syndrome and newly diagnosed diabetes in the ELSA-Brasil study. Diabetol. Metab. Syndr. 2017, 9, 13. [Google Scholar] [CrossRef]
- Hekmatdoost, A.; Shamsipour, A.; Meibodi, M.; Gheibizadeh, N.; Eslamparast, T.; Poustchi, H. Adherence to the Dietary Approaches to Stop Hypertension (DASH) and risk of Nonalcoholic Fatty Liver Disease. Int. J. Food Sci. Nutr. 2016, 67, 1024–1029. [Google Scholar] [CrossRef]
- Pistollato, F.; Iglesias, R.C.; Ruiz, R.; Aparicio, S.; Crespo, J.; Lopez, L.D.; Manna, P.P.; Giampieri, F.; Battino, M. Nutritional patterns associated with the maintenance of neurocognitive functions and the risk of dementia and Alzheimer’s disease: A focus on human studies. Pharmacol. Res. 2018, 131, 32–43. [Google Scholar] [CrossRef]
- Solfrizzi, V.; Custodero, C.; Lozupone, M.; Imbimbo, B.P.; Valiani, V.; Agosti, P.; Schilardi, A.; D’Introno, A.; La Montagna, M.; Calvani, M.; et al. Relationships of Dietary Patterns, Foods, and Micro- and Macronutrients with Alzheimer’s Disease and Late-Life Cognitive Disorders: A Systematic Review. J. Alzheimers Dis. 2017, 59, 815–849. [Google Scholar] [CrossRef]
- Berendsen, A.A.M.; Kang, J.H.; van de Rest, O.; Feskens, E.J.M.; de Groot, L.C.P.G.M.; Grodstein, F. The Dietary Approaches to Stop Hypertension Diet, Cognitive Function, and Cognitive Decline in American Older Women. J. Am. Med. Dir. Assoc. 2017, 18, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Roussell, M.A.; Hill, A.M.; Gaugler, T.L.; West, S.G.; Heuvel, J.P.; Alaupovic, P.; Gillies, P.J.; Kris-Etherton, P.M. Beef in an Optimal Lean Diet study: Effects on lipids, lipoproteins, and apolipoproteins. Am. J. Clin. Nutr. 2012, 1, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Roussell, M.A.; Hill, A.M.; Gaugler, T.L.; West, S.G.; Ulbrecht, J.S.; Vanden Heuvel, J.P.; Gillies, P.J.; Kris-Etherton, P.M. Effects of a DASH-like diet containing lean beef on vascular health. J. Hum. Hypertens. 2014, 10, 600–605. [Google Scholar] [CrossRef] [PubMed]
- 2015–2020 Dietary Guidelines: Appendix 2. Estimated Calorie Needs Per Day, by Age, Sex, and Physical Activity Level. Available online: https://health.gov/dietaryguidelines/2015/guidelines/appendix-2/ (accessed on 7 March 2017).
- National Heart, Lung, and Blood Institute, DASH Eating Plan. Available online: https://www.nhlbi.nih.gov/health-topics/dash-eating-plan (accessed on 7 March 2017).
- Kenny, R.A.; Rubenstein, L.Z.; Tinetti, M.E.; Brewer, K.; Cameron, K.A.; Capezuti, E.A.; John, D.P.; Lamb, S.; Martin, F.; Rockey, P.H.; et al. Summary of the Updated American Geriatrics Society/British Geriatrics Society Clinical Practice Guideline for Prevention of Falls in Older Persons. J. Am. Geriatr. Soc. 2011, 59, 148–157. [Google Scholar] [CrossRef]
- Stevens, J.A.; Phelan, E.A. Development of STEADI: A Fall Prevention Resource for Health Care Providers. Health Promot. Pract. 2013, 14, 706–714. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Lippincott Williams and Wilkins: Baltimore, MD, USA, 2017. [Google Scholar]
- Robergs, R.A.; Dwyer, A.D.; Astorino, T. Recommendations for Improved Data Processing from Expired Gas Analysis Indirect Calorimetry. Sports Med. 2010, 40, 95–111. [Google Scholar] [CrossRef]
- Jones, C.J.; Rikli, R.E.; Beam, W.C. A 30-s Chair-stand Test as a Measure of Lower Body Strength in Community-residing Older Adults. Res. Q. Exerc. Sport. 1999, 70, 113–119. [Google Scholar] [CrossRef]
- Tinetti, M.E. Performance-oriented Assessment of Mobility Problems in Elderly Patients. J. Am. Geriatr. Soc. 1986, 34, 119–126. [Google Scholar] [CrossRef]
- Hernandez, D.; Rose, D.J. Predicting Which Older Adults Will or Will not Fall Using the Fullerton Advanced Balance scale. Arch. Phys. Med. Rehabil. 2008, 89, 2309–2315. [Google Scholar] [CrossRef]
- Klein, P.J.; Fiedler, R.C.; Rose, D.J. Rasch Analysis of the Fullerton Advanced Balance (FAB) Scale. Physiother. Can. 2011, 63, 115–125. [Google Scholar] [CrossRef]
- Protas, E.J.; Harri, C.; Moch, C.; Rusk, M. Sensitivity of a Clinical Scale of Balance and Gait in Frail Nursing Home Residents. Disabil. Rehabil. 2000, 22, 372–378. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention. Losing Weight. Available online: https://www.cdc.gov/healthyweight/losing_weight/index.html (accessed on 17 July 2019).
- Blackburn, G. Effect of degree of weight loss on health benefits. Obes. Res. 1995, 3, 211S–216S. [Google Scholar] [CrossRef] [PubMed]
- JafariNasabian, P.; Inglis, J.E.; Wendimere, R.; Kelly, O.J.; Ilich, J.Z. Aging human body: Changes in bone, muscle and body fat with consequent changes in nutrient intake. J. Endocrinol. 2017, 234, R37–R51. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Physical Status: The Use and Interpretation of Anthropometry: Report of a WHO Expert Committee; WHO Technical Report Series 854: Geneva, Switzerland, 1995; Available online: http://whqlibdoc.who.int/trs/WHO_TRS_854.pdf (accessed on 14 November 2019).
- Liu, P.-Y.; Ilich, J.Z.; Brummel-Smith, K.; Ghosh, S. New insight into fat, muscle and bone relationship in women: Determining the threshold at which body fat assumes negative relationship with bone mineral density. Int. J. Prev. Med. 2014, 5, 1452–1463. [Google Scholar] [PubMed]
- Klein, S.; Allison, D.B.; Heymsfield, S.B.; Kelley, D.E.; Leibel, R.L.; Nonas, C. Waist Circumference and Cardiometabolic Risk. Diabetes Care 2007, 30, 1647–1652. [Google Scholar] [CrossRef]
- Borruel, S.; Moltó, J.F.; Alpañes, M.; Fernández-Durán, E.; Álvarez-Blasco, F.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Surrogate Markers of Visceral Adiposity in Young Adults: Waist Circumference and Body Mass Index Are More Accurate than Waist Hip Ratio, Model of Adipose Distribution and Visceral Adiposity Index. PLoS ONE 2014, 9, e114112. [Google Scholar] [CrossRef]
- Ormsbee, M.J.; Prado, C.M.; Ilich, J.Z.; Purcell, S.; Siervo, M.; Folsom, A.; Panton, L. Osteosarcopenic obesity: The role of bone, muscle and fat in health. J. Cachexia Sarcopenia Muscle 2014, 3, 183–192. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Adult Obesity Facts. Available online: https://www.cdc.gov/obesity/data/adult.html (accessed on 17 July 2019).
- Chronic Disease Management. National Council on Aging. Available online: https://www.ncoa.org/healthy-aging/chronic-disease/ (accessed on 17 July 2019).
- Cawley, J.; Meyerhoefer, C. The medical costs of obesity: An instrumental variables approach. J. Health Econ. 2012, 31, 219–230. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Ibrahim, K.; May, C.; Patel, H.P.; Baxter, M.; Sayer, A.A.; Roberts, H. A feasibility study of implementing grip strength measurement into routine hospital practice (GRImP): Study protocol. Pilot Feasibility Stud. 2016, 2, 27. [Google Scholar] [CrossRef]
- Leong, D.P.; Teo, K.K.; Rangarajan, S.; Lopez-Jaramillo, P.; Avezum, A., Jr.; Orlandini, A.; Seron, P.; Ahmed, S.H.; Rosengren, A.; Kelishadi, R.; et al. Prognostic value of grip strength: Findings from the Prospective Urban Rural Epidemiology (PURE) study. Lancet 2015, 386, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Abellan van Kan, G.; Rolland, Y.; Andrieu, S.; Bauer, J.; Beauchet, O.; Bonnefoy, M.; Cesari, M.; Donini, L.M.; Gillette-Guyonnet, S.; Inzitari, M.; et al. Gait speed at usual pace as a predictor of adverse outcomes in community—dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J. Nutr. Health Aging 2009, 13, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Peel, N.M.; Kuys, S.S.; Klein, K. Gait speed as a measure in geriatric assessment in clinical settings: A systematic review. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Studenski, S.; Perera, S.; Patel, K.; Rosano, C.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Connor, E.B.; et al. Gait speed and survival in older adults. JAMA 2011, 305, 50–58. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Ferrucci, L.; Pieper, C.F.; Leveille, S.G.; Markides, K.S.; Ostir, G.V.; Studenski, S.; Berkman, L.F.; Wallace, R.B. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef]
| Food Group | DASH Recommended Servings | Study Diet |
|---|---|---|
| Grains | 6 | 7 |
| Vegetables | 4–5 | 5 |
| Fruits | 4–5 | 4 |
| Dairy | 2–3 | 3 |
| Lean Meat | 6 or less | 4.5 |
| Legumes | 4 per week | 4 per week |
| Fats/Oil | 2–3 | 3 |
| Sweets | 5 or less | 0 |
| Sodium | 2300 mg/day | 1895 mg/day |
| Calories | 1800 | 1800 cal/day |
| Macronutrients | Carbohydrates: 45%–65% | 59% |
| Fat: 20%–30% | 21% | |
| Protein: 15%–25% | 20% | |
| Saturated Fat | <6%–10% | 8% |
| Variables | Total (n = 36) | Male (n = 15) | Female (n = 21) | p Value |
|---|---|---|---|---|
| Age (year) | 70.7 | 71.5 | 70.1 | 0.473 |
| Height (cm) | 169.1 | 177.3 | 163.2 | <0.0001 |
| Weight (kg) | 91.2 | 98.7 | 85.9 | 0.033 |
| BMI (kg/m2) | 32.0 | 31.4 | 32.5 | 0.646 |
| Waist (cm) | 100.7 | 108.1 | 95.4 | 0.020 |
| Hip (cm) | 115.3 | 111.0 | 118.3 | 0.125 |
| WHR | 0.874 | 0.974 | 0.803 | <0.0001 |
| Body Fat (%) | 37.2 | 30.7 | 41.8 | <0.0001 |
| AFM (kg) | 34.5 | 30.5 | 37.3 | 0.116 |
| FFM (kg) | 56.7 | 68.1 | 48.6 | <0.0001 |
| LBM (kg) | 64.0 | 71.1 | 58.9 | <0.0001 |
| SMM (kg) | 31.4 | 38.0 | 26.6 | <0.0001 |
| TBW (kg) | 40.7 | 49.7 | 34.2 | <0.0001 |
| REE (kcal/day) | 1600.3 | 1785.9 | 1442.1 | 0.002 |
| REE (kcal/day/kg) | 17.8 | 18.2 | 17.5 | 0.504 |
| SBP (mmHg) | 132.9 | 131.2 | 134.1 | 0.671 |
| DBP (mmHg) | 76.3 | 79.9 | 73.7 | 0.056 |
| Handgrip (kg) | 62.6 | 78.9 | 51.1 | <0.0001 |
| Sit/Stand (reps) | 11.4 | 11.9 | 11.1 | 0.364 |
| Gait score | 11.6 | 11.8 | 11.5 | 0.167 |
| Balance score | 14.4 | 14.1 | 14.7 | 0.365 |
| Weeks of Intervention | ||||||
|---|---|---|---|---|---|---|
| Variable | 0 | 3 | 6 | 9 | 12 | p-Value + |
| Weight (kg) | ||||||
| All Participants | * 91.2 (18.0) | 88.6 (17.3) | 87.3 (16.9) | 86.4 (16.6) | * 85.5 (16.3) | <0.001 |
| Females | * 85.9 (19.9) | 84.0 (19.3) | 82.9 (18.9) | 82.0 (18.4) | * 81.3 (18.2) | <0.001 |
| Males | * 98.7 (11.8) | 95.0 (11.9) | 93.4 (11.6) | 92.5 (11.5) | * 91.5 (11.1) | <0.001 |
| BMI (kg/m2) | ||||||
| All Participants | * 32.0 (6.9) | 31.2 (6.7) | 30.7 (6.6) | 30.4 (6.5) | * 30.1 (6.4) | <0.001 |
| Females | * 32.5 (8.5) | 31.8 (8.3) | 31.4 (8.2) | 31.0 (8.0) | * 30.8 (7.9) | <0.001 |
| Males | * 31.4 (3.6) | 30.3 (3.6) | 29.7 (3.5) | 29.4 (3.5) | * 29.1 (3.3) | <0.001 |
| Waist (cm) | ||||||
| All Participants | * 101 (16.4) | 99.1 (15.7) | 97.9 (15.9) | 97.0 (15.5) | * 96.3 (15.1) | <0.001 |
| Females | * 95.4 (18.4) | 94.3 (17.8) | 93.2 (18.0) | 92.3 (17.7) | * 92.1 (17.4) | <0.001 |
| Males | * 108.1 (9.2) | 105.8 (8.7) | 104.5 (9.2) | 103.7 (8.7) | * 102.2 (8.6) | <0.001 |
| Hip (cm) | ||||||
| All Participants | * 115 (14.0) | 114 (14.1) | 113 (14.3) | 112 (13.2) | * 111 (14.1) | <0.001 |
| Females | * 118 (16.7) | 117 (16.6) | 116 (17.0) | 114 (15.6) | * 114 (16.6) | <0.001 |
| Males | * 111.0 (7.9) | 109.7 (8.2) | 109.0 (8.2) | 108.9 (8.2) | * 107.3 (8.3) | <0.05 |
| WHR | ||||||
| All Participants | 0.87 (0.10) | 0.87 (0.10) | 0.87 (0.10) | 0.86 (0.09) | 0.86 (0.09) | 0.06 |
| Females | 0.80 (0.06) | 0.80 (0.06) | 0.80 (0.06) | 0.80 (0.06) | 0.80 (0.06) | >0.05 |
| Males | * 0.97 (0.05) | 0.96 (0.05) | 0.96 (0.05) | 0.95 (.05) | * 0.95 (0.05) | <0.05 |
| Body Fat (%) | ||||||
| All Participants | * 37.2 (9.8) | 36.9 (10.1) | 35.7 (10.1) | 35.3 (10.4) | * 34.7 (10.3) | <0.001 |
| Females | * 41.8 (9.5) | 41.7 (9.7) | 40.4 (9.8) | 40.3 (9.9) | * 39.8 (9.6) | <0.001 |
| Males | * 30.7 (5.8) | 30.2 (6.1) | 29.2 (6.2) | 28.2 (6.5) | * 27.6 (6.4) | <0.001 |
| AFM (kg) | ||||||
| All Participants | * 34.5 (12.7) | 33.4 (12.9) | 31.9 (12.6) | 31.1 (12.7) | * 30.3 (12.4) | <0.001 |
| Females | * 37.3 (14.8) | 36.4 (14.8) | 34.9 (14.6) | 34.4 (14.4) | * 33.7 (14.1) | <0.001 |
| Males | * 30.5 (7.9) | 29.1 (8.3) | 27.5 (7.8) | 26.4 (8.0) | * 25.5 (7.7) | <0.001 |
| FFM (kg) | ||||||
| All Participants | 56.7 (12.2) | 55.3 (11.3) | 55.4 (11.2) | 55.3 (11.4) | 55.2 (11.3) | <0.05 |
| Females | 48.6 (7.0) | 47.6 (6.3) | 48.0 (6.1) | 47.6 (5.9) | 47.5 (5.8) | <0.05 |
| Males | 68.1 (8.0) | 65.9 (7.1) | 65.9 (7.7) | 66.1 (7.9) | 66.0 (7.8) | <0.05 |
| LBM (kg) | ||||||
| All Participants | * 64.0 (10.5) | 62.3 (9.6) | 62.4 (9.5) | 62.2 (9.7) | * 62.0 (9.6) | <0.001 |
| Females | * 58.9 (9.0) | 57.7 (8.2) | 57.9 (8.0) | 57.4 (7.8) | * 57.3 (7.6) | <0.001 |
| Males | 71.1 (8.2) | 68.8 (7.4) | 68.7 (8.0) | 68.9 (8.2) | 68.7 (8.0) | <0.001 |
| TBW (kg) | ||||||
| All Participants | 41.5 (8.8) | 40.5 (8.3) | 40.7 (8.2) | 40.6 (8.4) | 40.5 (8.3) | <0.001 |
| Females | 35.7 (5.2) | 34.9 (4.6) | 35.2 (4.5) | 34.9 (4.4) | 34.9 (4.3) | 0.007 |
| Males | 49.7 (5.9) | 48.4 (5.2) | 48.3 (5.6) | 48.5 (5.8) | 48.3 (5.7) | 0.003 |
| Weeks of Intervention | ||||||
|---|---|---|---|---|---|---|
| Variable | 0 | 3 | 6 | 9 | 12 | +p-Value |
| Skeletal muscle (kg) | ||||||
| All Participants | * 31.4 (7.2) | 30.7 (6.7) | 30.8 (6.6) | 30.7 (6.8) | * 30.6 (6.8) | <0.001 |
| Females | 26.6 (4.1) | 26.1 (3.6) | 26.4 (3.5) | 26.0 (3.4) | 26.0 (3.4) | <0.05 |
| Males | 38.0 (4.9) | 37.1 (4.3) | 37.0 (4.6) | 37.2 (4.8) | 37.0 (4.8) | <0.05 |
| Handgrip (kg) | ||||||
| All Participants | 62.6 (19.4) | 62.8 (17.1) | 64.1 (18.0) | 62.9 (17.3) | 64.7 (17.5) | >0.05 |
| Females | 51.1 (9.7) | 51.8 (7.7) | 52.5 (8.6) | 51.6 (9.0) | 53.0 (7.1) | 0.058 |
| Males | 78.8 (18.0) | 78.3 (14.6) | 80.4 (14.8) | 78.8 (13.1) | 81.0 (14.2) | >0.05 |
| Handgrip (per kg mass) | ||||||
| All Participants | * 0.70 (0.21) | 0.73 (0.20) | 0.75 (0.20) | 0.74 (0.19) | * 0.77 (0.19) | <0.0001 |
| Females | * 0.62 (0.18) | 0.65 (0.17) | 0.66 (0.17) | 0.66 (0.17 | * 0.68 (0.16) | <0.0001 |
| Males | * 0.81 (0.21) | 0.84 (0.19) | 0.87 (0.19) | 0.86 (0.17) | * 0.90 (0.17) | <0.0001 |
| Sit/Stand (reps) | ||||||
| All Participants | * 11.4 (2.3) | 11.9 (2.4) | 12.9 (2.5) | 13.1 (2.9) | * 13.8 (2.5) | <0.001 |
| Females | * 11.1 (2.1) | 11.6 (2.3) | 12.5 (2.3) | 12.8 (2.8) | * 13.4 (2.1) | <0.001 |
| Males | * 11.9 (2.6) | 12.4 (2.4) | 13.4 (2.8) | 13.5 (3.0) | * 14.3 (3.0) | <0.001 |
| Gait score | ||||||
| All Participants | 11.6 (0.7) | 11.6 (0.7) | 11.8 (0.5) | 11.9 (0.4) | 11.8 (0.5) | >0.05 |
| Females | 11.5 (0.8) | 11.5 (0.9) | 11.8 (0.5) | 11.9 (0.3) | 11.9 (0.5) | >0.05 |
| Males | 11.8 (0.4) | 11.8 (0.4) | 11.9 (0.4) | 11.8 (0.6) | 11.8 (0.6) | >0.05 |
| Balance score | ||||||
| All Participants | 14.4 (1.7) | 15.0 (1.3) | 15.3 (0.8) | 15.1 (1.1) | 15.1 (1.1) | >0.05 |
| Females | 14.7 (1.4) | 15.2 (0.9) | 15.3 (0.9) | 15.1 (1.1) | 15.0 (1.2) | >0.05 |
| Males | 14.1 (2.1) | 14.7 (1.6) | 15.3 (0.8) | 15.1 (1.1) | 15.3 (1.0) | >0.05 |
| REE (kcal/day) | ||||||
| All Participants | 1600 (321) | 1597 (277) | 1579 (280) | 1604 (273) | 1534 (302) | >0.05 |
| Females | 1442 (273) | 1469 (218) | 1446 (229) | 1478 (178) | 1386 (159) | >0.05 |
| Males | 1786 (271) | 1768 (260) | 1756 (247) | 1772 (293) | 1732 (338) | >0.05 |
| REE (kcal/day/kg) | ||||||
| All Participants | 17.8 (3.1) | 18.6 (3.7) | 18.6 (3.5) | 19.2 (3.8) | 18.4 (3.9) | >0.05 |
| Females | 17.5 (3.0) | 18.4 (4.0) | 18.4 (3.8) | 18.9 (3.4) | 18.0 (3.9) | >0.05 |
| Males | 18.2 (3.5) | 18.9 (3.3) | 19.0 (3.2) | 19.5 (4.5) | 19.1 (4.1) | >0.05 |
| REE (kcal/day/AFM) | ||||||
| All Participants | * 53.8 (23.1) | 57.4 (27.1) | 60.0 (30.2) | 63.5 (34.3) | * 61.8 (31.3) | <0.0001 |
| Females | * 47.2 (23.4) | 51.3 (29.4) | 53.1 (31.7) | 55.2 (33.2) | * 52.7 (31.2) | <0.01 |
| Males | * 62.5 (20.3) | 65.5 (22.1) | 69.2 (26.3) | 74.7 (33.5) | * 74.0 (27.9) | <0.01 |
| Systolic BP (mmHg) | ||||||
| All Participants | * 133 (20) | 125 (16.1) | 120 (15.4) | 119 (16.1) | * 120 (16.8) | <0.001 |
| Females | * 134 (16.5) | 127 (16.1) | 117 (14.0) | 119 (12.6) | * 122 (14.0) | <0.001 |
| Males | 131 (24.8) | 121 (17.8) | 122 (17.3) | 118 (20.6) | 117 (20.2) | <0.05 |
| Diastolic BP (mmHg) | ||||||
| All Participants | * 76.3 (9.7) | 72.6 (8.8) | 69.0 (8.9) | 70.8 (8.1) | * 70.5 (10.9) | <0.001 |
| Females | 73.7 (6.6) | 70.5 (7.9) | 67.3 (8.2) | 68.9 (7.9) | 70.1 (8.8) | <0.05 |
| Males | * 80 (12.2) | 75.7 (9.4) | 71.3 (9.6) | 73.4 (7.9) | * 71.1 (13.6) | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perry, C.A.; Van Guilder, G.P.; Kauffman, A.; Hossain, M. A Calorie-Restricted DASH Diet Reduces Body Fat and Maintains Muscle Strength in Obese Older Adults. Nutrients 2020, 12, 102. https://doi.org/10.3390/nu12010102
Perry CA, Van Guilder GP, Kauffman A, Hossain M. A Calorie-Restricted DASH Diet Reduces Body Fat and Maintains Muscle Strength in Obese Older Adults. Nutrients. 2020; 12(1):102. https://doi.org/10.3390/nu12010102
Chicago/Turabian StylePerry, Cydne A., Gary P. Van Guilder, Alyssa Kauffman, and Mosharraf Hossain. 2020. "A Calorie-Restricted DASH Diet Reduces Body Fat and Maintains Muscle Strength in Obese Older Adults" Nutrients 12, no. 1: 102. https://doi.org/10.3390/nu12010102
APA StylePerry, C. A., Van Guilder, G. P., Kauffman, A., & Hossain, M. (2020). A Calorie-Restricted DASH Diet Reduces Body Fat and Maintains Muscle Strength in Obese Older Adults. Nutrients, 12(1), 102. https://doi.org/10.3390/nu12010102





