Dietary Supplementation with Hazelnut Oil Reduces Serum Hyperlipidemia and Ameliorates the Progression of Nonalcoholic Fatty Liver Disease in Hamsters Fed a High-Cholesterol Diet
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Analytical Determination of Supplemented Dietary Fat
2.3. Experimental Design
2.4. Sample Collection and Preparation
2.5. Determination of Serum Lipid and Lipoprotein Cholesterol
2.6. Hepatic and Fecal Lipid Analysis
2.7. Liver Antioxidant and Paroxidant Analysis
2.8. Histopathological Analysis
2.9. Statistical Analysis
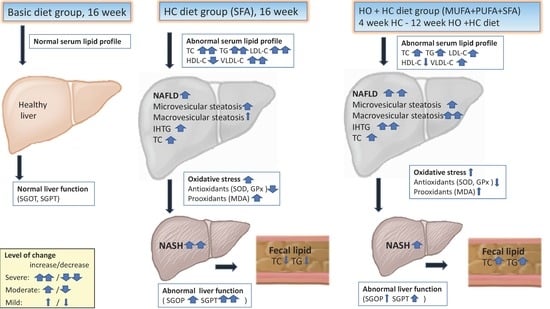
3. Results
3.1. Food Ingestion and Weight Gain
3.2. Serum Lipid Profile
3.3. Liver Function
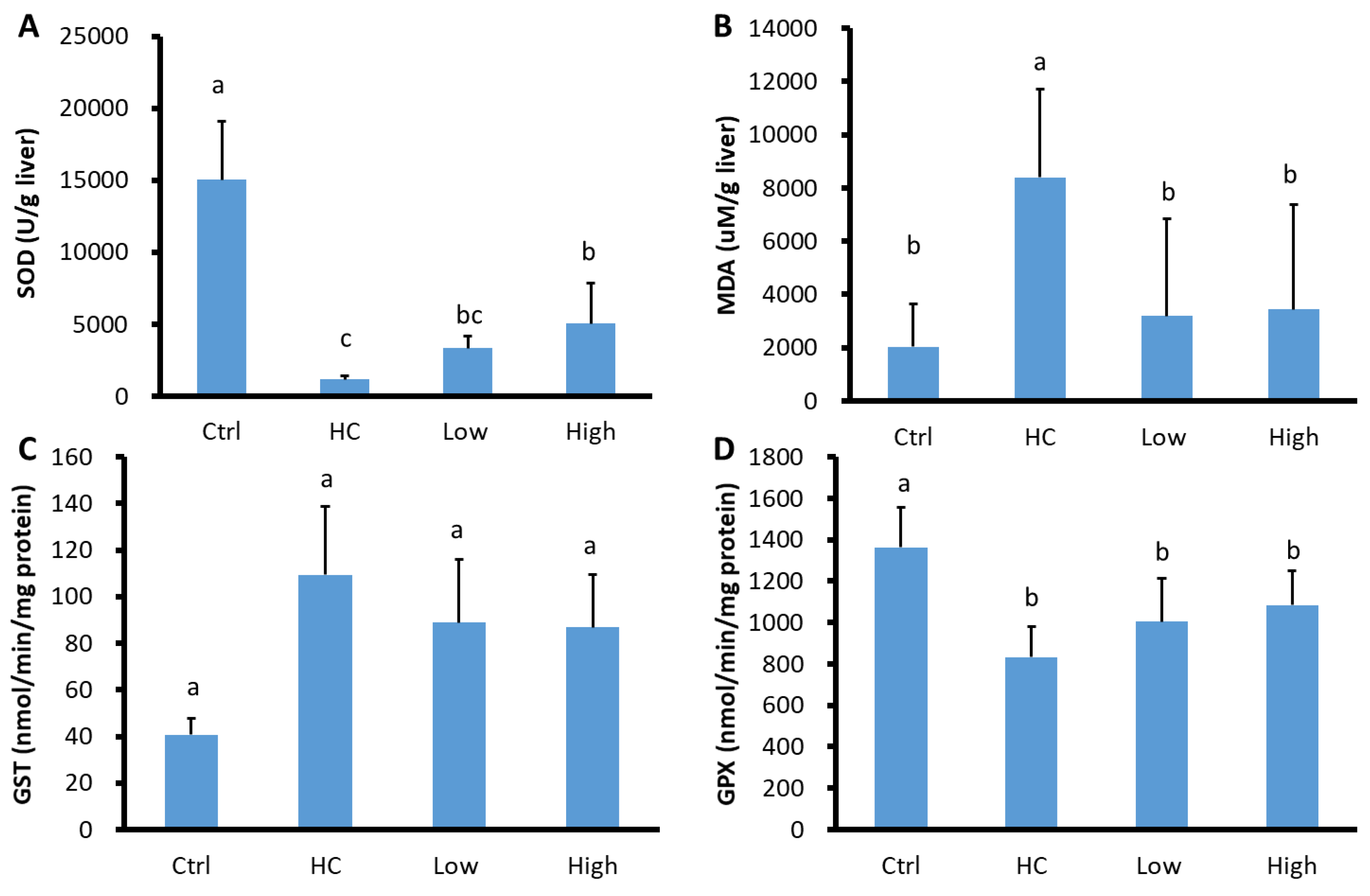
3.4. Hepatic Antioxidant and Prooxidant Analysis
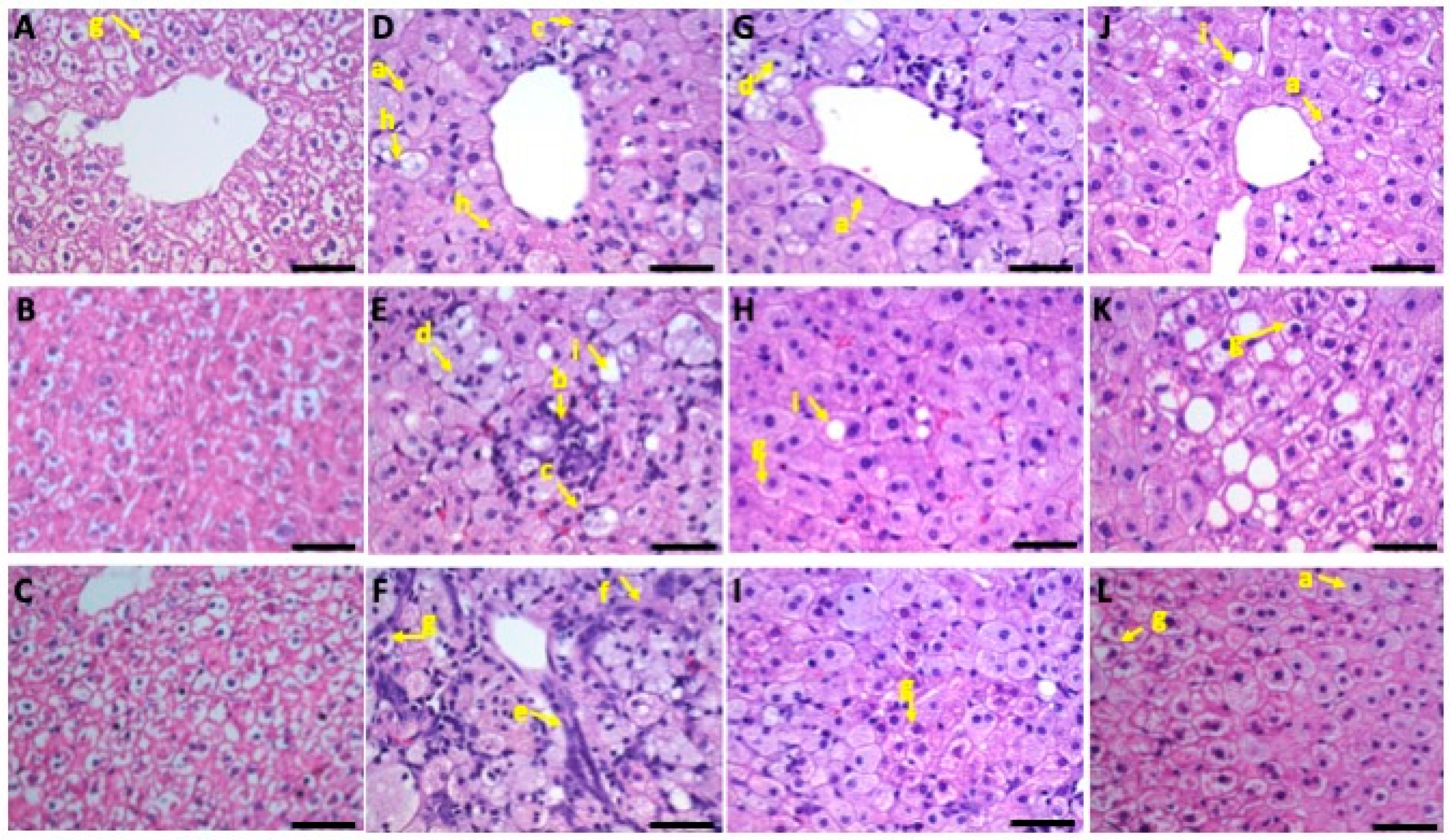
3.5. Liver Histopathology
3.5.1. Microvesicular Steatosis
3.5.2. Macrovesicular Steatosis
3.5.3. Inflammatory Changes
3.5.4. Fibrotic Changes
3.5.5. Glycogen Accumulation
3.6. Lipid Contents in Liver Parenchymal and Fecal Material
4. Discussion
4.1. Serum Lipid Profile in the HC + HO Groups
4.2. Hepatic Steatosis in the HC + HO Groups
4.3. Steatohepatitis in the HC + HO Groups
5. Conclusions
6. Limitation and Future Works
Author Contributions
Funding
Conflicts of Interest
References
- Siri-Tarino, P.W.; Sun, Q.; Hu, F.B.; Krauss, R.M. Saturated fatty acids and risk of coronary heart disease: Modulation by replacement nutrients. Curr. Atheroscler. Rep. 2010, 12, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Seppanen, C.M.; Cho, H.; Csallany, A.S. Comparison between High-PUFA and Low-PUFA Fats on Lipid Peroxidation and LDL Oxidation. Food Nutr. Sci. 2013, 4, 572–579. [Google Scholar] [CrossRef]
- Serviddio, G.; Bellanti, F.; Villani, R.; Tamborra, R.; Zerbinati, C.; Blonda, M.; Ciacciarelli, M.; Poli, G.; Vendemiale, G.; Iuliano, L. Effects of dietary fatty acids and cholesterol excess on liver injury: A lipidomic approach. Redox Biol. 2016, 9, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Barona, J.; Fernandez, M.L. Dietary cholesterol affects plasma lipid levels, the intravascular processing of lipoproteins and reverse cholesterol transport without increasing the risk for heart disease. Nutrients 2012, 4, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Ooi, E.M.; Watts, G.F.; Ng, T.W.; Barrett, P.H. Effect of dietary Fatty acids on human lipoprotein metabolism: A comprehensive update. Nutrients 2015, 7, 4416–4425. [Google Scholar] [CrossRef]
- Mawatari, S.; Ohnishi, Y.; Kaji, Y.; Maruyama, T.; Murakami, K.; Tsutsui, K.; Fujino, T. High-cholesterol diets induce changes in lipid composition of rat erythrocyte membrane including decrease in cholesterol, increase in alpha-tocopherol and changes in fatty acids of phospholipids. Biosci. Biotechnol. Biochem. 2003, 67, 1457–1464. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lawrence, G.D. Dietary fats and health: Dietary recommendations in the context of scientific evidence. Adv. Nutr. (Bethesda, MD) 2013, 4, 294–302. [Google Scholar] [CrossRef]
- Ishiyama, J.; Taguchi, R.; Akasaka, Y.; Shibata, S.; Ito, M.; Nagasawa, M.; Murakami, K. Unsaturated FAs prevent palmitate-induced LOX-1 induction via inhibition of ER stress in macrophages. J. Lipid Res. 2011, 52, 299–307. [Google Scholar] [CrossRef]
- Siri-Tarino, P.W.; Chiu, S.; Bergeron, N.; Krauss, R.M. Saturated Fats Versus Polyunsaturated Fats Versus Carbohydrates for Cardiovascular Disease Prevention and Treatment. Annu. Rev. Nutr. 2015, 35, 517–543. [Google Scholar] [CrossRef]
- Hooper, L.; Martin, N.; Abdelhamid, A.; Smith, G.D. Reduction in saturated fat intake for cardiovascular disease. Cochrane Database Syst. Rev. 2015, 6, CD011737. [Google Scholar] [CrossRef]
- Thomas, J.; Thomas, C.J.; Radcliffe, J.; Itsiopoulos, C. Omega-3 Fatty Acids in Early Prevention of Inflammatory Neurodegenerative Disease: A Focus on Alzheimer’s Disease. BioMed Res. Int. 2015, 2015, 172801. [Google Scholar] [CrossRef] [PubMed]
- Varela-Lopez, A.; Quiles, J.L.; Cordero, M.; Giampieri, F.; Bullon, P. Oxidative Stress and Dietary Fat Type in Relation to Periodontal Disease. Antioxidants (Basel, Switzerland) 2015, 4, 322–344. [Google Scholar] [CrossRef] [PubMed]
- Yucesan, F.B.; Orem, A.; Kural, B.V.; Orem, C.; Turan, I. Hazelnut consumption decreases the susceptibility of LDL to oxidation, plasma oxidized LDL level and increases the ratio of large/small LDL in normolipidemic healthy subjects. Anadolu Kardiyol. Derg. 2010, 10, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Lima, R.P.A.; do Nascimento, R.A.F.; Luna, R.C.P.; Persuhn, D.C.; da Silva, A.S.; Gonçalves, M.D.R.; de Almeida, A.T.C.; de Moraes, R.M.; Junior, E.V.; Fouilloux-Meugnier, E.; et al. Effect of a diet containing folate and hazelnut oil capsule on the methylation level of the ADRB3 gene, lipid profile and oxidative stress in overweight or obese women. Clin. Epigenet. 2017, 9, 110. [Google Scholar] [CrossRef] [PubMed]
- Hatipoglu, A.; Kanbagli, O.; Balkan, J.; Kucuk, M.; Cevikbas, U.; Aykac-Toker, G.; Berkkan, H.; Uysal, M. Hazelnut oil administration reduces aortic cholesterol accumulation and lipid peroxides in the plasma, liver, and aorta of rabbits fed a high-cholesterol diet. Biosci. Biotechnol. Biochem. 2004, 68, 2050–2057. [Google Scholar] [CrossRef] [PubMed]
- Perna, S.; Giacosa, A.; Bonitta, G.; Bologna, C.; Isu, A.; Guido, D.; Rondanelli, M. Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis. Nutrients 2016, 8, 747. [Google Scholar] [CrossRef] [PubMed]
- Mercanligil, S.M.; Arslan, P.; Alasalvar, C.; Okut, E.; Akgul, E.; Pinar, A.; Geyik, P.O.; Tokgozoglu, L.; Shahidi, F. Effects of hazelnut-enriched diet on plasma cholesterol and lipoprotein profiles in hypercholesterolemic adult men. Eur. J. Clin. Nutr. 2007, 61, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.C.; Chisholm, A.W.; Delahunty, C.M.; Gray, A.R.; Williams, S.M. Effects of different forms of hazelnuts on blood lipids and alpha-tocopherol concentrations in mildly hypercholesterolemic individuals. Eur. J. Clin. Nutr. 2011, 65, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Skeaff, C.M.; Chisholm, W.A. The effect of replacing dietary saturated fat with polyunsaturated or monounsaturated fat on plasma lipids in free-living young adults. Eur. J. Clin. Nutr. 2001, 55, 908–915. [Google Scholar] [CrossRef]
- Poorghasemi, M.; Seidavi, A.; Qotbi, A.A.; Laudadio, V.; Tufarelli, V. Influence of dietary fat source on growth performance responses and carcass traits of broiler chicks. Asian Aust. J. Anim. Sci. 2013, 26, 705–710. [Google Scholar] [CrossRef]
- Yanai, H.; Katsuyama, H.; Hamasaki, H.; Abe, S.; Tada, N.; Sako, A. Effects of Dietary Fat Intake on HDL Metabolism. J. Clin. Med. Res. 2015, 7, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Tufarelli, V.; Bozzo, G.; Perillo, A.; Laudadio, V. Effects of feeding different lipid sources on hepatic histopathology features and growth traits of broiler chickens. Acta Histochem. 2015, 117, 780–783. [Google Scholar] [CrossRef] [PubMed]
- Issaoui, M.; Dabbou, S.; Mechri, B.; Nakbi, A.; Chehab, H.; Hammami, M. Fatty acid profile, sugar composition, and antioxidant compounds of table olives as affected by different treatments. Eur. Food Res. Technol. 2011, 232, 867–876. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Shackelford, C.; Long, G.; Wolf, J.; Okerberg, C.; Herbert, R. Qualitative and quantitative analysis of nonneoplastic lesions in toxicology studies. Toxicol. Pathol. 2002, 30, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Natoli, S.; McCoy, P. A review of the evidence: Nuts and body weight. Asia Pac. J. Clin. Nutr. 2007, 16, 588–597. [Google Scholar]
- Sabate, J. Nut consumption and body weight. Am. J. Clin. Nutr. 2003, 78, 647S–650S. [Google Scholar] [CrossRef]
- Piscopo, S. The Mediterranean diet as a nutrition education, health promotion and disease prevention tool. Public Health Nutr. 2009, 12, 1648–1655. [Google Scholar] [CrossRef]
- Mukuddem-Petersen, J.; Oosthuizen, W.; Jerling, J.C. A systematic review of the effects of nuts on blood lipid profiles in humans. J. Nutr. 2005, 135, 2082–2089. [Google Scholar] [CrossRef]
- Hansen, K.S.; Ballmer-Weber, B.K.; Sastre, J.; Lidholm, J.; Andersson, K.; Oberhofer, H.; Lluch-Bernal, M.; Ostling, J.; Mattsson, L.; Schocker, F.; et al. Component-resolved in vitro diagnosis of hazelnut allergy in Europe. J. Allergy Clin. Immunol. 2009, 123, 1134–1141.e3. [Google Scholar] [CrossRef]
- Laudadio, V.; Ceci, E.; Lastella, N.M.B.; Tufarelli, V. Dietary high-polyphenols extra-virgin olive oil is effective in reducing cholesterol content in eggs. Lipids Health Dis. 2015, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Tufarelli, V.; Laudadio, V.; Casalino, E. An extra-virgin olive oil rich in polyphenolic compounds has antioxidant effects in meat-type broiler chickens. Environ. Sci. Pollut. Res. Int. 2016, 23, 6197–6204. [Google Scholar] [CrossRef] [PubMed]
- Daumerie, C.M.; Woollett, L.A.; Dietschy, J.M. Fatty acids regulate hepatic low density lipoprotein receptor activity through redistribution of intracellular cholesterol pools. Proc. Natl. Acad. Sci. USA 1992, 89, 10797–10801. [Google Scholar] [CrossRef] [PubMed]
- Spady, D.K.; Woollett, L.A.; Dietschy, J.M. Regulation of plasma LDL-cholesterol levels by dietary cholesterol and fatty acids. Annu. Rev. Nutr. 1993, 13, 355–381. [Google Scholar] [CrossRef] [PubMed]
- Gorinstein, S.; Zachwieja, Z.; Katrich, E.; Pawelzik, E.; Haruenkit, R.; Trakhtenberg, S.; Martín-Belloso, O. Comparison of the contents of the main antioxidant compounds and the antioxidant activity of white grapefruit and his new hybrid. LWT Food Sci. Technol. 2004, 37, 337–343. [Google Scholar] [CrossRef]
- Kawano, Y.; Cohen, D.E. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J. Gastroenterol. 2013, 48, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Gray, A.R.; Chisholm, A.W.; Delahunty, C.M.; Brown, R.C. The dose of hazelnuts influences acceptance and diet quality but not inflammatory markers and body composition in overweight and obese individuals. J. Nutr. 2013, 143, 1254–1262. [Google Scholar] [CrossRef]
- Damavandi, R.D.; Eghtesadi, S.; Shidfar, F.; Heydari, I.; Foroushani, A.R. Effects of hazelnuts consumption on fasting blood sugar and lipoproteins in patients with type 2 diabetes. J. Res. Med. Sci. 2013, 18, 314–321. [Google Scholar]
- Conlon, B.A.; Beasley, J.M.; Aebersold, K.; Jhangiani, S.S.; Wylie-Rosett, J. Nutritional management of insulin resistance in nonalcoholic fatty liver disease (NAFLD). Nutrients 2013, 5, 4093–4114. [Google Scholar] [CrossRef]
- Tandra, S.; Yeh, M.M.; Brunt, E.M.; Vuppalanchi, R.; Cummings, O.W.; Unalp-Arida, A.; Wilson, L.A.; Chalasani, N. Presence and significance of microvesicular steatosis in nonalcoholic fatty liver disease. J. Hepatol. 2011, 55, 654–659. [Google Scholar] [CrossRef]
- Duavy, S.M.P.; Salazar, G.J.T.; Leite, G.O.; Ecker, A.; Barbosa, N.V. Effect of dietary supplementation with olive and sunflower oils on lipid profile and liver histology in rats fed high cholesterol diet. Asian Pac. J. Trop. Med. 2017, 10, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Savy, V.; Zara, V. Olive oil increases the hepatic triacylglycerol content in mice by a distinct influence on the synthesis and oxidation of fatty acids. Biosci. Biotechnol. Biochem. 2008, 72, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Gutierrez, V.; Perez-Espinosa, A.; Vazquez, C.M.; Santa-Maria, C. Effects of dietary fats (fish, olive and high-oleic-acid sunflower oils) on lipid composition and antioxidant enzymes in rat liver. Br. J. Nutr. 1999, 82, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Giudetti, A.M.; Sabetta, S.; di Summa, R.; Leo, M.; Damiano, F.; Siculella, L.; Gnoni, G.V. Differential effects of coconut oil- and fish oil-enriched diets on tricarboxylate carrier in rat liver mitochondria. J. Lipid Res. 2003, 44, 2135–2141. [Google Scholar] [CrossRef] [PubMed]
- Koliwad, S.K.; Streeper, R.S.; Monetti, M.; Cornelissen, I.; Chan, L.; Terayama, K.; Naylor, S.; Rao, M.; Hubbard, B.; Farese, R.V., Jr. DGAT1-dependent triacylglycerol storage by macrophages protects mice from diet-induced insulin resistance and inflammation. J. Clin. Investig. 2010, 120, 756–767. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Yang, L.; McCall, S.; Huang, J.; Yu, X.X.; Pandey, S.K.; Bhanot, S.; Monia, B.P.; Li, Y.X.; Diehl, A.M. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology (Baltimore, MD) 2007, 45, 1366–1374. [Google Scholar] [CrossRef]
- Gentile, C.L.; Pagliassotti, M.J. The role of fatty acids in the development and progression of nonalcoholic fatty liver disease. J. Nutr. Biochem. 2008, 19, 567–576. [Google Scholar] [CrossRef]
- Melgert, B.N.; Olinga, P.; van der Laan, J.M.; Weert, B.; Cho, J.; Schuppan, D.; Groothuis, G.M.; Meijer, D.K.; Poelstra, K. Targeting dexamethasone to Kupffer cells: Effects on liver inflammation and fibrosis in rats. Hepatology (Baltimore, MD) 2001, 34 Pt 1, 719–728. [Google Scholar] [CrossRef]
- El Latif, H.A. Oval Cells: Potential Role in Liver Regeneration. Biomed. J. Sci. Tech. Res. 2018, 2. [Google Scholar] [CrossRef]
- Geisler, C.E.; Renquist, B.J. Hepatic lipid accumulation: Cause and consequence of dysregulated glucoregulatory hormones. J. Endocrinol. 2017, 234, R1–R21. [Google Scholar] [CrossRef]
- Yang, J.; Fernandez-Galilea, M.; Martinez-Fernandez, L.; Gonzalez-Muniesa, P.; Perez-Chavez, A.; Martinez, J.A.; Moreno-Aliaga, M.J. Oxidative Stress and Non-Alcoholic Fatty Liver Disease: Effects of Omega-3 Fatty Acid Supplementation. Nutrients 2019, 11, 872. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology (Baltimore, MD) 2010, 52, 1836–1846. [Google Scholar] [CrossRef] [PubMed]
- Dhibi, M.; Brahmi, F.; Mnari, A.; Houas, Z.; Chargui, I.; Bchir, L.; Gazzah, N.; Alsaif, M.A.; Hammami, M. The intake of high fat diet with different trans fatty acid levels differentially induces oxidative stress and non alcoholic fatty liver disease (NAFLD) in rats. Nutr. Metab. 2011, 8, 65. [Google Scholar] [CrossRef] [PubMed]
| Type | Fatty Acid Name | Composition | Percentage (%) | Amount (%) |
|---|---|---|---|---|
| Saturated Fatty Acid | Myristic Acid | C14:0 | 0.03 | 7.52 |
| Pentadecanoic Acid | C15:0 | 0.01 | ||
| Palmitic Acid | C16:0 | 4.96 | ||
| Margaric Acid | C17:0 | 0.04 | ||
| Stearic Acid | C18:0 | 2.46 | ||
| Lignoceric Acid | C24:0 | 0.01 | ||
| Monounsaturated Fatty Acid | Oleic Acid (ω-9) | C18:1 | 80.6 | 80.9 |
| Palmitoleic Acid | C16:1 | 0.15 | ||
| Gadoleic Acid | C20:1 | 0.19 | ||
| Polyunsaturated Fatty Acid | Linoleic Acid (ω-6) | C18:2 | 11.2 | 11.5 |
| Linolenic Acid (ω-3) | C18:3 | 0.27 | ||
| Trans Fat | Conjugated Linolenic Acid Trans-Octadecatrienoic acid | Trans C18:2 Trans C18:3 | 0.13 | 0.13 |
| Per 100 g | Control | HC | Low Dose HC + HO | High Dose HC + HO |
|---|---|---|---|---|
| Energy (Kcal) | 409 | 480 | 496 | 513 |
| Protein (g) | 29.39 | 26.8 | 25.3 | 23.8 |
| Total Fat (g) | 5.78 | 20.1 | 23.9 | 27.7 |
| Saturated Fatty Acid (g) | 1.22 | 6.6 | 7 | 7.4 |
| Unsaturated Fatty Acid (g) | 2.89 | 2.3 | 5.9 | 9.6 |
| Monounsaturated Fatty Acid (g) | 1.2 | 1 | 4.2 | 7.5 |
| Oleic Acid (g) | 0 | 0 | 3.3 | 6.6 |
| Polyunsaturated Fatty Acid (g) | 1.69 | 1.3 | 1.7 | 2.1 |
| Linolenic Acid (g) | 1.54 | 1.2 | 1.6 | 2 |
| Alpha-Linolenic acid (g) | 0.13 | 0.1 | 0.1 | 0.1 |
| Cholic Acid (g) | 0.22 | 0.38 | 0.36 | 0.35 |
| Cholesterol (g) | 0.03 | 0.22 | 0.22 | 0.22 |
| Phytochemicals (g) | 0 | 0 | 0.005 | 0.01 |
| Carbohydrate (g) | 59.84 | 48.04 | 45.04 | 42.05 |
| Fiber (g) | 4.1 | 3.26 | 3.06 | 2.85 |
| Sodium (mg) | 260 | 219 | 206 | 193 |
| Vitamin E (mg-αTE) | 4.1 | 3.2 | 4.7 | 6.2 |
| Group | Initial | WK0 | WK4 | WK8 | WK12 | |
|---|---|---|---|---|---|---|
| Body weight (g) | Control | 115.04 ± 6.13 | 140.05 ± 12.81 a | 157.59±15.56 a | 170.43±19.54 a | 175.08 ± 17.66 a |
| HC | 120.16 ± 6.04 | 135.25 ± 5.51 a | 152.99±9.39 a | 154.44±8.00 a,b | 154.82 ± 9.78 b | |
| Low dose HO + HC | 114.6 ± 12.2 | 143.93 ± 8.42 a | 144.62 ± 10.88 b | 147.70 ± 12.65 b | 144.55 ± 11.38 b | |
| High dose HO + HC | 128.9 ± 7.6 | 150.05 ± 10.52 a | 154.84 ± 10.98 a,b | 175.61 ± 13.69 a | 184.26 ± 13.83 a | |
| Ingestion of food (g) | Control | 7.33 ± 0.95 | 8.43±0.62 b | 7.45±0.71 a,b | 7.77±0.89 a | 7.56±0.68 a |
| HC | 8.13 ± 0.13 | 9.62 ± 0.66 b | 8.08 ± 0.86 a | 7.56 ± 0.92 a | 7.48 ± 1.03 a | |
| Low dose HO + HC | 10.3 ± 1.39 | 11.84 ± 0.50 a | 6.67 ± 0.51 b | 6.49 ± 0.39 b | 5.29 ± 0.73 a,b | |
| High dose HO + HC | 9.2 ± 0.07 | 11.81 ± 0.47 a, b | 4.87 ± 1.77 b | 7.25 ± 1.01 a,b | 6.26 ± 0.44 a | |
| Feed efficiency | Control | 2.97 ± 1.16 | 2.36 ± 0.72 | 1.65 ± 0.58 | 0.62 ± 0.52 | |
| HC | 1.57 ± 0.61 | 2.20 ± 1.18 | 0.19 ± 1.16 | 0.05 ± 0.42 | ||
| Low dose HO + HC | 2.55 ± 1.07 | 0.11 ± 0.77 | 0.46 ± 0.57 | -0.57 ± 0.46 | ||
| High dose HO + HC | 1.79 ± 0.49 | 1.10 ± 1.34 | 2.78 ± 1.06 | 1.37 ± 0.73 |
| Group | Initial | WK0 | WK4 | WK8 | WK12 | |
|---|---|---|---|---|---|---|
| TC (mg/dL) | Control | 148.00 ± 20.67 | 108.50 ± 22.83 | 117.80 ± 22.07 | 88.50 ± 5.91 | 95.90 ± 7.11 |
| HC | 143.3 ± 18.87 | 595.00 ± 158.41 b | 749.60 ± 133.07 a | 729.20 ± 116.32 a | 700.20 ± 106.26 a | |
| Low dose HC + HO | 146.3 ± 12.38 | 885.20 ± 245.06 a | 395.10 ± 125.29 b | 336.30 ± 94.18 b | 451.80 ± 127.56 b | |
| High dose HC + HO | 144.2 ± 17.61 | 1028.00 ± 327.41 a | 279.50 ± 30.87 c | 339.30 ± 58.23 b | 400.20 ± 83.65 b | |
| TG (mg/dL) | Control | 186.60 ± 76.09 | 154.20 ± 51.23 | 142.70 ± 63.64 | 116.50 ± 48.72 | 151.10 ± 40.22 |
| HC | 142.90 ± 49.48 | 223.4 ± 27.7794 | 971.7 ± 146.7485 | 576.2 ± 221.73 | 475.9 ± 190.9092 | |
| Low-dose HC + HO | 81.99 ± 34.09 | 1008.50 ± 496.96 a | 371.20 ± 252.58 a,b | 262.10 ± 163.20 c | 224.50 ± 142.73 b | |
| High-dose HC + HO | 127.37 ± 98.72 | 1474.20 ± 734.42 a | 313.40 ± 65.71 b | 384.40 ± 59.45 b | 363.80 ± 141.04 a | |
| LDL-C (mg/dL) | Control | 20.31 ± 7.87 | 14.29 ± 6.72 | 19.64 ± 6.99 | 9.85 ± 1.43 | 13.70 ± 3.04 |
| HC | 18.70 ± 4.73 | 205.80 ± 90.13 a | 229.58 ± 40.06 a | 228.83 ± 37.68 a | 223.92 ± 38.06 a | |
| Low-dose HC + HO | 41.30 ± 7.70 | 239.20 ± 72.79 a | 75.60 ± 44.96 b | 59.67 ± 33.05 b | 117.80 ± 60.29 b | |
| High-dose HC + HO | 38.80 ± 9.84 | 302.62 ± 132.52 a | 28.73 ± 8.17 c | 67.19 ± 24.67 b | 91.95 ± 33.36 b | |
| HDL-C (mg/dL) | Control | 71.91 ± 9.43 | 59.69 ± 8.03 | 58.01 ± 4.58 | 55.00 ± 3.89 | 53.35 ± 3.83 |
| HC | 74.15 ± 5.60 | 88.85 ± 5.77 a | 86.50 ± 8.47 b | 102.29 ± 7.61 b | 106.62 ± 5.08 b | |
| Low-dose HC + HO | 95.61 ± 8.94 | 82.71 ± 8.40 a,b | 123.47 ± 11.35 a | 124.27 ± 12.09 a | 119.11 ± 11.37 a | |
| High-dose HC + HO | 89.39 ± 8.51 | 79.39 ± 8.64 b | 131.00 ± 11.08 a | 117.89 ± 12.26 a | 117.90 ± 11.22 a | |
| LDL-C/HDL-C Ratio | Control | 0.28 ± 0.83 | 0.24 ± 0.84 c | 0.34 ± 0.53 | 0.18 ± 0.37 b | 0.26 ± 0.79 b |
| HC | 0.25 ± 0.84 | 2.35 ± 1.14 b | 2.70 ± 0.64 a | 2.26 ± 0.49 a | 2.10 ± 0.38 a | |
| Low-dose HC + HO | 0.43 ± 0.86 | 2.90 ± 1.14 a,b | 0.62 ± 0.37 b | 0.49 ± 0.28 b | 1.01 ± 0.55 b | |
| High-dose HC + HO | 0.43 ± 1.16 | 3.98 ± 2.10 a | 0.22 ± 0.07 c | 0.58 ± 0.24 b | 0.79 ± 0.32 b | |
| VLDL-C | Control | 55.78 ± 14.95 | 34.52 ± 10.99 c | 40.15 ± 14.03 c | 23.65 ± 3.65 c | 28.85 ± 3.77 c |
| HC | 50.46 ± 11.06 | 300.35 ± 116.30 a,b | 433.52 ± 102.25 a | 398.08 ± 86.03 a | 369.66 ± 72.25 a | |
| Low-dose HC + HO | 9.37 ± 6.27 | 563.29 ± 179.58 a | 196.03 ± 82.08 b | 152.36 ± 63.65 b | 214.89 ± 72.94 b | |
| High-dose HC + HO | 15.98 ± 13.38 | 645.99 ± 207.69 a | 119.77 ± 28.91 b | 154.22 ± 37.22 b | 190.35 ± 53.39 b |
| Group | Initial | WK0 | WK4 | WK8 | WK12 | |
|---|---|---|---|---|---|---|
| Control | 54.60 ± 10.96 | 61.10 ± 9.69 | 63.40 ± 7.81 | 56.10 ± 6.40 | 50.50 ± 6.29 | |
| GOT (U/L) | HC | 59.9 ± 24.5 | 172.90 ± 80.78 a | 137.30 ± 36.17 a | 113.90 ± 23.36 a | 96.70 ± 16.23 a |
| Low dose HC + HO | 58.5 ± 8.80 | 90.80 ± 26.55 b | 58.90 ± 17.34 b | 63.70 ± 18.82 b | 71.70 ± 24.84 b | |
| High dose HC + HO | 61.9 ± 12.53 | 81.00 ± 21.10 b | 51.60 ± 13.18 b | 58.10 ± 12.42 b | 62.60 ± 27.01 b,c | |
| Control | 85.80 ± 40.18 | 81.00 ± 25.94 | 84.10 ± 22.44 | 66.80 ± 13.65 | 81.90 ± 24.07 | |
| GPT (U/L) | HC | 78.4 ± 21.17 | 688.00 ± 269.41 a | 452.60 ± 142.17 a | 379.40 ± 94.72 a | 311.00 ± 83.76 a |
| Low dose HC + HO | 95.3 ± 23.44 | 349.30 ± 110.07 b | 151.50 ± 57.80 b | 189.40 ± 73.45 b | 226.80 ± 91.77 b | |
| High dose HC + HO | 102.8 ± 28.19 | 349.70 ± 141.63 b | 93.50 ± 20.39 b | 168.60 ± 35.15 b | 156.80 ± 38.18 c |
| Histopathological Lesions | Group | |||
|---|---|---|---|---|
| Control | HC | Low-Dose HC + HO | High-Dose HC + HO | |
| Fatty change, microvesicular, hepatocyte, diffuse, moderate/severe to severe/high | 0 ± 0 | 4.9 ± 0.3 a | 4.9 ± 0.3 a | 4.4 ± 0.5 b |
| Fatty change, macrovesicular, hepatocyte, multiple, minimal to moderate | 0 ± 0 | 1.0 ± 0.0 b,c | 1.0 ± 0.0 c | 1.6 ± 0.5 a,b |
| Infiltration, mononuclear cell and neutrophil, multiple, minimal to slight | 0 ± 0 | 2.2 ± 0.4 a | 1.9 ± 0.3 a,b | 1.6 ± 0.5 b,c |
| Hyperplasia, Kupffer cell, multiple, minimal to moderate | 0 ± 0 | 2.2 ± 0.4 a | 2.0 ± 0.5 a,b | 2.0 ± 0.0 a,b |
| Hyperplasia, oval cell, multiple, minimal to slight | 0 ± 0 | 2.1 ± 0.3 a | 1.7 ± 0.5 b | 1.2 ± 0.4 c |
| Fibrosis, multiple, minimal to slight | 0 ± 0 | 2.1 ± 0.3 a | 1.7 ± 0.5 b | 1.2 ± 0.4 c |
| Accumulation, glycogen, multiple, minimal to moderate | 3.2 ± 1.55 | 2.1 ± 0.3 a | 1.1 ± 0.3 b | 1.9 ± 0.7 a |
| Necrosis, hepatocyte, focal, minimal | 0 ± 0 | 1.0 ± 0.0 a | 1.0 ± 0.0 a | 1.0 ± 0.0 a |
| Group | Hepatic Lipid (mg/g Liver) | Fecal Lipid (mg/g Feces) | ||
|---|---|---|---|---|
| TC | TG | TC | TG | |
| Control | 2.49 ± 1.19 | 1.69 ± 0.44 | 1.62 ± 0.04 a | 1.35 ± 0.31 a,b |
| HC | 13.59 ± 2.91 a | 3.26 ± 0.85 c | 1.57 ± 0.76 a | 1.08 ± 0.41 b |
| Low-dose HC + HO | 13.51 ± 3.59 a | 5.69 ± 1.53 b | 3.33 ± 2.41 a | 2.05 ± 0.18 a |
| High-dose HC + HO | 11.87 ± 4.10 a | 7.49 ± 2.70 a,b | 1.70 ± 0.67 a | 1.25 ± 0.70 a,b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, J.-H.; Hsia, K.; Lin, C.-H.; Chen, C.-C.; Yang, H.-Y.; Lin, M.-H. Dietary Supplementation with Hazelnut Oil Reduces Serum Hyperlipidemia and Ameliorates the Progression of Nonalcoholic Fatty Liver Disease in Hamsters Fed a High-Cholesterol Diet. Nutrients 2019, 11, 2224. https://doi.org/10.3390/nu11092224
Lu J-H, Hsia K, Lin C-H, Chen C-C, Yang H-Y, Lin M-H. Dietary Supplementation with Hazelnut Oil Reduces Serum Hyperlipidemia and Ameliorates the Progression of Nonalcoholic Fatty Liver Disease in Hamsters Fed a High-Cholesterol Diet. Nutrients. 2019; 11(9):2224. https://doi.org/10.3390/nu11092224
Chicago/Turabian StyleLu, Jen-Her, Kai Hsia, Chih-Hsun Lin, Chien-Chin Chen, Hsin-Yu Yang, and Ming-Huei Lin. 2019. "Dietary Supplementation with Hazelnut Oil Reduces Serum Hyperlipidemia and Ameliorates the Progression of Nonalcoholic Fatty Liver Disease in Hamsters Fed a High-Cholesterol Diet" Nutrients 11, no. 9: 2224. https://doi.org/10.3390/nu11092224
APA StyleLu, J.-H., Hsia, K., Lin, C.-H., Chen, C.-C., Yang, H.-Y., & Lin, M.-H. (2019). Dietary Supplementation with Hazelnut Oil Reduces Serum Hyperlipidemia and Ameliorates the Progression of Nonalcoholic Fatty Liver Disease in Hamsters Fed a High-Cholesterol Diet. Nutrients, 11(9), 2224. https://doi.org/10.3390/nu11092224