Selenium Status and Hemolysis in Sickle Cell Disease Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Blood Collection
2.3. Hematological Parameters and Hemolysis Markers
2.4. Inflammatory Markers
2.5. Antioxidant Micronutrients
2.6. Cut-Off Values to Determine Micronutrient Deficiency
2.7. Food Consumption
2.8. Statistical Analysis
2.9. Ethical Aspects
3. Results
3.1. Participants’ Characteristics
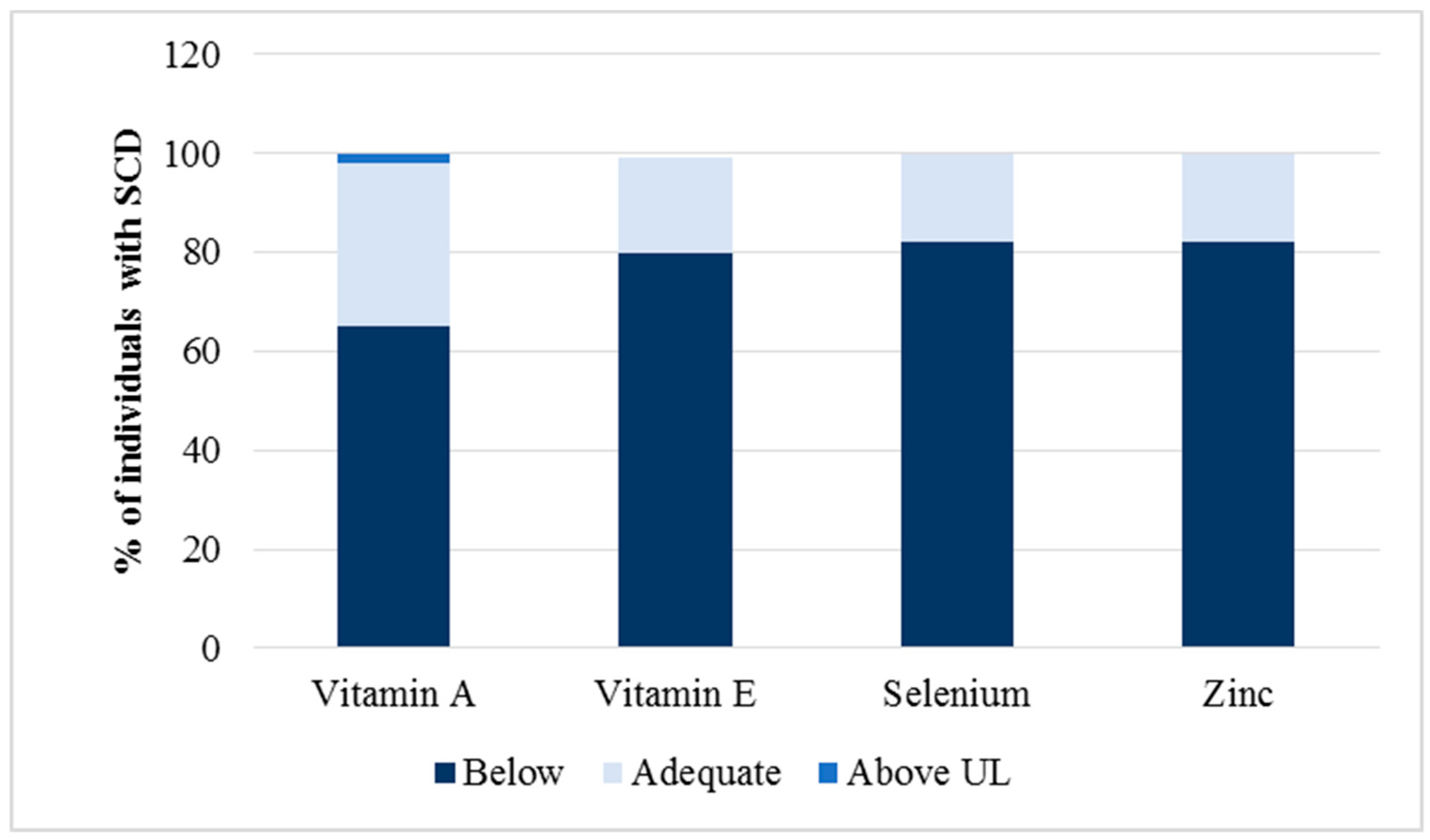
3.2. Relationship between Antioxidant Micronutrients and Hematological Parameters
3.3. Food Consumption
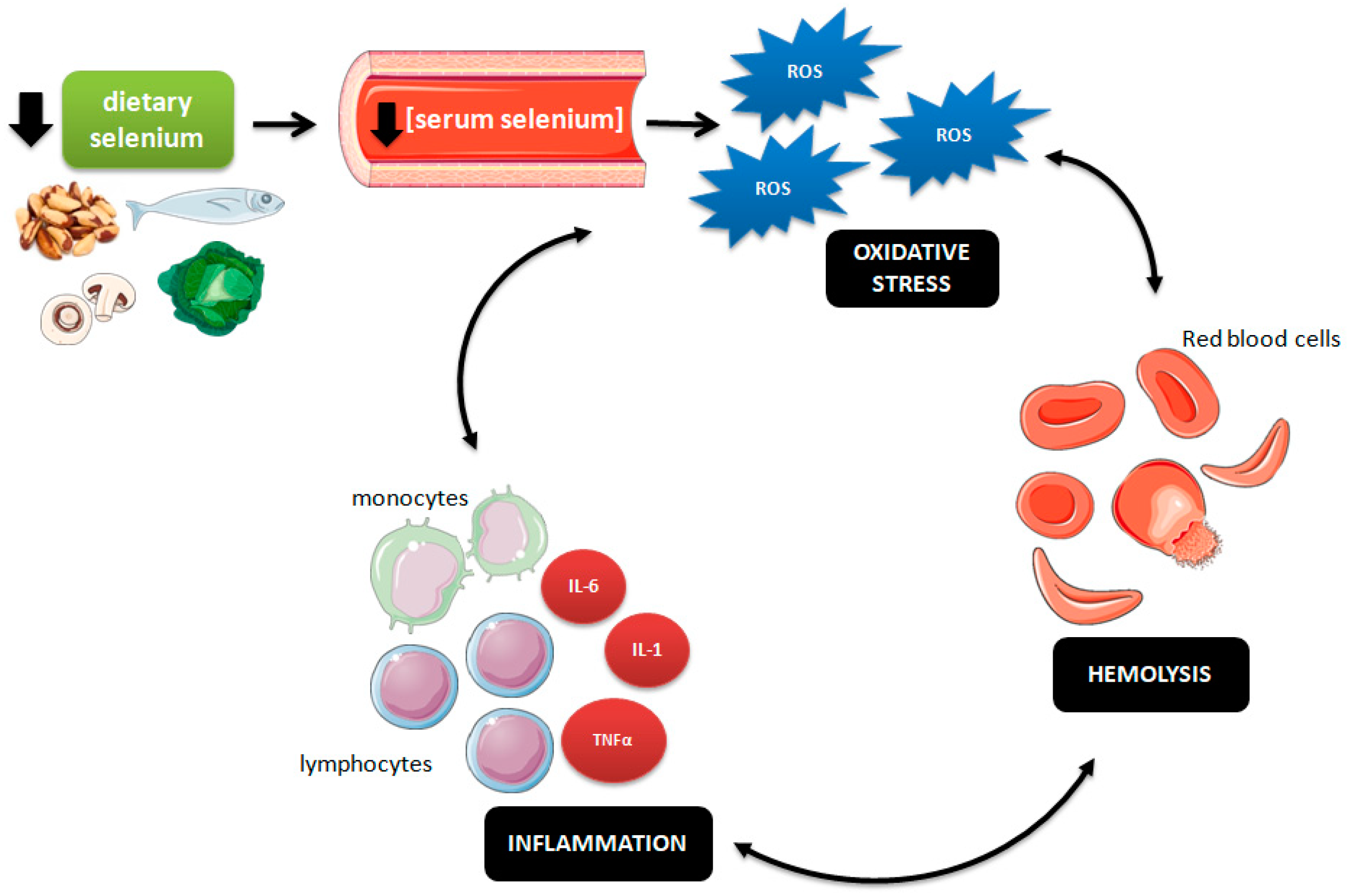
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kato, G.J.; Piel, F.B.; Reid, C.D.; Gaston, M.H.; Ohene-Frempong, K.; Krishnamurti, L.; Smith, W.R.; Panepinto, J.A.; Weatherall, D.J.; Costa, F.F.; et al. Sickle cell disease. Nat. Ver. Dis. Primers. 2018, 4, 1–22. [Google Scholar] [CrossRef]
- Ware, R.E.; Montalembert, M.; Tshilolo, L.; Abbound, M.R. Sickle cell disease. Lancet 2017, 390, 311–323. [Google Scholar] [CrossRef]
- Quinn, C.T.; Smith, E.P.; Arbabi, S.; Khera, P.K.; Lindsell, C.J.; Niss, O.; Joiner, C.H.; Franco, R.S.; Cohen, R.M. Biochemical surrogate markers of hemolysis do not correlate with directly measured erythrocyte survival in sickle cell anemia. Am. J. Hematol. 2016, 91, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Alayash, A.I. Oxidative pathways in the sickle cell and beyond. Blood Cells Mol. Dis. 2017, 70, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Hebbel, R.P.; Eaton, J.W.; Balasingam, M.; Steinberg, M.H. Spontaneous oxygen radical generation by sickle erythrocytes. J. Clin. Invest. 1982, 70, 1253–1259. [Google Scholar] [CrossRef]
- Hebbel, R.P.; Morgan, W.T.; Eaton, J.W.; Hedlund, B.E. Accelerated autoxidation and heme loss due to instability of sickle hemoglobin. Proc. Natl. Acad. Sci. USA 1988, 85, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Kanias, T.; Acker, J.P. Biopreservation of red blood cells—The struggle with haemoglobin oxidation. FEBS J. 1988, 277, 343–356. [Google Scholar] [CrossRef]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef]
- Oliveros, L.; Veja, V.; Anzulovich, A.C.; Ramirez, D.; Giménez, M.F. Vitamin A deficiency modifies antioxidant defenses and essential element contents in rat heart. Nutr. Res. 2000, 20, 1139–1150. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, A.; Li, Y.; Han, X.; Wang, Q.; Liang, H. Vitamin E supplementation protects erythrocyte membranes from oxidative stress in healthy Chinese middle-aged and elderly people. Nutr. Res. 2012, 32, 328–334. [Google Scholar] [CrossRef]
- Gammoh, N.Z.; Rink, L. Zinc in infection and inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.K.; Chen, C.J. Dietary selenium and age-related susceptibility of rat erythrocytes to oxidative damage. J. Nutr. 1980, 110, 2460–2466. [Google Scholar] [CrossRef] [PubMed]
- Nagababu, E.; Chrest, F.J.; Rifkind, J.M. Hydrogen-peroxide-induced heme degradation in red blood cells: The protective roles of catalase and glutathione peroxidase. Biochim. Biophys. Acta 2003, 1620, 211–217. [Google Scholar] [CrossRef]
- Liao, C.; Hardison, R.C.; Kennett, M.J.; Carlson, B.A.; Paulson, R.F.; Prabhu, K.S. Selenoproteins regulate stress erythroid progenitors and spleen microenvironment during stress erythropoiesis. Blood 2018, 131, 2568–2580. [Google Scholar] [CrossRef] [PubMed]
- McKerrell, T.D.H.; Cohen, H.W.; Billett, H.H. The older sickle cell patient. J. Hematol. 2004, 76, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.B.; Kesse-Adu, R.; Breen, C.; Murphy, P.B.; Chambers, J.; Holmes, P.; Howard, J. Descriptive study of the characteristics of older adults with sickle cell disease. Am. J. Hematol. 2017, 93, e38–e40. [Google Scholar] [CrossRef] [PubMed]
- Lobo, C.L.C.; Nascimento, E.M.D.; Jesus, L.J.C.; Freitas, T.G.; Lugon, J.R.; Ballas, S.K. Mortality in children, adolescents and adults with sickle cell anemia in Rio de Janeiro, Brazil. Hematol. Transf. Cell Ther. 2018, 40, 37–42. [Google Scholar] [CrossRef]
- Omena, J.; Cople-Rodrigues, C.D.S.; Cardoso, J.D.D.A.; Soares, A.R.; Fleury, M.K.; Brito, F.D.S.B.; Koury, J.C.; Citelli, M. Serum hepcidin concentration in individuals with sickle cell anemia: Basis for the dietary recommendation of iron. Nutrients 2018, 10, e498. [Google Scholar] [CrossRef]
- World Health Organization—WHO. Available online: http://www.who.int/nutrition/publications/micronutrients/vitamin_a_deficiency/WHONUT96.10.pdf?ua=1 (accessed on 8 November 2018).
- Clark, S.F. Vitamins and trace elements. In The ASPEN Nutrition Support Care Curriculum: A Case-Based Approach—The Adult Patient; Gottschlich, M.M., Ed.; American Society for Parenteral and Enteral Nutrition: Silver Spring, MD, USA, 2007; pp. 129–162. [Google Scholar]
- Langley, G. Fluid, electrolytes, and acid-base disorders. In The ASPEN Nutrition Support Care Curriculum: A Case-Based Approach—The Adult Patient; Gottschlich, M.M., Ed.; American Society for Parenteral and Enteral Nutrition: Silver Spring, MD, USA, 2007; pp. 104–128. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes (DRI) for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Kratz, A.; Ferraro, M.; Sluss, P.M.; Lewandrowski, K.B. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Laboratory reference values. N. Engl. J. Med. 2004, 351, 1548–1563. [Google Scholar] [CrossRef]
- Santos, I.N.; Damião, J.J.; Fonseca, M.J.M.; Cople-Rodrigues, C.S.; Aguiar, O.B. Food insecurity and social support in families of children with sickle-cell disease. J. Pediatr. 2019, 95, 306–313. [Google Scholar] [CrossRef]
- Thein, M.S.; Igbineweka, N.E.; Thein, S.L. Sickle cell disease in the older adult. Pathology 2017, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zachara, B.A. Selenium and selenium-dependent antioxidants in chronic kidney disease. Adv. Clin. Chem. 2015, 68, 131–151. [Google Scholar] [CrossRef] [PubMed]
- Yuzbasiyan-Gurkan, V.A.; Brewer, G.J.; Vander, A.J.; Guenther, M.J.; Prasad, A.S. Net renal tubular reabsorption of zinc in healthy man and impaired handling in sickle cell anemia. Am. J. Hematol. 1989, 31, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Claster, S.; Wood, J.C.; Noetzli, L.; Carson, S.M.; Hofstra, T.C.; Khanna, R.; Coates, T.D. Nutritional deficiencies in iron overloaded patients with hemoglobinopathies. Am. J. Hematol. 2009, 84, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.B.; Hafeman, D.G.; Hoekstra, W.G. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, E.; Gromadzinska, J.; Reszka, E.; Wasowicz, W.; Sobala, W.; Szeszenia-Dabrowska, N.; Boffetta, P. Association between GPx1 Pro198Leu polymorphism, GPx1 activity and plasma selenium concentration in humans. Eur. J. Nutr. 2009, 48, 383–386. [Google Scholar] [CrossRef]
- Natta, C.L.; Chen, L.C.; Chow, C.K. Selenium and glutathione peroxidase levels in sickle cell anemia. Acta Haematol. 1990, 83, 130–132. [Google Scholar] [CrossRef]
- Hamdy, M.M.; Mosallam, D.S.; Jamal, A.M.; Rabie, W.A. Selenium and vitamin E as antioxidants in chronic hemolytic anemia: Are they deficient? A case-control study in a group of Egyptian children. J. Adv. Res. 2015, 6, 1071–1077. [Google Scholar] [CrossRef]
- Liao, C.; Carlson, B.A.; Paulson, R.F.; Prabhu, K.S. The intricate role of selenium and selenoproteins in erythropoiesis. Free Radic. Biol. Med. 2018, 127, 165–171. [Google Scholar] [CrossRef]
- Schomburg, L. Dietary selenium and human health. Nutrients 2016, 9, 22. [Google Scholar] [CrossRef]
- Zhu, X.; Oseghale, A.R.; Nicole, L.H.; Li, B.; Pace, B.S. Mechanisms of NRF2 activation to mediate fetal hemoglobin induction and protection against oxidative stress in sickle cell disease. Exp. Biol. Med. 2019, 244, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Niki, E. Role of vitamin E as a lipid-soluble peroxyl radical scavenger: In vitro and in vivo evidence. Free Radic. Biol. Med. 2014, 66, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Iyer, M.K.; Nayak, R.; Colah, R.; Chattopadhyay, S. Attenuation of oxidative hemolysis of human red blood cells by the natural phenolic compound, allylpyrocatechol. Free Radic. Res. 2013, 47, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, R.C.; Prasad, A.S.; Rabbani, P.I.; Cossack, Z.T. Zinc in plasma, neutrophils, lymphocytes, and erythrocytes as determined by flameless atomic absorption spectrophotometry. Clin. Chem. 1982, 28, 475–480. Available online: http://clinchem.aaccjnls.org/content/28/3/475.long (accessed on 8 November 2018). [PubMed]
- Bao, B.; Prasad, A.S.; Beck, F.W.J.; Snell, D.; Suneja, A.; Sarkar, F.H.; Doshi, N.; Fitzgerald, J.T.; Swerdlow, P. Zinc supplementation decreases oxidative stress, incidence of infection, and generation of inflammatory cytokines in sickle cell disease patients. Transl. Res. 2008, 152, 67–80. [Google Scholar] [CrossRef]
- Cantor, A.B.; Orkin, S.H. Transcriptional regulation of erythropoiesis: An affair involving multiple partners. Oncogene 2002, 21, 3368–3376. [Google Scholar] [CrossRef] [PubMed]
- Voskou, S.; Aslanb, M.; Fanis, P.; Phylactides, M.; Kleanthous, M. Oxidative stress in β-thalassaemia and sickle cell disease. Redox Biol. 2015, 6, 226–239. [Google Scholar] [CrossRef]
| n (%) | |
|---|---|
| Genotype | |
| HbSS | 36 (70.6%) |
| HbSC | 15 (29.4%) |
| Sex | |
| female | 28 (54.9%) |
| male | 23 (45.1%) |
| Age (mean ± SD a) | 49.3 ± 6.57 |
| Skin colour | |
| White | 2 (3.9%) |
| Black | 27 (52.9%) |
| Mixed race | 22 (43.1%) |
| Use of hydroxyurea | |
| Yes | 21 (41.2%) |
| No | 30 (58.8%) |
| Reference Value * | Median | Minimum; Maxi mum | P25; P75 | |
|---|---|---|---|---|
| ERY (1012/L) | Fem 4.0–5.2 | 2.83 | 1.63; 5.03 | 2.31; 3.73 |
| Male 4.5–5.9 | ||||
| Hb (g/dL) | Fem 12–16 | 9.10 | 5.50; 14.70 | 7.50; 10.10 |
| Male 13.5–17.5 | ||||
| HT (%) | Fem 36–46 | 27.8 | 16.70; 46.40 | 23.40; 32.10 |
| Male 41–53 | ||||
| MCV (fl) | 80–100 | 96.00 | 63.00; 135.00 | 86.00; 102.00 |
| MCH (pg) | 27–32 | 31.00 | 19.00; 44.10 | 27.30; 33.20 |
| RET (% red cells) | 0.5–2.5 | 5.50 | 1.60; 18.30 | 0.30; 9.40 |
| RDW (%) | 11.5–14.5 | 15.00 | 11.10; 20.90 | 13.20; 17.70 |
| TB (mg/dL) | 0.3–1.0 | 1.37 | 0.50; 7.28 | 0.94; 2.51 |
| DB (mg/dL) | 0.1–0.3 | 0.58 | 0.20; 1.91 | 0.32; 0.86 |
| IB (mg/dL) | <0.8 | 0.96 | 0.10; 5.97 | 0.62; 1.70 |
| LDH (U/L) | 100–190 | 818.50 | 119.00; 2594.00 | 541.00; 1127.00 |
| HbF (%) | 0–2.0 | 4.40 | 0.20; 27.40 | 1.20; 12.40 |
| HbS (%) | – | 77.35 | 23.50; 95.10 | 49.40; 88.42 |
| Reference Value * | Median | Minimum; Maximum | P25; P75 | |
|---|---|---|---|---|
| Vitamin A (µmol/L) | >0.35 | 0.50 | 0.20; 1.30 | 0.30; 0.80 |
| Vitamin E (µg/mL) | >5 | 7.90 | 4.20; 20.6 | 6.70; 11.45 |
| Selenium (µg/dL) | >80 | 33.50 | 15.00; 130.00 | 27.00; 41.00 |
| Zinc (µg/dL) | >70 | 96.00 | 62.80; 145.70 | 83.70; 117.20 |
| Leukocytes (cells/mm³) | 4500–10,000 | 8200 | 3500; 25900 | 6600; 10200 |
| us-CRP (mg/dL) | <0.5 | 0.55 | 0.09; 2.79 | 0.25; 1.13 |
| Antioxidant Nutrients | Hematological Parameters | R2 | β | CI | p Value a |
|---|---|---|---|---|---|
| Selenium | LDH (U/L) | 0.62 | −13.90 | −22.79; −5.02 | 0.003 |
| ERY (1012/L) | 0.30 | 0.02 | 0.00; 0.04 | 0.010 | |
| Hb (g/dL) | 0.38 | 0.06 | 0.01; 0.10 | 0.006 | |
| HT (%) | 0.37 | 0.19 | 0.06; 0.32 | 0.006 | |
| RET (%) | 0.27 | −0.09 | −0.19; 0.00 | 0.040 | |
| TB (mg/dL) | 0.54 | −0.05 | −0.08; −0.02 | 0.001 | |
| DB (mg/dL) | 0.56 | −0.01 | −0.02; 0.00 | 0.007 | |
| IB (mg/dL) | 0.42 | −0.04 | −0.07; −0.01 | 0.007 | |
| HbF (%) | 0.41 | −0.22 | −0.40; −0.04 | 0.017 | |
| Vitamin E | RDW (%) | 0.28 | −0.26 | −0.43; −0.08 | 0.005 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delesderrier, E.; Cople-Rodrigues, C.S.; Omena, J.; Kneip Fleury, M.; Barbosa Brito, F.; Costa Bacelo, A.; Correa Koury, J.; Citelli, M. Selenium Status and Hemolysis in Sickle Cell Disease Patients. Nutrients 2019, 11, 2211. https://doi.org/10.3390/nu11092211
Delesderrier E, Cople-Rodrigues CS, Omena J, Kneip Fleury M, Barbosa Brito F, Costa Bacelo A, Correa Koury J, Citelli M. Selenium Status and Hemolysis in Sickle Cell Disease Patients. Nutrients. 2019; 11(9):2211. https://doi.org/10.3390/nu11092211
Chicago/Turabian StyleDelesderrier, Emília, Cláudia S. Cople-Rodrigues, Juliana Omena, Marcos Kneip Fleury, Flávia Barbosa Brito, Adriana Costa Bacelo, Josely Correa Koury, and Marta Citelli. 2019. "Selenium Status and Hemolysis in Sickle Cell Disease Patients" Nutrients 11, no. 9: 2211. https://doi.org/10.3390/nu11092211
APA StyleDelesderrier, E., Cople-Rodrigues, C. S., Omena, J., Kneip Fleury, M., Barbosa Brito, F., Costa Bacelo, A., Correa Koury, J., & Citelli, M. (2019). Selenium Status and Hemolysis in Sickle Cell Disease Patients. Nutrients, 11(9), 2211. https://doi.org/10.3390/nu11092211





