Black Raspberry (Rubus coreanus Miquel) Promotes Browning of Preadipocytes and Inguinal White Adipose Tissue in Cold-Induced Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Sample Preparation
2.3. Cell Culture and Beige Adipocyte Differentiation
2.4. Cell Cytotoxicity Assay
2.5. Oil Red O staining
2.6. RNA Isolation and Real-Time Reverse Transcription—Polymerase Chain Reaction (RT-PCR)
2.7. Animal Experiments
2.8. Hematoxylin and Eosin (H&E) Staining
2.9. Western Blot Analysis
2.10. Immunofluorescence Assay
2.11. Statistical Analysis
3. Results
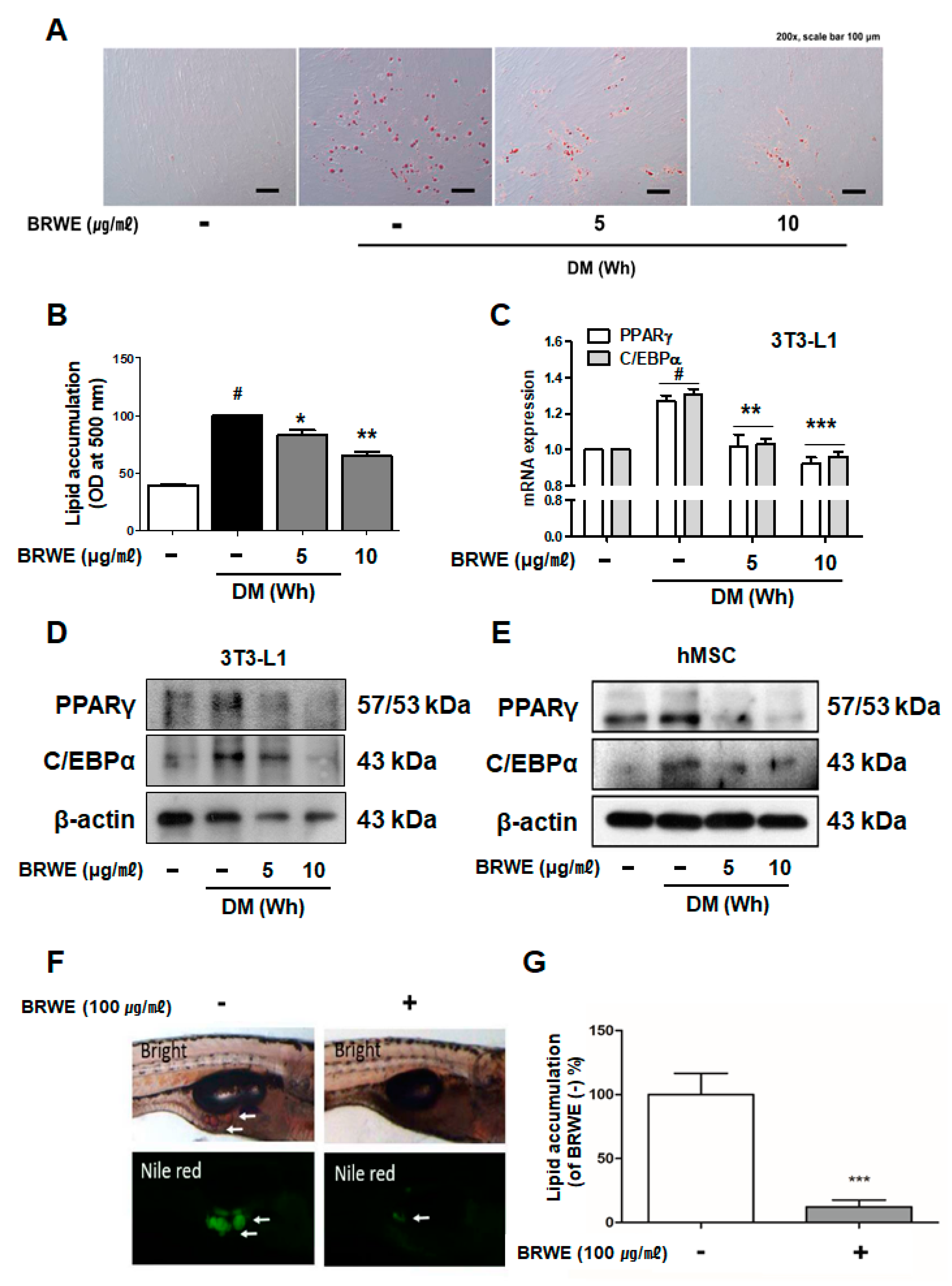
3.1. Effect of BRWE on Adipocyte Differentiation in hMSCs Cells and Zebrafish
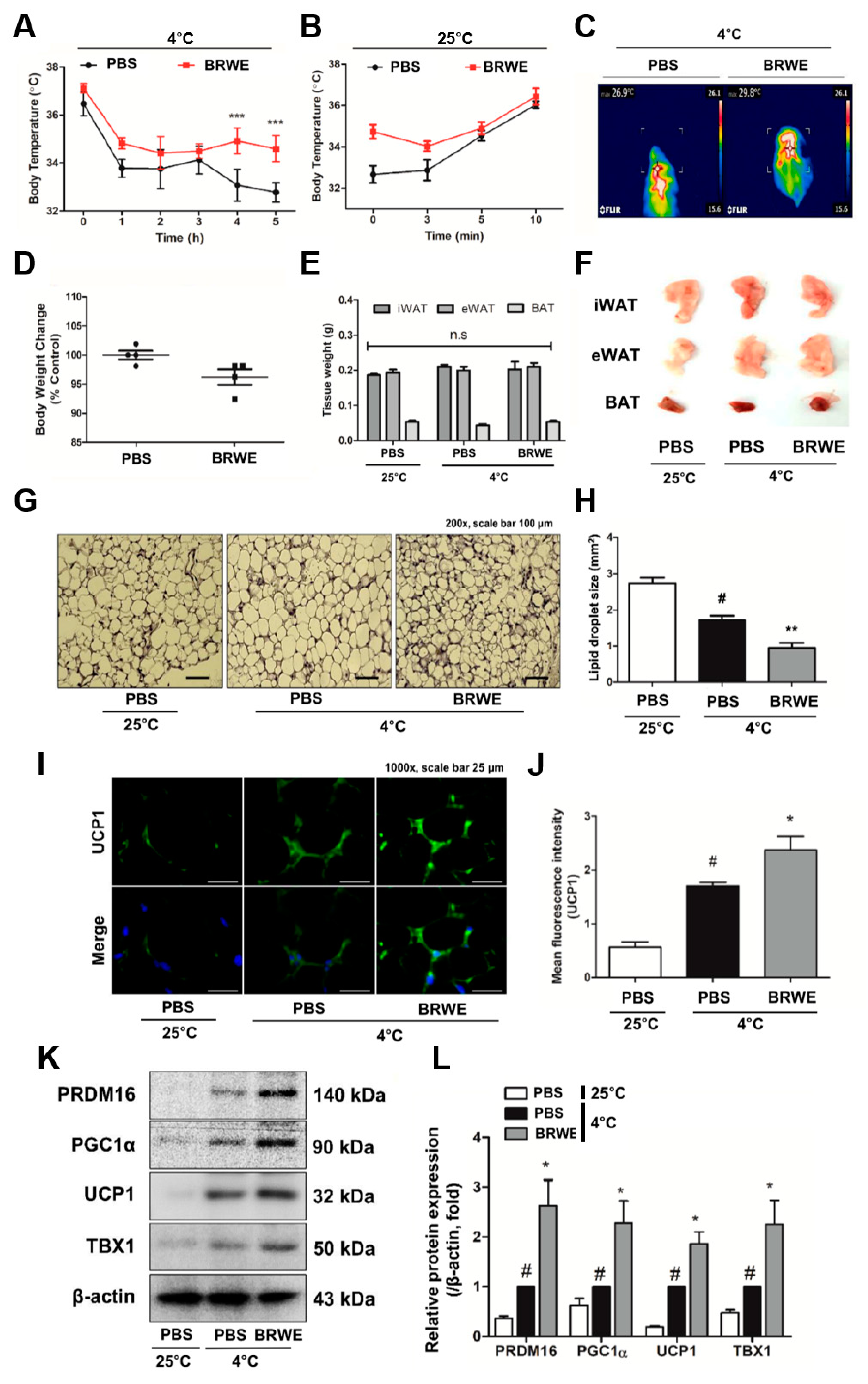
3.2. Effect of BRWE on Inguinal WAT (iWAT) Browning in Cold-Exposed Mice
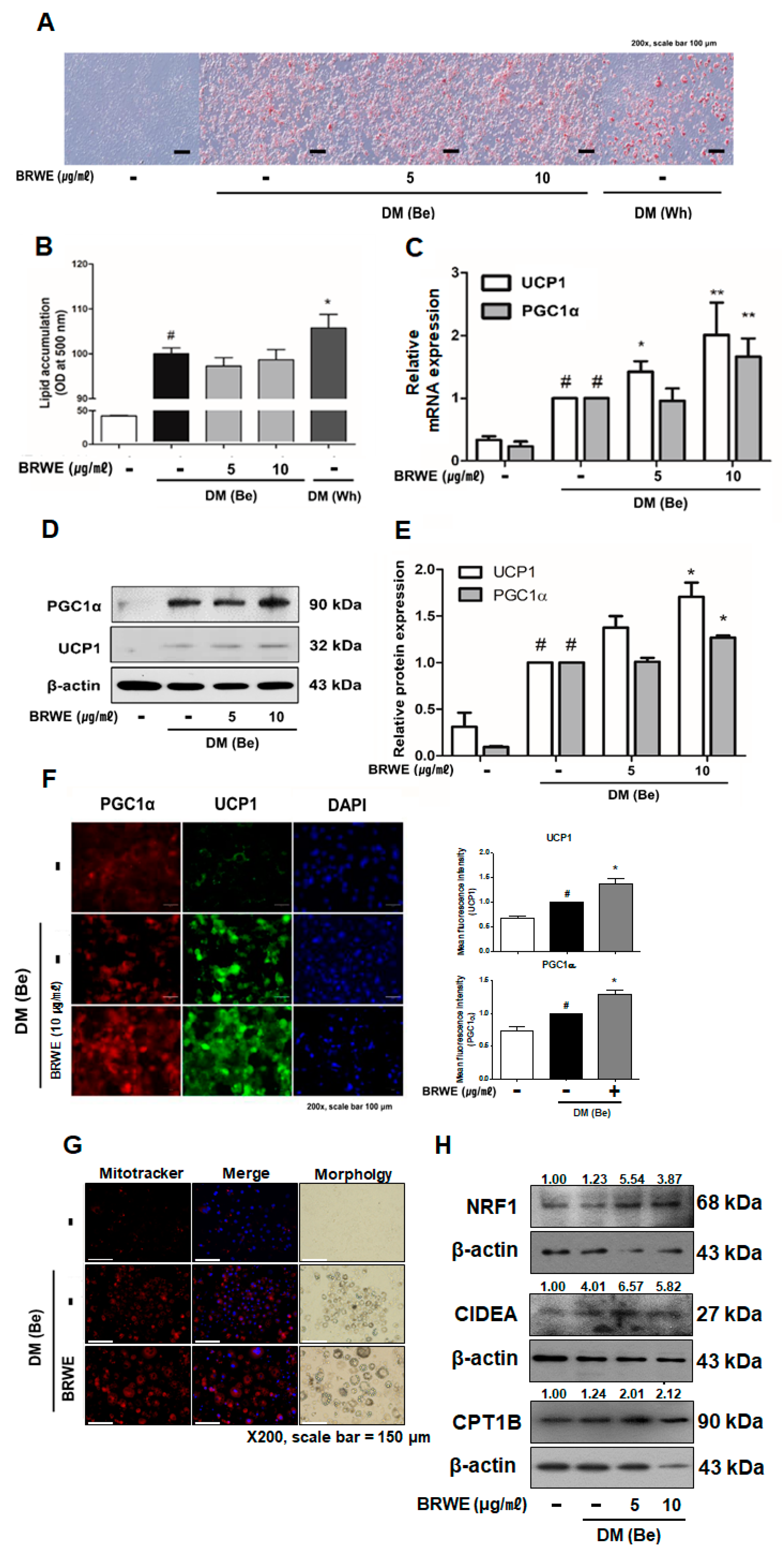
3.3. Effect of BRWE on Beige Adipocyte-Specific Markers in 3T3-L1 Adipocytes
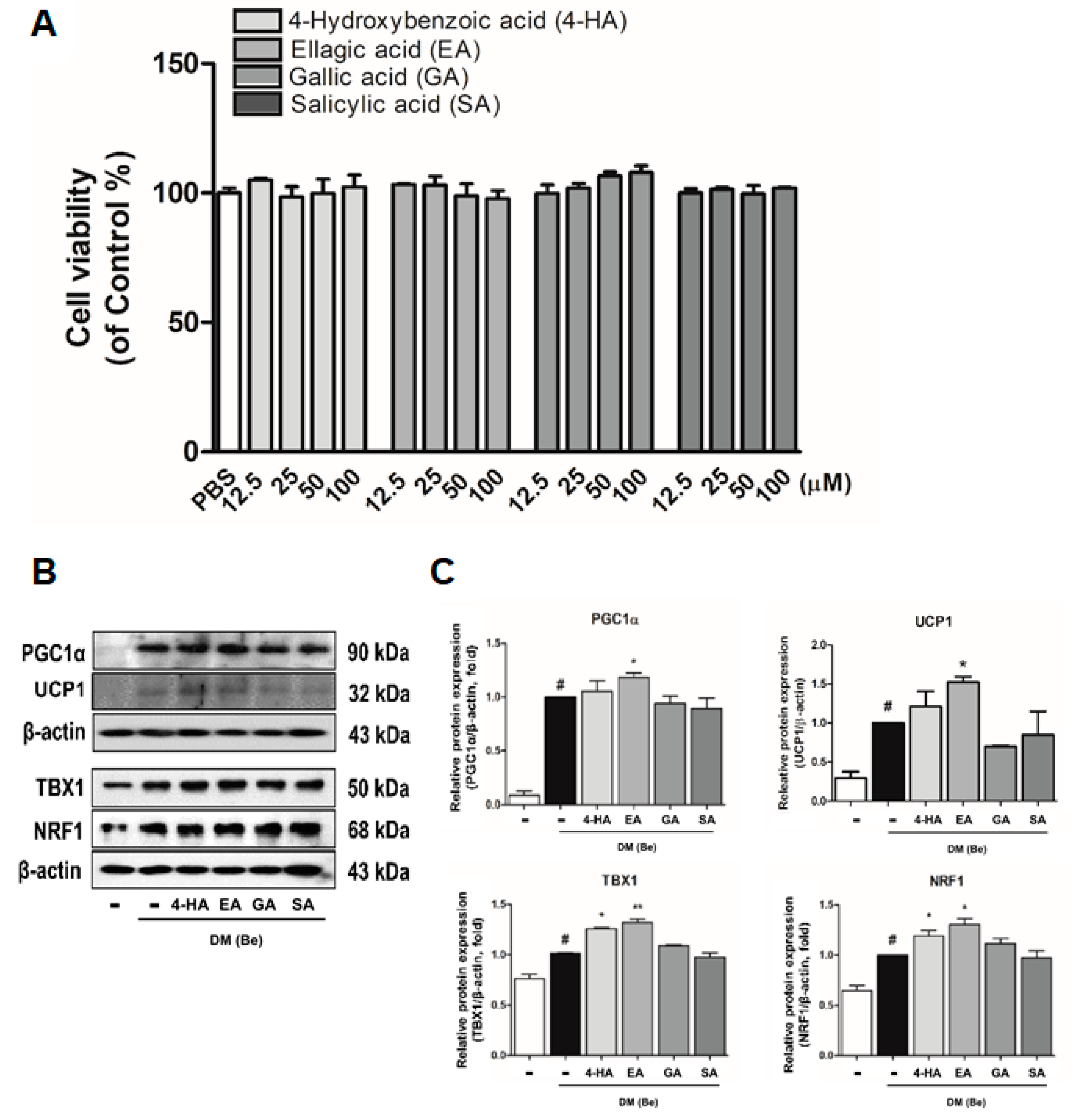
3.4. Effect of Phytochemicals Derived from BRWE on Beige Markers in 3T3-L1 Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- El-shiekh, R.A.; Al-Mahdy, D.A.; Mouneir, S.M.; Hifnawy, M.S.; Abdel-Sattar, E.A. Anti-obesity effect of argel (Solenostemma argel) on obese rats fed a high fat diet. J. Ethnopharmacol. 2019, 238, 111893. [Google Scholar] [CrossRef]
- Kopelman, P.G. Obesity as a medical problem. Nature 2000, 404, 635. [Google Scholar] [CrossRef]
- Gu, X.; Hong, Y.; Lin, Y.; Si, Q.; Yang, L.; Dong, W. Ginsenoside Rb2 alleviates obesity by activation of brown fat and induction of browning of white fat. Front. Endocrinol. 2019, 10, 153. [Google Scholar]
- Shabalina, I.G.; Petrovic, N.; de Jong, J.M.; Kalinovich, A.V.; Cannon, B.; Nedergaard, J. UCP1 in brite/beige adipose tissue mitochondria is functionally thermogenic. Cell Rep. 2013, 5, 1196–1203. [Google Scholar] [CrossRef]
- Chou, Y.-C.; Ho, C.-T.; Pan, M.-H. Immature Citrus reticulata extract promotes browning of beige adipocytes in high-fat diet-induced C57BL/6 mice. J. Agric. Food Chem. 2018, 66, 9697–9703. [Google Scholar] [CrossRef]
- Lo, K.A.; Sun, L. Turning WAT into BAT: A review on regulators controlling the browning of white adipocytes. Biosci. Rep. 2013, 33, e00065. [Google Scholar] [CrossRef]
- Wang, W.; Seale, P. Control of brown and beige fat development. Nat. Rev. Mol. Cell Biol. 2016, 17, 691. [Google Scholar] [CrossRef]
- Nedergaard, J.; Golozoubova, V.; Matthias, A.; Asadi, A.; Jacobsson, A.; Cannon, B. UCP1: The only protein able to mediate adaptive non-shivering thermogenesis and metabolic inefficiency. Biochim. Biophys. Acta-Bioenerg. 2001, 1504, 82–106. [Google Scholar] [CrossRef]
- Concha, F.; Prado, G.; Quezada, J.; Ramirez, A.; Bravo, N.; Flores, C.; Herrera, J.; Lopez, N.; Uribe, D.; Duarte-Silva, L. Nutritional and non-nutritional agents that stimulate white adipose tissue browning. Rev. Endocr. Metab. Disord. 2019, 20, 161–171. [Google Scholar] [CrossRef]
- Lim, J.W.; Jeong, J.T.; Shin, C.S. Component analysis and sensory evaluation of Korean black raspberry (Rubus coreanus Mique) wines. Int. J. Food Sci. Technol. 2012, 47, 918–926. [Google Scholar] [CrossRef]
- Jeong, M.-Y.; Kim, H.-L.; Park, J.; An, H.-J.; Kim, S.-H.; Kim, S.-J.; So, H.-S.; Park, R.; Um, J.-Y.; Hong, S.-H. Rubi fructus (Rubus coreanus) inhibits differentiation to adipocytes in 3T3-L1 cells. Evid.-Based Complement Altern. Med. 2013, 2013, 475386. [Google Scholar] [CrossRef]
- Jeong, M.; Kim, H.; Park, J.; Jung, Y.; Youn, D.; Lee, J.; Jin, J.; So, H.; Park, R.; Kim, S. Rubi Fructus (Rubus coreanus) activates the expression of thermogenic genes in vivo and in vitro. Int. J. Obes. 2015, 39, 456. [Google Scholar] [CrossRef]
- Kim, H.-L.; Park, J.; Park, H.; Jung, Y.; Youn, D.-H.; Kang, J.; Jeong, M.-Y.; Um, J.-Y. Platycodon grandiflorum A. de candolle ethanolic extract inhibits adipogenic regulators in 3T3-L1 cells and induces mitochondrial biogenesis in primary brown preadipocytes. J. Agric. Food Chem. 2015, 63, 7721–7730. [Google Scholar] [CrossRef]
- Kim, H.-L.; Park, J.; Jung, Y.; Ahn, K.S.; Um, J.-Y. Platycodin D, a novel activator of AMP-activated protein kinase, attenuates obesity in db/db mice via regulation of adipogenesis and thermogenesis. Phytomedicine 2019, 52, 254–263. [Google Scholar] [CrossRef]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Ablain, J.; Zon, L.I. Of fish and men: Using zebrafish to fight human diseases. Trends Cell Biol. 2013, 23, 584–586. [Google Scholar] [CrossRef]
- MacDonald, M.E.; Stainier, D.Y.R. Lessons from “Lower” Organisms: What Worms, Flies, and Zebrafish Can Teach Us about Human Energy Metabolism. PLoS Genet. 2007, 3, e199. [Google Scholar]
- Oka, T.; Nishimura, Y.; Zang, L.; Hirano, M.; Shimada, Y.; Wang, Z.; Umemoto, N.; Kuroyanagi, J.; Nishimura, N.; Tanaka, T. Diet-induced obesity in zebrafish shares common pathophysiological pathways with mammalian obesity. BMC Physiol. 2010, 10, 21. [Google Scholar] [CrossRef]
- Anderson, J.L.; Carten, J.D.; Farber, S.A. Zebrafish lipid metabolism: From mediating early patterning to the metabolism of dietary fat and cholesterol. Methods Cell Biol. 2011, 101, 111–141. [Google Scholar]
- Imrie, D.; Sadler, K.C. White adipose tissue development in zebrafish is regulated by both developmental time and fish size. Dev. Dyn. 2010, 239, 3013–3023. [Google Scholar] [CrossRef]
- Jung, Y.; Park, J.; Kim, H.-L.; Sim, J.-E.; Youn, D.-H.; Kang, J.; Lim, S.; Jeong, M.-Y.; Yang, W.M.; Lee, S.-G. Vanillic acid attenuates obesity via activation of the AMPK pathway and thermogenic factors in vivo and in vitro. FASEB J. 2017, 32, 1388–1402. [Google Scholar] [CrossRef]
- Ikeda, K.; Maretich, P.; Kajimura, S. The Common and Distinct Features of Brown and Beige Adipocytes. Trends Endocrinol. Metab. 2018, 29, 191–200. [Google Scholar] [CrossRef]
- Chang, S.-H.; Song, N.-J.; Choi, J.H.; Yun, U.J.; Park, K.W. Mechanisms underlying UCP1 dependent and independent adipocyte thermogenesis. Obes. Rev. 2019, 20, 241–251. [Google Scholar] [CrossRef]
- Nishimoto, Y.; Tamori, Y. CIDE Family-Mediated Unique Lipid Droplet Morphology in White Adipose Tissue and Brown Adipose Tissue Determines the Adipocyte Energy Metabolism. J. Atheroscler. Thromb. 2017, 24, 989–998. [Google Scholar] [CrossRef]
- Bartelt, A.; Widenmaier, S.B.; Schlein, C.; Johann, K.; Goncalves, R.L.S.; Eguchi, K.; Fischer, A.W.; Parlakgül, G.; Snyder, N.A.; Nguyen, T.B.; et al. Brown adipose tissue thermogenic adaptation requires Nrf1-mediated proteasomal activity. Nat. Med. 2018, 24, 292–303. [Google Scholar] [CrossRef]
- Calderon-Dominguez, M.; Sebastián, D.; Fucho, R.; Weber, M.; Mir, J.F.; García-Casarrubios, E.; Obregón, M.J.; Zorzano, A.; Valverde, Á.M.; Serra, D.; et al. Carnitine Palmitoyltransferase 1 Increases Lipolysis, UCP1 Protein Expression and Mitochondrial Activity in Brown Adipocytes. PLoS ONE 2016, 11, e0159399. [Google Scholar] [CrossRef]
- Fu, J.; Oveisi, F.; Gaetani, S.; Lin, E.; Piomelli, D. Oleoylethanolamide, an endogenous PPAR-α agonist, lowers body weight and hyperlipidemia in obese rats. Neuropharmacology 2005, 48, 1147–1153. [Google Scholar] [CrossRef]
- González-Castejón, M.; Rodriguez-Casado, A. Dietary phytochemicals and their potential effects on obesity: A review. Pharmacol. Res. 2011, 64, 438–455. [Google Scholar] [CrossRef]
- Kim, H.-L.; Sim, J.-E.; Choi, H.-M.; Choi, I.-Y.; Jeong, M.-Y.; Park, J.; Jung, Y.; Youn, D.-H.; Cho, J.-H.; Kim, J.-H. The AMPK pathway mediates an anti-adipogenic effect of fruits of Hovenia dulcis Thunb. Food Funct. 2014, 5, 2961–2968. [Google Scholar] [CrossRef]
- Horton, J.D.; Goldstein, J.L.; Brown, M.S. SREBPs: Activators of the complete program of cholesterol and fatty acid synthesis in the liver. J. Clin. Investig. 2002, 109, 1125–1131. [Google Scholar] [CrossRef]
- Lowell, B.B. An essential regulator of adipogenesis and modulator of fat cell function: PPARγ. Cell 1999, 99, 239–242. [Google Scholar] [CrossRef]
- Sell, H.; Deshaies, Y.; Richard, D. The brown adipocyte: Update on its metabolic role. Int. J. Biochem. Cell Biol. 2004, 36, 2098–2104. [Google Scholar] [CrossRef]
- Kajimura, S.; Spiegelman, B.M.; Seale, P. Brown and beige fat: Physiological roles beyond heat generation. Cell Metab. 2015, 22, 546–559. [Google Scholar] [CrossRef]
- Cannon, B.; Nedergaard, J. Brown adipose tissue: Function and physiological significance. Physiol. Rev. 2004, 84, 277–359. [Google Scholar] [CrossRef]
- Wu, Z.; Boss, O. Targeting PGC-1α to control energy homeostasis. Expert Opin. Ther. Targets 2007, 11, 1329–1338. [Google Scholar] [CrossRef]
- Lee, P.; Linderman, J.D.; Smith, S.; Brychta, R.J.; Wang, J.; Idelson, C.; Perron, R.M.; Werner, C.D.; Phan, G.Q.; Kammula, U.S. Irisin and FGF21 are cold-induced endocrine activators of brown fat function in humans. Cell Metab. 2014, 19, 302–309. [Google Scholar] [CrossRef]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.-H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef]
- Zhao, J.; Cannon, B.; Nedergaard, J. Thermogenesis is β3-but not β1-adrenergically mediated in rat brown fat cells, even after cold acclimation. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1998, 275, R2002–R2011. [Google Scholar] [CrossRef]
- Frühbeck, G.; Méndez-Giménez, L.; Fernández-Formoso, J.-A.; Fernández, S.; Rodriguez, A. Regulation of adipocyte lipolysis. Nutr. Res. Rev. 2014, 27, 63–93. [Google Scholar] [CrossRef]
- Kajimura, S.; Seale, P.; Kubota, K.; Lunsford, E.; Frangioni, J.V.; Gygi, S.P.; Spiegelman, B.M. Initiation of myoblast to brown fat switch by a PRDM16–C/EBP-β transcriptional complex. Nature 2009, 460, 1154. [Google Scholar] [CrossRef]
- Matsukawa, T.; Villareal, M.O.; Motojima, H.; Isoda, H. Increasing cAMP levels of preadipocytes by cyanidin-3-glucoside treatment induces the formation of beige phenotypes in 3T3-L1 adipocytes. J. Nutr. Biochem. 2017, 40, 77–85. [Google Scholar] [CrossRef]
- Kang, N.H.; Mukherjee, S.; Yun, J.W. Trans-Cinnamic Acid Stimulates White Fat Browning and Activates Brown Adipocytes. Nutrients 2019, 11, 577. [Google Scholar] [CrossRef]
- Palacios-González, B.; Vargas-Castillo, A.; Velázquez-Villegas, L.A.; Vasquez-Reyes, S.; López, P.; Noriega, L.G.; Aleman, G.; Tovar-Palacio, C.; Torre-Villalvazo, I.; Yang, L.-J. Genistein increases the thermogenic program of subcutaneous WAT and increases energy expenditure in mice. J. Nutr. Biochem. 2019, 68, 59–68. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, W.Y.; Choe, S.-K.; Park, J.; Um, J.-Y. Black Raspberry (Rubus coreanus Miquel) Promotes Browning of Preadipocytes and Inguinal White Adipose Tissue in Cold-Induced Mice. Nutrients 2019, 11, 2164. https://doi.org/10.3390/nu11092164
Park WY, Choe S-K, Park J, Um J-Y. Black Raspberry (Rubus coreanus Miquel) Promotes Browning of Preadipocytes and Inguinal White Adipose Tissue in Cold-Induced Mice. Nutrients. 2019; 11(9):2164. https://doi.org/10.3390/nu11092164
Chicago/Turabian StylePark, Woo Yong, Seong-Kyu Choe, Jinbong Park, and Jae-Young Um. 2019. "Black Raspberry (Rubus coreanus Miquel) Promotes Browning of Preadipocytes and Inguinal White Adipose Tissue in Cold-Induced Mice" Nutrients 11, no. 9: 2164. https://doi.org/10.3390/nu11092164
APA StylePark, W. Y., Choe, S.-K., Park, J., & Um, J.-Y. (2019). Black Raspberry (Rubus coreanus Miquel) Promotes Browning of Preadipocytes and Inguinal White Adipose Tissue in Cold-Induced Mice. Nutrients, 11(9), 2164. https://doi.org/10.3390/nu11092164





