Palm Oil on the Edge
Abstract
1. Introduction: Justification and Need of the Consensus
2. Sustainability of Oil Palm Crops
- Management and conservation of natural resources.
- Efficient use of natural resources for production.
- Protection of rural livelihoods.
- Improvement of equity and rural welfare.
- Building the resilience of individuals and communities.
- Establishment of accountable and effective governance mechanisms, strengthening institutions and investment.
3. Importance of Fats in Foods
3.1. Fat Intake in Spain
3.2. Complexity of Fat Metabolism
4. Nutritional Composition of Palm Oil
Palmitic Acid
5. Palm Oil and Its Effects on the Lipid Profile
Palm Oil and Microbiota
6. Palm Oil in Animal Nutrition
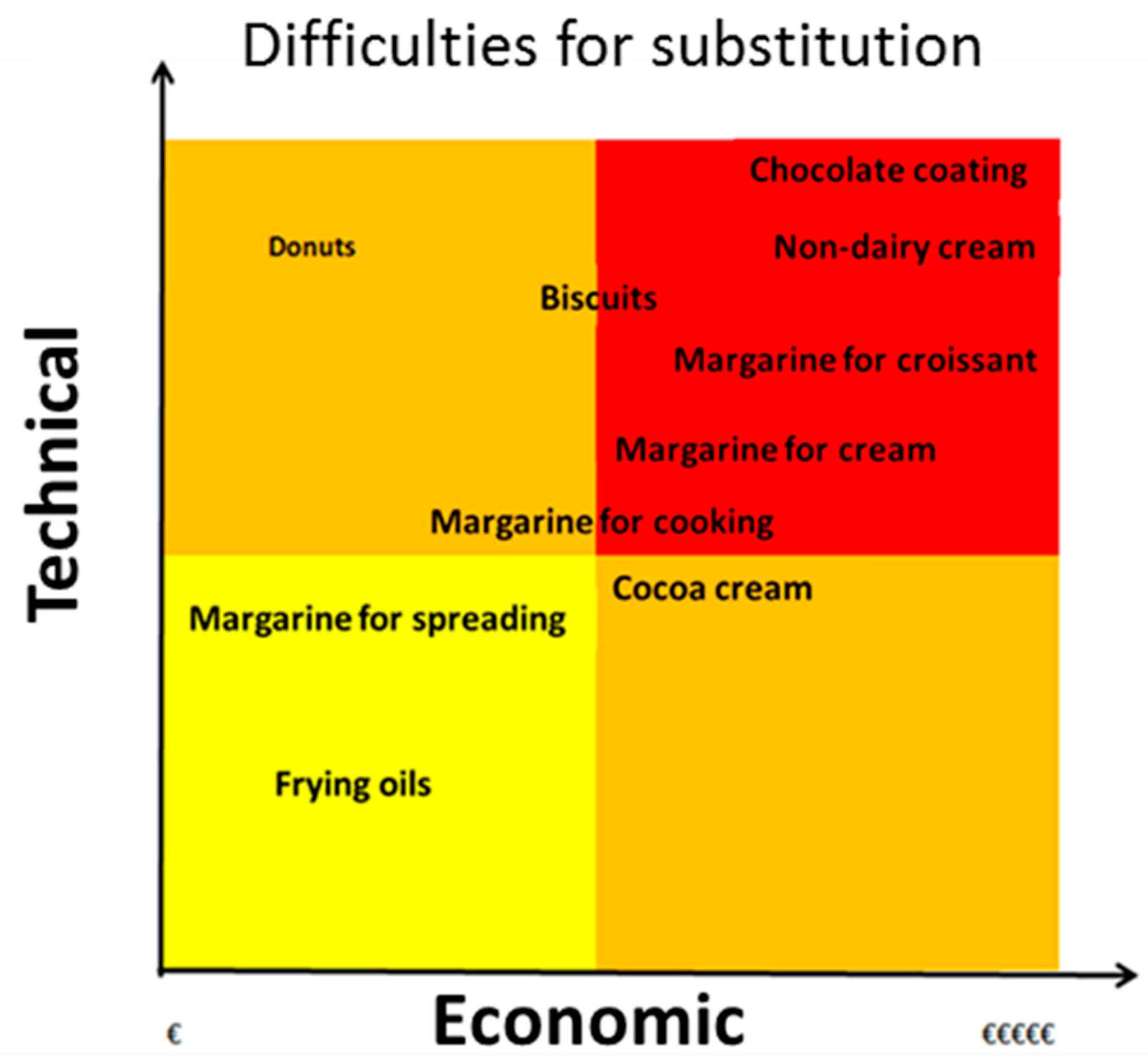
7. The Use of Palm Oil in the Food Industry
7.1. Justification for the Inclusion of Palm Oil in the Composition of Processed Foods
7.2. Role of Industry in Sustainable Palm Oil
8. Food Safety and Toxicological Aspects
8.1. Definition, Formation, and Possibility of Reduction of the Contaminants Free and Esterified 2-MCPD and 3-MCPD and Glycidil Esters (GE).
8.2. Risk Assessment of Contaminants
8.3. Palm Oil and Risk of Cancer
9. Palm Oil as an Example of the Complexity of Communication in Nutrition
10. Conclusions
10.1. Sustainability
10.2. Nutrition
10.3. Industry
10.4. Safety
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- China Soybean Imports LoweredImports down 16 Million Tons SinceJune, United States Department of Agriculture (USDA). Oilseeds: World Markets and Trade; United States Department of Agriculture: Foreign Agricultural Service: Washington, DC, USA, 2018; Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/tx31qh68h/ht24wn965/x920g124z/oilseeds.pdf (accessed on 13 February 2019).
- Mba, O.I.; Dumont, M.J.; Ngadi, M. Palm oil: Processing, characterization and utilization in the food industry—A review. Food Biosci. 2015, 10, 26–41. [Google Scholar] [CrossRef]
- Marangoni, F.; Galli, C.; Ghiselli, A.; Lercker, G.; La Vecchia, C.; Maffeis, C.; Agostoni, C.; Ballardini, D.; Brignoli, O.; Faggiano, P.; et al. Palm oil and human health. Meeting Report of NFI: Nutrition Foundation of Italy symposium. Int. J. Food Sci. Nutr. 2017, 68, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Boletín Oficial del Estado (BOE). Real Decreto 1011/1981 de 10 de abril por el que se aprueba la reglamentación técnico sanitaria para la elaboración, circulación y comercio de grasas comestibles (animales, vegetales y anhidras), margarinas, minarinas y preparados grasos. BOE nº 130. 01/06/1981. pp. 12071–12076. Available online: https://www.boe.es/eli/es/rd/1981/04/10/1011/dof/spa/pdf (accessed on 22 June 2019).
- Keys, A.; Menotti, A.; Karvonen, M.J.; Aravanis, C.; Blackburn, H.; Buzina, R.; Djordjevic, B.S.; Dontas, A.S.; Fidanza, F.; Keys, M.H.; et al. The diet and 15-year death rate in the Seven Countries Study. Am. J. Epidemiol. 1986, 124, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P.; Katan, M.B. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler. Thromb. 1992, 12, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Muniz, F.J.; Bastida, S. Lípidos. In Libro Blanco de la Nutrición en España; Fundación Española de la Nutrición (FEN): Madrid, Spain, 2013; pp. 113–124. [Google Scholar]
- Picó, C. Ácidos grasos trans. In Consenso Sobre las Grasas y Aceites en la Alimentación de la Población Española Adulta; Federación Española de Sociedades de Nutrición, Alimentación y Dietética (FESNAD): Madrid, Spain, 2015; pp. 28–34. Available online: http://www.fesnad.org/resources/files/Publicaciones/Consenso_sobre_las_grasas_y_aceites_2015.pdf (accessed on 22 July 2019).
- European Food Safety Authority (EFSA). Scientific and Technical Assistance on Trans Fatty Acids; Technical report; European Food Safety Authority (EFSA): Parma, Italy, 2018. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Replace Trans-Fat. 2018. WHO/NMH/NHD/18.4. Available online: https://www.who.int/nutrition/topics/replace-transfat (accessed on 22 July 2019).
- European Food Information Council (EUFIC). From Farm to Fork. 2006. Available online: https://www.eufic.org/en/food-production/article/from-farm-to-fork (accessed on 20 September 2018).
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. Closing the Loop—An EU Action Plan for the Circular Economy; European Commission: Brussels, Belgium, 2015; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/DOC/?uri=CELEX:52015DC0614&from=EN (accessed on 22 September 2018).
- United Nations (UN). Report of the World Commission on Environment and Development: Our Common Future. 1987. Available online: https://sustainabledevelopment.un.org/content/documents/5987our-common-future.pdf (accessed on 20 September 2018).
- Food and Agriculture Organization (FAO) Sustainable Diets and Biodiversity. Directions and Solutions for Policy, Research and Action. Rome, 2012. Available online: http://www.fao.org/docrep/016/i3004e/i3004e.pdf (accessed on 21 September 2018).
- High Level Panel of Experts on Food Security and Nutrition (HLPE). Food Losses and Waste in the Context of Sustainable Food Systems. Rome, 2014. Available online: http://www.fao.org/3/a-i3901e.pdf (accessed on 20 September 2018).
- United Nations (UN). Sustainable Development Goals. 2015. Available online: https://www.un.org/sustainabledevelopment/sustainable-development-goals/ (accessed on 20 September 2018).
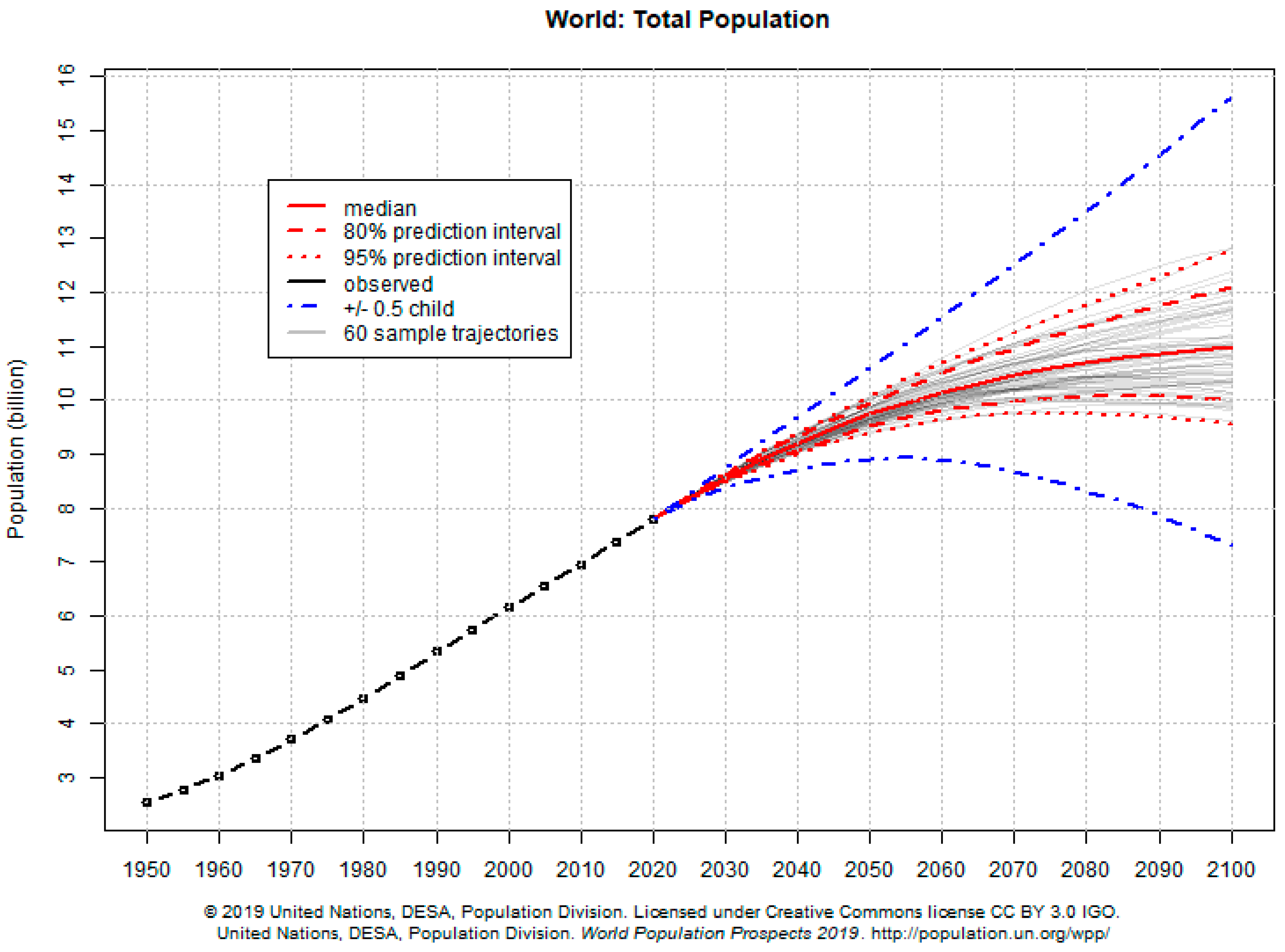
- United Nations (UN). World Population Prospects 2019. Available online: https://population.un.org/wpp/Graphs/Probabilistic/POP/TOT/900 (accessed on 22 May 2019).
- Roundtable for Sustainable Palm Oil (RSPO) Principles and Criteria for the Production of Sustainable Palm Oil. 2018. Available online: https://rspo.org/resources/certification/rspo-principles-criteria-certification (accessed on 20 June 2019).
- Roundtable for Sustainable Palm Oil (RSPO) RSPO Guide to Supply Chain Certification. Available online: https://rspo.org/resources/certification/supply-chain-certification (accessed on 20 June 2019).
- Voigt, M.; Wich, S.A.; Ancrenaz, M.; Wells, J.; Wilson, K.A.; Kühl, H.S. Global demand for natural resources eliminated more than 100,000 Bornean orangutans. Curr. Biol. 2018, 28, 761–769. [Google Scholar] [CrossRef] [PubMed]
- European Parliament Resolution of 4 April 2017 on Palm Oil and Deforestation of Rainforests (2016/2222(INI)). Available online: http://www.europarl.europa.eu/doceo/document/TA-8-2017-0098_EN.pdf (accessed on 20 September 2018).
- Bayona Rodríguez, C.J.; Ávila Diazgranados, R.A.; Rincón Numpaque, A.H.; Romero Angulo, H.M. Emisiones de CO2 de suelo bajo diferentes métodos de renovación en palma de aceite. Rev. Fac. Nac. Agron. Medellín. 2015, 68, 7619–7625. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. Climate change affecting oil palm agronomy, and oil palm cultivation increasing climate change, require amelioration. Ecol. Evol. 2018, 8, 452–461. [Google Scholar] [CrossRef]
- The Amsterdam Declaration in Support of a Fully Sustainable Palm Oil Supply Chain by 2020. Available online: https://www.euandgvc.nl/documents/publications/2015/december/7/declarations-palm-oil (accessed on 22 September 2018).
- European Sustainable Palm Oil Organization (ESPO). Making Sustainable Palm Oil the Norm in Europe. 2017. Available online: https://www.mvo.nl/media/making_sustainable_palm_oil_the_norm_in_europe_-_progress_report_2017.pdf (accessed on 21 September 2018).
- Arum Estrategias Internacionalizacion SL. Informe Económico del Aceite de Palma en España 2018. Available online: https://aceitedepalmasostenible.es/wp-content/uploads/2018/12/Informe-econ%C3%B3mico-del-aceite-de-palma.pdf (accessed on 09 January 2019).
- European Parliament and Council of the European Union. Directive 2009/28/EC of the European Parliament and of the Council of 23 April 2009 on the promotion of the use of energy from renewable sources and amending and subsequently repealing directives 2001/77/EC and 2003/30/EC. Off. J. Eur. Union 2009, 5, 2009. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2009:140:0016:0062:en:PDF (accessed on 22 September 2018).
- Food and Agriculture Organization (FAO). Food Wastage Footprint. Impacts on Natural Resources. 2013. Available online: http://www.fao.org/docrep/018/i3347e/i3347e.pdf (accessed on 25 September 2018).
- Asociación Española de Codificación Comercial (AECOC) 6º Punto de Encuentro AECOC Contra el Desperdicio Alimentario. 2018. Available online: https://www.aecoc.es/minisite/punto-de-encuentro-contra-el-desperdicio-alimentario/ (accessed on 21 September 2018).
- Villadiego, L. La Huella Que Deja el Arroz Que Comes: Del Agua al Efecto Invernadero. El DIARIO 11/01/2018. Available online: https://www.eldiario.es/desalambre/contaminacion-arroz-Asia_0_727227673.html (accessed on 10 October 2018).
- Round Table on Responsible Soy (RTRS) RTRS Standard for Responsible Soy Production. 2017. Available online: http://www.responsiblesoy.org/wpdm-package/rtrs-standard-responsible-soy-production-v3-1/?lang=en (accessed on 23 May 2019).
- Round Table on Responsible Soy (RTRS) RTRS Annual Summary 2018. Available online: http://www.responsiblesoy.org/resumen-anual-de-rtrs-2018/?lang=en (accessed on 23 May 2019).
- Strong Brazilian Soybean Sales Expected to Slow Rebound in Argentine Exports. United States Department of Agriculture (USDA). Oilseeds: World Markets and Trade. United States Department of Agriculture: Foreign Agricultural Service, 2018. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/tx31qh68h/ng451n501/gt54ks07t/oilseeds.pdf (accessed on 24 March 2019).
- UTZ Standard. Available online: https://utz.org/what-we-offer/certification/the-standard/ (accessed on 23 May 2019).
- Drewnowski, A. Why do we like fat? J. Am. Diet. Assoc. 1997, 97 (Suppl. 7), S58–S62. [Google Scholar] [CrossRef]
- WHO/FAO. Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; WHO Technical Report Series 916; WHO: Geneva, Switzerland, 2003; Available online: https://apps.who.int/iris/bitstream/handle/10665/42665/WHO_TRS_916.pdf?ua=1 (accessed on 23 March 2019).
- WHO/FAO. Fats and Fatty Acids in Human Nutrition Report of an Expert Consultation; FAO Food and Nutrition Paper 91; FAO: Rome, Italy, 2010; Available online: https://www.who.int/nutrition/publications/nutrientrequirements/fatsandfattyacids_humannutrition/en/ (accessed on 23 March 2019).
- Valenzuela Báez, R.; Larqué Daza, E.; Valenzuela Bonomo, A. Funciones y metabolismo de los ácidos grasos esenciales y de sus derivados activos. In Tratado de Nutrición, 3rd ed.; Gil, A., Ed.; Editorial Médica Panamericana: Buenos Aires, Argentina, 2017; Volume I, pp. 155–171. [Google Scholar]
- Gesteiro, E.; Galera-Gordo, J.; González-Gross, M. Palm oil and cardiovascular health: Considerations to evaluate the literature critically. Nutr. Hosp. 2018, 35, 1229–1242. [Google Scholar] [CrossRef]
- Krauss, R.M.; Eckel, R.H.; Howard, B.; Appel, L.J.; Daniels, S.R.; Deckelbaum, R.J.; Erdman, J.W., Jr.; Kris-Etherton, P.; Goldberg, I.J.; Kotchen, T.A.; et al. AHA Dietary Guidelines revision 2000: A statement for healthcare professionals from the Nutrition Committee of the American Heart Association. Stroke 2000, 31, 2751–2766. [Google Scholar] [CrossRef]
- FESNAD. Consenso Sobre las Grasas y Aceites en la Alimentación de la Población Española Adulta; Federación Española de Sociedades de Nutrición, Alimentación y Dietética (FESNAD): Madrid, Spain, 2015; pp. 67–70. Available online: http://www.fesnad.org/resources/files/Publicaciones/Consenso_sobre_las_grasas_y_aceites_2015.pdf (accessed on 22 March 2019).
- Ruiz, E.; Ávila, J.M.; Castillo, A.; Valero, T.; del Pozo, S.; Rodriguez, P.; Aranceta-Bartrina, J.; Gil, A.; González-Gross, M.; Ortega, R.M.; et al. Energy intake, profile, and dietary sources in the Spanish population: Findings of the ANIBES Study. Nutrients 2015, 7, 4739–4762. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Pesca y Alimentación (MAPA). Informe de Consumo de Alimentación en España 2018. Available online: https://www.mapa.gob.es/es/alimentacion/temas/consumo-y-comercializacion-y-distribucion-alimentaria/20190624_informedeconsumo2018pdf_tcm30-510816.pdf (accessed on 30 June 2019).
- Manore, M.M.; Larson-Meyer, D.E.; Lindsey, A.R.; Hongu, N.; Houtkooper, L. Dynamic energy balance: An integrated framework for discussing diet and physical activity in obesity prevention—Is it more than eating less and exercising more? Nutrients 2017, 9, 905. [Google Scholar] [CrossRef]
- Ministerio de Educación, Cultura y Deporte. Agencia Española de Protección de la Salud en el Deporte. Actividad Física y Prevalencia de Patologías en la Población Española. 2016. Available online: https://sede.educacion.gob.es/publiventa/descarga.action?f_codigo_agc=17739 (accessed on 16 May 2019).
- Mielgo-Ayuso, J.; Aparicio-Ugarriza, R.; Castillo, A.; Ruiz, E.; Ávila, J.M.; Aranceta-Batrina, J.; Gil, A.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; et al. Physical activity patterns of the Spanish population are mostly determined by sex and age: Findings in the ANIBES Study. PLoS ONE 2016, 11, e0149969. [Google Scholar] [CrossRef]
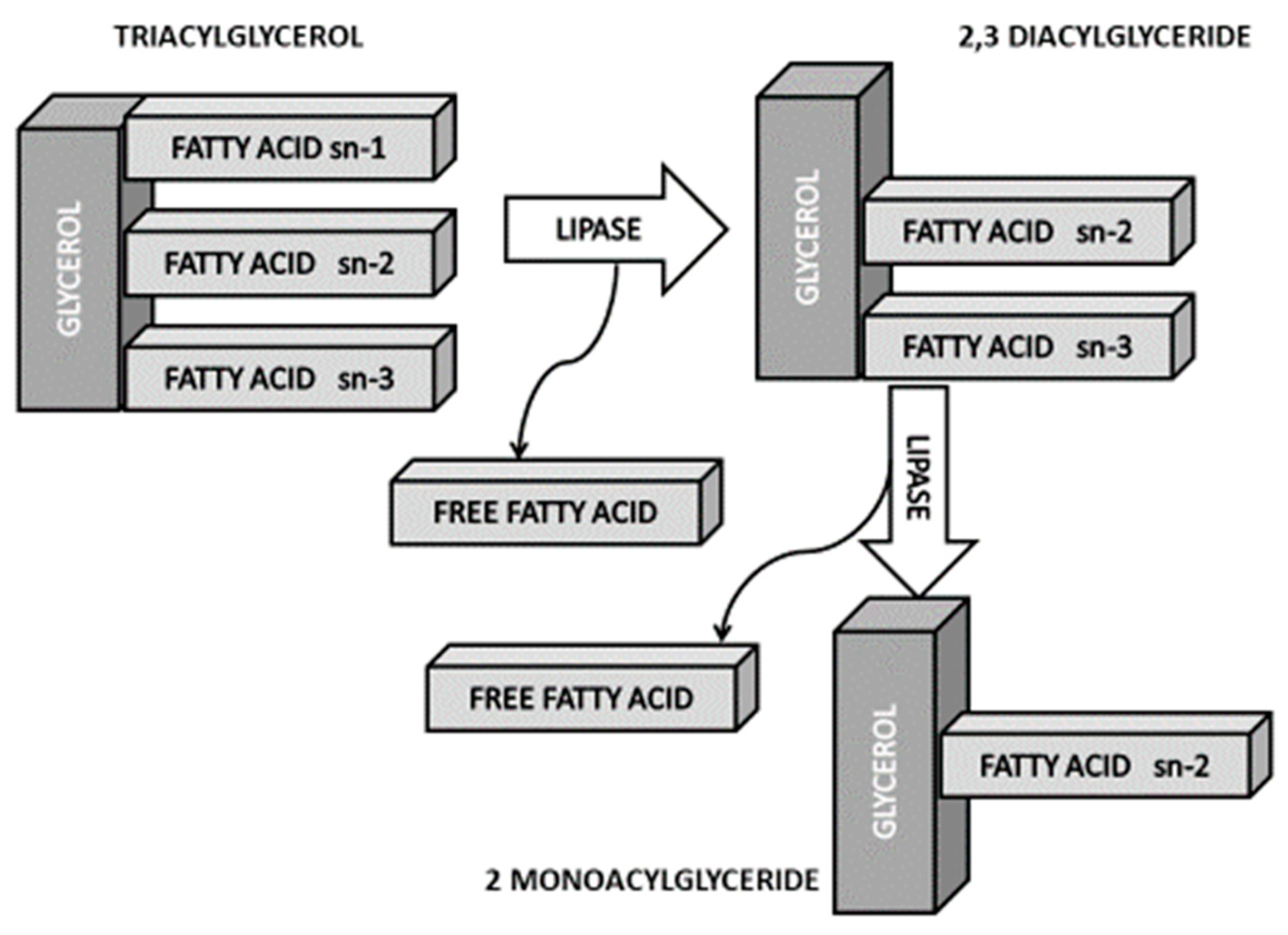
- Berry, S.E. Triacylglycerol structure and interesterification of palmitic and stearic acid-rich fats: An overview and implications for cardiovascular disease. Nutr. Res. Rev. 2009, 22, 3–17. [Google Scholar] [CrossRef]
- Sin Teh, S.; Ong, A.S.H.; Choo, Y.M.; Mah, S.H. sn-2 Hypothesis: A review of the effects of palm oil on blood lipid levels. J. Oleo Sci. 2018, 67, 697–706. [Google Scholar] [CrossRef]
- Codex Alimentarius. Codex Standard for Named Vegetable Oils. Adopted 1999. CODEX STAN 210-1999. 2001. Available online: http://www.fao.org/3/y2774e/y2774e04.htm (accessed on 15 September 2018).
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cudrado, C. Tablas de Composición de Alimentos. Ed Pirámide. 2013. Available online: https://catedraalimentacioninstitucional.files.wordpress.com/2014/09/3-l-tablas_de_composicion_de_alimentos.pdf (accessed on 12 November 2019).
- Ministry of Agriculture, Fisheries and Food (MAFF). Fatty acids. Seventh supplement to 5th edition of McCance and Widdowson’s The Composition of Foods; The Royal Society of Chemistry: Cambridge, UK, 1998. [Google Scholar]
- Fattore, E.; Bosetti, B.; Brighenti, F.; Agostoni, C.; Fattore, G. Palm oil and blood lipid-related markers of cardiovascular disease: A systematic review and meta-analysis of dietary intervention trials. Am. J. Clin. Nutr. 2014, 99, 1331–1350. [Google Scholar] [CrossRef]
- Lucci, P.; Borrero, M.; Ruiz, A.; Pacetti, D.; Frega, N.G.; Diez, O.; Ojeda, M.; Gagliardi, R.; Parra, L.; Angel, M. Palm oil and cardiovascular disease: A randomized trial of the effects of hybrid palm oil supplementation on human plasma lipid patterns. Food Funct. 2016, 7, 347–354. [Google Scholar] [CrossRef]
- Agostoni, C.; Moreno, L.; Shamir, R. Palmitic acid and health: Introduction. Crit. Rev. Food Sci. Nutr. 2016, 56, 1941–1942. [Google Scholar] [CrossRef]
- Innis, S.M. Palmitic acid in early human development. Crit. Rev. Food Sci. Nutr. 2016, 56, 1952–1959. [Google Scholar] [CrossRef]
- Carta, G.; Murru, E.; Banni, S.; Manca, C. Palmitic acid: Physiological role, metabolism and nutritional implications. Front. Physiol. 2017, 8, 902. [Google Scholar] [CrossRef]
- Pascual, G.; Avgustinova, A.; Mejetta, S.; Martín, M.; Castellanos, A.; Attolini, C.S.; Berenguer, A.; Prats, N.; Toll, A.; Hueto, J.A.; et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 2017, 541, 41–45. [Google Scholar] [CrossRef]
- Temme, E.; Mensink, R.P.; Hornstra, G. Effects of medium chain fatty acids (MCFA), myristic acid, and oleic acid on serum lipoproteins in healthy subjects. J. Lipid Res. 1997, 38, 1746–1754. [Google Scholar]
- Chen, B.K.; Seligman, B.; Farquhar, J.W.; Goldhaber-Fiebert, J.D. Multi-country analysis of palm oil consumption and cardiovascular disease mortality for countries at different stages of economic development: 1980–1997. Glob. Health 2011, 7, 45. [Google Scholar] [CrossRef]
- Sun, Y.; Neelakantan, N.; Wu, Y.; Lote-Oke, R.; Pan, A.; van Dam, R.M. Palm oil consumption increases LDL cholesterol compared with vegetable oils low in saturated fat in a meta-analysis of clinical trials. J. Nutr. 2015, 145, 1549–1558. [Google Scholar] [CrossRef]
- Ismail, S.R.; Maarof, S.K.; Siedar Ali, S.; Ali, A. Systematic review of palm oil consumption and the risk of cardiovascular disease. PLoS ONE 2018, 13, e0193533. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Bogensberger, B.; Benčič, A.; Knüppel, S.; Boeing, H.; Hoffmann, G. Effects of oils and solid fats on blood lipids: A systematic review and network meta-analysis. J. Lipid Res. 2018, 59, 1771–1782. [Google Scholar] [CrossRef]
- Di Genova, L.; Cerquiglini, L.; Penta, L.; Biscarini, A.; Esposito, S. Pediatric age palm oil consumption. Int. J. Environ. Res. Public Health 2018, 15, e651. [Google Scholar] [CrossRef]
- WHO. Noncommunicable Diseases Country Profiles 2018. Available online: https://www.who.int/nmh/countries/en/ (accessed on 8 May 2019).
- Sánchez-Muniz, F.J.; Sanz Pérez, B. Hidratación y dieta en la prevención y tratamiento de la obesidad. In Tercer Curso Avanzado Sobre Obesidad; Anales de la Real Academia Nacional de Farmacia: Madrid, Spain, 2016; Volume 82, pp. 106–128. [Google Scholar]
- Zock, P.L.; de Vries, J.H.; Katan, M.B. Impact of myristic acid versus palmitic acid on serum lipid and lipoprotein levels in healthy women and men. Arterioscler. Thromb. 1994, 14, 567–575. [Google Scholar] [CrossRef]
- Forsythe, C.E.; French, M.A.; Goh, Y.K.; Clandinin, M.T. Cholesterolaemic influence of palmitic acid in the sn-1, 3 v. the sn-2 position with high or low dietary linoleic acid in healthy young men. Br. J. Nutr. 2007, 98, 337–344. [Google Scholar] [CrossRef]
- Nestel, P.J.; Noakes, M.; Belling, G.B.; McArthur, R.; Clifton, P.M. Effect on plasma lipids of interesterifying a mix of edible oils. Am. J. Clin. Nutr. 1995, 62, 950–955. [Google Scholar] [CrossRef]
- Cuesta, C.; Sánchez-Muniz, F.J.; Ródenas, S.; Rodríguez-Gil, S.; Merinero, M.C. Lipoprotein profile and serum peroxide levels of aged women consuming palmolein or oleic acid-rich sunflower oil diets. Eur. J. Clin. Nutr. 1998, 52, 675–683. [Google Scholar] [CrossRef][Green Version]
- Steinberg, D.; Parthasarathy, S.; Carew, T.E.; Khoo, J.C.; Witztum, J.L. Beyond cholesterol: Modification of low density lipoproteins that increase its atherogenicity. N. Engl. J. Med. 1989, 320, 915–924. [Google Scholar]
- Ross, R. Atherosclerosis: An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Merinero, M.C.; Rodríguez-Gil, S.; Ordovas, J.M.; Ródenas, S.; Cuesta, C. Dietary fat saturation affects apolipoprotein AII levels and HDL composition in postmenopausal women. J. Nutr. 2002, 132, 50–54. [Google Scholar] [CrossRef]
- Fuchart, J.C.; de Gesteire, C.; Delfly, B.; Castro, G.R. Apolipoprotein A1-containing particles and reverse cholesterol transport: Evidence for connection between cholesterol efflux and atherosclerosis risk. Atherosclerosis 1994, 110, S35–S39. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Oubiña, P.; Ródenas, S.; Benedí, J.; Cuesta, C. Platelet aggregation, thromboxane production and thrombogenic ratio in postmenopausal women consuming high oleic acid-sunflower oil or palm olein. Eur. J. Nutr. 2003, 32, 299–306. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Nus, M. Importancia de la interacción dieta-genética en la prevención cardiovascular. In Genética, Nutrición y Enfermedad; Vaquero, P., Médicos, S.A., Eds.; Instituto Tomás Pascual Sanz y CSIC EDIMSA: Madrid, Spain, 2008; pp. 125–144. [Google Scholar]
- Corella, D.; Ordovás, J.M. Basic concepts in molecular biology related to genetics and epigenetics. Rev. Esp. Cardiol. 2017, 70, 744–753. [Google Scholar] [CrossRef]
- Sun, G.; Xia, H.; Yang, Y.; Ma, S.; Zhou, H.; Shu, G.; Wang, S.; Yang, X.; Tang, H.; Wang, F.; et al. Effects of palm olein and olive oil on serum lipids in a Chinese population: A randomized, double-blind, cross-over trial. Asia Pac. J. Clin. Nutr. 2018, 27, 572–580. [Google Scholar] [CrossRef]
- Bester, D.; Esterhuyse, A.J.; Truter, E.J.; van Rooyen, J. Cardiovascular effects of edible oils: A comparison between four popular edible oils. Nutr. Res. Rev. 2010, 23, 334–348. [Google Scholar] [CrossRef]
- Teng, K.T.; Nagapan, G.; Cheng, H.M.; Nesaretnam, K. Palm olein and olive oil cause a higher increase in postprandial lipemia compared with lard but had no effect on plasma glucose, insulin and adipocytokines. Lipids 2011, 46, 381–388. [Google Scholar] [CrossRef]
- Fattore, E.; Fanelli, R. Palm oil and palmitic acid: A review on cardiovascular effects and carcinogenicity. Int. J. Food Sci. Nutr. 2013, 64, 648–659. [Google Scholar] [CrossRef]
- Voon, P.T.; NG, T.K.W.; Lee, V.K.M.; Nesaretnam, K. Virgin olive oil, palm olein and coconut oil diets do not raise cell adhesion molecules and thrombogenicity indices in healthy Malaysian adults. Eur. J. Clin. Nutr. 2015, 69, 712–716. [Google Scholar] [CrossRef]
- Odia, O.J.; Ofori, S.; Maduka, O. Palm oil and the heart: A review. World J. Cardiol. 2015, 7, 144–149. [Google Scholar] [CrossRef]
- Imoisi, O.B.; Ilori, G.E.; Agho, I.; Ekhator, J.O. Palm oil, its nutritional and health implications. J. Appl. Sci. Environ. Manag. 2015, 19, 127–133. [Google Scholar] [CrossRef]
- Mancini, A.; Imperlini, E.; Nigro, E.; Montagnese, C.; Daniele, A.; Orrú, S.; Buono, P. Biological and nutritional properties of palm oil and palmitic acid: Effects on health. Molecules 2015, 20, 17339–17361. [Google Scholar] [CrossRef]
- Kübeck, R.; Bonet-Ripoll, C.; Hoffmann, C.; Walker, A.; Müller, V.M.; Schüppel, V.L.; Lagkouvardos, I.; Scholz, B.; Engel, K.H.; Daniel, H.; et al. Dietary fat and gut microbiota interactions determine diet-induced obesity in mice. Mol. Metab. 2016, 5, 1162–1174. [Google Scholar] [CrossRef]
- Just, S.; Mondot, S.; Ecker, J.; Wegner, K.; Rath, E.; Gau, L.; Streidl, T.; Hery-Arnaud, G.; Schmidt, S.; Lesker, T.R.; et al. The gut microbiota drives the impact of bile acids and fat source in diet on mouse metabolism. Microbiome 2018, 6, 134. [Google Scholar] [CrossRef]
- European Feed Manufacturers’ Federation (FEFAC) Annual Report 2017–2018. Available online: https://www.fefac.eu/files/83625.pdf (accessed on 24 February 2019).
- Ministerio de Agricultura, Pesca y Alimentación (MAPA). Datos de Producción de Piensos 2017. Available online: https://www.mapa.gob.es/es/ganaderia/temas/alimentacion-animal/informedatosdeproducciondepiensosenespana2017_tcm30-459584.pdf (accessed on 24 February 2019).
- Ley 17/2011, de 5 de Julio, de Seguridad Alimentaria y Nutrición. Available online: https://www.boe.es/buscar/act.php?id=BOE-A-2011-11604 (accessed on 2 April 2019).
- Report from the Commission to the European Parliament and the Council Regarding Trans Fats in Foods and in the Overall Diet of the Union Population. 2015. Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/fs_labelling-nutrition_trans-fats-report_en.pdf (accessed on 15 January 2019).
- Commission Regulation (EU) 2019/649 of 24 April 2019 Amending Annex III to Regulation (EC) No 1925/2006 of the European Parliament and of the Council as Regards Trans Fat, Other Than Trans Fat Naturally Occurring in Fat of Animal Origin. Available online: http://data.europa.eu/eli/reg/2019/649/oj (accessed on 15 January 2019).
- European Parliament Resolution of 26 October 2016 on Trans Fats (TFAs) (2016/2637(RSP)). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52016IP0417&from=EN (accessed on 15 January 2019).
- Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the Provision of Food Information to Consumers, Amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, and Repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No 608/2004. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32011R1169&from=ES (accessed on 6 November 2019).
- Boletin Oficial del Estado (BOE) Orden de 26 de enero de 1989 por la que se aprueba la Norma de Calidad para los Aceites y Grasas Calentados. BOE nº 26 31/01/1989. pp. 2665–2667. Available online: https://www.boe.es/eli/es/o/1989/01/26/(1)/con (accessed on 24 July 2019).
- Arroyo, R.; Cuesta, C.; Sánchez-Montero, J.M.; Sánchez-Muniz, F.J. High performance size exclusion chromatography of palm olein used for frying. Fat Scid. Technol. 1995, 97, 292–296. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Bastida, S.; Márquez-Ruiz, G.; Dobarganes, M.C. Effects of heating and frying on oil and food fatty acids. In Fatty Acids in Foods and Their Health Implications, 3rd ed.; Chow, C.K., Ed.; CRC Press, Taylor and Francis Book: Boca Ratón, FL, USA, 2008; pp. 511–543. [Google Scholar]
- Bastida, S.; Sánchez-Muniz, F.J. Frying a cultural way of cooking in the Mediterranean diet. In The Mediterranean Diet: A Comprehensive Approach; Preedy, V.R., Watson, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 217–234. [Google Scholar]
- Sánchez-Muniz, F.J.; Benedí, J.; Bastida, S.; Olivero-David, R.; González-Muñoz, M.J. Enzymes and thermally oxidized oils and fats. In Frying of Foods: Oxidation, Nutrient and Non-Nutrient Antioxidants, Biologically Active Compounds and High Temperatures, 2nd ed.; Boskou, D., Elmadfa, I., Eds.; CRC Press, Taylor and Francis Group LLC: Boca Ratón, FL, USA, 2011; pp. 105–155. [Google Scholar]
- Olivero-David, R.; Sánchez-Muniz, F.J.; Bastida, S.; Benedí, J.; González-Muñoz, M.J. Gastric emptying and short-term digestibility of thermally oxidized sunflower oil used for frying in fasted and non-fasted rats. J. Agric. Food Chem. 2010, 58, 9242–9248. [Google Scholar] [CrossRef]
- MacMahon, S.; Begley, T.H.; Diachenko, G.W. Occurrence of 3-MCPD and glycidyl esters in edible oils in the United States. Food Addit. Contam. Part A 2013, 30, 2081–2092. [Google Scholar] [CrossRef]
- Nagy, K.; Sandoz, L.; Craft, B.D.; Destaillats, F. Mass-defect filtering of isotope signatures to reveal the source of chlorinated palm oil contaminants. Food Addit. Contam. Part A 2011, 28, 1492–1500. [Google Scholar] [CrossRef]
- Joint FAO/WHO Expert Comitee on Food Aditives; 83rd Meeting Rome, 8–17 November 2016. Summary and Conclusions JECFA/83/SC. 2016. Available online: http://www.fao.org/3/a-bq821e.pdf (accessed on 17 January 2019).
- European Food Safety Authority (EFSA). Panel on Contaminants in the Food Chain (CONTAM) Risks for human health related to the presence of 3- and 2-monochloropropanediol (MCPD), and their fatty acid esters, and glycidyl fatty acid esters in food. EFSA J. 2016, 14, 4426. [Google Scholar]
- Matthäus, B.; Pudel, F.; Fehling, P.; Vosmann, K.; Freudenstein, A. Strategies for the reduction of 3-MCPD esters and related compounds in vegetable oils. Eur. J. Lipid Sci. Technol. 2011, 113, 380–386. [Google Scholar] [CrossRef]
- Willits, J. Los 3-MCPD y su importancia en la industria del aceite de palma. Palmas 2013, 34, 165–173. [Google Scholar]
- Custodio-Mendoza, J.A.; Carro, A.M.; Lage-Yusty, M.A.; Herrero, A.; Valente, I.M.; Rodrigues, J.A.; Lorenzo, R.A. Occurrence and exposure of 3-monochloropropanedioldiesters in edible oils and oil-based foodstuffs from Spanish market. Food Chem. 2019, 270, 214–222. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Analysis of occurrence of 3-monochloropropane-1, 2-diol (3-MCPD) in food in Europe in the years 2009–2011 and preliminary exposure assessment. EFSA J. 2013, 11, 3381. [Google Scholar] [CrossRef]
- Wong, Y.H.; Muhamad, H.; Abas, F.; Lai, O.M.; Nyam, K.L.; Tan, C.P. Effects of temperature and NaCl on the formation of 3-MCPD esters and glycidyl esters in refined, bleached and deodorized palm olein during deep-fat frying of potato chips. Food Chem. 2017, 219, 126–130. [Google Scholar] [CrossRef]
- Velayuthan, A. Procesamiento y control del aceite de palma. Palmas 1986, 7, 19–33. [Google Scholar]
- Oot, C.K.; Choo, Y.M.; Yap, S.C.; Ma, A.N. Refinación del aceite rojo de palma. Palmas 1997, 18, 79–85. [Google Scholar]
- European Food Safety Authority (EFSA) Panel on Contaminants in the Food Chain (CONTAM) Update of the risk assessment on 3-monochloropropane diol and its fatty acid esters. EFSA J. 2018, 16, 508.
- Ruiz, E.; Ávila, J.M.; Valero, T.; del Pozo, S.; Rodriguez, P.; Aranceta-Bartrina, J.; Gil, A.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; et al. Macronutrient distribution and dietary sources in the Spanish population: Findings from the ANIBES study. Nutrients 2016, 8, 177. [Google Scholar] [CrossRef]
- Agencia Española de Consumo, Seguridad Alimentaria y Nutrición (AECOSAN). Encuesta ENALIA. Encuesta Nacional de Alimentación en la población Infantil y Adolescente. Available online: http://www.aecosan.msssi.gob.es/AECOSAN/web/seguridad_alimentaria/ampliacion/enalia.htm (accessed on 19 January 2019).
- Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006R1881&from=ES (accessed on 17 January 2019).
- Commission Regulation (EU) 2018/290 of 26 February 2018 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Glycidyl Fatty Acid Esters in Vegetable Oils and Fats, Infant Formula, Follow-on Formula and Foods for Special Medical Purposes Intended for Infants and Young Children. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32018R0290&qid=1562972106866&from=EN (accessed on 17 January 2019).
- IARC Monographs Data Base 101. pp. 349–374. Available online: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono101-010.pdf (accessed on 12 January 2019).
- IARC Monographs Data Base 77. pp. 469–486. Available online: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono77-19.pdf (accessed on 12 January 2019).
- Cheng, W.; Liu, G.; Wang, L.; Liu, Z. Glycidyl fatty acid esters in refined edible oils: A review on formation, occurrence, analysis, and elimination methods. Compr. Rev. Food Sci. Food Saf. 2017, 16, 263–281. [Google Scholar] [CrossRef]
- Šmidrkal, J.; Tesařová, M.; Hrádková, I.; Berčíková, M.; Adamčíková, A.; Filip, V. Mechanism of formation of 3-chloropropan-1,2-diol (3-MCPD) esters under conditions of the vegetable oil refining. Food Chem. 2016, 211, 124–129. [Google Scholar] [CrossRef]
- Clemens, R.; Wallace Hayes, A.; Sundram, K.; Pressman, P. Palm oil and threats to a critically important food source: The chloropropanols—Caution, controversy, and correction. Toxicol. Res. Appl. 2017, 1, 1–9. [Google Scholar] [CrossRef]
- Asociación Española de Pediatría. Comunicado AEP y SEGNP Aceite de Palma y Ácido Palmítico en la Alimentación Infantil. 2017. Available online: http://www.aeped.es/comite-nutricion/noticias/comunicado-sobre-aceite-palma-y-acido-palmitico-en-alimentacion-infantil (accessed on 13 January 2019).
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406, Erratum in 2014, 160, 658. [Google Scholar] [CrossRef]
- Bier, D.M. Saturated fats and cardiovascular disease: Interpretations not as simple as they once were. Crit. Rev. Food Sci. Nutr. 2016, 56, 1943–1946. [Google Scholar] [CrossRef]
- Nettleton, J.A.; Brouwer, I.A.; Mensink, R.P.; Diekman, C.; Hornstra, G. Fats in foods: Current evidence for dietary advice. Ann. Nutr. Metab. 2018, 72, 248–254. [Google Scholar] [CrossRef]
- Stamler, J. Diet-heart: A problematic revisit. Am. J. Clin. Nutr. 2010, 91, 497–499. [Google Scholar] [CrossRef]
- Szajewska, H.; Szajewski, T. Saturated fat controversy: Importance of systematic reviews and meta-analyses. Crit. Rev. Food Sci. Nutr. 2016, 56, 1947–1951. [Google Scholar] [CrossRef]
- Mensink, R.P.; Zock, P.L.; Kester, A.D.; Katan, M.B. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: A meta-analysis of 60 controlled trials. Am. J. Clin. Nutr. 2003, 77, 1146–1155. [Google Scholar] [CrossRef]
| Goal 1 | Goal 7 | Goal 13 |
| No poverty | Affordable and clean energy | Climate action |
| Goal 2 | Goal 8 | Goal 14 |
| Zero hunger | Decent work and economic growth | Life below water |
| Goal 3 | Goal 9 | Goal 15 |
| Good health and well-being | Industry, innovation, and infrastructure | Life on land |
| Goal 4 | Goal 10 | Goal 16 |
| Quality education | Reducing inequality | Peace, justice, and strong institutions |
| Goal 5 | Goal 11 | Goal 17 |
| Gender equality | Sustainable cities and communities | Partnerships for the goals |
| Goal 6 | Goal 12 | |
| Clean water and sanitation | Responsible consumption and production |
| 1. Behave ethically and transparently |
| 2. Operate legally and respect rights |
| 3. Optimize productivity, efficiency, positive impacts and resilience |
| 4. Respect community and human rights and deliver benefits |
| 5. Support smallholder inclusion |
| 6. Respect workers’ rights and conditions |
| 7. Protect, conserve and enhance ecosystems and the environment |
| WHO/FAO (2010) [37] | FESNAD (2015) [41] | AHA (2000) [40] | |
|---|---|---|---|
| Total fat | 20%–35% | 20%–40% | 30% |
| SFA | <10% | The recommendations are reducing the SFA-rich foods intake. | <10% |
| MUFA | Calculated by difference | 12%–30% (27–67 g/day) 20%–25% (45–55 g/day) * | Calculated by difference |
| PUFA | 6%–11% | <10% | |
| PUFA W3 Alfa linolenic acid DHA + EPA | 0.1%–1% (0.25–2.25 g/day) 0.5–1% | ||
| PUFA W6 | 0.5%–2% | 5%–10% (10–20 g/day) | |
| Trans-FAs | <1% | 2%–3% Reduce the intake as much as possible | |
| Cholesterol | <300 mg/day |
| Fatty Acid | Palm Oil | Palm Stearin | Palm Olein | Palm Superolein | Palm Kernel Oil | Palm Kernel Stearin | Palm Kernel Olein |
|---|---|---|---|---|---|---|---|
| C6:0 | ND | ND | ND | ND | ND-0.8 | ND-0.2 | ND-0.7 |
| C8:0 | ND | ND | ND | ND | 2.4–6.2 | 1.3–3.0 | 2.9–6.3 |
| C10:0 | ND | ND | ND | ND | 2.6–5.0 | 2.4–3.3 | 2.7–4.5 |
| C12:0 | ND-0.5 | 0.1–0.5 | 0.1–0.5 | 0.1–0.5 | 45.0–55.0 | 52.0–59.7 | 39.7–47.0 |
| C14:0 | 0.5–2.0 | 1.0–2.0 | 0.5–1.5 | 0.5–1.5 | 14.0–18.0 | 20.0–25.0 | 11.5–15.5 |
| C16:0 | 39.3–47.5 | 48.0–74.0 | 38.0–43.5 | 30.0–39.0 | 6.5–10.0 | 6.7–10.0 | 6.2–10.6 |
| C16:1 | ND-0.6 | ND-0.2 | ND-0.6 | ND-0.5 | ND-0.2 | ND | ND-0.1 |
| C17:0 | ND-0.2 | ND-0.2 | ND-0.2 | ND-0.1 | ND | ND | ND |
| C17:1 | ND | ND-0.1 | ND-0.1 | ND | ND | ND | ND |
| C18:0 | 3.5–6.0 | 3.9–6.0 | 3.5–5.0 | 2.8–4.5 | 1.0–3.0 | 1.0–3.0 | 1.7–3.0 |
| C18:1 | 36.0–44.0 | 15.5–36.0 | 39.8–46.0 | 43.0–49.5 | 12.0–19.0 | 4.1–8.0 | 14.4–24.6 |
| C18:2 | 9.0–12.0 | 3.0–10.0 | 10.0–13.5 | 10.5–15.0 | 1.0–3.5 | 0.5–1.5 | 2.4–4.3 |
| C18:3 | ND-0.5 | ND-0.5 | ND-0.6 | 0.2–1.0 | ND-0.2 | ND-0.1 | ND-0.3 |
| C20:0 | ND-1.0 | ND-1.0 | ND-0.6 | ND-0.4 | ND-0.2 | ND-0.5 | ND-0.5 |
| C20:1 | ND-0.4 | ND-0.4 | ND-0.4 | ND-0.2 | ND-0.2 | ND-0.1 | ND-0.2 |
| C20:2 | ND | ND | ND | ND | ND | ND | ND |
| C22:0 | ND-0.2 | ND-0.2 | ND-0.2 | ND-0.2 | ND-0.2 | ND | ND |
| C22:1 | ND | ND | ND | ND | ND | ND | ND |
| C22:2 | ND | ND | ND | ND | ND | ND | ND |
| C24: 0 | ND | ND | ND | ND | ND | ND | ND |
| C24:1 | ND | ND | ND | ND | ND | ND | ND |
| Reference | Type of Study and Aims | Results and Conclusions |
|---|---|---|
| Bester et al. [78] | Review. To identify the benefits and risks of four edible oils (olive, sunflower, fish and palm oils) for cardiovascular health. | The studies reviewed here suggest that all four oils could be suitable for inclusion in a healthy diet. Beneficial effects have been identified for each one, although in some cases there remains some dispute. |
| In the case of palm oil, the bulk of the studies have addressed its effects on the serum lipid profile, concluding that the lipid profile is unaffected, and can even reduce atherosclerosis and prevent cardiac ischaemia. The few studies addressing the effect of palm oil on arrhythmogenesis are inconclusive, although it can apparently have a slight anti-arrhythmogenetic effect. | ||
| The studies performed with extracts of the tocotrienol-rich fraction of palm oil suggest that it may offer protection against myocardial ischaemia-reperfusion injury. However, these effects require further study with dietary supplementation models. | ||
| Studies with red palm oil (RPO), which contains the same components as refined oil but also contains more micronutrients, confirm the above-described effects of palm oil, enhanced by the presence of more micronutrients. It has also been shown that dietary supplementation with RPO offers protection against myocardial ischaemia-reperfusion injury. Again, however, further studies with RPO are needed to fully confirm these effects on risk factors of ischaemia and cardiac arrhythmias. | ||
| In conclusion, palm oil has shown only a mild effect on the lipid profile, and so there is no saying whether its consumption would have any beneficial effect in persons at cardiovascular risk (CVR). Nonetheless, it could be useful for some people to consume this oil, since it is one of the few oils to have demonstrated protective effect against myocardial ischaemia-reperfusion injury. | ||
| Teng et al. [79] | Single-blind randomized crossover trial (n = 10 healthy males) comparing the effects of high intake of fat (50 g) rich in palmitic acid of both plant (palm olein) and animal (pork lard) origin versus consumption of a fat rich in oleic acid (virgin olive oil) on lipaemia, plasma glucose, insulin and adipocytokines. | The serum triglyceride (TG) concentration was significantly lower after consumption of pork lard than after consumption of olive oil or palm olein (meal effect p = 0.003; time effect p < 0.001). There was also a greater reduction in plasma levels of non-esterified free fatty acids (FFA) in the group that consumed pork lard than in the group that consumed olive oil (p < 0.05). The differences in the physical and structural characteristics of pork lard TGs may account to some extent for the fact that the increase of postprandial lipaemia was smaller than in the case of palm olein. The effects of palm olein and olive oil on postprandial lipaemia were similar. |
| Levels of plasma glucose, insulin and adipocytokines [interleukin-6 (IL-6), tumoral necrosis factor-α (TNF-α), interleukine-1β (IL-1β) and leptin] were unaffected by the type of fat consumed. In the case of plasma IL-1β, there was such a change over time after taking 3 meals with a high-fat diet (p = 0.036). These results could suggest that it is a high fat intake, rather than the type of fatty acid (FA), that affects the response of pro-inflammatory markers. | ||
| Fattore and Fanelli [80] | Review of the scientific evidence on the relationship between palm oil and adverse health effects. | The main reason why consumption of palm oil is associated with adverse health effects is that it contains relatively high concentrations of saturated fatty acids (SFAs), particularly palmitic acid, which in turn have been associated with increased risk of coronary heart disease (CHD) and some types of tumor. However, recent research on the subject has reconsidered the negative role of dietary SFAs as a CVR factor and has shown that not only the type of fat but also the structure of the TGs plays an important part in cholesterolaemia. |
| Some studies have concluded that modification of the type of fat, or modification combined with reduction (but not only reduction) appear to reduce the incidence of cardiovascular events in high-risk subjects. Moreover, most studies posit a possible protective effect from partial replacement of SFAs by polyunsaturated (PUFAs) as opposed to the adverse effect of SFAs per se. This is indicative of the importance of continuing research to improve the balance between SFAs and PUFAs in foods. | ||
| The process of interesterification, which causes a reordering of palmitic acid and does not occur naturally in native oil, could be associated with potentially adverse health effects and hence is to be discouraged. | ||
| As for its role in the development of cancer, the studies are few and the evidence unconvincing. | ||
| Despite these uncertainties, this review does not demonstrate a negative role of palmitic acid in health, and much less native palm oil, which is a complex alimentary matrix in which palmitic acid is only one of the components. Palm oil also contains other FAs, chiefly oleic acid, along with antioxidant compounds, which may have compensatory effects. | ||
| Fattore et al. [52] | Systematic review and meta-analysis of dietary intervention assays, to evaluate the effect on lipid markers for CHD and cardiovascular disease (CVD) of replacing palm oil with other primary dietary fats. The review includes a total of 51 intervention studies ranging in duration from 2 to 16 weeks, in which palm oil-rich diets were compared with diets rich in other fats, and in which at least one or more of the following CHD or CVD biomarkers were determined: total cholesterol (TC), cholesterol transported by low density lipoproteins (LDLc), cholesterol transported by high density lipoproteins (HDLc), TC/HDLc ratio, TG, apolipoprotein (Apo) A-I and Apo B. | Levels of TC, LDLc, Apo B, HDLc and Apo A-I were significantly higher in palm oil-rich diets than in diets rich in stearic acid, monounsaturated fatty acids (MUFAS) and PUFAs whereas most of these biomarkers were significantly lower than in diets rich in myristic and lauric acids. |
| Comparison of the effect of palm oil-rich diets with diets rich in trans-FAs showed that HDLc and Apo-A-I concentrations were significantly higher, while Apo B, and TG concentrations and TC/HDLc ratio were lower. | ||
| This meta-analysis indicates that replacement of palm oil by other fats (SFAs, MUFAs, PUFAs) produces both favorable and unfavorable changes in biomarkers for CVD and CHD. However, when palm oil was replaced by trans-FAs, only favorable changes were found. | ||
| The results do not support an association between palm oil replacement and reduction of mortality from CVD. They rather suggest a need for fresh comparative research and also call for caution in the development of policies for the general population that promote the use of some specific fats over others. More solid evidence is required as to the effects of palm oil on health and on the socio-economic consequences before embarking on such policies. | ||
| Voon et al. [81] | Randomized crossover trial of 5 weeks with a total of 45 volunteers. To assess the effects of a (typical Malaysian) diet containing a high proportion of protein and supplemented with virgin olive oil, palm olein or coconut oil on arteriosclerosis markers (cell adhesion molecules, lipid inflammatory mediators and thrombogenicity indices) in healthy adults. | SFA-enriched diets (palm olein and coconut oil), and also a diet with virgin olive oil, containing high levels of oleic acid, have similar effects on arteriosclerosis markers, such as cell adhesion molecules and prostaglandin E2 (PGE2) and thromboxane B2 (TXB2)/Prostaglandin F1α (PGF1α) thrombogenicity indices. |
| Only the diet supplemented with virgin olive oil exhibited a lower level of the leukotriene B4 (LTB4) pro-inflammatory marker than the other two diets assayed. | ||
| The results indicate that it could be premature labelling palm olein and coconut oil as “bad oils” because of their contribution to increased CVR based solely on their effects on the serum lipid profile. | ||
| Odia et al. [82] | Review of experimental studies in animals and humans on the association between palm oil and its constituents and serum lipid profile and CVD. | Many scientific studies, both in animals and human beings, clearly show that consumption of palm oil does not cause a rise in serum TC levels and that it is not atherogenic. Apart from palmitic acid, palm oil is composed of oleic and linoleic acids and also contains vitamins A and E, which are powerful antioxidants. It has been demonstrated scientifically that palm oil protects the heart and blood vessels from plaque and ischaemic lesions. |
| Consumed as part of a healthy, balanced diet, it does not cause increased CVR. Replacing this oil with others rich in MUFAs and PUFAs would not produce any additional benefit. | ||
| This review concludes that more longitudinal studies are required to assess the impact on CVR of diets containing palm oil in comparison with diets containing other oils accepted as “beneficial for cardiovascular health”, such as olive oil, analyzing lipid parameters as risk markers. | ||
| Imoisi et al. [83] | Review on palm oil: composition and health implications. | Prospective epidemiological studies have shown that the level of plasma TGs, especially after eating, is a major factor in the pathogenesis of CHD. Palm oil is considered as to raise TC and to increase the CVR because of its SFA content (44% palmitic acid and 5% stearic acid). However, there is a growing body of scientific evidence indicating that the effect of palm oil on TC is relatively neutral compared to other fats and oils. Palm oil raises TC only when an excess of dietary cholesterol is present in the diet. |
| Palm oil stimulates the synthesis of HDLc and the elimination of LDLc. | ||
| Palm oil is rich in vitamin E (particularly tocotrienols), which can reduce serum TC concentrations and has powerful antioxidant effects. These benefits occur especially from non-oxidized palm oil. Oxidized palm oil can have an adverse effect on lipid profile, FFAs and phospholipids. | ||
| In conclusion, consumption of palm oil as a fat source in the diet does not pose any additional risk of CHD when consumed in realistic quantities as part of a healthy diet. Consumption of oxidized palm oil should be avoided owing to its adverse effects on lipid profiles. | ||
| Mancini et al. [84] | Review on the functional role of palm oil and palmitic acid in the development of obesity, type 2 diabetes mellitus, CVD, and cancer. It also discusses the atherogenic potential of palmitic acid and its stereospecific position in TGs. | As to the possible effects of palm oil-rich diets on CVR in humans, the studies reviewed here report conflicting results. The main criticisms of these may be summarized as follows: (a) considerable qualitative and quantitative heterogeneity of FA concentrations in the diets; (b) differences in selection criteria used to form the trial and control groups; (c) wide age range addressed in the studies; and (d) little attention is paid to other dietary components which could confound the direct effects of FAs on blood lipid markers. |
| To date no clear evidence has been found to demonstrate beyond a doubt the association of consumption of palm oil with increased CVR, particularly in normo-cholesterolaemic subjects, assuming the recommended intake of PUFAs. | ||
| Moreover, the percentage of palmitic acid at position sn-2 on the TG is smaller in palm oil than in animal fats, which supports the hypothesis that palm oil has little atherogenic potential in the context of a balanced diet, and that the alleged adverse effects may be due to a dose-response ratio. | ||
| This review posit the need to carry out more rigorous research to define the advantages and disadvantages of palm oil consumption as regards CVR. | ||
| Sun et al. [60]. | Systematic review on the effect of palm oil consumption on blood lipids compared with other cooking oils, based on data from clinical trials (minimum duration 2 weeks, comparing the effects of palm oil consumption with that of other oils such as; low-SFA vegetable oils, partially hydrogenated vegetable oils containing trans-FAs and animal fats). | Palm oil significantly increased LDLc by 0.24 mmol/L (95% CI: 0.13, 0.35 mmol/L; I(2) = 83.2%) compared with other low-SFA vegetable oils. This effect was observed in randomized but not in non-randomized trials. Moreover, among randomized trials, only modest differences in study results were observed regarding the effects of palm oil versus LDLc. SFAs from palm oil appear to have the same effects on LDLc as animal fat. |
| Palm oil increased HDLc by 0.02 mmol/L compared with the low-SFA vegetable oils, and by 0.09 mmol/L compared with oils containing trans-FAs. | ||
| In conclusion, the consumption of palm oil results in a higher level of LDLc than that of low-SFA vegetable oils, and in a higher level of HDLc if compared with the effect of oils containing trans-SFAs. This would tend to support the recommendation to reduce the use of palm oil and replace it with low-SFA and trans-FA vegetable oils. | ||
| In the case of foods that are consumed in small quantities which are made using trans-FAs for their sensory characteristics, palm oil can be an alternative thanks to its better effects on HDLc. | ||
| Marangoni et al. [3] | Summary of the symposium‘s conclusions on the use of palm oil in the food industry, produced by experts from several Italian medical and nutritional scientific societies and collected by the nutrition foundation of Italy. Toxicological and environmental issues were not considered. | The main conclusions of the Symposium may be summarized thus: |
| ||
| ||
| ||
| Palm oil intake does not appear to be a public health priority in Italy, in light of its overall effects and available consumption data. |
| Mean Content (mg/kg) (2009) | Maximum Content (mg/kg) (2009) | Mean Content (ppm) (2011) | Maximum Content (ppm) (2011) | Mean Content (mg/kg) (2019) | Maximum Content (mg/kg) (2019) | |
|---|---|---|---|---|---|---|
| Canola oil | 0.3 | 1.5 | 1 | 1 | 1.03 (*) | N |
| Soya oil | N | N | 0.5 | 0.6 | N | N |
| Sunflower oil | 1 | 5.7 | 2 | 4 | 1.82 | 2.05 |
| Corn oil | 2.8 | 7 | 7 | 9 | 0.9 | 1.55 |
| Coconut oil | N | N | 7 | 7.5 | Nd | Nd |
| Palm oil | 4.5 | 13 | 6 | 14 | 6.22 (*) | N |
| Sesame oil | N | N | N | N | 1.6 | 1.63 |
| Safflower oil | N | N | N | N | 2.78 (*) | N |
| Extra virgin olive oil | N | N | N | N | Nd | Nd |
| Refined olive oil 1° | N | N | N | N | 0.78 | 1.11 |
| Refined olive oil 0.4° | N | N | N | N | 0.95 | 1.22 |
| Fried extra virgin olive oil | N | N | N | N | 0.93 | 1.72 |
| Foodstuffs | Maximum Level (μg/kg) | |
|---|---|---|
| 4.1 | 3-Monochloropropanediol (3-MCPD) | |
| 4.1.1 | Hydrolysed vegetable protein | 20 |
| 4.1.2 | Soy sauce | 20 |
| 4.2 | Glycidyl Fatty Acid Esters Expressed as Glycidol | |
| 4.2.1 | Vegetable oils and fats placed on the market for the final consumer or for use as an ingredient in food with the exception of the foods referred to in 4.2.2 | 1000 |
| 4.2.2 | Vegetable oils and fats destined for the production of baby food and processed cereal-based food for infant and young children | 500 |
| 4.2.3 | Infant formula, follow-on formula and foods for special medical purposes intended for infants and young children (powder) | 75 until 30 June 2019. 50 as from 1 July 2019 |
| 4.2.4 | Infant formula, follow-on formula and foods for special medical purposes intended for infants and young children (liquid) | 10.0 until 30 June 2019. 6.0 as from 1 July 2019 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gesteiro, E.; Guijarro, L.; Sánchez-Muniz, F.J.; Vidal-Carou, M.d.C.; Troncoso, A.; Venanci, L.; Jimeno, V.; Quilez, J.; Anadón, A.; González-Gross, M. Palm Oil on the Edge. Nutrients 2019, 11, 2008. https://doi.org/10.3390/nu11092008
Gesteiro E, Guijarro L, Sánchez-Muniz FJ, Vidal-Carou MdC, Troncoso A, Venanci L, Jimeno V, Quilez J, Anadón A, González-Gross M. Palm Oil on the Edge. Nutrients. 2019; 11(9):2008. https://doi.org/10.3390/nu11092008
Chicago/Turabian StyleGesteiro, Eva, Luis Guijarro, Francisco J. Sánchez-Muniz, María del Carmen Vidal-Carou, Ana Troncoso, Lluis Venanci, Vicente Jimeno, Joan Quilez, Arturo Anadón, and Marcela González-Gross. 2019. "Palm Oil on the Edge" Nutrients 11, no. 9: 2008. https://doi.org/10.3390/nu11092008
APA StyleGesteiro, E., Guijarro, L., Sánchez-Muniz, F. J., Vidal-Carou, M. d. C., Troncoso, A., Venanci, L., Jimeno, V., Quilez, J., Anadón, A., & González-Gross, M. (2019). Palm Oil on the Edge. Nutrients, 11(9), 2008. https://doi.org/10.3390/nu11092008





