Associations between the Prenatal Diet and Neonatal Outcomes—A Secondary Analysis of the Cluster-Randomised GeliS Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
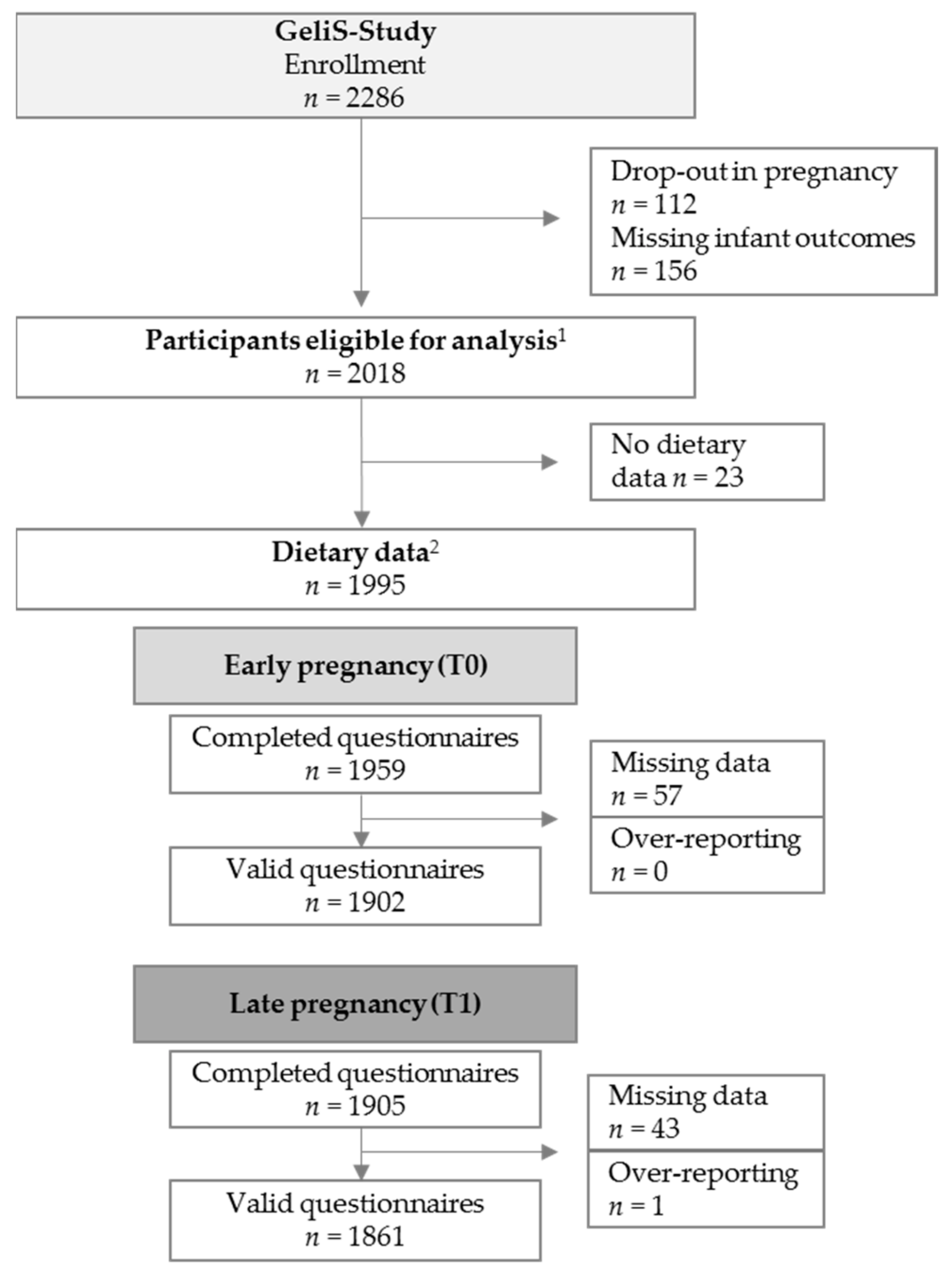
2.2. Study Participants
2.3. The Lifestyle Intervention Program
2.4. Study Outcomes
2.5. Data Collection and Processing
2.6. Statistical Analysis
3. Results
3.1. Study Participants and Baseline Characteristics
3.2. Associations between the Maternal Diet and Infant Weight and Weight-Related Parameters
3.3. Diet Quality and Infant Weight Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schellong, K.; Schulz, S.; Harder, T.; Plagemann, A. Birth weight and long-term overweight risk: Systematic review and a meta-analysis including 643,902 persons from 66 studies and 26 countries globally. PLoS ONE 2012, 7, e47776. [Google Scholar] [CrossRef]
- Harder, T.; Rodekamp, E.; Schellong, K.; Dudenhausen, J.W.; Plagemann, A. Birth weight and subsequent risk of type 2 diabetes: A meta-analysis. Am. J. Epidemiol. 2007, 165, 849–857. [Google Scholar] [CrossRef]
- Kim, S.Y.; Sharma, A.J.; Sappenfield, W.; Wilson, H.G.; Salihu, H.M. Association of maternal body mass index, excessive weight gain, and gestational diabetes mellitus with large-for-gestational-age births. Obstet. Gynecol. 2014, 123, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Lau, E.Y.; Liu, J.; Archer, E.; McDonald, S.M.; Liu, J. Maternal weight gain in pregnancy and risk of obesity among offspring: A systematic review. J. Obes. 2014, 2014, 524939. [Google Scholar] [CrossRef] [PubMed]
- Mamun, A.A.; Mannan, M.; Doi, S.A.R. Gestational weight gain in relation to offspring obesity over the life course: A systematic review and bias-adjusted meta-analysis. Obes. Rev. 2014, 15, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Fraser, A.; Tilling, K.; Macdonald-Wallis, C.; Sattar, N.; Brion, M.-J.; Benfield, L.; Ness, A.; Deanfield, J.; Hingorani, A.; Nelson, S.M.; et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 2010, 121, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Morales, M.E.; Bacardi-Gascon, M.; Jimenez-Cruz, A. Association of excessive GWG with adiposity indicators and metabolic diseases of their offspring: Systematic review. Nutr. Hosp. 2015, 31, 1473–1480. [Google Scholar] [CrossRef]
- Grieger, J.A.; Clifton, V.L. A review of the impact of dietary intakes in human pregnancy on infant birthweight. Nutrients 2014, 7, 153–178. [Google Scholar] [CrossRef]
- Abu-Saad, K.; Fraser, D. Maternal nutrition and birth outcomes. Epidemiol. Rev. 2010, 32, 5–25. [Google Scholar] [CrossRef]
- Imdad, A.; Bhutta, Z.A. Effect of balanced protein energy supplementation during pregnancy on birth outcomes. BMC Public Health 2011, 11 (Suppl. 3), S17. [Google Scholar] [CrossRef]
- Brantsæter, A.L.; Olafsdottir, A.S.; Forsum, E.; Olsen, S.F.; Thorsdottir, I. Does milk and dairy consumption during pregnancy influence fetal growth and infant birthweight? A systematic literature review. Food Nutr. Res. 2012, 56. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M.; Stettler, N.; Smith, K.M.; Reiss, R. Associations of consumption of fruits and vegetables during pregnancy with infant birth weight or small for gestational age births: A systematic review of the literature. Int. J. Womens Health 2014, 6, 899–912. [Google Scholar] [CrossRef] [PubMed]
- Leventakou, V.; Roumeliotaki, T.; Martinez, D.; Barros, H.; Brantsaeter, A.-L.; Casas, M.; Charles, M.-A.; Cordier, S.; Eggesbø, M.; van Eijsden, M.; et al. Fish intake during pregnancy, fetal growth, and gestational length in 19 European birth cohort studies. Am. J. Clin. Nutr. 2014, 99, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Phelan, S.; Hart, C.; Phipps, M.; Abrams, B.; Schaffner, A.; Adams, A.; Wing, R. Maternal behaviors during pregnancy impact offspring obesity risk. Exp. Diabetes Res. 2011, 2011, 985139. [Google Scholar] [CrossRef] [PubMed]
- International Weight Management in Pregnancy (i-WIP) Collaborative Group. Effect of diet and physical activity based interventions in pregnancy on gestational weight gain and pregnancy outcomes: Meta-analysis of individual participant data from randomised trials. BMJ 2017, 358, j3119. [Google Scholar] [CrossRef]
- Rauh, K.; Kunath, J.; Rosenfeld, E.; Kick, L.; Ulm, K.; Hauner, H. Healthy living in pregnancy: A cluster-randomized controlled trial to prevent excessive gestational weight gain—Rationale and design of the GeliS study. BMC Pregnancy Childbirth 2014, 14, 119. [Google Scholar] [CrossRef]
- Kunath, J.; Günther, J.; Rauh, K.; Hoffmann, J.; Stecher, L.; Rosenfeld, E.; Kick, L.; Ulm, K.; Hauner, H. Effects of a lifestyle intervention during pregnancy to prevent excessive gestational weight gain in routine care—The cluster-randomised GeliS trial. BMC Med. 2019, 17, 5. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Bauer, C.-P.; Bung, P.; Cremer, M.; Flothkötter, M.; Hellmers, C.; Kersting, M.; Krawinkel, M.; Przyrembel, H.; Rasenack, R.; et al. Ernährung in der Schwangerschaft—Teil 2. Handlungsempfehlungen des Netzwerks “Gesund ins Leben—Netzwerk Junge Familie”. Deutsche Medizinische Wochenschrift 2012, 137, 1366–1372. [Google Scholar] [CrossRef]
- National Research Council. Weight Gain during Pregnancy. Reexamining the Guidelines; Rasmussen, K.M., Yaktine, A.L., Eds.; National Academies Press: Washington, DC, USA, 2009; ISBN 9780309131131. [Google Scholar]
- Haftenberger, M.; Heuer, T.; Heidemann, C.; Kube, F.; Krems, C.; Mensink, G.B.M. Relative validation of a food frequency questionnaire for national health and nutrition monitoring. Nutr. J. 2010, 9, 36. [Google Scholar] [CrossRef]
- Meltzer, H.M.; Brantsaeter, A.L.; Ydersbond, T.A.; Alexander, J.; Haugen, M. Methodological challenges when monitoring the diet of pregnant women in a large study: Experiences from the Norwegian Mother and Child Cohort Study (MoBa). Matern. Child Nutr. 2008, 4, 14–27. [Google Scholar] [CrossRef]
- Kuhn, D.-A. Entwicklung eines Index zur Bewertung der Ernährungsqualität in der Studie zur Gesundheit Erwachsener in Deutschland (DEGS1), German (“Development of a dietary quality index in the German Health Examination Survey for Adults”). Master’s Thesis, Robert Koch Institute, Berlin, Germany, 2017. [Google Scholar]
- Kleinwechter, H.; Schäfer-Graf, U.; Bührer, C.; Hoesli, I.; Kainer, F.; Kautzky-Willer, A.; Pawlowski, B.; Schunck, K.; Somville, T.; Sorger, M. Gestationsdiabetes mellitus (GDM)—Diagnostik, Therapie und Nachsorge. Diabetologie und Stoffwechsel 2016, 11, S182–S194. [Google Scholar] [CrossRef]
- Metzger, B.E.; Gabbe, S.G.; Persson, B.; Buchanan, T.A.; Catalano, P.A.; Damm, P.; Dyer, A.R.; de Leiva, A.; Hod, M.; Kitzmiler, J.L.; et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Bernal, C.L.; Rebagliato, M.; Iñiguez, C.; Vioque, J.; Navarrete-Muñoz, E.M.; Murcia, M.; Bolumar, F.; Marco, A.; Ballester, F. Diet quality in early pregnancy and its effects on fetal growth outcomes: The Infancia y Medio Ambiente (Childhood and Environment) Mother and Child Cohort Study in Spain. Am. J. Clin. Nutr. 2010, 91, 1659–1666. [Google Scholar] [CrossRef] [PubMed]
- Abubakari, A.; Jahn, A. Maternal Dietary Patterns and Practices and Birth Weight in Northern Ghana. PLoS ONE 2016, 11, e0162285. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Bishwajit, G.; Yaya, S.; Cheng, Z.; Zou, D.; Zhou, Y. Prevalence of low birth weight and its association with maternal body weight status in selected countries in Africa: A cross-sectional study. BMJ Open 2018, 8, e020410. [Google Scholar] [CrossRef] [PubMed]
- Kjøllesdal, M.K.R.; Holmboe-Ottesen, G. Dietary Patterns and Birth Weight-a Review. AIMS Public Health 2014, 1, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Lenders, C.M.; Hediger, M.L.; Scholl, T.O.; Khoo, C.S.; Slap, G.B.; Stallings, V.A. Gestational age and infant size at birth are associated with dietary sugar intake among pregnant adolescents. J. Nutr. 1997, 127, 1113–1117. [Google Scholar] [CrossRef]
- Grundt, J.H.; Eide, G.E.; Brantsaeter, A.-L.; Haugen, M.; Markestad, T. Is consumption of sugar-sweetened soft drinks during pregnancy associated with birth weight? Matern. Child Nutr. 2017, 13. [Google Scholar] [CrossRef]
- Mamluk, L.; Edwards, H.B.; Savović, J.; Leach, V.; Jones, T.; Moore, T.H.M.; Ijaz, S.; Lewis, S.J.; Donovan, J.L.; Lawlor, D.; et al. Low alcohol consumption and pregnancy and childhood outcomes: Time to change guidelines indicating apparently ’safe’ levels of alcohol during pregnancy? A systematic review and meta-analyses. BMJ Open 2017, 7, e015410. [Google Scholar] [CrossRef]
- Günther, J.; Hoffmann, J.; Kunath, J.; Spies, M.; Meyer, D.; Stecher, L.; Rosenfeld, E.; Kick, L.; Rauh, K.; Hauner, H. Effects of a Lifestyle Intervention in Routine Care on Prenatal Dietary Behavior—Findings from the Cluster-Randomized GeliS Trial. JCM 2019, 8, 960. [Google Scholar] [CrossRef]
- Ledikwe, J.H.; Blanck, H.M.; Kettel Khan, L.; Serdula, M.K.; Seymour, J.D.; Tohill, B.C.; Rolls, B.J. Dietary energy density is associated with energy intake and weight status in US adults. Am. J. Clin. Nutr. 2006, 83, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.M.; Simpson, J.M.; Rissel, C.; Baur, L.A. Maternal “junk food” diet during pregnancy as a predictor of high birthweight: Findings from the healthy beginnings trial. Birth 2013, 40, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Günther, J.; Geyer, K.; Stecher, L.; Kunath, J.; Meyer, D.; Spies, M.; Rosenfeld, E.; Kick, L.; Rauh, K.; et al. Effects of prenatal physical activity on neonatal and obstetric outcomes—Findings from the cluster-randomised GeliS trial. BMC Pregnancy Childbirth 2019. under review. [Google Scholar]
- Brei, C.; Stecher, L.; Meyer, D.M.; Young, V.; Much, D.; Brunner, S.; Hauner, H. Impact of Dietary Macronutrient Intake during Early and Late Gestation on Offspring Body Composition at Birth, 1, 3, and 5 Years of Age. Nutrients 2018, 10, 579. [Google Scholar] [CrossRef] [PubMed]
| Maternal and Neonatal Characteristics | Total (n = 2018) |
|---|---|
| Maternal characteristics | |
| Pre-pregnancy age, years | 30.3 ± 4.4 a |
| Pre-pregnancy weight, kg | 68.2 ± 13.4 a |
| Pre-pregnancy BMI, kg/m2 | 24.4 ± 4.5 a |
| Pre-pregnancy BMI category | |
| BMI 18.5–24.9 kg/m2 | 1311/2018 (65.0%) |
| BMI 25.0–29.9 kg/m2 | 464/2018 (23.0%) |
| BMI 30.0–40.0 kg/m2 | 243/2018 (12.0%) |
| Educational level | |
| General secondary school | 320/2014 (15.9%) |
| Intermediate secondary school | 856/2014 (42.5%) |
| (Technical) High school | 838/2014 (41.6%) |
| Country of birth | |
| Germany | 1790/2014 (88.9%) |
| Others | 224/2014 (11.1%) |
| Further maternal characteristics | |
| Nulliparous | 1162/2018 (57.6%) |
| Living with a partner | 1939/2011 (96.4%) |
| Full-time employed | 1056/1996 (52.9%) |
| Gestational diabetes mellitus b | 209/1940 (10.8%) |
| Pregnancy hypertension c | 161/2015 (8.0%) |
| Preterm delivery d | 131/2016 (6.5%) |
| Neonatal characteristics | |
| Birth weight, g | 3337.6 ± 517.8 a |
| Birth length, cm | 51.3 ± 2.6 a |
| Head circumference, cm | 34.7 ± 1.6 a |
| BMI, kg/m2 | 12.7 ± 1.3 a |
| SGA | 172/2016 (8.5%) |
| LGA | 148/2016 (7.3%) |
| Low birth weight | 101/2018 (5.0%) |
| High birth weight | 169/2018 (8.4%) |
| Food Groups | Birth Weight (g) | BMI (kg/m2) | ||
|---|---|---|---|---|
| Adjusted Effect Size a (95% CI) | Adjusted p Value a | Adjusted Effect Size a (95% CI) | Adjusted p Value a | |
| Soft drinks b (200 mL/day) | ||||
| T0 | −10.90 (−18.17, −3.64) | 0.003 | −0.02 (−0.03,0.00) | 0.076 |
| T1 | −8.19 (−16.26, −0.11) | 0.047 | −0.01 (−0.03,0.01) | 0.437 |
| Light drinks c (200 mL/day) | ||||
| T0 | −5.70 (−17.59,6.19) | 0.347 | −0.00 (−0.03,0.03) | 0.919 |
| T1 | −5.89 (−18.34,6.55) | 0.353 | 0.01 (−0.02,0.04) | 0.577 |
| Vegetables (150 g/day) | ||||
| T0 | 41.28 (17.87,64.70 | 0.001 | 0.07 (0.01,0.13) | 0.017 |
| T1 | 36.67 (15.95,57.39) | 0.001 | 0.09 (0.03,0.14) | 0.002 |
| Fruit (150 g/day) | ||||
| T0 | 5.55 (−5.44,16.55) | 0.322 | 0.02 (−0.01,0.05) | 0.172 |
| T1 | 15.25 (3.67,26.83) | 0.010 | 0.04 (0.01,0.07) | 0.009 |
| Dairy products (200 g/day) | ||||
| T0 | 8.41 (−5.60,22.41) | 0.239 | 0.00 (−0.03,0.04) | 0.838 |
| T1 | 3.13 (−9.37,15.62) | 0.624 | 0.02 (−0.01,0.05) | 0.233 |
| Meat and meat products (150 g/day) | ||||
| T0 | 6.79 (−54.36,67.94) | 0.828 | 0.00 (−0.15,0.15) | 0.993 |
| T1 | 3.35 (−60.86,67.55) | 0.919 | 0.00 (−0.16,0.17) | 0.966 |
| Sweets and snacks (50 g/day) | ||||
| T0 | 11.69 (−8.07,31.44) | 0.246 | 0.00 (−0.05,0.05) | 0.895 |
| T1 | −4.97 (−21.12,11.18) | 0.547 | 0.00 (−0.04,0.05) | 0.855 |
| Fast food (250 g/day) | ||||
| T0 | 9.08 (−153.36,171.52) | 0.913 | 0.08 (−0.32,0.48) | 0.702 |
| T1 | −95.15 (−273.51,83.22) | 0.296 | −0.17 (−0.63,0.29) | 0.470 |
| Food Groups | Low Birth Weight | High Birth Weight | SGA | LGA | ||||
|---|---|---|---|---|---|---|---|---|
| Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | |
| Soft drinks b (200 mL/day) | ||||||||
| T0 | 1.04(0.99,1.09) | 0.150 | 0.95(0.88,1.02) | 0.149 | 1.03(0.99,1.08) | 0.123 | 0.94(0.87,1.02) | 0.155 |
| T1 | 1.01(0.94,1.09) | 0.766 | 0.95(0.88,1.03) | 0.221 | 1.00(0.94,1.07) | 0.973 | 0.95(0.87,1.03) | 0.212 |
| Light drinks c (200 mL/day) | ||||||||
| T0 | 0.98(0.87,1.11) | 0.778 | 0.99(0.91,1.08) | 0.858 | 1.01(0.93,1.10) | 0.783 | 1.00(0.92,1.09) | 0.971 |
| T1 | 1.01(0.90,1.14) | 0.864 | 1.01(0.94,1.10) | 0.763 | 1.05(0.98,1.12) | 0.213 | 1.02(0.94,1.10) | 0.627 |
| Vegetables (150 g/day) | ||||||||
| T0 | 0.85(0.67,1.08) | 0.184 | 1.09(0.94,1.27) | 0.262 | 0.98(0.84,1.16) | 0.841 | 1.13(0.96,1.32) | 0.138 |
| T1 | 0.81(0.63,1.05) | 0.113 | 1.14(1.00,1.31) | 0.058 | 0.88(0.74,1.05) | 0.146 | 1.14(0.98,1.32) | 0.081 |
| Fruit (150 g/day) | ||||||||
| T0 | 1.05(0.96,1.14) | 0.284 | 1.06(0.99,1.13) | 0.094 | 0.97(0.89,1.05) | 0.460 | 1.08(1.02,1.15) | 0.016 |
| T1 | 1.07(0.96,1.18) | 0.224 | 1.05(0.97,1.14) | 0.197 | 0.90(0.81,1.01) | 0.063 | 1.06(0.97,1.14) | 0.187 |
| Dairy products (200 g/day) | ||||||||
| T0 | 0.95(0.82,1.10) | 0.458 | 1.04(0.95,1.14) | 0.387 | 1.05(0.96,1.14) | 0.294 | 1.06(0.97,1.16) | 0.177 |
| T1 | 0.98(0.85,1.13) | 0.772 | 0.95(0.85,1.06) | 0.321 | 0.95(0.84,1.06) | 0.343 | 0.99(0.90,1.10) | 0.903 |
| Meat and meat products (150 g/day) | ||||||||
| T0 | 1.40(0.87,2.25) | 0.172 | 1.20(0.82,1.76) | 0.349 | 0.85(0.54,1.33) | 0.474 | 1.16(0.77,1.72) | 0.481 |
| T1 | 1.52(0.82,2.82) | 0.183 | 1.25(0.79,1.98) | 0.347 | 1.00(0.61,1.63) | 0.998 | 1.19(0.73,1.94) | 0.486 |
| Sweets and snacks (50 g/day) | ||||||||
| T0 | 1.07(0.91,1.25) | 0.423 | 1.02(0.90,1.17) | 0.722 | 0.97(0.84,1.12) | 0.683 | 1.06(0.93,1.20) | 0.424 |
| T1 | 1.14(0.99,1.30) | 0.066 | 0.96(0.84,1.09) | 0.496 | 1.00(0.88,1.13) | 0.963 | 0.93(0.81,1.07) | 0.308 |
| Fast food (250 g/day) | ||||||||
| T0 | 1.35(0.36,5.09) | 0.655 | 3.14(1.26,7.84) | 0.014 | 1.10(0.37,3.25) | 0.862 | 2.31(0.85,6.33) | 0.103 |
| T1 | 1.87(0.34,10.36) | 0.473 | 2.21(0.64,7.61) | 0.210 | 1.68(0.47,5.97) | 0.427 | 1.04(0.26,4.16) | 0.955 |
| Energy and Macronutrient Intake | Birth Weight (g) | BMI (kg/m2) | ||
|---|---|---|---|---|
| Adjusted Effect Size a (95% CI) | Adjusted p Value a | Adjusted Effect Size a (95% CI) | Adjusted p Value a | |
| Energy [100 kcal/day] | ||||
| T0 | −0.04 (−3.89,3.80) | 0.984 | −0.01 (−0.02,0.00) | 0.224 |
| T1 | 3.15 (−0.45,6.76) | 0.087 | 0.01 (−0.00,0.02) | 0.218 |
| Carbohydrates [10 E%] | ||||
| T0 | −8.69 (−39.35,21.97) | 0.579 | 0.01 (−0.06,0.09) | 0.769 |
| T1 | 9.16 (−22.26,40.58) | 0.568 | 0.01 (−0.07,0.09) | 0.764 |
| Saccharose [10 g/day] | ||||
| T0 | −8.27 (−15.83,−0.70) | 0.032 | −0.02 (−0.04,0.00) | 0.074 |
| T1 | 3.72 (−3.90,11.34) | 0.339 | 0.01 (−0.01,0.03) | 0.271 |
| Protein [10 E%] | ||||
| T0 | 48.88 (−28.19,125.95) | 0.214 | 0.08 (−0.12,0.27) | 0.439 |
| T1 | 14.36 (−60.91,89.64) | 0.708 | 0.08 (−0.11,0.27) | 0.425 |
| Fat [10 E%] | ||||
| T0 | 5.35 (−33.39,44.08) | 0.787 | −0.03 (−0.13,0.06) | 0.489 |
| T1 | −17.00 (−55.49,21.50) | 0.387 | −0.04 (−0.14,0.06) | 0.458 |
| Alcohol [g] | ||||
| T0 | −15.32 (−29.83,−0.80) | 0.039 | −0.01 (−0.05,0.02) | 0.439 |
| T1 | −40.67 (−105.97,24.63) | 0.222 | −0.11 (−0.28,0.05) | 0.182 |
| Caffeine [100 mg] | ||||
| T0 | −6.57 (−32.54,19.41) | 0.620 | −0.03 (−0.10,0.03) | 0.313 |
| T1 | −12.20 (−40.04,15.64) | 0.390 | −0.05 (−0.13,0.02) | 0.139 |
| HEI [10 points] | ||||
| T0 | 39.26 (12.29,66.22) | 0.004 | 0.06 (−0.01,0.13) | 0.081 |
| T1 | 42.76 (15.98,69.55) | 0.002 | 0.10 (0.03,0.16) | 0.006 |
| Energy and Macronutrient Intake | Low Birth Weight | High Birth Weight | SGA | LGA | ||||
|---|---|---|---|---|---|---|---|---|
| Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | Adjusted Odds Ratio a (95% CI) | Adjusted p Value a | |
| Energy [100 kcal/day] | ||||||||
| T0 | 1.01 (0.98,1.05) | 0.560 | 1.01 (0.98,1.04) | 0.509 | 1.00 (0.97,1.03) | 0.854 | 1.01 (0.98,1.04) | 0.630 |
| T1 | 0.99 (0.95,1.03) | 0.594 | 1.01 (0.98,1.03) | 0.659 | 0.98 (0.96,1.01) | 0.267 | 0.99 (0.96,1.02) | 0.604 |
| Carbohydrates [10 E%] | ||||||||
| T0 | 0.96 (0.73,1.26) | 0.761 | 0.94 (0.76,1.17) | 0.587 | 0.92 (0.74,1.13) | 0.408 | 1.03 (0.83,1.29) | 0.780 |
| T1 | 1.05 (0.76,1.45) | 0.786 | 1.01 (0.79,1.27) | 0.966 | 0.83 (0.65,1.06) | 0.131 | 1.09 (0.85,1.39) | 0.501 |
| Saccharose [10 g/day] | ||||||||
| T0 | 1.07 (1.01,1.13) | 0.026 | 0.97 (0.92,1.03) | 0.358 | 1.01 (0.96,1.07) | 0.627 | 0.99 (0.94,1.05) | 0.702 |
| T1 | 0.98 (0.90,1.07) | 0.664 | 0.98 (0.92,1.04) | 0.513 | 0.96 (0.90,1.02) | 0.210 | 0.96 (0.90,1.03) | 0.244 |
| Protein [10 E%] | ||||||||
| T0 | 0.77 (0.38,1.55) | 0.457 | 1.31 (0.76,2.23) | 0.331 | 0.95 (0.56,1.61) | 0.852 | 1.02 (0.58,1.81) | 0.940 |
| T1 | 1.11 (0.51,2.40) | 0.802 | 1.32 (0.76,2.30) | 0.329 | 0.98 (0.55,1.75) | 0.948 | 1.49 (0.84,2.66) | 0.173 |
| Fat [10 E%] | ||||||||
| T0 | 1.14 (0.80,1.61) | 0.467 | 1.05 (0.80,1.38) | 0.735 | 1.14 (0.88,1.49) | 0.329 | 0.94 (0.71,1.25) | 0.673 |
| T1 | 0.91 (0.61,1.36) | 0.647 | 0.92 (0.69,1.23) | 0.584 | 1.33 (0.99,1.79) | 0.057 | 0.79 (0.58,1.07) | 0.131 |
| Alcohol [g] | ||||||||
| T0 | 1.01 (0.88,1.15) | 0.905 | 0.77 (0.55,1.07) | 0.115 | 1.05 (0.98,1.13) | 0.181 | 1.03 (0.94,1.12) | 0.555 |
| T1 | 1.05 (0.53,2.07) | 0.887 | 0.85 (0.46,1.58) | 0.616 | 0.74 (0.35,1.58) | 0.442 | 0.81 (0.42,1.59) | 0.544 |
| Caffeine [100 mg] | ||||||||
| T0 | 1.09 (0.88,1.34) | 0.431 | 0.94 (0.77,1.14) | 0.494 | 1.03 (0.86,1.24) | 0.739 | 1.03 (0.87,1.22) | 0.718 |
| T1 | 0.93 (0.68,1.27) | 0.638 | 0.84 (0.66,1.07) | 0.154 | 1.13 (0.93,1.37) | 0.210 | 0.87 (0.68,1.10) | 0.245 |
| HEI [10 points] | ||||||||
| T0 | 0.86 (0.68,1.09) | 0.220 | 1.11(0.92,1.34) | 0.290 | 0.84(0.70,1.02) | 0.076 | 1.10(0.90,1.35) | 0.363 |
| T1 | 0.80 (0.61,1.06) | 0.117 | 1.15(0.94,1.41) | 0.171 | 0.85(0.69,1.04) | 0.104 | 1.15(0.93,1.43) | 0.188 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Günther, J.; Hoffmann, J.; Spies, M.; Meyer, D.; Kunath, J.; Stecher, L.; Rosenfeld, E.; Kick, L.; Rauh, K.; Hauner, H. Associations between the Prenatal Diet and Neonatal Outcomes—A Secondary Analysis of the Cluster-Randomised GeliS Trial. Nutrients 2019, 11, 1889. https://doi.org/10.3390/nu11081889
Günther J, Hoffmann J, Spies M, Meyer D, Kunath J, Stecher L, Rosenfeld E, Kick L, Rauh K, Hauner H. Associations between the Prenatal Diet and Neonatal Outcomes—A Secondary Analysis of the Cluster-Randomised GeliS Trial. Nutrients. 2019; 11(8):1889. https://doi.org/10.3390/nu11081889
Chicago/Turabian StyleGünther, Julia, Julia Hoffmann, Monika Spies, Dorothy Meyer, Julia Kunath, Lynne Stecher, Eva Rosenfeld, Luzia Kick, Kathrin Rauh, and Hans Hauner. 2019. "Associations between the Prenatal Diet and Neonatal Outcomes—A Secondary Analysis of the Cluster-Randomised GeliS Trial" Nutrients 11, no. 8: 1889. https://doi.org/10.3390/nu11081889
APA StyleGünther, J., Hoffmann, J., Spies, M., Meyer, D., Kunath, J., Stecher, L., Rosenfeld, E., Kick, L., Rauh, K., & Hauner, H. (2019). Associations between the Prenatal Diet and Neonatal Outcomes—A Secondary Analysis of the Cluster-Randomised GeliS Trial. Nutrients, 11(8), 1889. https://doi.org/10.3390/nu11081889





