A Randomized Double-Blind, Cross-Over Trial of very Low-Calorie Diet in Overweight Migraine Patients: A Possible Role for Ketones?
Abstract
1. Introduction
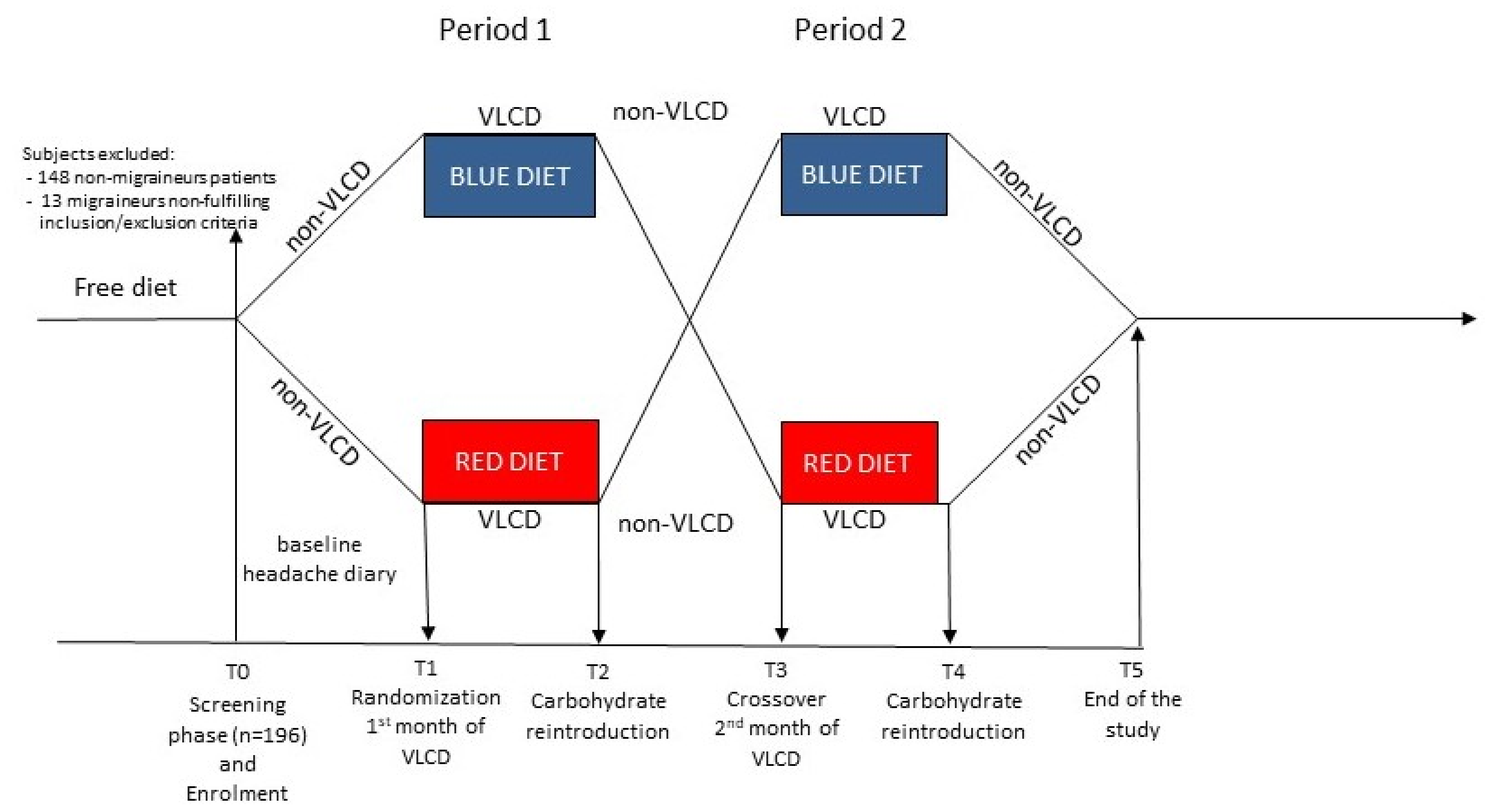
2. Materials and Methods
2.1. Randomization and Blinding
2.2. Study Population
2.3. Outcome Measures
2.4. Statistical Analysis
3. Results
3.1. Participants
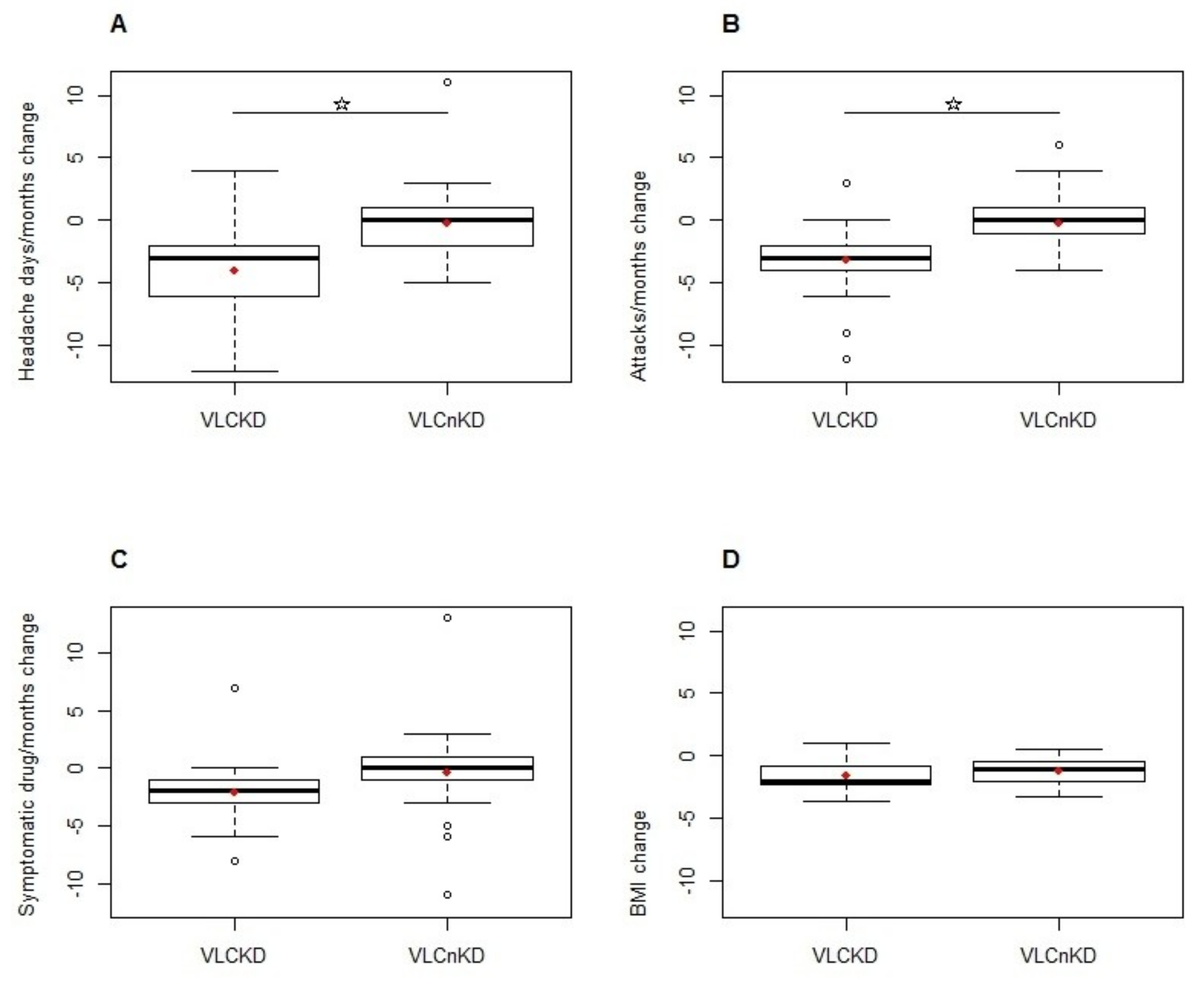
3.2. Efficacy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AcAc | Acetoacetate |
| BHB | Beta-Hydroxybutyrate |
| BMI | Body Mass Index |
| CSD | Cortical Spreading Depression |
| DRI | Dietary Reference Intake |
| EFSA | European Food Safety Authority |
| ITT | Intention-to-Treat |
| KD | Ketogenic Diet |
| LMM | Linear Mixed Model |
| PSMF | Protein Sparing Modified Fast |
| VLCD | Very Low-Calorie Diet |
| VLCKD | Very Low-Calorie Ketogenic Diet |
| VLCnKD | Very Low-Calorie non-Ketogenic Diet |
References
- Winesett, S.P.; Bessone, S.K.; Kossoff, E.H.W. The ketogenic diet in pharmacoresistant childhood epilepsy. Expert Rev. Neurother. 2015, 15, 621–628. [Google Scholar] [CrossRef] [PubMed]
- McDonald, T.; Cervenka, M. The Expanding Role of Ketogenic Diets in Adult Neurological Disorders. Brain Sci. 2018, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Schnabel, T. An Experience with a Ketogenic Dietary in Migraine. Ann. Intern. Med. 1928, 2, 341–347. [Google Scholar]
- Barborka, C. Migrain: Results of treatment by ketogenic diet in fifty cases. JAMA 1930, 95, 1825–1828. [Google Scholar] [CrossRef]
- Strahlman, R.S. Can ketosis help migraine sufferers? A case report. Headache 2006, 46, 182. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Currà, A.; Sirianni, G.; Coppola, G.; Bracaglia, M.; Cardillo, A.; De Nardis, L.; Pierelli, F. Diet transiently improves migraine in two twin sisters: Possible role of ketogenesis? Funct. Neurol. 2013, 28, 305–308. [Google Scholar]
- Cervenka, M.C.; Kossoff, E.H. Dietary treatment of intractable epilepsy. Continuum (Minneap. Minn). 2013, 19, 756–766. [Google Scholar] [CrossRef]
- Owen, O.E.; Felig, P.; Morgan, A.P.; Wahren, J.; Cahill, G.F. Liver and kidney metabolism during prolonged starvation. J. Clin. Invest. 1969, 48, 574–583. [Google Scholar] [CrossRef]
- Anderson, J.W.; Konz, E.C.; Frederich, R.C.; Wood, C.L. Long-term weight-loss maintenance: A meta-analysis of US studies. Am. J. Clin. Nutr. 2001, 74, 579–584. [Google Scholar] [CrossRef]
- Saris, W.H. Very-low-calorie diets and sustained weight loss. Obes. Res. 2001, 9, 295S–301S. [Google Scholar] [CrossRef]
- Thomas, D.D.; Istfan, N.W.; Bistrian, B.R.; Apovian, C.M. Protein sparing therapies in acute illness and obesity: A review of George Blackburn’s contributions to nutrition science. Metabolism. 2018, 79, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Goday, A.; Bellido, D.; Sajoux, I.; Crujeiras, A.B.; Burguera, B.; García-Luna, P.P.; Oleaga, A.; Moreno, B.; Casanueva, F.F. Short-term safety, tolerability and efficacy of a very low-calorie-ketogenic diet interventional weight loss program versus hypocaloric diet in patients with type 2 diabetes mellitus. Nutr. Diabetes 2016, 6, e230. [Google Scholar] [CrossRef] [PubMed]
- Moreno, B.; Bellido, D.; Sajoux, I.; Goday, A.; Saavedra, D.; Crujeiras, A.B.; Casanueva, F.F. Comparison of a very low-calorie-ketogenic diet with a standard low-calorie diet in the treatment of obesity. Endocrine 2014, 47, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.L.; Kaiser, D.L. Effects of calorie restriction and weight loss on glucose and insulin levels in obese humans. J. Am. Coll. Nutr. 1985, 4, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, C.; Coppola, G.; Sirianni, G.; Di Lorenzo, G.; Bracaglia, M.; Di Lenola, D.; Siracusano, A.; Rossi, P.; Pierelli, F. Migraine improvement during short lasting ketogenesis: A proof-of-concept study. Eur. J. Neurol. 2015, 22, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Bigal, M.E.; Liberman, J.N.; Lipton, R.B. Obesity and migraine: A population study. Neurology 2006, 66, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Hamilton, C.C.; Brinkman-Kaplan, V. Benefits and risks of an intensive very-low-calorie diet program for severe obesity. Am. J. Gastroenterol. 1992, 87, 6–15. [Google Scholar]
- Scientific Opinion on the essential composition of total diet replacements for weight control. EFSA J. 2015, 13, 3957. [CrossRef]
- Cousins, G.; Hijazze, S.; Van de Laar, F.A.; Fahey, T. Diagnostic accuracy of the ID Migraine: A systematic review and meta-analysis. Headache 2011, 51, 1140–1148. [Google Scholar] [CrossRef]
- The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808. [CrossRef]
- Silberstein, S.; Tfelt-Hansen, P.; Dodick, D.W.; Limmroth, V.; Lipton, R.B.; Pascual, J.; Wang, S.J. Task Force of the International Headache Society Clinical Trials Subcommittee Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults. Cephalalgia 2008, 28, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bosco, G.; Camporesi, E.M.; Mangar, D. Ketosis, ketogenic diet and food intake control: A complex relationship. Front. Psychol. 2015, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Verrotti, A.; Di Fonzo, A.; Penta, L.; Agostinelli, S.; Parisi, P. Obesity and headache/migraine: The importance of weight reduction through lifestyle modifications. Biomed. Res. Int. 2014, 2014, 420858. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, U.; Sukhotinsky, I.; Eikermann-Haerter, K.; Ayata, C. Glucose modulation of spreading depression susceptibility. J. Cereb. Blood Flow Metab. 2013, 33, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Rainero, I.; Govone, F.; Gai, A.; Vacca, A.; Rubino, E. Is Migraine Primarily a Metaboloendocrine Disorder? Curr. Pain Headache Rep. 2018, 22, 36. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, C.; Coppola, G.; Bracaglia, M.; Di Lenola, D.; Evangelista, M.; Sirianni, G.; Rossi, P.; Di Lorenzo, G.; Serrao, M.; Parisi, V.; et al. Cortical functional correlates of responsiveness to short-lasting preventive intervention with ketogenic diet in migraine: A multimodal evoked potentials study. J. Headache Pain 2016, 17, 58. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, C.; Coppola, G.; Bracaglia, M.; Di Lenola, D.; Sirianni, G.; Rossi, P.; Di Lorenzo, G.; Parisi, V.; Serrao, M.; Cervenka, M.C.; et al. A ketogenic diet normalizes interictal cortical but not subcortical responsivity in migraineurs. BMC Neurol. 2019, 19, 136. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Di Lorenzo, C.; Schoenen, J.; Pierelli, F. Habituation and sensitization in primary headaches. J. Headache Pain 2013, 14, 65. [Google Scholar] [CrossRef]
- Kim, D.; Davis, L.M.; Sullivan, P.G.; Maalouf, M.; Simeone, T.A.; van, B.; Rho, J.M. Ketone bodies are protective against oxidative stress in neocortical neurons. J. Neurochem. 2007, 101, 1316–1326. [Google Scholar] [CrossRef]
- Cullingford, T.E. The ketogenic diet; fatty acids, fatty acid-activated receptors and neurological disorders. Prostaglandins. Leukot. Essent. Fatty Acids 2004, 70, 253–264. [Google Scholar] [CrossRef]
- Gross, E.; Putananickal, N.; Orsini, A.-L.; Schmidt, S.; Vogt, D.R.; Cichon, S.; Sandor, P.; Fischer, D. Efficacy and safety of exogenous ketone bodies for preventive treatment of migraine: A study protocol for a single-centred, randomised, placebo-controlled, double-blind crossover trial. Trials 2019, 20, 61. [Google Scholar] [CrossRef] [PubMed]
- Montagna, P.; Cortelli, P.; Monari, L.; Pierangeli, G.; Parchi, P.; Lodi, R.; Iotti, S.; Frassineti, C.; Zaniol, P.; Lugaresi, E. 31P-magnetic resonance spectroscopy in migraine without aura. Neurology 1994, 44, 666–669. [Google Scholar] [CrossRef]
- Zaletel, M.; Strucl, M.; Bajrović, F.F.; Pogacnik, T. Coupling between visual evoked cerebral blood flow velocity responses and visual evoked potentials in migraneurs. Cephalalgia 2005, 25, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Schoenen, J. Deficient habituation of evoked cortical potentials in migraine: A link between brain biology, behavior and trigeminovascular activation? Biomed. Pharmacother. 1996, 50, 71–78. [Google Scholar] [CrossRef]
- Lisicki, M.; D’Ostilio, K.; Coppola, G.; Scholtes, F.; Maertens de Noordhout, A.; Parisi, V.; Schoenen, J.; Magis, D. Evidence of an increased neuronal activation-to-resting glucose uptake ratio in the visual cortex of migraine patients: A study comparing18FDG-PET and visual evoked potentials. J. Headache Pain 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- DeVivo, D.C.; Leckie, M.P.; Ferrendelli, J.S.; McDougal, D.B. Chronic ketosis and cerebral metabolism. Ann. Neurol. 1978, 3, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Cullingford, T.E.; Eagles, D.A.; Sato, H. The ketogenic diet upregulates expression of the gene encoding the key ketogenic enzyme mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase in rat brain. Epilepsy Res. 2002, 49, 99–107. [Google Scholar] [CrossRef]
- Maalouf, M.; Sullivan, P.G.; Davis, L.; Kim, D.Y.; Rho, J.M. Ketones inhibit mitochondrial production of reactive oxygen species production following glutamate excitotoxicity by increasing NADH oxidation. Neuroscience 2007, 145, 256–264. [Google Scholar] [CrossRef]
- Owen, O.E.; Caprio, S.; Reichard, G.A.; Mozzoli, M.A.; Boden, G.; Owen, R.S. Ketosis of starvation: A revisit and new perspectives. Clin. Endocrinol. Metab. 1983, 12, 359–379. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Coppola, G.; Di Lenola, D.; Evangelista, M.; Sirianni, G.; Rossi, P.; Di Lorenzo, G.; Serrao, M.; Pierelli, F. Efficacy of Modified Atkins Ketogenic Diet in Chronic Cluster Headache: An Open-label, Single-arm, Clinical Trial. Front. Neurol. 2018, 9, 64. [Google Scholar] [CrossRef]
- Sapir, D.G.; Owen, O.E. Renal conservation of ketone bodies during starvation. Metabolism 1975, 24, 23–33. [Google Scholar] [CrossRef]
- Urbain, P.; Bertz, H. Monitoring for compliance with a ketogenic diet: What is the best time of day to testfor urinary ketosis? Nutr. Metab. (Lond). 2016, 13, 77. [Google Scholar] [CrossRef] [PubMed]
| Micronutrient | Daily Dose | Micronutrient | Daily Dose |
|---|---|---|---|
| Vitamin A | 80 mg | Vitamin E | 12 mg |
| Vitamin B1 | 1.1 mg | Vitamin H | 50 mcg |
| Vitamin B2 | 1.4 mg | Ca2+ | 800 mg |
| Vitamin B3 | 16 mg | Cr2+ | 40 mcg |
| Vitamin B5 | 6 mg | Cu+ | 1 mg |
| Vitamin B6 | 2 mg | I- | 150 mcg |
| Vitamin B8 | 150 mcg | K+ | 2000 mg |
| Vitamin B9 | 200 mcg | Mg2+ | 375 mg |
| Vitamin B12 | 2,5 mcg | Mn2+ | 2 mg |
| Vitamin C | 60 mg | Se4+ | 55 mcg |
| Vitamin D | 5 mcg | Zn2+ | 10 mg |
| Variables | R→B | B→R | Statistics |
|---|---|---|---|
| Gender (women/men) | 15/3 | 14/3 | = 0.006, p = 1.000 |
| Age (years) | 41 (12) | 46 (7) | t33 = −1.661, p = 0.106 |
| Headache days/months | 6 (3) | 8 (4) | t33 = −1.227, p = 0.229 |
| Attacks/months | 4.83 (2.01) | 5.94 (2.49) | t33 = −1.454, p = 0.155 |
| Symptomatic /months | 3.94 (3.59) | 5.35 (3.94) | t33 = −1.107, p = 0.276 |
| BMI (Kg/m2) | 34.9 (5.4) | 35.9 (7.2) | t33 = −0.491, p = 0.627 |
| Clinical Variables | Estimate | Standard Error | 95% CI | df | t | P | |
|---|---|---|---|---|---|---|---|
| Headache days/months change | |||||||
| Treatment | −3.733 | 0.810 | −5.315, −2.148 | 27 | −4.607 | 0.0001 | |
| Period | 0.150 | 0.810 | −1.433, 1.732 | 27 | 0.185 | 0.855 | |
| Gender | 0.382 | 1.313 | −2.088, 2.852 | 25 | 0.291 | 0.773 | |
| Age | −0.028 | 0.042 | −0.108, 0.051 | 25 | −0.676 | 0.505 | |
| BMI | −0.0004 | 0.073 | −0.138, 0.136 | 25 | −0.005 | 0.996 | |
| Attacks/months change | |||||||
| Treatment | −3.017 | 0.581 | −4.153, −1.880 | 27 | −5.191 | <0.00001 | |
| Period | 0.101 | 0.581 | −1.035, 1.236 | 27 | 0.173 | 0.864 | |
| Gender | 0.987 | 0.935 | −0.656, 2.630 | 25 | 1.056 | 0.301 | |
| Age | −0.011 | 0.030 | −0.064, 0.042 | 25 | −0.370 | 0.714 | |
| BMI | −0.030 | 0.052 | −0.121, 0.061 | 25 | −0.576 | 0.570 | |
| Symptomatic drugs/months change | |||||||
| Treatment | −1.485 | 0.903 | −3.189, 0.219 | 27 | −1.645 | 0.112 | |
| Period | 0.985 | 0.903 | −0.719, 2.689 | 27 | 1.091 | 0.285 | |
| Gender | 0.271 | 1.306 | −2.193, 2.736 | 25 | 0.208 | 0.837 | |
| Age | 0.006 | 0.042 | −0.073, 0.085 | 25 | 0.145 | 0.886 | |
| BMI | 0.004 | 0.073 | −0.133, 0.141 | 25 | 0.051 | 0.960 | |
| BMI change | |||||||
| Treatment | −0.204 | 0.216 | −0.606, 0.219 | 27 | −0.943 | 0.354 | |
| Period | 1.117 | 0.216 | 0.726, 1.511 | 27 | 5.162 | <0.00001 | |
| Gender | −0.487 | 0.394 | −0.996, 0.081 | 25 | −1.237 | 0.228 | |
| Age | 0.019 | 0.013 | 0.002, 0.037 | 25 | 1.466 | 0.155 | |
| BMI | −0.040 | 0.022 | −0.069, −0.009 | 25 | −1.844 | 0.077 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Lorenzo, C.; Pinto, A.; Ienca, R.; Coppola, G.; Sirianni, G.; Di Lorenzo, G.; Parisi, V.; Serrao, M.; Spagnoli, A.; Vestri, A.; et al. A Randomized Double-Blind, Cross-Over Trial of very Low-Calorie Diet in Overweight Migraine Patients: A Possible Role for Ketones? Nutrients 2019, 11, 1742. https://doi.org/10.3390/nu11081742
Di Lorenzo C, Pinto A, Ienca R, Coppola G, Sirianni G, Di Lorenzo G, Parisi V, Serrao M, Spagnoli A, Vestri A, et al. A Randomized Double-Blind, Cross-Over Trial of very Low-Calorie Diet in Overweight Migraine Patients: A Possible Role for Ketones? Nutrients. 2019; 11(8):1742. https://doi.org/10.3390/nu11081742
Chicago/Turabian StyleDi Lorenzo, Cherubino, Alessandro Pinto, Roberta Ienca, Gianluca Coppola, Giulio Sirianni, Giorgio Di Lorenzo, Vincenzo Parisi, Mariano Serrao, Alessandra Spagnoli, Annarita Vestri, and et al. 2019. "A Randomized Double-Blind, Cross-Over Trial of very Low-Calorie Diet in Overweight Migraine Patients: A Possible Role for Ketones?" Nutrients 11, no. 8: 1742. https://doi.org/10.3390/nu11081742
APA StyleDi Lorenzo, C., Pinto, A., Ienca, R., Coppola, G., Sirianni, G., Di Lorenzo, G., Parisi, V., Serrao, M., Spagnoli, A., Vestri, A., Schoenen, J., Donini, L. M., & Pierelli, F. (2019). A Randomized Double-Blind, Cross-Over Trial of very Low-Calorie Diet in Overweight Migraine Patients: A Possible Role for Ketones? Nutrients, 11(8), 1742. https://doi.org/10.3390/nu11081742








