A Population Survey of Dietary Attitudes towards Gluten
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Methods
2.3. Data Analysis
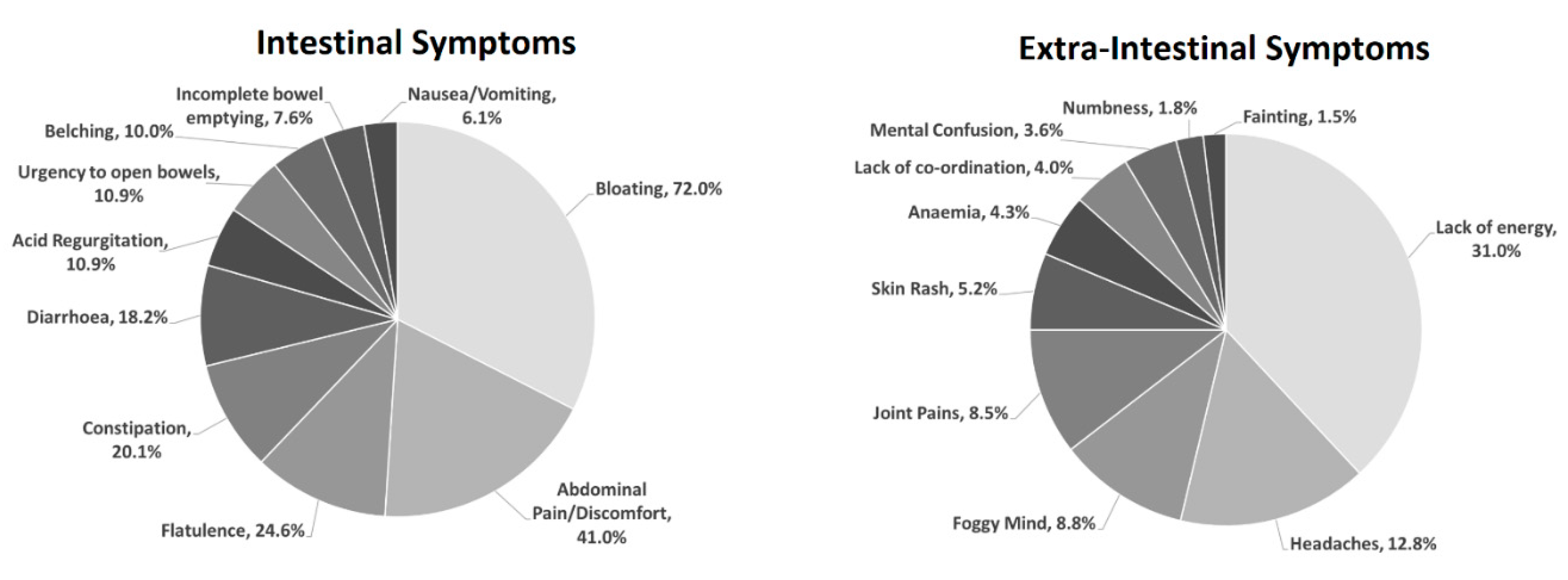
3. Results
3.1. Overall Population Characteristics
3.2. Self-Reported Gluten Sensitivity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gujral, N.; Freeman, H.J.; Thomson, A.B.R. Celiac disease: Prevalence, diagnosis, pathogenesis and treatment. World J. Gastroenterol. 2012, 18, 6036–6059. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Ludvigsson, J.F.; Brantner, T.L.; Murray, J.A.; Everhart, J.E. The prevalence of celiac disease in the United States. Am. J. Gastroenterol. 2012, 107, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.S.; Patel, D.; Stephenson, T.J.; Ward, A.M.; McCloskey, E.V.; Hadjivassiliou, M.; Lobo, A.J. A primary care cross-sectional study of undiagnosed adult coeliac disease. Eur. J. Gastroenterol. Hepatol. 2003, 15, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Terazono, E. Gluten Free: One of 3 Trends Shaking Up Commodities. Available online: https://www.ft.com/content/5348432e-1a13-11e7-bcac-6d03d067f81f (accessed on 3 July 2018).
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of non-celiac gluten sensitivity (NCGS): The salerno experts’ criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef] [PubMed]
- Lionetti, E.; Pulvirenti, A.; Vallorani, M.; Catassi, G.; Verma, A.K.; Gatti, S.; Catassi, C. Re-challenge Studies in Non-celiac Gluten Sensitivity: A Systematic Review and Meta-Analysis. Front. Physiol. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Tovoli, F.; Cicola, R.; Parisi, C.; Fabbri, A.; Piscaglia, M.; Fiorini, E.; Caio, G. Serological tests in gluten sensitivity (nonceliac gluten intolerance). J. Clin. Gastroenterol. 2012, 46, 680–685. [Google Scholar] [CrossRef]
- Sapone, A.; Lammers, K.M.; Casolaro, V.; Cammarota, M.; Giuliano, M.T.; De Rosa, M.; Stefanile, R.; Mazzarella, G.; Tolone, C.; Russo, M.I.; et al. Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: Celiac disease and gluten sensitivity. BMC Med. 2011, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Uhde, M.; Ajamian, M.; Caio, G.; De Giorgio, R.; Indart, A.; Green, P.H.; Verna, E.C.; Volta, U.; Alaedini, A. Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut 2016, 65, 1930–1937. [Google Scholar] [CrossRef]
- Jones, A.L. The Gluten-Free Diet: Fad or Necessity? Diabetes Spectr. 2017, 30, 118–123. [Google Scholar] [CrossRef]
- El Khoury, D.; Balfour-Ducharme, S.; Joye, I.J. A Review on the Gluten-Free Diet: Technological and Nutritional Challenges. Nutrients 2018, 10, 1410. [Google Scholar] [CrossRef]
- Mintel Health/Weight-Conscious Consumers Are Driving the Gluten-Free Market, Not Celiacs, Says Mintel. Available online: https://www.foodnavigator-usa.com/Article/2013/10/15/Healthy-eaters-dieters-not-celiacs-propelling-gluten-free-market# (accessed on 27 June 2018).
- MINTEL Non-Celiacs Drive Gluten-Free Market Growth. Available online: http://www.mintel.com/blog/food-market-news/gluten-free-consumption-trends (accessed on 27 June 2018).
- Harper, L.; Bold, J. An exploration into the motivation for gluten avoidance in the absence of coeliac disease. Gastroenterol. Hepatol. Bed Bench 2018, 11, 259–268. [Google Scholar] [PubMed]
- Catassi, C.; Alaedini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D.; et al. The overlapping area of non-celiac gluten sensitivity (NCGS) and wheat-sensitive irritable bowel syndrome (IBS): An update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef] [PubMed]
- Lerner, B.A.; Green, P.H.R.; Lebwohl, B. Going against the Grains: Gluten-Free Diets in Patients without Celiac Disease—Worthwhile or Not? Dig. Dis. Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Reilly, N.R. The Gluten-Free Diet: Recognizing Fact, Fiction, and Fad. J. Pediatr. 2016, 175, 206–210. [Google Scholar] [CrossRef] [PubMed]
- MINTEL Half of Americans Think Gluten-Free Diets Are a Fad While 25% Eat Gluten-Free Foods. Available online: http://www.mintel.com/press-centre/food-and-drink/half-of-americans-think-gluten-free-diets-are-a-fad-while-25-eat-gluten-free-foods (accessed on 4 July 2018).
- Aziz, I.; Sanders, D.S. Patients who avoid wheat and gluten: Is that health or lifestyle? Dig. Dis. Sci. 2014, 59, 1080–1082. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aziz, I.; Lewis, N.R.; Hadjivassiliou, M.; Winfield, S.N.; Rugg, N.; Kelsall, A.; Newrick, L.; Sanders, D.S. A UK study assessing the population prevalence of self-reported gluten sensitivity and referral characteristics to secondary care. Eur. J. Gastroenterol. Hepatol. 2014, 26, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.S.; Carter, M.J.; Hurlstone, D.P.; Pearce, A.; Ward, A.M.; McAlindon, M.E.; Lobo, A.J. Association of adult coeliac disease with irritable bowel syndrome: A case-control study in patients fulfilling ROME II criteria referred to secondary care. Lancet 2001, 358, 1504–1508. [Google Scholar] [CrossRef]
- van Gils, T.; Nijeboer, P.; Ijssennagger, C.E.; Sanders, D.S.; Mulder, C.J.J.; Bouma, G. Prevalence and characterization of self-reported gluten sensitivity in The Netherlands. Nutrients 2016, 8, 714. [Google Scholar] [CrossRef] [PubMed]
- Hopman, E.G.; Kiefte-de Jong, J.C.; le Cessie, S.; Moll, H.A.; Witteman, J.C.; Bleeker, S.E.; Mearin, M.L. Food questionnaire for assessment of infant gluten consumption. Clin. Nutr. 2007, 26, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Hopman, E.G.D.; Pruijn, R.; Tabben, E.H.; Le Cessie, S.; Mearin, M.L. Food questionnaire for the assessment of gluten intake by children 1 to 4 years old. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Molina-Infante, J.; Santolaria, S.; Sanders, D.S.; Fernández-Bañares, F. Systematic review: Noncoeliac gluten sensitivity. Aliment. Pharmacol. Ther. 2015, 41, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Canavan, C.; West, J.; Card, T. Epidemiology of Irritable Bowel Syndrome. Clin. Epidemiol. 2014, 6, 71–80. [Google Scholar] [PubMed]
- Ontiveros, N.; López-Gallardo, J.A.; Vergara-Jiménez, M.J.; Cabrera-Chávez, F. Self-reported prevalence of symptomatic adverse reactions to gluten and adherence to gluten-free diet in an adult mexican population. Nutrients 2015, 7, 6000–6015. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Chávez, F.; Granda-Restrepo, D.M.; Arámburo-Gálvez, J.G.; Franco-Aguilar, A.; Magaña-Ordorica, D.; de Vergara-Jiménez, M.J.; Ontiveros, N. Self-Reported Prevalence of Gluten-Related Disorders and Adherence to Gluten-Free Diet in Colombian Adult Population. Gastroenterol. Res. Pract. 2016, 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nachman, F.; Vazquez, H.; Gonzalez, A.; Andrenacci, P.; Compagni, L.; Reyes, H.; Sugai, E.; Moreno, M.L.; Smecuol, E.; Hwang, H.J.; et al. Gastroesophageal reflux symptoms in patients with celiac disease and the effects of a gluten-free diet. Clin. Gastroenterol. Hepatol. 2011, 9, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; Swift, G.L.; Card, T.R.; Sanders, D.S.; Ludvigsson, J.F.; Bai, J.C. Psychological morbidity of celiac disease: A review of the literature. United Eur. Gastroenterol. J. 2015, 3, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Zylberberg, H.M.; Demmer, R.T.; Murray, J.A.; Green, P.H.R.; Lebwohl, B. Depression and Insomnia among Individuals with Celiac Disease or on a Gluten-Free Diet in the United States: Results from a National Survey. Eur. J. Gastroenterol. Hepatol. 2017, 29, 1091–1096. [Google Scholar] [CrossRef]
- Niland, B.; Cash, B.D. Health benefits and adverse effects of a gluten-free diet in non-celiac disease patients. Gastroenterol. Hepatol. 2018, 14, 82–91. [Google Scholar]
| Variable | As Reported in 2012 (Overall N = 1002) | As Reported in The Current Study (Measured in 2015, Overall N = 1004) | Chi-Squared p Value (Where Conducted) |
|---|---|---|---|
| Prevalence of self-reported gluten sensitivity | 12.9% (N = 129) | 32.8% (N = 329) | p < 0.001 |
| Mean age (years) of people with self-reported gluten sensitivity | 39.5 ± 17.7 | 37.7 ± 16.0 | - |
| % Female within people with self-reported gluten sensitivity | 79% | 66% | - |
| Overall prevalence of coeliac disease diagnosis | 0.8% (N = 8) | 1.2% (N = 12) | p = 0.370 |
| Overall prevalence of people adhering to a GFD | 3.7% (N = 37) | 3.7% (N = 37) | p = 0.997 |
| Pre-existing conditions which gluten-sensitive individuals are more likely to have | Anxiety, Depression, Chronic fatigue syndrome, Food allergies/intolerances, IBS (rates not reported) | Anxiety (22.4%), Depression (14.7%), Chronic fatigue syndrome (3.5%), Myalgic encephalomyelitis (10.3%), Chronic headaches (9.0%), Acid reflux (11.2%), Food allergies/intolerances/IBS (3.2%–9.0%) | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Croall, I.D.; Trott, N.; Rej, A.; Aziz, I.; O’Brien, D.J.; George, H.A.; Hossain, M.Y.; Marks, L.J.S.; Richardson, J.I.; Rigby, R.; et al. A Population Survey of Dietary Attitudes towards Gluten. Nutrients 2019, 11, 1276. https://doi.org/10.3390/nu11061276
Croall ID, Trott N, Rej A, Aziz I, O’Brien DJ, George HA, Hossain MY, Marks LJS, Richardson JI, Rigby R, et al. A Population Survey of Dietary Attitudes towards Gluten. Nutrients. 2019; 11(6):1276. https://doi.org/10.3390/nu11061276
Chicago/Turabian StyleCroall, Iain D, Nick Trott, Anupam Rej, Imran Aziz, David J O’Brien, Harvey A George, Mohammed Y Hossain, Lauren J S Marks, Jessica I Richardson, Rebecca Rigby, and et al. 2019. "A Population Survey of Dietary Attitudes towards Gluten" Nutrients 11, no. 6: 1276. https://doi.org/10.3390/nu11061276
APA StyleCroall, I. D., Trott, N., Rej, A., Aziz, I., O’Brien, D. J., George, H. A., Hossain, M. Y., Marks, L. J. S., Richardson, J. I., Rigby, R., Hadjivassiliou, M., Hoggard, N., & Sanders, D. S. (2019). A Population Survey of Dietary Attitudes towards Gluten. Nutrients, 11(6), 1276. https://doi.org/10.3390/nu11061276






