The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Study Measures
2.3. Classification by Region of Residence and Latitude
2.4. Covariates
2.5. Statistics
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ES | Endocrine Society |
| IOM | Institute of Medicine |
| 25(OH)D | 25-hydroxyvitamin D |
References
- United Nations. World Population Prospects: The 2017 Revision, Key Findings and Advance Tables. 2017. Available online: https://esa.un.org/unpd/wpp/Publications/Files/WPP2017_KeyFindings.pdf (accessed on 8 August 2017).
- Public Health England. Health Profile for England. 2017. Available online: https://www.gov.uk/government/publications/health-profile-for-england (accessed on 28 June 2017).
- Heilbronn, L.K.; De Jonge, L.; Frisard, M.I.; DeLany, J.P.; Larson-Meyer, D.E.; Rood, J.; Nguyen, T.; Martin, C.K.; Volaufova, J.; Most, M.M.; et al. Effect of 6-month calorie restriction on biomarkers of longevity, metabolic adaptation, and oxidative stress in overweight individuals: A randomized controlled trial. JAMA 2006, 295, 1539–1548. [Google Scholar] [CrossRef]
- Krzymińska-Siemaszko, R.; Czepulis, N.; Lewandowicz, M.; Zasadzka, E.; Suwalska, A.; Witowski, J.; Wieczorowska-Tobis, K. The Effect of a 12-Week Omega-3 Supplementation on Body Composition, Muscle Strength and Physical Performance in Elderly Individuals with Decreased Muscle Mass. Int. J. Environ. Res. Public Health 2015, 12, 10558–10574. [Google Scholar] [CrossRef] [PubMed]
- Sahni, S.; Mangano, K.M.; McLean, R.R.; Hannan, M.T.; Kielm, D.P. Dietary approaches for bone health: Lessons from the Framingham Osteoporosis Study. Curr. Osteoporos Rep. 2015, 13, 245–255. [Google Scholar] [CrossRef]
- Maijo, M.; Ivory, K.; Clements, S.J.; Dainty, J.R.; Jennings, A.; Gillings, R.; Fairweather-Tait, S.; Gulisano, M.; Santoro, A.; Franceschi, C.; et al. One-year consumption of a mediterranean-like dietary pattern with vitamin D3 supplements induced small scale but extensive changes of immune cell phenotype, co-receptor expression and innate immune responses in healthy elderly subjects: Results from the United Kingdom arm of the NU-AGE trial. Front. Physiol. 2018, 9, 997. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; Ward, M.; McSorley, E.; Strain, J.J.; Wallace, J. Vitamin D and bone health; Potential mechanisms. Nutrients 2010, 2, 693–724. [Google Scholar] [CrossRef]
- Bischoff, H.A.; Stähelin, H.B.; Dick, W.; Akos, R.; Knecht, M.; Salis, C.; Nebiker, M.; Theiler, R.; Pfeifer, M.; Begerow, B.; et al. Effects of vitamin D and calcium supplementation on falls: A randomized controlled trial. J. Bone Miner. Res. 2003, 18, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Willett, W.C.; Staehelin, H.B.; Bazemore, M.G.; Zee, R.Y.; Wong, J.B. Effect of vitamin D on falls: A meta-analysis. JAMA 2004, 291, 1999–2006. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Giovannucci, E.; Dietrich, T.; Dawson-Hughes, B. Fracture prevention with vitamin D supplementation: A meta-analysis of randomized controlled trials. JAMA 2005, 293, 2257–2264. [Google Scholar] [CrossRef]
- Pludowski, P.; Holick, M.F.; Pilz, S.; Wagner, C.L.; Hollis, B.W.; Grant, W.B.; Shoenfeld, Y.; Lerchbaum, E.; Llewellyn, D.J.; Kienreich, K.; et al. Vitamin D effects on musculoskeletal health, immunity, autoimmunity, cardiovascular disease, cancer, fertility, pregnancy, dementia and mortality-a review of recent evidence. Autoimmun Rev. 2013, 12, 976–989. [Google Scholar] [CrossRef]
- Skaaby, T. The relationship of vitamin D status to risk of cardiovascular disease and mortality. Dan Med. J. 2015, 62. [Google Scholar]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.H.; Rasmussen, H.; Yu, W.W.; Epstein, S.R.; Russell, R.M. Modified MyPyramid for Older Adults. J. Nutr. 2008, 138, 5–11. [Google Scholar] [CrossRef]
- Hirani, V.; Tull, K.; Ali, A.; Mindell, J. Urgent action needed to improve vitamin D status among older people in England! Age Ageing 2010, 39, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; O’Halloran, A.M.; Carey, D.; Healy, M.; O’Connor, D.; Moore, P.; Shannon, T.; Molloy, A.M.; Kenny, R.A. The Prevalence of Vitamin D Deficiency and the Determinants of 25(OH)D Concentration in Older Irish Adults: Data From The Irish Longitudinal Study on Ageing (TILDA). J. Gerontol. Ser. A 2017, 73, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Hosking, D.; Lippuner, K.; Norquist, J.M.; Wehren, L.; Maalouf, G.; Ragi-Eis, S.; Chandler, J. The prevalence of vitamin D inadequacy amongst women with osteoporosis: An international epidemiological investigation. J. Intern. Med. 2006, 260, 245–254. [Google Scholar] [CrossRef]
- Macdonald, H.M.; Mavroeidi, A.; Barr, R.J.; Black, A.J.; Fraser, W.D.; Reid, D.M. Vitamin D status in postmenopausal women living at higher latitudes in the UK in relation to bone health, overweight, sunlight exposure and dietary vitamin D. Bone 2008, 42, 996–1003. [Google Scholar] [CrossRef]
- Tripkovic, L.; Wilson, L.R.; Hart, K.; Johnsen, S.; De Lusignan, S.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Elliott, R.; et al. Daily supplementation with 15ug vitamin D2 compared with vitamin D3 to increase wintertime 25-hydroxyvitamin D status in healthy South Asian and white European women: A 12-wk randomized, placebo-controlled food-fortification trial. Am. J. Clin. Nutr. 2017, 106, 481–490. [Google Scholar] [CrossRef]
- O’Neill, C.; Kazantzidis, A.; Ryan, M.; Barber, N.; Sempos, C.; Durazo-Arvizu, R.; Jorde, R.; Grimnes, G.; Eiriksdottir, G.; Gudnason, V.; et al. Seasonal changes in vitamin D-effective UVB availability in Europe and associations with population serum 25-Hydroxyvitamin, D. Nutrients 2016, 8, 533. [Google Scholar] [CrossRef]
- Hirani, V.; Primatesta, P. Vitamin D concentrations among people aged 65 years and over living in private households and institutions in England: Population survey. Age Ageing 2005, 34, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Institute Of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press (US): Washington, DC, USA, 2010. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Van Schoor, N.M.; Comijs, H.C.; Llewellyn, D.J.; Lips, P. Cross-sectional and longitudinal associations between serum 25-hydroxyvitamin D and cognitive functioning. Int. Psychogeriatr. 2016, 28, 759–768. [Google Scholar] [CrossRef]
- Smithers, G.; Finch, S.; Doyle, W.; Lowe, C.; Bates, C.J.; Prentice, A.; Clarke, P.C. The National Diet and Nutrition Survey: People aged 65 years and over. Nutr. Food Sci. 1998, 98, 133–134. [Google Scholar] [CrossRef]
- Van der Wielen, R.P.; De Groot, L.C.; Van Staveren, W.A.; Löwik, M.R.; Van den Berg, H.; Haller, J.; Moreiras, O. Serum vitamin D concentrations among elderly people in Europe. Lancet 1995, 346, 207–221. [Google Scholar] [CrossRef]
- Andersen, R.; Mølgaard, C.; Skovgaard, L.T.; Brot, C.; Cashman, K.D.; Chabros, E.; Charzewska, J.; Flynn, A.; Jakobsen, J.; Karkkainen, M.; et al. Teenage girls and elderly women living in northern Europe have low winter vitamin D status. Eur. J. Clin. Nutr. 2005, 59, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.R.; O'Brien, M.M.; Lamberg-Allardt, C.; Jakobsen, J.; Kiely, M.; Flynn, A.; Cashman, K.D. Vitamin D status of 51-75-year-old Irish women: Its determinants and impact on biochemical indices of bone turnover. Public Health Nutr. 2006, 9, 225–233. [Google Scholar] [CrossRef]
- McCarroll, K.; Beirne, A.; Casey, M.; McNulty, H.; Ward, M.; Hoey, L.; Molloy, A.; Laird, E.; Healy, M.; Strain, J.J.; et al. Determinants of 25-hydroxyvitamin D in older Irish adults. Age Ageing 2015, 44, 847–853. [Google Scholar] [CrossRef]
- Van Ballegooijen, A.J.; Visser, M.; Brouwer, I.A. Determinants of vitamin D supplement use among community-dwelling older adults. Tijdschr. Gerontol. Geriatr. 2015, 46, 282–289. [Google Scholar] [CrossRef]
- Laird, E.; Shannon, T.; Crowley, V.E.; Healy, M. The benefits of utilising geo-mapping for visualising the vitamin D status of Dublin city and the surrounding urban districts. Ir. J. Med. Sci. 2017, 186, 807–813. [Google Scholar] [CrossRef]
- Jääskeläinen, T.; Itkonen, S.T.; Lundqvist, A.; Erkkola, M.; Koskela, T.; Lakkala, K.; Dowling, K.G.; Hull, G.L.; Kröger, H.; Karppinen, J.; et al. The positive impact of general vitamin D food fortification policy on vitamin D status in a representative adult Finnish population: Evidence from an 11-y follow-up based on standardized 25-hydroxyvitamin D data. Am. J. Clin. Nutr. 2017, 105, 1512–1520. [Google Scholar] [CrossRef]
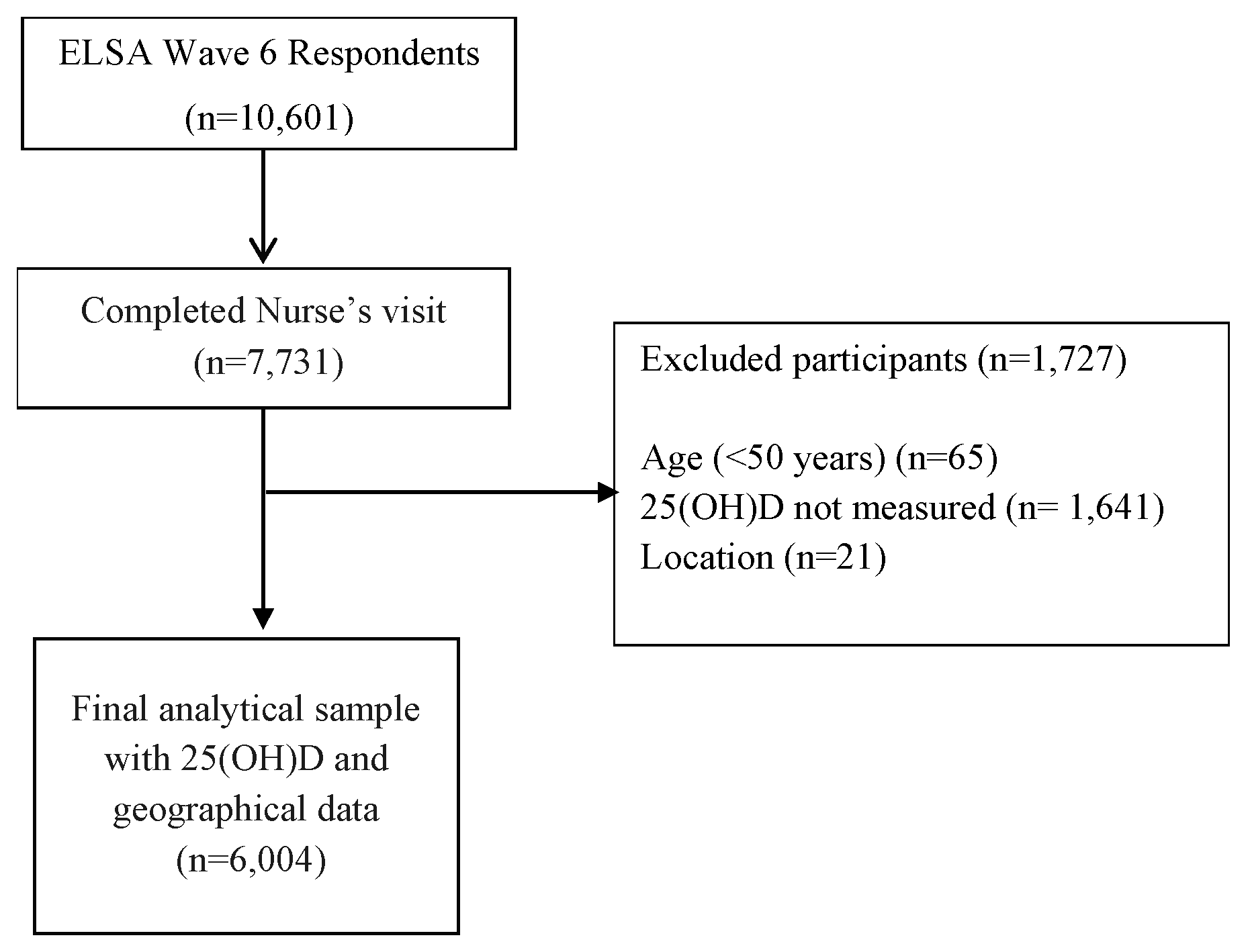
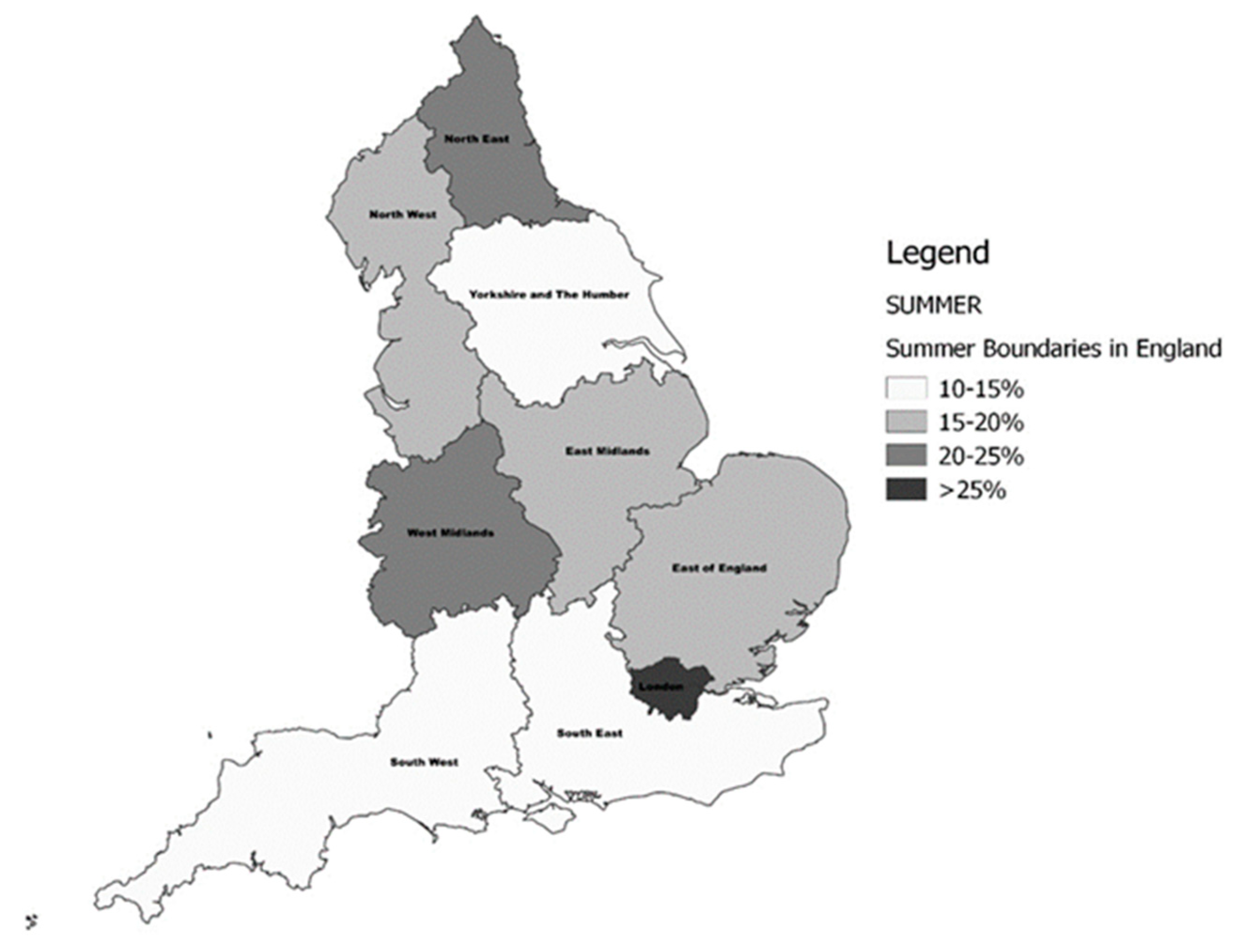
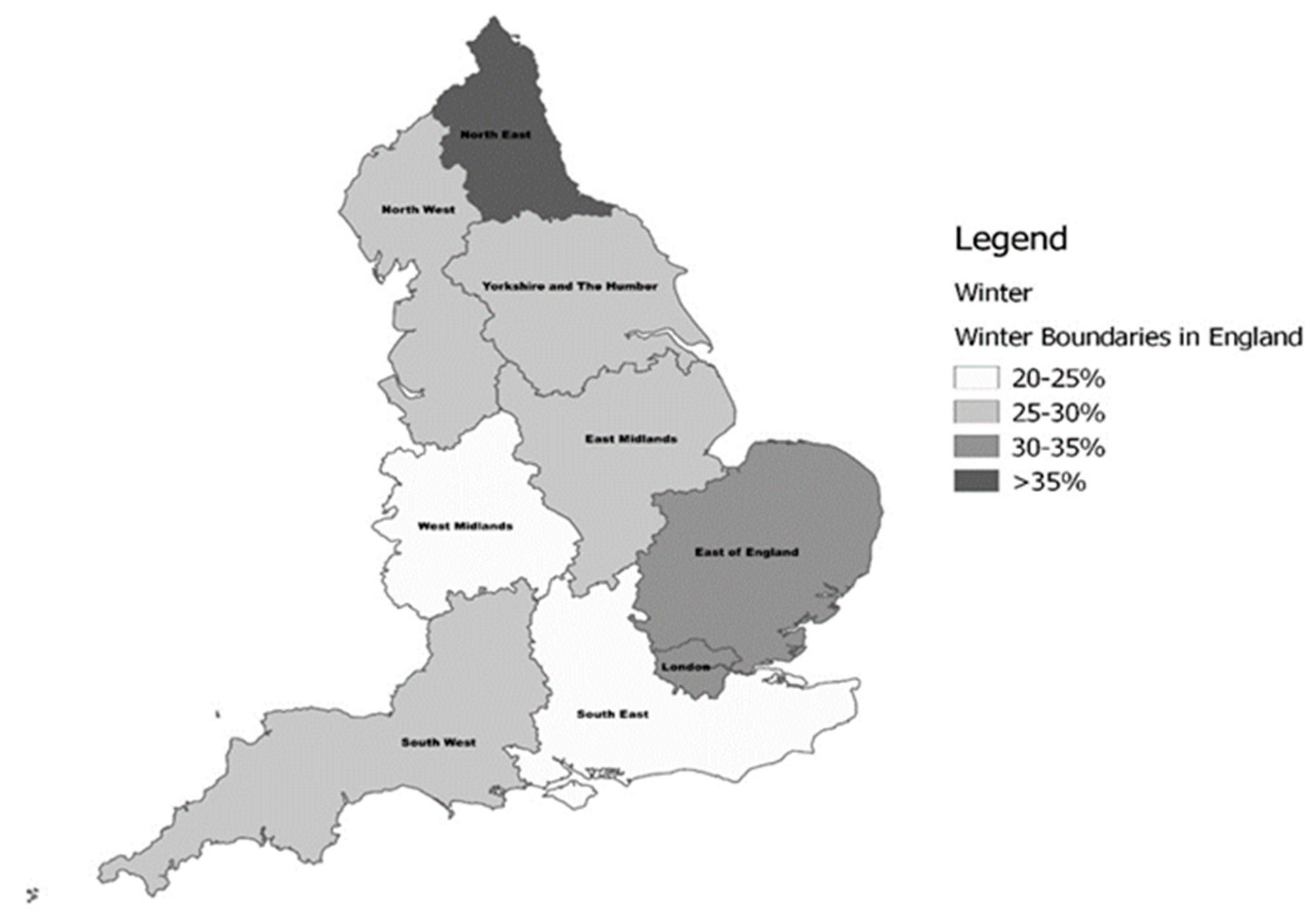
| Variables | North (n = 1692) | Midlands (n = 2036) | South (n = 2276) | All (n = 6004) |
|---|---|---|---|---|
| n (%) ± SD | n (%) ± SD | n (%) ± SD | n (%) ± SD | |
| Age, years | 66.3 ± 8.8 | 66.6 ± 8.9 | 66.3 ± 8.8 | 66.4 ± 8.8 |
| Female | 950 (56.2) | 1114 (54.7) | 1227 (53.9) | 3291 (54.8) |
| Education ≥O-level | 1183 (69.9) | 1420 (69.7) | 1789 (78.6) *** | 4392 (73.2%) |
| No qualification | 429 (25.4) | 521 (25.6) | 390 (17.1) *** | 1340 (22.4) |
| Marital Status | ||||
| a Married | 1111 (65.7) | 1357 (66.7) | 1566 (68.8) * | 3997 (66.6) |
| Single | 99 (5.9) | 116 (5.7) | 168 (7.4) * | 320 (5.3) |
| Widow | 250 (14.8) | 270 (13.3) | 279 (12.3) * | 763 (12.7) |
| Health and lifestyle factors | ||||
| BMI (kg/m²) | 28.3 ± 5.1 | 28.2 ± 4.9 | 27.7 ± 4.8 *** | 28.0 ± 4.9 |
| Obese | 541 (32.7) | 601 (30.7) | 589 (26.6) *** | 1731 (29.7) |
| Physical Activity | ||||
| Vigorous (>1/wk) | 376 (22.2) | 451 (22.2) | 532 (23.4) | 1359 (22.6) |
| Moderate (>1/wk) | 1055 (62.4) | 1332 (65.4) | 1546 (67.9) *** | 3933 (65.6) |
| Current smoker | 205 (12.1) | 244 (11.9) | 246 (10.8) | 695 (11.6) |
| Alcohol 5–6 days/week | 101 (5.9) | 99 (4.8) | 156 (6.9) | 356 (5.9) |
| SR Health | ||||
| Excellent | 189 (11.2) | 256 (12.6) | 327 (14.4) ** | 772 (12.9) |
| Poor | 114 (6.7) | 137 (6.7) | 109 (4.8) ** | 360 (6.0) |
| bLimiting illness | 576 (34.0) | 668 (32.8) | 643 (28.3) *** | 1887 (31.4) |
| Polypharmacy | 474 (28.0) | 538 (26.4) | 533 (23.4) *** | 1546 (25.7) |
| Predictors of Vitamin D Status | ||||
| c Winter blood sample | 1011 (59.7) | 1219 (59.9) | 1334 (58.6) | 3564 (59.4) |
| d Sun holiday travel | 953 (56.3) | 1200 (58.9) | 1304 (57.3) | 3457 (57.6) |
| VitD supp user | 84 (4.9) | 85 (4.2) | 93 (4.1) | 262 (4.4) |
| 25(OH)D, nmol/L | 47.4 ± 23.0 | 48.1 ± 23.9 | 50.0 ± 23.2 ** | 48.7 ± 23.4 |
| IOM Deficiency < 30 nmol/L (n = 1423) | ||||
| % | 50–59 (n = 469) | 60–69 (n = 492) | 70–79 (n = 325) | 80+ (n = 137) |
| Overall | 27.4 * | 20.4 *** | 22.2 *** | 32.0 |
| Males | 28.1 | 19.3 | 17.3 | 26.3 |
| Female | 26.9 | 21.5 | 26.1 | 36.8 |
| a Season-Winter | 32.9 | 24.3 | 27.8 | 35.6 |
| ES Deficiency <50 nmol/L (n = 3317) | ||||
| % | 50–59 (n = 1007) | 60–69 (n = 1251) | 70–79 (n = 785) | 80+ (n = 273) |
| Overall | 58.9 | 52.1 *** | 53.6 *** | 63.8 |
| Males | 59.0 | 51.8 | 50.1 | 58.2 |
| Female | 58.9 | 52.3 | 56.4 | 68.4 |
| a Season-Winter | 66.7 | 58.7 | 60.3 | 69.1 |
| Prevalence of vitamin D deficiency, stratified by age, among D supplement users b (n = 262) | ||||
| N (%) | 50–59 (n = 33) | 60–69 (n = 105) | 70–79 (n = 90) | 80+ (n = 34) |
| <30 nmol/L (n = 27) | 9 (27.3) | 8 (7.6) | 8 (8.9) | 2 (5.9) |
| <50 nmol/L (n = 72) | 15 (45.4) | 25 (23.8) | 19 (21.1) | 13 (38.2) |
| Unadjusted Model 1 | Adjusted Model 1 | |||
|---|---|---|---|---|
| Demographic Variables | OR | (95%CI) | OR | (95%CI) |
| Female | 1.21 ** | (1.04, 1.41) | 1.23 ** | (1.04, 1.44) |
| Age | ||||
| 50–59 (Reference) | [1] | [1] | ||
| 60–69 | 0.71 *** | (0.59, 0.84) | 0.85 | (0.69, 1.05) |
| 70–79 | 0.79 * | (0.66, 0.97) | 0.89 | (0.68, 1.16) |
| 80+ | 1.31 * | (1.01, 1.69) | 1.42 * | (1.01, 1.96) |
| Non-white ethnicity | 3.59 *** | (2.4, 5.4) | 3.8 *** | (2.39, 6.05) |
| Marital Status—Widow | 1.59 *** | (1.31, 1.93) | 1.44 *** | (1.15, 1.79) |
| Single | 1.37 * | (1.04, 1.82) | 1.33 | (0.98, 1.81) |
| Employment—retired | 0.79 ** | (0.69, 0.92) | 0.79 * | (0.65, 0.95) |
| Region—North | [1] | [1] | ||
| Midlands | 0.93 | (0.78, 1.11) | 0.95 | (0.79, 1.14) |
| South | 0.79 * | (0.66, 0.96) | 0.78 ** | (0.64, 0.95) |
| aLatitude (°N) | 1.11 *** | (1.05, 1.17) | 1.11 *** | (1.04, 1.17) |
| Modifiable Health and Lifestyle Factors | ||||
| BMI—Normal | 0.73 *** | (0.61, 0.87) | 0.81 * | (0.67, 1.00) |
| Obese | 1.59 *** | (1.36, 1.86) | 1.32 ** | (1.09, 1.58) |
| Current smoker | 2.18 *** | (1.78, 2.67) | 1.88 *** | (1.51, 2.34) |
| SR health—Excellent | [1] | [1] | ||
| Poor | 2.26 ** | (1.67, 3.06) | 1.49 * | (1.08, 2.07) |
| Fair | 3.49 *** | (2.43, 5.02) | 1.99 *** | (1.33, 2.96) |
| VitD supplement use | 0.39 *** | (0.25, 0.64) | 0.28 *** | (0.17, 0.45) |
| Season (summer) | 0.52 *** | (0.45, 0.61) | 0.47 *** | (0.40, 0.56) |
| Sun travel | 0.54 *** | (0.47, 0.63) | 0.74 *** | (0.63, 0.86) |
| PA-Vigorous (>1/week) | 0.46 *** | (0.37, 0.57) | 0.68 *** | (0.55, 0.86) |
| Moderate (>1/week) | 0.50 *** | (0.43, 0.58) | 0.74 *** | (0.62, 0.88) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aspell, N.; Laird, E.; Healy, M.; Shannon, T.; Lawlor, B.; O’Sullivan, M. The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA). Nutrients 2019, 11, 1253. https://doi.org/10.3390/nu11061253
Aspell N, Laird E, Healy M, Shannon T, Lawlor B, O’Sullivan M. The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA). Nutrients. 2019; 11(6):1253. https://doi.org/10.3390/nu11061253
Chicago/Turabian StyleAspell, Niamh, Eamon Laird, Martin Healy, Tom Shannon, Brian Lawlor, and Maria O’Sullivan. 2019. "The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA)" Nutrients 11, no. 6: 1253. https://doi.org/10.3390/nu11061253
APA StyleAspell, N., Laird, E., Healy, M., Shannon, T., Lawlor, B., & O’Sullivan, M. (2019). The Prevalence and Determinants of Vitamin D Status in Community-Dwelling Older Adults: Results from the English Longitudinal Study of Ageing (ELSA). Nutrients, 11(6), 1253. https://doi.org/10.3390/nu11061253






