High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years
Abstract
1. Introduction
2. Materials and Methods
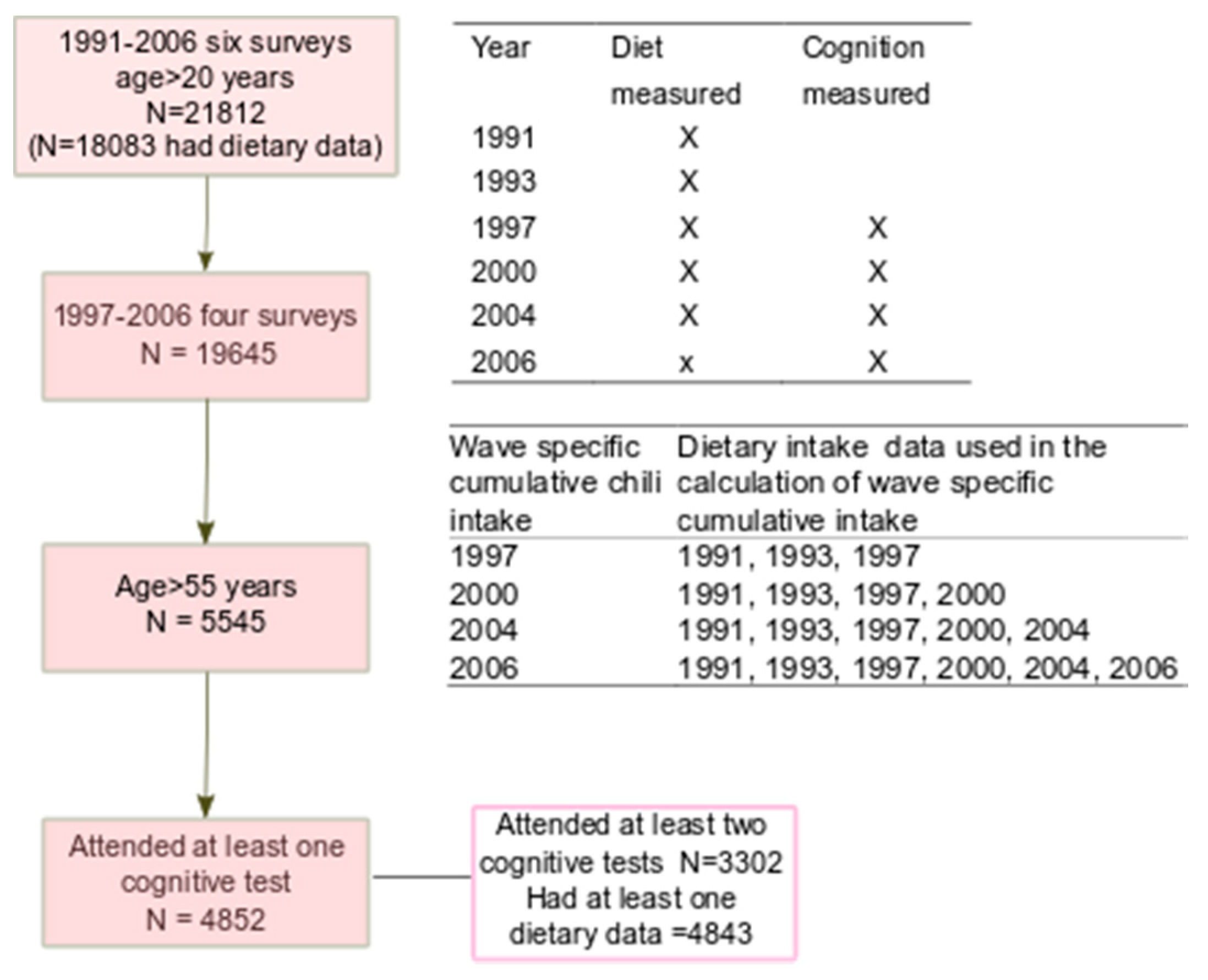
2.1. Study Design and Sample
2.2. Outcome Variable: Cognitive Function
2.3. Exposure Variable: Cumulative Mean Chili Intake
2.4. Covariates
2.5. Data Analyses
3. Results
3.1. Descriptive Results
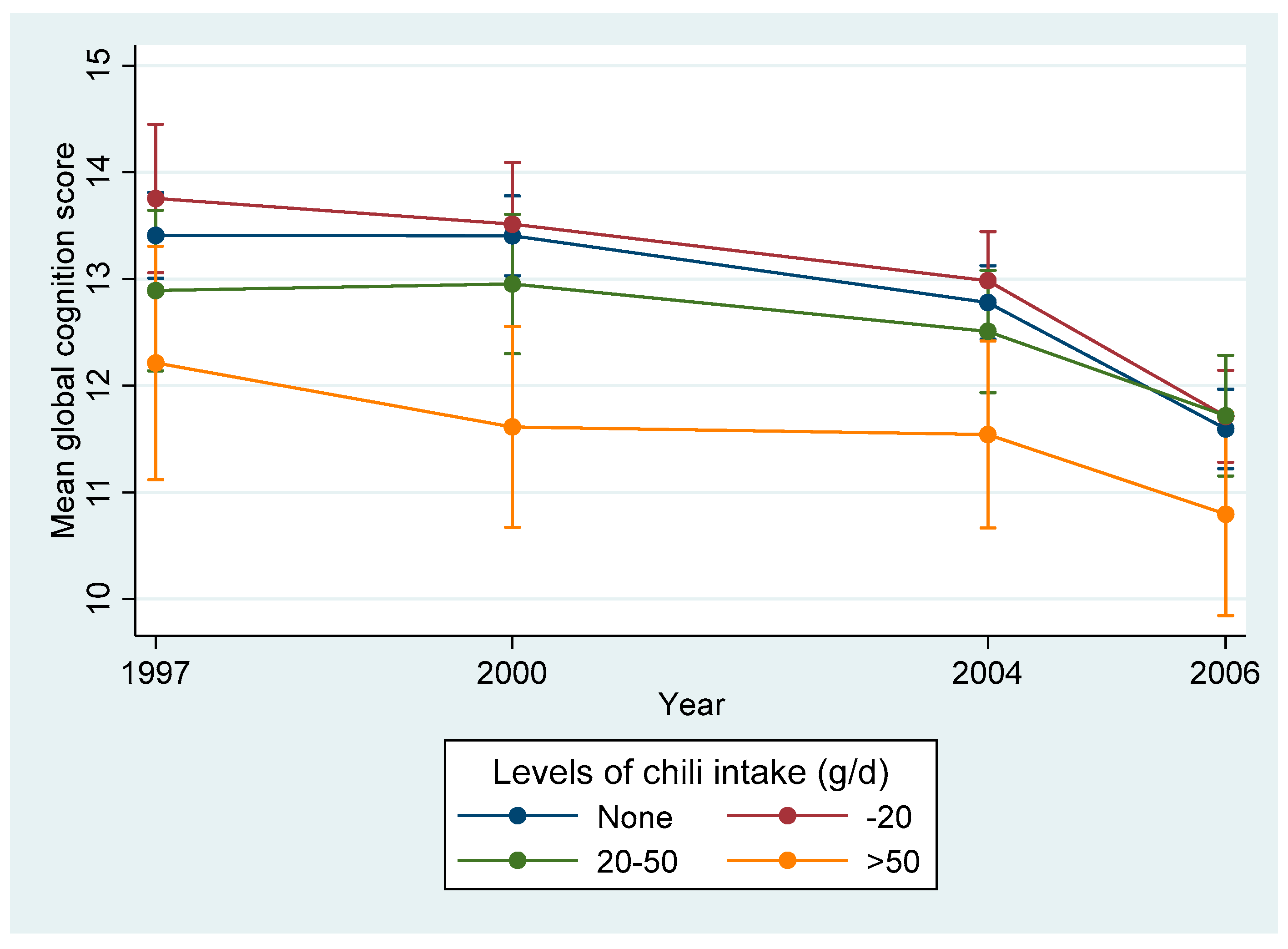
3.2. Association between Chili Intake and Cognitive Function
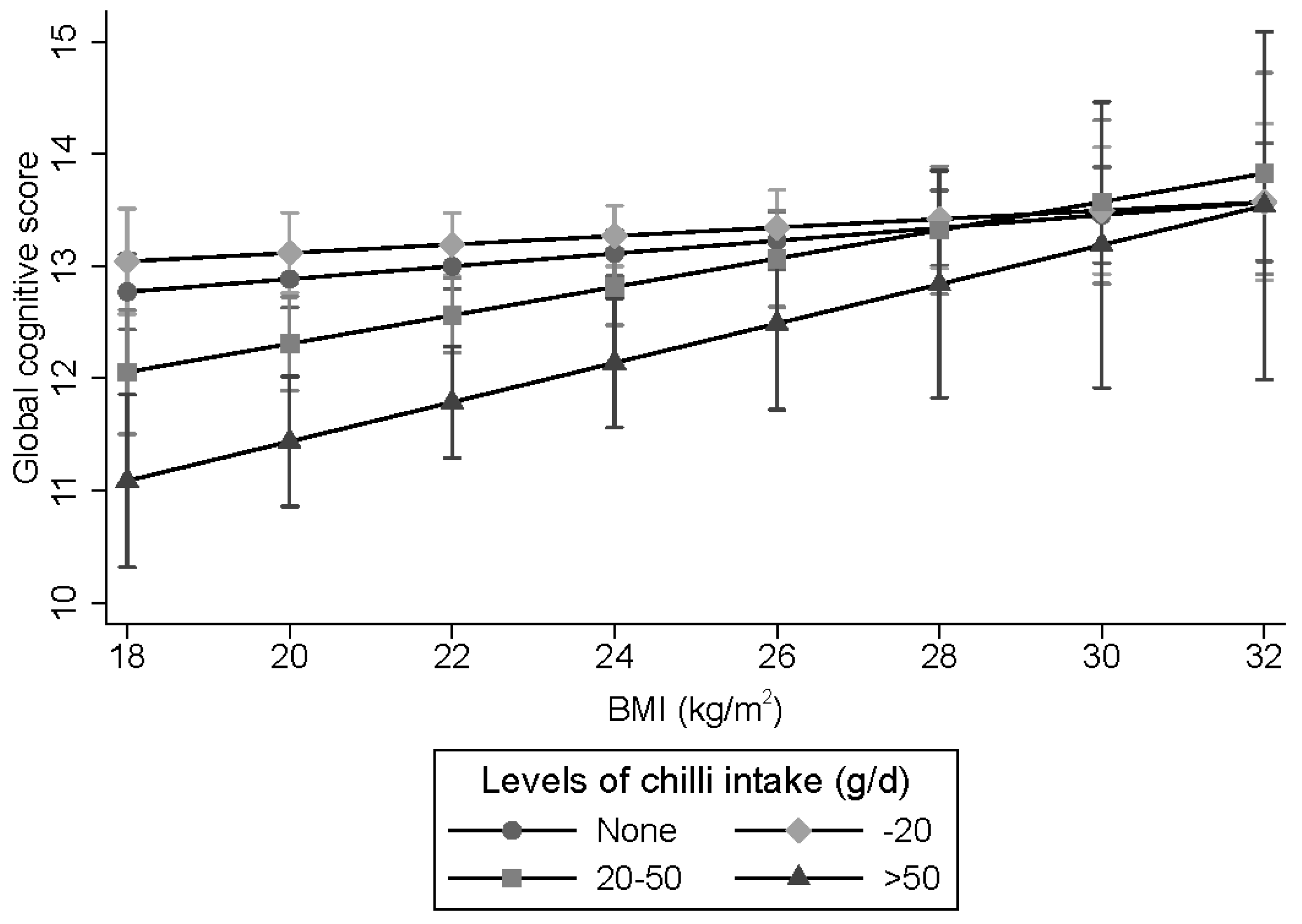
3.3. Weight Status Modifies the Association between Chili Intake and Cognitive Function
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prince, M.; Bryce, R.; Albanese, E.; Wimo, A.; Ribeiro, W.; Ferri, C.P. The global prevalence of dementia: A systematic review and metaanalysis. Alzheimers Dement. 2013, 9, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.T.; Ali, G.C.; Guerchet, M.; Prina, A.M.; Chan, K.Y.; Prince, M.; Brayne, C. Prevalence of dementia in mainland China, Hong Kong and Taiwan: An updated systematic review and meta-analysis. Int. J. Epidemiol. 2018, 47, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S.G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef]
- Van de Rest, O.; Berendsen, A.A.; Haveman-Nies, A.; de Groot, L.C. Dietary patterns, cognitive decline, and dementia: A systematic review. Adv. Nutr. 2015, 6, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Rozin, P.; Schiller, D. The nature and acquisition of a preference for chili pepper by humans. Motiv. Emot. 1980, 4, 77–101. [Google Scholar] [CrossRef]
- Astrup, A.; Kristensen, M.; Gregersen, N.T.; Belza, A.; Lorenzen, J.K.; Due, A.; Larsen, T.M. Can bioactive foods affect obesity? Ann. N. Y. Acad. Sci. 2010, 1190, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Qi, L.; Yu, C.; Yang, L.; Guo, Y.; Chen, Y.; Bian, Z.; Sun, D.; Du, J.; Ge, P.; et al. Consumption of spicy foods and total and cause specific mortality: Population based cohort study. BMJ 2015, 351, h3942. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, A.; Arguin, H.; Panahi, S. Capsaicinoids: A spicy solution to the management of obesity? Int. J. Obes. 2016, 40, 1198–1204. [Google Scholar] [CrossRef]
- Shi, Z.; Riley, M.; Brown, A.; Page, A. Chilli intake is inversely associated with hypertension among adults. Clin. Nutr. ESPEN 2018, 23, 67–72. [Google Scholar] [CrossRef]
- Sun, F.; Xiong, S.; Zhu, Z. Dietary Capsaicin Protects Cardiometabolic Organs from Dysfunction. Nutrients 2016, 8, 174. [Google Scholar] [CrossRef]
- Janssens, P.L.; Hursel, R.; Martens, E.A.; Westerterp-Plantenga, M.S. Acute effects of capsaicin on energy expenditure and fat oxidation in negative energy balance. PLoS ONE 2013, 8, e67786. [Google Scholar] [CrossRef]
- Whiting, S.; Derbyshire, E.J.; Tiwari, B. Could capsaicinoids help to support weight management? A systematic review and meta-analysis of energy intake data. Appetite 2014, 73, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Ludy, M.J.; Moore, G.E.; Mattes, R.D. The effects of capsaicin and capsiate on energy balance: Critical review and meta-analyses of studies in humans. Chem. Senses 2012, 37, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, M.; St-Pierre, S.; Drapeau, V.; Dionne, I.; Doucet, E.; Suzuki, M.; Tremblay, A. Effects of red pepper on appetite and energy intake. Br. J. Nutr. 1999, 82, 115–123. [Google Scholar] [CrossRef]
- Jiang, X.; Jia, L.W.; Li, X.H.; Cheng, X.S.; Xie, J.Z.; Ma, Z.W.; Xu, W.J.; Liu, Y.; Yao, Y.; Du, L.L.; et al. Capsaicin ameliorates stress-induced Alzheimer’s disease-like pathological and cognitive impairments in rats. J. Alzheimers Dis. 2013, 35, 91–105. [Google Scholar] [CrossRef]
- Xu, W.; Liu, J.; Ma, D.; Yuan, G.; Lu, Y.; Yang, Y. Capsaicin reduces Alzheimer-associated tau changes in the hippocampus of type 2 diabetes rats. PLoS ONE 2017, 12, e0172477. [Google Scholar] [CrossRef]
- Chard, P.S.; Bleakman, D.; Savidge, J.R.; Miller, R.J. Capsaicin-induced neurotoxicity in cultured dorsal root ganglion neurons: Involvement of calcium-activated proteases. Neuroscience 1995, 65, 1099–1108. [Google Scholar] [CrossRef]
- Jancso, G.; Kiraly, E.; Such, G.; Joo, F.; Nagy, A. Neurotoxic effect of capsaicin in mammals. Acta Physiol. Hung. 1987, 69, 295–313. [Google Scholar]
- Liu, C.H.; Bu, X.L.; Wang, J.; Zhang, T.; Xiang, Y.; Shen, L.L.; Wang, Q.H.; Deng, B.; Wang, X.; Zhu, C.; et al. The Associations between a Capsaicin-Rich Diet and Blood Amyloid-beta Levels and Cognitive Function. J. Alzheimers Dis. 2016, 52, 1081–1088. [Google Scholar] [CrossRef]
- Iadecola, C.; Yaffe, K.; Biller, J.; Bratzke, L.C.; Faraci, F.M.; Gorelick, P.B.; Gulati, M.; Kamel, H.; Knopman, D.S.; Launer, L.J.; et al. Impact of Hypertension on Cognitive Function: A Scientific Statement from the American Heart Association. Hypertension 2016, 68, e67–e94. [Google Scholar] [CrossRef] [PubMed]
- Qizilbash, N.; Gregson, J.; Johnson, M.E.; Pearce, N.; Douglas, I.; Wing, K.; Evans, S.J.W.; Pocock, S.J. BMI and risk of dementia in two million people over two decades: A retrospective cohort study. Lancet Diabetes Endocrinol. 2015, 3, 431–436. [Google Scholar] [CrossRef]
- Kim, S.; Kim, Y.; Park, S.M. Body Mass Index and Decline of Cognitive Function. PLoS ONE 2016, 11, e0148908. [Google Scholar] [CrossRef] [PubMed]
- Zhai, F.Y.; Du, S.F.; Wang, Z.H.; Zhang, J.G.; Du, W.W.; Popkin, B.M. Dynamics of the Chinese diet and the role of urbanicity, 1991–2011. Obes. Rev. 2014, 15 (Suppl. 1), 16–26. [Google Scholar] [CrossRef]
- Popkin, B.M.; Du, S.; Zhai, F.; Zhang, B. Cohort Profile: The China Health and Nutrition Survey--monitoring and understanding socio-economic and health change in China, 1989–2011. Int. J. Epidemiol. 2010, 39, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Plassman, B.L.; Welsh, K.A.; Helms, M.; Brandt, J.; Page, W.F.; Breitner, J.C. Intelligence and education as predictors of cognitive state in late life: A 50-year follow-up. Neurology 1995, 45, 1446–1450. [Google Scholar] [CrossRef]
- Lei, X.; Hu, Y.; McArdle, J.J.; Smith, J.P.; Zhao, Y. Gender Differences in Cognition among Older Adults in China. J. Hum. Resour. 2012, 47, 951–971. [Google Scholar]
- Hu, F.B.; Stampfer, M.J.; Rimm, E.; Ascherio, A.; Rosner, B.A.; Spiegelman, D.; Willett, W.C. Dietary fat and coronary heart disease: A comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am. J. Epidemiol. 1999, 149, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y. Chinese Food Composition Table 2004; Peking University Medical Press: Beijing, China, 2005. [Google Scholar]
- Yao, M.; McCrory, M.A.; Ma, G.; Tucker, K.L.; Gao, S.; Fuss, P.; Roberts, S.B. Relative influence of diet and physical activity on body composition in urban Chinese adults. Am. J. Clin. Nutr. 2003, 77, 1409–1416. [Google Scholar] [CrossRef]
- Shi, Z.; Taylor, A.W.; Riley, M.; Byles, J.; Liu, J.; Noakes, M. Association between dietary patterns, cadmium intake and chronic kidney disease among adults. Clin. Nutr. 2018, 37, 276–284. [Google Scholar] [CrossRef]
- Lebrun, B.; Bariohay, B.; Moyse, E.; Jean, A. Brain-derived neurotrophic factor (BDNF) and food intake regulation: A minireview. Auton. Neurosci. 2006, 126–127, 30–38. [Google Scholar] [CrossRef]
- Atake, K.; Nakamura, T.; Ueda, N.; Hori, H.; Katsuki, A.; Yoshimura, R. The Impact of Aging, Psychotic Symptoms, Medication, and Brain-Derived Neurotrophic Factor on Cognitive Impairment in Japanese Chronic Schizophrenia Patients. Front. Psychiatry 2018, 9, 232. [Google Scholar] [CrossRef] [PubMed]
- Komulainen, P.; Pedersen, M.; Hanninen, T.; Bruunsgaard, H.; Lakka, T.A.; Kivipelto, M.; Hassinen, M.; Rauramaa, T.H.; Pedersen, B.K.; Rauramaa, R. BDNF is a novel marker of cognitive function in ageing women: The DR’s EXTRA Study. Neurobiol. Learn. Mem. 2008, 90, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.J.; Kwon, D.Y.; Kim, M.J.; Kang, S.; Moon, N.R.; Daily, J.W.; Park, S. Red peppers with moderate and severe pungency prevent the memory deficit and hepatic insulin resistance in diabetic rats with Alzheimer’s disease. Nutr. Metab. 2015, 12, 9. [Google Scholar] [CrossRef]
- Ha, S.O.; Yoo, H.J.; Park, S.Y.; Hong, H.S.; Kim, D.S.; Cho, H.J. Capsaicin effects on brain-derived neurotrophic factor in rat dorsal root ganglia and spinal cord. Brain Res. Mol. Brain Res. 2000, 81, 181–186. [Google Scholar] [CrossRef]
- Cunha, C.; Brambilla, R.; Thomas, K.L. A simple role for BDNF in learning and memory? Front. Mol. Neurosci. 2010, 3, 1. [Google Scholar] [CrossRef] [PubMed]
- Pal, M.; Angaru, S.; Kodimuthali, A.; Dhingra, N. Vanilloid receptor antagonists: Emerging class of novel anti-inflammatory agents for pain management. Curr. Pharm. Des. 2009, 15, 1008–1026. [Google Scholar] [CrossRef]
- Suemoto, C.K.; Gilsanz, P.; Mayeda, E.R.; Glymour, M.M. Body mass index and cognitive function: The potential for reverse causation. Int. J. Obes. 2015, 39, 1383–1389. [Google Scholar] [CrossRef]
| None | 1–20 | 20–50 | >50 | p-value | |
|---|---|---|---|---|---|
| n | 2662 | 975 | 718 | 346 | |
| Age (years) | 64.3 (7.9) | 62.4 (7.8) | 62.4 (7.4) | 62.2 (6.9) | <0.001 |
| Sex | 0.008 | ||||
| Men | 1217 (46.4%) | 461 (47.3%) | 374 (52.1%) | 185 (53.5%) | |
| Women | 1405 (53.6%) | 514 (52.7%) | 344 (47.9%) | 161 (46.5%) | |
| Income | <0.001 | ||||
| Low | 776 (30.0%) | 310(32.1%) | 256(35.9%) | 121 (35.3%) | |
| Medium | 724 (28.0%) | 304(31.5%) | 232(32.5% | 136 (39.7%) | |
| High | 1090 (42.1%) | 351(36.4%) | 225(31.6%) | 86 (25.1%) | |
| Education | 0.003 | ||||
| Low | 1681 (73.0%) | 625 (68.2%) | 486 (72.5%) | 256 (80.5%) | |
| Medium | 333 (14.5%) | 161 (17.6%) | 93 (13.9%) | 36 (11.3%) | |
| High | 290 (12.6%) | 130 (14.2%) | 91 (13.6%) | 26 (8.2%) | |
| Urbanization | <0.001 | ||||
| Low | 648 (24.7%) | 244 (25.0%) | 181 (25.2%) | 110 (31.8%) | |
| Medium | 634 (24.2%) | 295 (30.3%) | 245 (34.1%) | 124 (35.8%) | |
| High | 1340 (51.1%) | 436 (44.7%) | 292 (40.7%) | 112 (32.4%) | |
| Smoking | <0.001 | ||||
| Non smoker | 1837 (70.2%) | 656 (67.5%) | 442 (61.6%) | 197 (57.1%) | |
| Ex-smokers | 84 (3.2%) | 34 (3.5%) | 27 (3.8%) | 25 (7.2%) | |
| Current smokers | 695 (26.6%) | 282 (29.0%) | 248 (34.6%) | 123 (35.7%) | |
| Survey year | <0.001 | ||||
| 1997 | 1219 (46.5%) | 379 (38.9%) | 318 (44.3%) | 136 (39.3%) | |
| 2000 | 462 (17.6%) | 159 (16.3%) | 107 (14.9%) | 69 (19.9%) | |
| 2004 | 621 (23.7%) | 241 (24.7%) | 173 (24.1%) | 71 (20.5%) | |
| 2006 | 320 (12.2%) | 196 (20.1%) | 120 (16.7%) | 70 (20.2%) | |
| Alcohol drinking | 737 (28.7%) | 308 (32.2%) | 256 (36.2%) | 131 (38.4%) | <0.001 |
| Physical activity (MET/week) | 79.2 (90.8) | 89.8 (102.5) | 100.2 (110.3) | 121.7 (115.8) | <0.001 |
| BMI (kg/m2) | 23.2 (3.7) | 23.1 (3.6) | 22.7 (3.6) | 22.2 (3.2) | <0.001 |
| BMI>24 (kg/m2) | 967 (39.9%) | 330 (36.6%) | 223 (32.9%) | 81 (24.5%) | <0.001 |
| Energy intake (kcal/day) | 2038.2 (613.6) | 2103.8 (617.0) | 2160.0 (621.2) | 2342.5 (711.8) | <0.001 |
| Fat intake (g/day) | 66.2 (36.4) | 67.0 (35.5) | 66.6 (36.7) | 70.1 (41.6) | 0.32 |
| Protein intake (g/day) | 62.9 (23.2) | 63.6 (21.7) | 63.8 (21.8) | 68.4 (27.5) | <0.001 |
| Carbohydrate intake (g/day) | 292.4 (103.1) | 305.3 (104.8) | 320.2 (109.6) | 347.7 (110.2) | <0.001 |
| Traditional southern dietary pattern score | −0.2 (0.9) | −0.0 (0.9) | 0.1 (0.8) | 0.2 (0.9) | <0.001 |
| Modern dietary pattern score | 0.0 (0.9) | −0.1 (0.8) | −0.2 (0.7) | −0.4 (0.6) | <0.001 |
| Chili intake (g/day) | 0.0 (0.0) | 9.8 (5.4) | 33.5 (8.4) | 75.3 (23.8) | <0.001 |
| Hypertension | 948 (38.2%) | 311 (33.9%) | 215 (31.3%) | 92 (27.8%) | <0.001 |
| Diabetes | 89 (3.5%) | 33 (3.4%) | 22 (3.1%) | 5 (1.5%) | 0.260 |
| Stroke | 62 (2.4) | 26 (2.7%) | 6 (0.8%) | 6 (1.8%) | 0.046 |
| Self-reported poor memory | 561(21.5%) | 189(19.6%) | 116(16.4%) | 98(28.6%) | <0.001 |
| Self-reported memory decline | 998(39.3%) | 353(37.0%) | 260(37.2%) | 168(50.0%) | <0.001 |
| None | 1–20 | 20–50 | >50 | p value | |
|---|---|---|---|---|---|
| Global cognitive function | |||||
| Model 1 | 0.00 | 0.16 (−0.15–0.47) | −0.60 (−0.96–0.24) | −1.87 (−2.39–1.35) | <0.001 |
| Model 2 | 0.00 | 0.27 (−0.05-0.58) | −0.27 (−0.64–0.10) | −1.10 (−1.62–0.57) | 0.001 |
| Model 3 | 0.00 | 0.28 (−0.04-0.60) | −0.26 (−0.63–0.12) | −1.12 (−1.64–0.59) | 0.001 |
| Model 4 | 0.00 | 0.18 (−0.14-0.51) | −0.36 (−0.74–0.02) | −1.17 (−1.70–0.63) | <0.001 |
| Model 5 | 0.00 | 0.17 (−0.18-0.52) | −0.31 (−0.72–0.10) | −1.13 (−1.71–0.54) | 0.001 |
| None | 1–20 | 20–50 | >50 | p value | |
|---|---|---|---|---|---|
| Self-reported poor memory | |||||
| Model 1 | 1.00 | 1.17 (1.02–1.34) | 1.07 (0.91–1.26) | 2.26 (1.82–2.81) | <0.001 |
| Model 2 | 1.00 | 1.14 (0.98–1.32) | 1.02 (0.85–1.21) | 2.03 (1.61–2.56) | <0.001 |
| Model 3 | 1.00 | 1.13 (0.97–1.31) | 1.00 (0.84–1.19) | 1.98 (1.56–2.50) | <0.001 |
| Model 4 | 1.00 | 1.23 (1.05–1.43) | 1.06 (0.89–1.27) | 2.17 (1.69–2.77) | <0.001 |
| Model 5 | 1.00 | 1.20 (1.02–1.42) | 1.10 (0.90–1.33) | 2.12 (1.63–2.77) | 0.001 |
| Self-reported memory decline | |||||
| Model 1 | 1.00 | 1.07 (0.96–1.20) | 1.13 (0.99–1.29) | 1.75 (1.45–2.11) | <0.001 |
| Model 2 | 1.00 | 1.05 (0.93–1.20) | 1.10 (0.95–1.28) | 1.61 (1.31–1.99) | <0.001 |
| Model 3 | 1.00 | 1.04 (0.92–1.18) | 1.08 (0.93–1.25) | 1.54 (1.25–1.90) | <0.001 |
| Model 4 | 1.00 | 1.06 (0.93–1.21) | 1.10 (0.95–1.29) | 1.56 (1.26–1.94) | 0.001 |
| Model 5 | 1.00 | 1.08 (0.93–1.24) | 1.12 (0.95–1.33) | 1.56 (1.23–1.97) | 0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Z.; El-Obeid, T.; Riley, M.; Li, M.; Page, A.; Liu, J. High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years. Nutrients 2019, 11, 1183. https://doi.org/10.3390/nu11051183
Shi Z, El-Obeid T, Riley M, Li M, Page A, Liu J. High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years. Nutrients. 2019; 11(5):1183. https://doi.org/10.3390/nu11051183
Chicago/Turabian StyleShi, Zumin, Tahra El-Obeid, Malcolm Riley, Ming Li, Amanda Page, and Jianghong Liu. 2019. "High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years" Nutrients 11, no. 5: 1183. https://doi.org/10.3390/nu11051183
APA StyleShi, Z., El-Obeid, T., Riley, M., Li, M., Page, A., & Liu, J. (2019). High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years. Nutrients, 11(5), 1183. https://doi.org/10.3390/nu11051183








