Hypovitaminosis D: Is It Time to Consider the Use of Calcifediol?
Abstract
1. Introduction
1.1. Biosynthesis of Vitamin D
1.2. Vitamin-D-Binding Protein (DBP)
1.3. Cytochrome P-450 Enzymes (CYPs) with 25-Hydroxylase Activity
1.4. Diseases/Conditions Associated with Mutations/Variants of CYP Genes
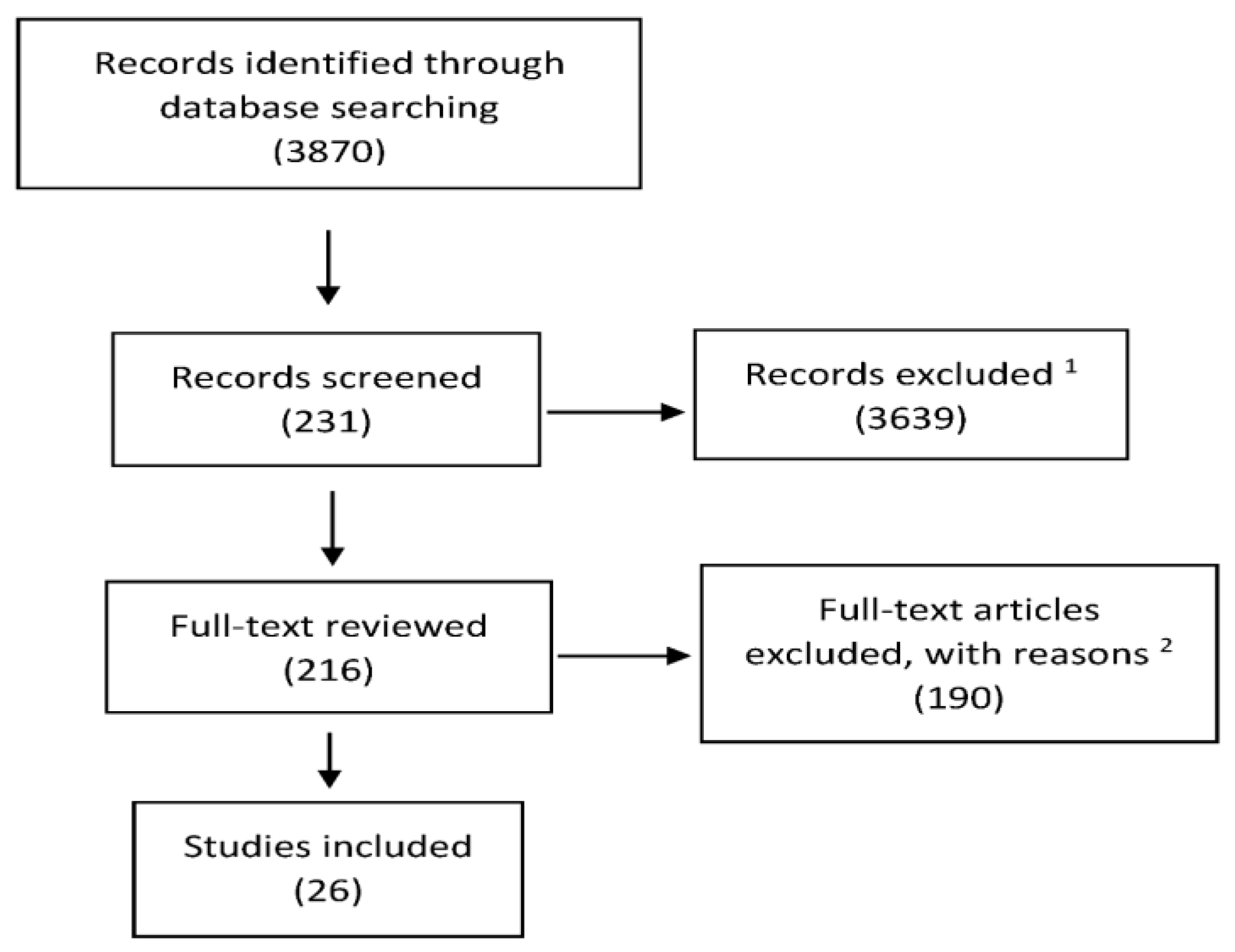
2. Materials and Methods
3. Vitamin D and Pathophysiological Related Effects on Calcium Absorption, Secondary Hyperparathyroidism, and Bone
3.1. Calcium Absorption
3.2. Secondary Hyperparathyroidism (SHPT)
3.3. Bone, Osteomalacia, Osteoporosis, and Fractures
4. Pharmacokinetics of Calcifediol
4.1. General Remarks
4.2. Randomized Double-Blind Controlled Trials
4.2.1. Bischoff-Ferrari et al.
4.2.2. Cashman et al.
4.2.3. Jetter et al.
4.2.4. Vaes et al.
4.3. Open-Label Trials
4.3.1. Barger-Lux et al.
4.3.2. Rossini et al.
4.3.3. Navarro-Valverde et al.
4.3.4. Shieh et al.
5. Calcifediol in Different Clinical Settings
5.1. Calcifediol in SHPT
5.2. Calcifediol and Increased Risk of Falls
5.3. Calcifediol and Malabsorption
5.4. Calcifediol and Obesity
5.5. Drugs Interfering with Calcifediol Metabolism
6. Conclusions: Time to Consider Calcifediol as an Adequate Supplement of Vitamin D?
Author Contributions
Funding
Conflicts of Interest
References
- Holick, M.F.; Siris, E.S.; Binkley, N.; Beard, M.K.; Khan, A.; Katzer, J.T.; Petruschke, R.A.; Chen, E.; De Papp, A.E. Prevalence of vitamin D inadequacy among postmenopausal north American women receiving osteoporosis therapy. J. Clin. Endocrinol. Metab. 2005, 90, 3215–3224. [Google Scholar] [CrossRef]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin d from the institute of medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Cesareo, R.; Iozzino, M.; D’Onofrio, L.; Terrinoni, I.; Maddaloni, E.; Casini, A.; Campagna, G.; Santonati, A.; Palermo, A. Effectiveness and safety of calcium and vitamin D treatment for postmenopausal osteoporosis. Minerva Endocrinol. 2015, 40, 231–237. [Google Scholar] [PubMed]
- Bouillon, R. Extra-skeletal effects of vitamin D. Front. Horm. Res. 2018, 50, 72–88. [Google Scholar] [PubMed]
- Quesada-Gómez, J.M.; Bouillon, R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos. Int. 2018, 29, 1697–1711. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; Kline, L.; Holick, M.F. Influence of season and latitude on the cutaneous synthesis of vitamin D3: Exposure to winter sunlight in boston and edmonton will not promote vitamin D3 synthesis in human skin. J. Clin. Endocrinol. Metab. 1988, 67, 373–378. [Google Scholar] [CrossRef]
- Holick, M.F. Resurrection of vitamin D deficiency and rickets. J. Clin. Investig. 2006, 116, 2062–2072. [Google Scholar] [CrossRef] [PubMed]
- Plum, L.A.; DeLuca, H.F. The functional metabolism and molecular biology of vitamin D action. Clin. Rev. Bone Miner. Metab. 2009, 7, 21–41. [Google Scholar] [CrossRef]
- Cashman, K.D.; Hayes, A.; Galvin, K.; Merkel, J.; Jones, G.; Kaufmann, M.; Hoofnagle, A.N.; Carter, G.D.; Durazo-Arvizu, R.A.; Sempos, C.T. Significance of serum 24,25-dihydroxyvitamin D in the assessment of vitamin D status: A double-edged sword? Clin. Chem. 2015, 61, 636–645. [Google Scholar] [CrossRef]
- DeLuca, H.F. Evolution of our understanding of vitamin D. Nutr. Rev. 2008, 66, S73–S87. [Google Scholar] [CrossRef] [PubMed]
- Vieth, R. The mechanisms of vitamin D toxicity. Bone Miner. 1990, 11, 267–272. [Google Scholar] [CrossRef]
- Verboven, C.; Bogaerts, I.; Waelkens, E.; Rabijns, A.; Van Baelen, H.; Bouillon, R.; De Ranter, C. Actin-DBP: The perfect structural fit? Acta Crystallogr. D Biol. Crystallogr. 2003, 59, 263–273. [Google Scholar] [CrossRef]
- Van Hoof, H.J.; De Sévaux, R.G.; Van Baelen, H.; Swinkels, L.M.; Klipping, C.; Ross, H.A.; Sweep, C.G. Relationship between free and total 1,25-dihydroxyvitamin D in conditions of modified binding. Eur. J. Endocrinol. 2001, 144, 391–396. [Google Scholar] [CrossRef]
- Hollis, B.W. Comparison of equilibrium and disequilibrium assay conditions for ergocalciferol, cholecalciferol and their major metabolites. J. Steriod Biochem. 1984, 21, 81–86. [Google Scholar] [CrossRef]
- Smith, J.E.; Goodman, D.S. The turnover and transport of vitamin D and of a polar metabolite with the properties of 25-hydroxycholecalciferol in human plasma. J. Clin. Investig. 1971, 50, 2159–2167. [Google Scholar] [CrossRef] [PubMed]
- Christakos, S.; Ajibade, D.V.; Dhawan, P.; Fechner, A.J.; Mady, L.J. Vitamin D: Metabolism. Rheum. Dis. Clin. N. Am. 2012, 38, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Fu, L.; Juras, D.J.; Karmali, M.; Wong, B.Y.; Gozdzik, A.; Cole, D.E. Common variants of the vitamin D binding protein gene and adverse health outcomes. Crit. Rev. Clin. Lab. Sci. 2013, 50, 1–22. [Google Scholar] [CrossRef]
- Lauridsen, A.L.; Vestergaard, P.; Hermann, A.P.; Brot, C.; Heickendorff, L.; Mosekilde, L.; Nexo, E. Plasma concentrations of 25-hydroxy-vitamin D and 1,25-dihydroxy-vitamin D are related to the phenotype of Gc (vitamin D-binding protein): A cross-sectional study on 595 early postmenopausal women. Calcif. Tissue Int. 2005, 77, 15–22. [Google Scholar] [CrossRef]
- Bouillon, R. Genetic and racial differences in the vitamin D endocrine system. Endocrinol. Metab. Clin. N. Am. 2017, 46, 1119–1135. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.B.; Motola, D.L.; Mangelsdorf, D.J.; Russell, D.W. De-orphanization of cytochrome P450 2R1: A microsomal vitamin D 25-hydroxilase. J. Biol. Chem. 2003, 278, 38084–38093. [Google Scholar] [CrossRef]
- Roizen, J.D.; Long, C.; Casella, A.; O’Lear, L.; Caplan, I.; Lai, M.; Sasson, I.; Singh, R.; Makowski, A.J.; Simmons, R.; et al. Obesity decreases hepatic 25-hydroxylase activity causing low serum 25-hydroxyvitamin D. J. Bone Miner. Res. 2019, e3686. [Google Scholar] [CrossRef] [PubMed]
- Aatsinki, S.M.; Elkhwanky, M.S.; Kummu, O.; Karpale, M.; Buler, M.; Viitala, P.; Rinne, V.; Mutikainen, M.; Tavi, P.; Franko, A. Fasting-induced transcription factors repress vitamin D bioactivation, a mechanism for vitamin D deficiency in diabetes. Diabetes 2019, 68, 918–931. [Google Scholar] [CrossRef]
- Chakraborty, S.; Sarkar, A.K.; Bhattacharya, C.; Krishnan, P.; Chakraborty, S. A nontoxic case of vitamin D toxicity. Lab. Med. 2015, 46, 146–149. [Google Scholar] [CrossRef]
- Dusso, A.S.; Gomez-Alonso, C.; Cannata-Andia, J.B. The hypercalcaemia of CYP24A1 inactivation: new ways to improve diagnosis and treatment. Clin. Kidney J. 2015, 8, 456–458. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-wide association study of circulating vitamin D levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; Van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Anderson, D.; Holt, B.J.; Pennell, C.E.; Holt, P.G.; Hart, P.H.; Blackwell, J.M. Genome-wide association study of vitamin D levels in children: replication in the Western Australian Pregnancy Cohort (Raine) study. Genes Immun. 2014, 15, 578–583. [Google Scholar] [CrossRef]
- Molin, A.; Wiedemann, A.; Demers, N.; Kaufmann, M.; Do Cao, J.; Mainard, L.; Dousset, B.; Journeau, P.; Abeguile, G.; Coudray, N.; et al. Vitamin D-Dependent Rickets Type 1B (25-Hydroxylase Deficiency): A Rare Condition or a Misdiagnosed Condition? J. Bone Miner. Res. 2017, 32, 1893–1899. [Google Scholar] [CrossRef]
- Bu, F.X.; Armas, L.; Lappe, J.; Zhou, Y.; Gao, G.; Wang, H.W.; Recker, R.; Zhao, L.J. Comprehensive association analysis of nine candidate genes with serum 25-hydroxy vitamin D levels among healthy Caucasian subjects. Hum. Genet. 2010, 128, 549–556. [Google Scholar] [CrossRef]
- Elkum, N.; Alkayal, F.; Noronha, F.; Ali, M.M.; Melhem, M.; Al-Arouj, M.; Bennakhi, A.; Behbehani, K.; Alsmadi, O.; Abubaker, J. Vitamin D Insufficiency in Arabs and South Asians Positively Associates with Polymorphisms in GC and CYP2R1 Genes. PLoS ONE 2014, 9, e113102. [Google Scholar] [CrossRef] [PubMed]
- Nissen, J.; Rasmussen, L.B.; Ravn-Haren, G.; Andersen, E.W.; Hansen, B.; Andersen, R.; Mejborn, H.; Madsen, K.H.; Vogel, U. Common Variants in CYP2R1 and GC Genes Predict Vitamin D Concentrations in Healthy Danish Children and Adults. PLoS ONE 2014, 9, e89907. [Google Scholar] [CrossRef] [PubMed]
- Nissen, J.; Vogel, U.; Ravn-Haren, G.; Andersen, E.W.; Madsen, K.H.; Nexø, B.A.; Andersen, R.; Mejborn, H.; Bjerrum, P.J.; Rasmussen, L.B.; et al. Common variants in CYP2R1 and GC genes are both determinants of serum 25-hydroxyvitamin D concentrations after UVB irradiation and after consumption of vitamin D3–fortified bread and milk during winter in Denmark. Am. J. Clin. Nutr. 2015, 101, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.L.; Rees, J.R.; Peacock, J.L.; Mott, L.A.; Amos, C.I.; Bostick, R.M.; Figueiredo, J.C.; Ahnen, D.J.; Bresalier, R.S.; Burke, C.A.; et al. Genetic Variants in CYP2R1, CYP24A1, and VDR Modify the Efficacy of Vitamin D3 Supplementation for Increasing Serum 25-Hydroxyvitamin D Levels in a Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2014, 99, E2133–E2137. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhao, L.J.; Xu, X.; Ye, A.; Travers-Gustafson, D.; Zhou, B.; Wang, H.-W.; Zhang, W.; Hamm, L.L.; Deng, H.-W.; et al. DNA methylation levels of CYP2R1 and CYP24A1 predict vitamin D response variation. J. Steroid Biochem. Mol. Biol. 2014, 144, 207–214. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Zorzela, L.; Loke, Y.K.; Ioannidis, J.P.; Golder, S.; Santaguida, P.; Altman, D.G.; Moher, D.; Vohra, S.; PRISMA harms group. PRISMA harms checklist: Improving harms reporting in systematic reviews. BMJ 2016, 352, i157. [Google Scholar] [CrossRef]
- Need, A.G.; O’Loughlin, P.D.; Morris, H.A.; Coates, P.S.; Horowitz, M.; Nordin, B.E. Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J. Bone Miner. Res. 2008, 23, 1859–1863. [Google Scholar] [CrossRef]
- Heaney, R.P.; Dowell, M.S.; Hale, C.A.; Bendich, A. Calcium Absorption Varies within the Reference Range for Serum 25-Hydroxyvitamin D. J. Am. Coll. Nutr. 2003, 22, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, J.C.; Yalamanchili, V.; Smith, L.M. The Effect of Vitamin D on Calcium Absorption in Older Women. J. Clin. Endocrinol. Metab. 2012, 97, 3550–3556. [Google Scholar] [CrossRef] [PubMed]
- Aloia, J.F.; Dhaliwal, R.; Shieh, A.; Mikhail, M.; Fazzari, M.; Ragolia, L.; Abrams, S.A. Vitamin D supplementation increases calcium absorption without a threshold effect. Am. J. Clin. Nutr. 2014, 99, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Chapuy, M.C.; Schott, A.M.; Garnero, P.; Hans, D.; Delmas, P.D.; Meunier, P.J. Healthy elderly French women living at home have secondary hyperparathyroidism and high bone turnover in winter. EPIDOS Study Group. J. Clin. Endocrinol. Metab. 1996, 81, 1129–1133. [Google Scholar] [PubMed]
- Thomas, M.K.; Lloyd-Jones, D.M.; Thadhani, R.I.; Shaw, A.C.; Deraska, D.J.; Kitch, B.T.; Vamvakas, E.C.; Dick, I.M.; Prince, R.L.; Finkelstein, J.S. Hypovitaminosis D in medical inpatients. N. Engl. J. Med. 1998, 338, 777–783. [Google Scholar] [CrossRef]
- Munns, C.F.; Shaw, N.; Kiely, M.; Specker, B.L.; Thacher, T.D.; Ozono, K.; Michigami, T.; Tiosano, D.; Mughal, M.Z.; Mäkitie, O.; et al. Global Consensus Recommendations on Prevention and Management of Nutritional Rickets. J. Clin. Endocrinol. Metab. 2016, 101, 394–415. [Google Scholar] [CrossRef]
- Atapattu, N.; Shaw, N.; Högler, W. Relationship between serum 25-hydroxyvitamin D and parathyroid hormone in the search for a biochemical definition of vitamin D deficiency in children. Pediatr. Res. 2013, 74, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Chiang, C.; Sikaris, K.; Lu, Z.; Bui, M.; Zebaze, R.; Seeman, E. Serum 25-Hydroxyvitamin D Insufficiency in Search of a Bone Disease. J. Clin. Endocrinol. Metab. 2017, 102, 2321–2328. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Van Schoor, N.M.; Gielen, E.; Boonen, S.; Mathieu, C.; Vanderschueren, D.; Lips, P. Optimal Vitamin D Status: A Critical Analysis on the Basis of Evidence-Based Medicine. J. Clin. Endocrinol. Metab. 2013, 98, E1283–E1304. [Google Scholar] [CrossRef]
- Malabanan, A.; Veronikis, I.E.; Holick, M.F. Redefining vitamin D insufficiency. Lancet 1998, 351, 805–806. [Google Scholar] [CrossRef]
- Lips, P.; Duong, T.; Oleksik, A.; Black, D.; Cummings, S.; Cox, D.; Nickelsen, T. A Global Study of Vitamin D Status and Parathyroid Function in Postmenopausal Women with Osteoporosis: Baseline Data from the Multiple Outcomes of Raloxifene Evaluation Clinical Trial. J. Clin. Endocrinol. Metab. 2001, 86, 1212–1221. [Google Scholar] [CrossRef]
- Lotito, A.; Teramoto, M.; Cheung, M.; Becker, K.; Sukumar, D. Serum Parathyroid Hormone Responses to Vitamin D Supplementation in Overweight/Obese Adults: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Nutrients 2017, 9, 241. [Google Scholar] [CrossRef]
- Priemel, M.; von Domarus, C.; Klatte, T.O.; Kessler, S.; Schlie, J.; Meier, S.; Proksch, N.; Pastor, F.; Netter, C.; Streichert, T.; et al. Bone mineralization defects and vitamin D deficiency: Histomorphometric analysis of iliac crest bone biopsies and circulating 25-hydroxyvitamin D in 675 patients. J. Bone Miner. Res. 2010, 25, 305–331. [Google Scholar] [CrossRef]
- Hansen, K.E.; Johnson, R.E.; Chambers, K.R.; Johnson, M.G.; Lemon, C.C.; Vo, T.N.T.; Marvdashti, S. Treatment of vitamin D insufficiency in postmenopausal women: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1612–1621. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Giovannucci, E.; Dietrich, T.; Dawson-Hughes, B. Fracture prevention with vitamin D supplementation: A meta-analysis of randomized controlled trials. JAMA 2005, 293, 2257–2264. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Stuck, A.E.; Staehelin, H.B.; Orav, E.J.; Thoma, A.; Kiel, D.P.; Henschkowski, J. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: A meta-analysis of randomized controlled trials. Arch. Intern. Med. 2009, 169, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Orav, E.J.; Oray, E.J.; Lips, P.; Meunier, P.J.; Lyons, R.A.; Flicker, L.; Wark, J.; Jackson, R.D.; et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N. Engl. J. Med. 2012, 367, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Avenell, A.; Gillespie, W.J.; Gillespie, L.D.; O’Connell, D. Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst. Rev. 2005, 30, CD000227. [Google Scholar]
- Bolland, M.J.; Grey, A.; Avenell, A. Effects of vitamin D supplementation on musculoskeletal health: A systematic review, meta-analysis, and trial sequential analysis. Lancet Diabetes Endocrinol. 2018, 6, 847–858. [Google Scholar] [CrossRef]
- Stamp, T. C Intestinal absorption of 25-hydroxycholecalciferol. Lancet 1974, 2, 121–123. [Google Scholar] [CrossRef]
- Heaney, R.P.; Horst, R.L.; Cullen, D.M.; Armas, L.A. Vitamin D3 distribution and status in the body. J. Am. Nutr. 2009, 28, 252–256. [Google Scholar] [CrossRef]
- Heaney, R.P.; Armas, L.A.; Shary, J.R.; Bell, N.H.; Binkley, N.; Hollis, B.W. 25-Hydroxylation of vitamin D3: relation to circulating vitamin D3 under various input conditions. Am. J. Clin. Nutr. 2008, 87, 1738–1742. [Google Scholar] [CrossRef]
- Minisola, S.; Cianferotti, L.; Biondi, P.; Cipriani, C.; Fossi, C.; Franceschelli, F.; Giusti, F.; Leoncini, G.; Pepe, J.; Bischoff-Ferrari, H.A.; et al. Correction of vitamin D status by calcidiol: pharmacokinetic profile, safety, and biochemical effects on bone and mineral metabolism of daily and weekly dosage regimens. Osteoporos. Int. 2017, 28, 3239–3249. [Google Scholar] [CrossRef]
- Cianferotti, L.; Cricelli, C.; Kanis, J.A.; Nuti, R.; Reginster, J.Y.; Ringe, J.D.; Rizzoli, R.; Brandi, M.L. The clinical use of vitamin D metabolites and their potential developments: A position statement from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) and the International Osteoporosis Foundation (IOF). Endocrine 2015, 50, 12–26. [Google Scholar] [CrossRef] [PubMed]
- Vaes, A.M.M.; Tieland, M.; De Regt, M.F.; Wittwer, J.; Van Loon, L.J.C.; De Groot, L.C.G.P. Dose–response effects of supplementation with calcifediol on serum 25-hydroxyvitamin D status and its metabolites: A randomized controlled trial in older adults. Clin. Nutr. 2018, 37, 808–814. [Google Scholar] [CrossRef]
- Wagner, D.; Hanwell, H.E.; Schnabl, K.; Yazdanpanah, M.; Kimball, S.; Fu, L.; Sidhom, G.; Rousseau, D.; Cole, D.E.; Vieth, R. The ratio of serum 24,25-dihydroxyvitamin D3 to 25-hydroxyvitamin D3 is predictive of 25-hydroxyvitamin D3 response to vitamin D3 supplementation. J. Steriod. Biochem. Mol. Biol. 2011, 126, 72–77. [Google Scholar] [CrossRef] [PubMed]
- De Boer, I.H.; Sachs, M.C.; Chonchol, M.; Himmelfarb, J.; Hoofnagle, A.N.; Ix, J.H.; Kremsdorf, R.A.; Lin, Y.S.; Mehrotra, R.; Robinson-Cohen, C.; et al. Estimated GFR and circulating 24,25-dihydroxyvitamin D3 concentration: A participant-level analysis of 5 cohort studies and clinical trials. Am. J. Kidney Dis. 2014, 64, 187–197. [Google Scholar] [CrossRef]
- Stamp, T.C.; Haddad, J.G.; Twigg, C.A. Comparison of oral 25-hydroxycholecalciferol, vitamin D, and ultraviolet light as determinants of circulating 25-hydroxyvitamin D. Lancet 1977, 1, 1341–1343. [Google Scholar] [CrossRef]
- Shieh, A.; Ma, C.; Chun, R.F.; Witzel, S.; Rafison, B.; Contreras, H.T.M.; Wittwer-Schegg, J.; Swinkels, L.; Huijs, T.; Hewison, M.; et al. Effects of Cholecalciferol vs Calcifediol on Total and Free 25-Hydroxyvitamin D and Parathyroid Hormone. J. Clin. Endocrinol. Metab. 2017, 102, 1133–1140. [Google Scholar] [CrossRef]
- Navarro-Valverde, C.; Sosa-Henríquez, M.; Alhambra-Expósito, M.R.; Quesada-Gómez, J.M. Vitamin D3 and calcidiol are not equipotent. J. Steriod. Biochem. Mol. Biol. 2016, 164, 205–208. [Google Scholar] [CrossRef]
- Rossini, M.; Viapiana, O.; Gatti, D.; James, G.; Girardello, S.; Adami, S. The long term correction of vitamin D deficiency: Comparison between different treatments with vitamin D in clinical practice. Minerva Med. 2005, 96, 1–7. [Google Scholar]
- Barger-Lux, M.J.; Heaney, R.P.; Dowell, S.; Chen, T.C.; Holick, M.F. Vitamin D and its major metabolites: serum levels after graded oral dosing in healthy men. Osteoporos. Int. 1998, 8, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Jetter, A.; Egli, A.; Dawson-Hughes, B.; Staehelin, H.B.; Stoecklin, E.; Goessl, R.; Henschkowski, J.; Bischoff-Ferrari, H.A. Pharmacokinetics of oral vitamin D3 and calcifediol. Bone 2014, 59, 14–19. [Google Scholar] [CrossRef]
- Cashman, K.D.; Seamans, K.M.; Lucey, A.J.; Stöcklin, E.; Weber, P.; Kiely, M.; Hill, T.R. Relative effectiveness of oral 25-hydroxyvitamin D3 and vitamin D3 in raising wintertime serum 25-hydroxyvitamin D in older adults. Am. J. Clin. Nutr. 2012, 95, 1350–1356. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Stöcklin, E.; Sidelnikov, E.; Willett, W.C.; Edel, J.O.; Stähelin, H.B.; Wolfram, S.; Jetter, A.; Schwager, J.; et al. Oral supplementation with 25(OH)D 3 versus vitamin D3: Effects on 25(OH)D levels, lower extremity function, blood pressure, and markers of innate immunity. J. Bone Miner. Res. 2012, 27, 160–169. [Google Scholar] [CrossRef]
- Alvarez, J.A.; Law, J.; Coakley, K.E.; Zughaier, S.M.; Hao, L.; Shahid Salles, K.; Wasse, H.; Gutiérrez, O.M.; Ziegler, T.R.; Tangpricha, V. High-dose cholecalciferol reduces parathyroid hormone in patients with early chronic kidney disease: a pilot, randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2012, 96, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Li, Y.C.; Zhao, S.M.; Li, Z.-X. Evaluation of responses to vitamin D3 (cholecalciferol) in patients on dialysis: a systematic review and meta-analysis. J. Investig. Med. 2016, 64, 1050–1059. [Google Scholar] [CrossRef]
- Loh, H.H.; Lim, L.L.; Yee, A.; Loh, H.S.; Vethakkan, S.R. Effect of vitamin D replacement in primary hyperparathyroidism with concurrent vitamin D deficiency: A systematic review and meta-analysis. Minerva Endocrinol. 2017. [Google Scholar]
- Ketteler, M.; Block, G.A.; Evenepoel, P.; Fukagawa, M.; Herzog, C.A.; McCann, L.; Moe, S.M.; Shroff, R.; Tonelli, M.A.; Toussaint, N.D.; et al. Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder: Synopsis of the Kidney Disease: Improving Global Outcomes 2017 Clinical Practice Guideline Update. Ann. Intern. Med. 2018, 168, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Petkovich, M.; Melnick, J.; White, J.; Tabash, S.; Strugnell, S.; Bishop, C.W. Modified-release oral calcifediol corrects vitamin D insufficiency with minimal CYP24A1 upregulation. J. Steroid Biochem. Mol. Biol. 2015, 148, 283–289. [Google Scholar] [CrossRef]
- Sprague, S.M.; Silva, A.L.; Al-Saghir, F.; Damle, R.; Tabash, S.P.; Petkovich, M.; Messner, E.J.; White, J.A.; Melnick, J.Z.; Bishop, C.W. Modified-Release Calcifediol Effectively Controls Secondary Hyperparathyroidism Associated with Vitamin D Insufficiency in Chronic Kidney Disease. Am. J. Nephrol. 2014, 40, 535–545. [Google Scholar] [CrossRef]
- Galassi, A.; Bellasi, A.; Ciceri, P.; Pivari, F.; Conte, F.; Cozzolino, M. Calcifediol to treat secondary hyperparathyroidism in patients with chronic kidney disease. Expert Rev. Clin. Pharmacol. 2017, 10, 1073–1084. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Orav, E.J.; Staehelin, H.B.; Meyer, O.W.; Theiler, R.; Dick, W.; Willett, W.C.; Egli, A. Monthly high-dose vitamin D treatment for the prevention of functional decline: A randomized clinical trial. JAMA Intern. Med. 2016, 176, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Nechama, H.; Noff, D.; Edelstein, S.; Harell, A. The intestinal absorption of cholecalciferol metabolites in the rat. Harefuah 1978, 95, 3–5. [Google Scholar] [PubMed]
- Davies, M.; Mawer, E.B.; Krawitt, E.L. Comparative absorption of vitamin D3 and 25-hydroxyvitamin D3 in intestinal disease. Gut 1980, 21, 287–292. [Google Scholar] [CrossRef]
- Cianferotti, L.; Bertoldo, F.; Bischoff-Ferrari, H.A.; Bruyere, O.; Cooper, C.; Cutolo, M.; Kanis, J.A.; Kaufman, J.M.; Reginster, J.Y.; Rizzoli, R.; et al. Vitamin D supplementation in the prevention and management of major chronic diseases not related to mineral homeostasis in adults: research for evidence and a scientific statement from the European society for clinical and economic aspects of osteoporosis and osteoarthritis (ESCEO). Endocrine 2017, 56, 245–261. [Google Scholar] [PubMed]
- Cesareo, R.; Attanasio, R.; Caputo, M.; Castello, R.; Chiodini, I.; Falchetti, A.; Guglielmi, R.; Papini, E.; Santonati, A.; Scillitani, A.; et al. Italian association of clinical endocrinologists (AME) and Italian AACE chapter position statement: clinical management of vitamin D deficiency in adults. Nutrients 2018, 10, 546. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E. The Importance of β-Cell Failure in the Development and Progression of Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2001, 86, 4047–4058. [Google Scholar]
- Ludwig, D.S. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Pourshahidi, L.K. Vitamin D and obesity: Current perspectives and future directions. Proc. Nutr. Soc. 2015, 74, 115–124. [Google Scholar] [CrossRef]
- Biondi, P.; Pepe, J.; Biamonte, F.; Occhiuto, M.; Parisi, M.; Demofonti, C.; Baffa, V.; Minisola, S.; Cipriani, C. Oral calcidiol is a good form of vitamin D supplementation. Clin. Cases Miner. Bone Metab. 2017, 14, 207–208. [Google Scholar] [CrossRef] [PubMed]
- Di Nisio, A.; De Toni, L.; Sabovic, I.; Rocca, M.S.; De Filippis, V.; Opocher, G.; Azzena, B.; Vettor, R.; Plebani, M.; Foresta, C. Impaired Release of Vitamin D in Dysfunctional Adipose Tissue: New Cues on Vitamin D Supplementation in Obesity. J. Clin. Endocrinol. Metab. 2017, 102, 2564–2574. [Google Scholar] [CrossRef]
- Khandalavala, B.N.; Hibma, P.P.; Fang, X. Prevalence and Persistence of Vitamin D Deficiency in Biliopancreatic Diversion Patients: A Retrospective Study. Obes. Surg. 2010, 20, 881–884. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.T.; Souza, F.I.; Fonseca, F.L.; Lazaretti-Castro, M.; Sarni, R.O. [Changes in bone metabolism markers in women after Roux-en-Y gastric bypass]. Arq. Bras. Endocrinol. Metabol. 2012, 56, 376–382. [Google Scholar] [CrossRef]
- Heber, D.; Greenway, F.L.; Kaplan, L.M.; Livingston, E.; Salvador, J.; Still, C.; Endocrine Society. Endocrine and nutritional management of the post-bariatric surgery patient: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2010, 95, 4823–4843. [Google Scholar] [CrossRef]
- Mechanick, J.I.; Youdim, A.; Jones, D.B.; Garvey, W.T.; Hurley, D.L.; McMahon, M.M.; Heinberg, L.J.; Kushner, R.; Adams, T.D.; Shikora, S.; et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient—2013 update: Cosponsored by American association of clinical endocrinologists, the obesity society, and american society for metabolic & bariatric surgery. Endocr. Pract. 2013, 19, 337–372. [Google Scholar]
- Fried, M.; Yumuk, V.; Oppert, J.M.; Scopinaro, N.; Torres, A.; Weiner, R.; Yashkov, Y.; Frühbeck, G.; International Federation for Surgery of Obesity and Metabolic Disorders-European Chapter (IFSO-EC); European Association for the Study of Obesity (EASO); et al. Interdisciplinary European guidelines on metabolic and bariatric surgery. Obes. Surg. 2014, 24, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Brancatella, A.; Cappellani, D.; Vignali, E.; Canale, D.; Marcocci, C. Calcifediol Rather Than Cholecalciferol for a Patient Submitted to Malabsortive Bariatric Surgery: A Case Report. J. Endocr. Soc. 2017, 1, 1079–1084. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, R.H.; Lyles, K.W.; Colón-Emeric, C. A Review of the Effect of Anticonvulsant Medications on Bone Mineral Density and Fracture Risk. Am. J. Geriatr. Pharmacother. 2010, 8, 34–46. [Google Scholar] [CrossRef]
- Gröber, U.; Kisters, K. Influence of drugs on vitamin D and calcium metabolism. Dermato-Endocrinology 2012, 4, 158–166. [Google Scholar] [CrossRef]
- Arora, E.; Singh, H.; Gupta, Y.K. Impact of antiepileptic drugs on bone health: Need for monitoring, treatment, and prevention strategies. J. Fam. Med. Prim. Care 2016, 5, 248–253. [Google Scholar]
- Petty, S.J.; Wilding, H.; Wark, J.D. Osteoporosis Associated with Epilepsy and the Use of Anti-Epileptics—A Review. Curr. Osteoporos. Rep. 2016, 14, 54–65. [Google Scholar] [CrossRef]
- Boerman, R.; Cohen, D.; Schulte, P.F.; Nugter, A. Prevalence of Vitamin D Deficiency in Adult Outpatients With Bipolar Disorder or Schizophrenia. J. Clin. Psychopharmacol. 2016, 36, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Belvederi Murri, M.; Respino, M.; Masotti, M.; Innamorati, M.; Mondelli, V.; Pariante, C.; Amore, M. Vitamin D and psychosis: Mini meta-analysis. Schizophr. Res. 2013, 150, 235–239. [Google Scholar] [CrossRef]
- Krivoy, A.; Onn, R.; Vilner, Y.; Hochman, E.; Weizman, S.; Paz, A.; Hess, S.; Sagy, R.; Kimhi-Nesher, S.; Kalter, E.; et al. Vitamin D Supplementation in chronic schizophrenia patients treated with clozapine: A randomized, double-blind, placebo-controlled clinical trial. EBioMedicine 2017, 26, 138–145. [Google Scholar] [CrossRef]
- Tombini, M.; Palermo, A.; Assenza, G.; Pellegrino, G.; Benvenga, A.; Campana, C.; Naciu, A.M.; Assenza, F.; Lazzaro, V.D. Calcium metabolism serum markers in adult patients with epilepsy and the effect of vitamin D supplementation on seizure control. Seizure 2018, 58, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, H.; Mohammed, H.T.; Patel, T. Vitamin D supplementation for bone health in adults with epilepsy: A systematic review. Epilepsia 2018, 59, 885–896. [Google Scholar] [CrossRef]
- Balvers, M.G.; Brouwer-Brolsma, E.M.; Endenburg, S.; de Groot, L.C.; Kok, F.J.; Gunnewiek, J.K. Recommended intakes of vitamin D to optimise health, associated circulating 25-hydroxyvitamin D concentrations, and dosing regimens to treat deficiency: workshop report and overview of current literature. J. Nutr. Sci. 2015, 4, e23. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Ernst, J.B.; Gummert, J.F.; Borgermann, J. Vitamin D supplementation, body weight and human serum 25-hydroxyvitamin D response: A systematic review. Eur. J. Nutr. 2014, 53, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.S.; Schoenmakers, I.; Bluck, L.J.; Ding, S.; Prentice, A. Plasma appearance and disappearance of an oral dose of 25-hydroxyvitamin D2 in healthy adults. Br. J. Nutr. 2012, 107, 1128–1137. [Google Scholar] [CrossRef]
- Stamp, T.C.B.; Round, J.M.; Haddad, J.G. Effect of oral vitamin D, 25-hydroxycholecalciferol (25-HCC) and whole-body ultra-violet irradiation on plasma 25-HCC levels in man. Clin. Sci. 1973, 44, 3P–4P. [Google Scholar] [CrossRef] [PubMed]
- Haddad, J.G.; Stamp, T.C.B. Circulating 25-hydroxyvitamin D in man. Am. J. Med. 1974, 57, 57–62. [Google Scholar] [CrossRef]
- Ortego-Jurado, M.; Callejas-Rubio, J.-L.; Rios-Fernandez, R.; González-Moreno, J.; Ramírez, A.R.G.; González-Gay, M.A.; Ortego-Centeno, N. Oral calcidiol is more effective than cholecalciferol supplementation to reach adequate 25(OH)D levels in patients with autoimmune diseases chronically treated with low doses of glucocorticoids: A “real-life” study. J. Osteoporos. 2015, 2015, 729451. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, S.B. 25-Hydroxyvitamin D3 treatment of bone disease in primary biliary cirrhosis. Gastroenterology 1982, 83, 137–140. [Google Scholar] [PubMed]
- Compston, J.E. Hepatic osteodystrophy: vitamin D metabolism in patients with liver disease. Gut 1986, 27, 1073–1090. [Google Scholar] [CrossRef] [PubMed]
| Authors | Shieh [67] | Navarro-Valverde [68] | Rossini [69] | Barger-Lux [70] | Vaes [63] | Jetter [71] | Cashman [72] | Bischoff-Ferrari [73] |
|---|---|---|---|---|---|---|---|---|
| Type of study | Open-label RCT | Open-label RCT | Open-label RCT | Prospective open-label study | Double-blind RCT | Double-blind RCT | Double-blind, placebo-controlled RCT | Double-blind RCT |
| Study population | 35 multiethnic healthy adults ≥18 years of age with a mean BMI of 26.5 kg/m2 | 40 postmenopausal osteopenic women with an average age of 67 years and a BMI of 26.4 ± 4 kg/m2 | 271 postmenopausal osteopenic or osteoporotic women | 116 healthy adults with a mean age of 28 years | 59 subjects: men and women aged >65 years with a BMI between 20 and 35 kg/m2. | 35 healthy women aged 50–70 years with a baseline BMI between 18 and 29 kg/m2 | 56 healthy adults aged ≥50 years with a mean BMI of 28.3 ± 4.8 kg/m2 | 20 healthy postmenopausal women with a mean age of 61.5 ± 7.2 years and a BMI between 18 and 29 kg/m2 |
| Baseline Mean 25(OH)D level Assay | 16.6 ± 3.1 ng/mL chemiluminescence immunoassay | 37.5 ± 10 nmol/L HPLC and ultraviolet detection method | 22 nmol/L radioimmunoassay | 67 ± 25 nmol/L competitive protein-binding assay with chromatography | 39.4 ± 11.9 nmol/L IDXLC-MS/MS | 12.54 ± 3.51 ng/mL HPLC–MS/MS | 43.6 ± 12.3 nmol/L ELISA | 13.2 ± 3.9 ng/mL HPLC–MS/MS |
| Intervention | 60 μg chol/day 20 μg calcif/day for 16 weeks | 20 μg chol/day, 20 μg calcif/day, 266 μg calcif/week, 266 μg calcif/every other week for one year | 4000 IU calcif/week, 800–880 IU chol/day for one year | 25 μg chol/day, 250 μg chol/day, 1250 μg chol/day for 8 weeks; 10 μg calcif/day, 20 μg calcif/day, 50 μg calcif/day for 4 weeks 0.5 μg calcit/day, 1.0 μg calcit/day, 1.5 μg calcit/day for 2 weeks. | 20 μg chol/day; 5 μg calcif/day, 10 μg calcif/day, 15 μg calcif/day for 24 weeks | 20 μg chol/day; 20 μg calcif/day, 140 μg calcif/week, 140 μg chol/week, for 15 weeks, bolus 140 μg calcif, bolus 140 μg chol both bolus (chol and calcif) | placebo, 20 μg chol/day, 7 μg calcif/day, 20 μg calcif/day, for 10 weeks | 20 μg chol/day; 20 μg calcif/day, for 4 months |
| Results | The mean total 25(OH)D significantly increased to ≥30 ng/mL by 4 weeks of calcif, while among the chol group, the mean total 25(OH)D remained <30 ng/mL for the entire study. | Calcif increased to significantly higher 25(OH)D serum levels compared with daily chol. The increase in 25(OH)D serum levels was almost 2 times higher in the group treated with weekly calcif. | The compliance with the weekly calcif was over 90%, and determined serum levels of 25(OH)D were similar to those obtained with chol daily. | Treatment with calcif significantly increased 25(OH)D serum levels more than chol. | Calcif significantly elevated serum 25(OH)D concentrations more rapidly compared with chol. | 20 μg calcif given daily or 140 μg given weekly appeared to significantly correct vitamin D deficiency more rapidly and reliably than the same dose of daily or weekly chol. | 20 μg calcif daily significantly increased 25(OH)D serum levels more than either 20 μg chol or 7 μg calcif daily. | Immediate sustained and significant increase in 25(OH)D serum levels with calcif. |
| Notes | After 12 months, calcif was between 3 and 5 times more potent than chol. | Calcif was 3–6 times more potent in increasing 25(OH) D serum levels compared with chol. | The potency of calcif versus chol in increasing 25 (OH)D was 1.66. This study aimed to evaluate the compliance of patients and not the efficacy. | The potency in increasing 25(OH)D levels of calcif was 3.3–3.5 times more than chol at a low dosage and 7–8 times more for the highest dosages of both products. | 5 μg of calcif had a potency of about 1.04 versus chol, whereas for both other dosages (10–15 μg), the potency was about 3. | Daily calcif was 2–3 times more potent than chol, and weekly calcif was 5–6 times more potent than chol. | Calcif seemed to be 4.2–5 times more potent than chol. Both dosages of calcif reduced serum PTH vs. placebo. | Potency of calcif vs. chol was 3.4. Calcif demonstrated a decrease of 5.7 mmHg SBP. |
| Osteomalacia |
| Osteoporosis (particularly if bone-active drugs are to be used) |
| Older adults with a history of falls |
| Older adults with a history of non-traumatic fractures |
| Pregnant and lactating women |
| Obese children and adults |
| People with insufficient sun exposure |
| Malabsorption syndromes (congenital or acquired) and bariatric surgery |
| Chronic kidney disease |
| Hepatic failure |
| Cystic fibrosis |
| Hyperparathyroidism |
| People taking drugs that interfere with vitamin D metabolism (antiseizure medications, glucocorticoids, AIDS medications, antifungals, cholestyramine) |
| Granulomatous disorders and some lymphomas |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cesareo, R.; Falchetti, A.; Attanasio, R.; Tabacco, G.; Naciu, A.M.; Palermo, A. Hypovitaminosis D: Is It Time to Consider the Use of Calcifediol? Nutrients 2019, 11, 1016. https://doi.org/10.3390/nu11051016
Cesareo R, Falchetti A, Attanasio R, Tabacco G, Naciu AM, Palermo A. Hypovitaminosis D: Is It Time to Consider the Use of Calcifediol? Nutrients. 2019; 11(5):1016. https://doi.org/10.3390/nu11051016
Chicago/Turabian StyleCesareo, Roberto, Alberto Falchetti, Roberto Attanasio, Gaia Tabacco, Anda Mihaela Naciu, and Andrea Palermo. 2019. "Hypovitaminosis D: Is It Time to Consider the Use of Calcifediol?" Nutrients 11, no. 5: 1016. https://doi.org/10.3390/nu11051016
APA StyleCesareo, R., Falchetti, A., Attanasio, R., Tabacco, G., Naciu, A. M., & Palermo, A. (2019). Hypovitaminosis D: Is It Time to Consider the Use of Calcifediol? Nutrients, 11(5), 1016. https://doi.org/10.3390/nu11051016






