Dietary Protein Intake Level Modulates Mucosal Healing and Mucosa-Adherent Microbiota in Mouse Model of Colitis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Diets
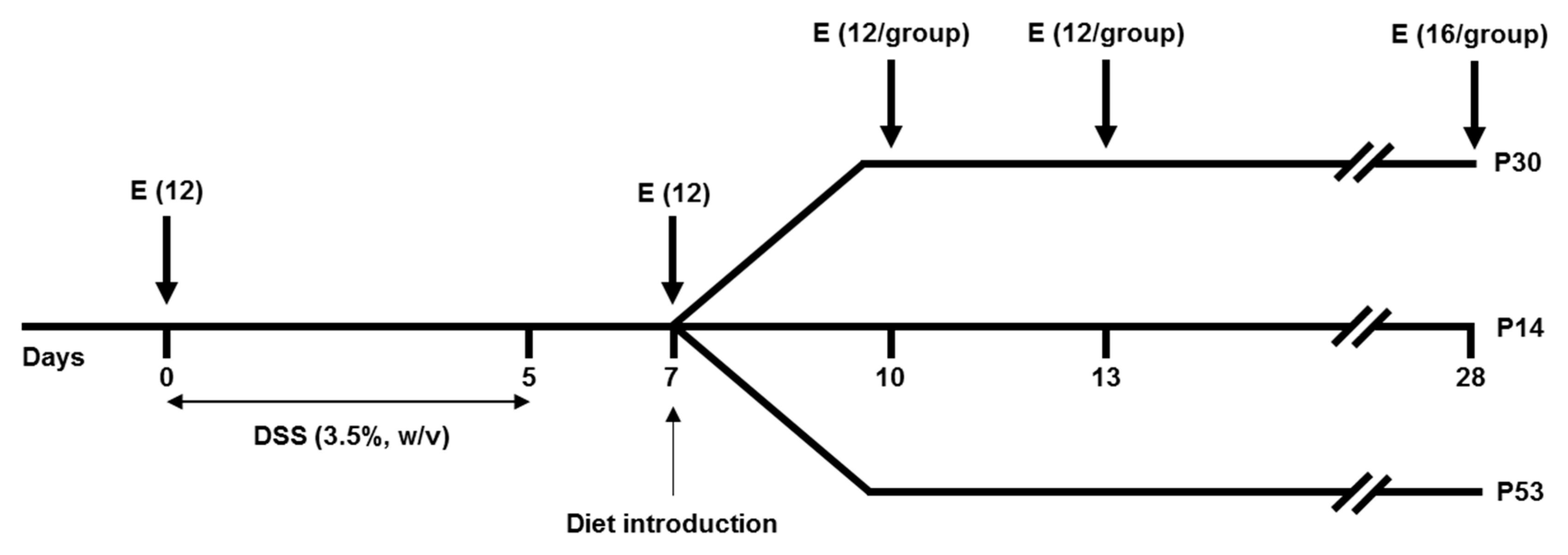
2.2. Experimental Design
2.3. Intestinal Permeability Assessment
2.4. Tissue Collection
2.4.1. Determination of Local and Systemic Inflammatory Markers
2.4.2. Histological Analysis
2.4.3. RNA Isolation and Quantitative Real-Time PCR
2.4.4. Western-Blot Analysis
2.4.5. Evaluation of Adherent Mucosal Microbiota Composition
2.5. Statistical Analysis
3. Results
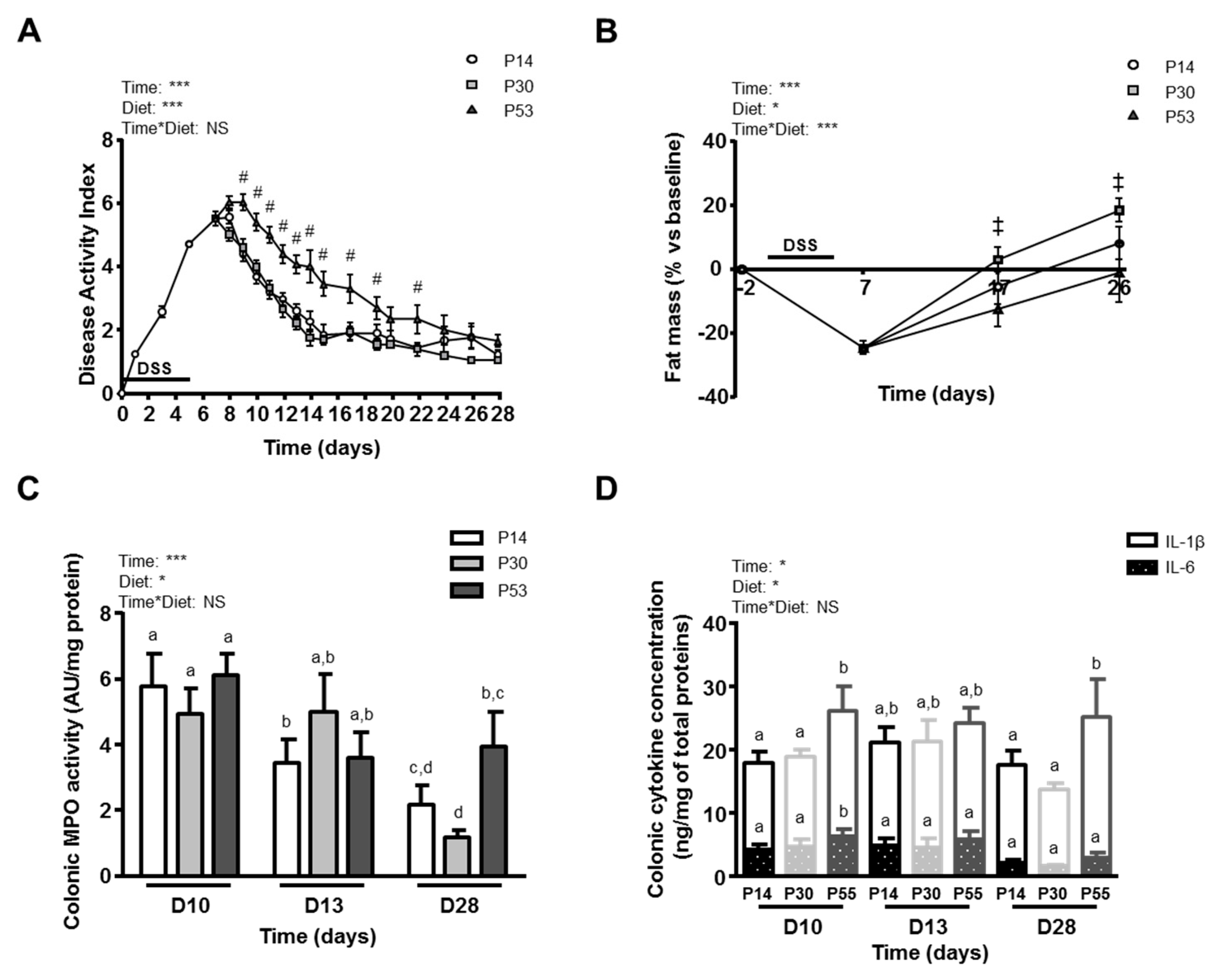
3.1. Dietary Protein Level Influences Colon Inflammation and Body Weight Recovery
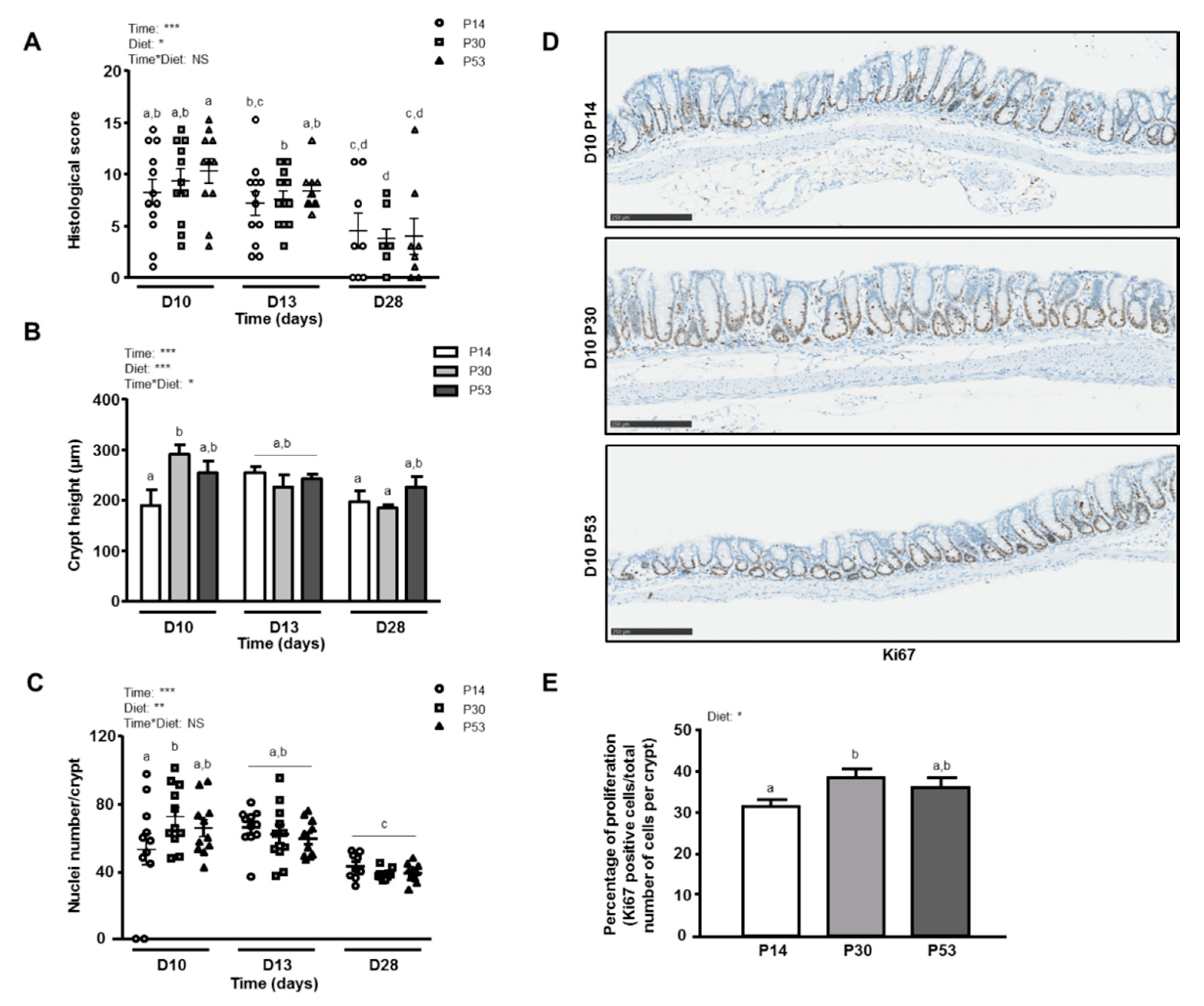
3.2. P30 Diet Enhances Epithelial Repair Features
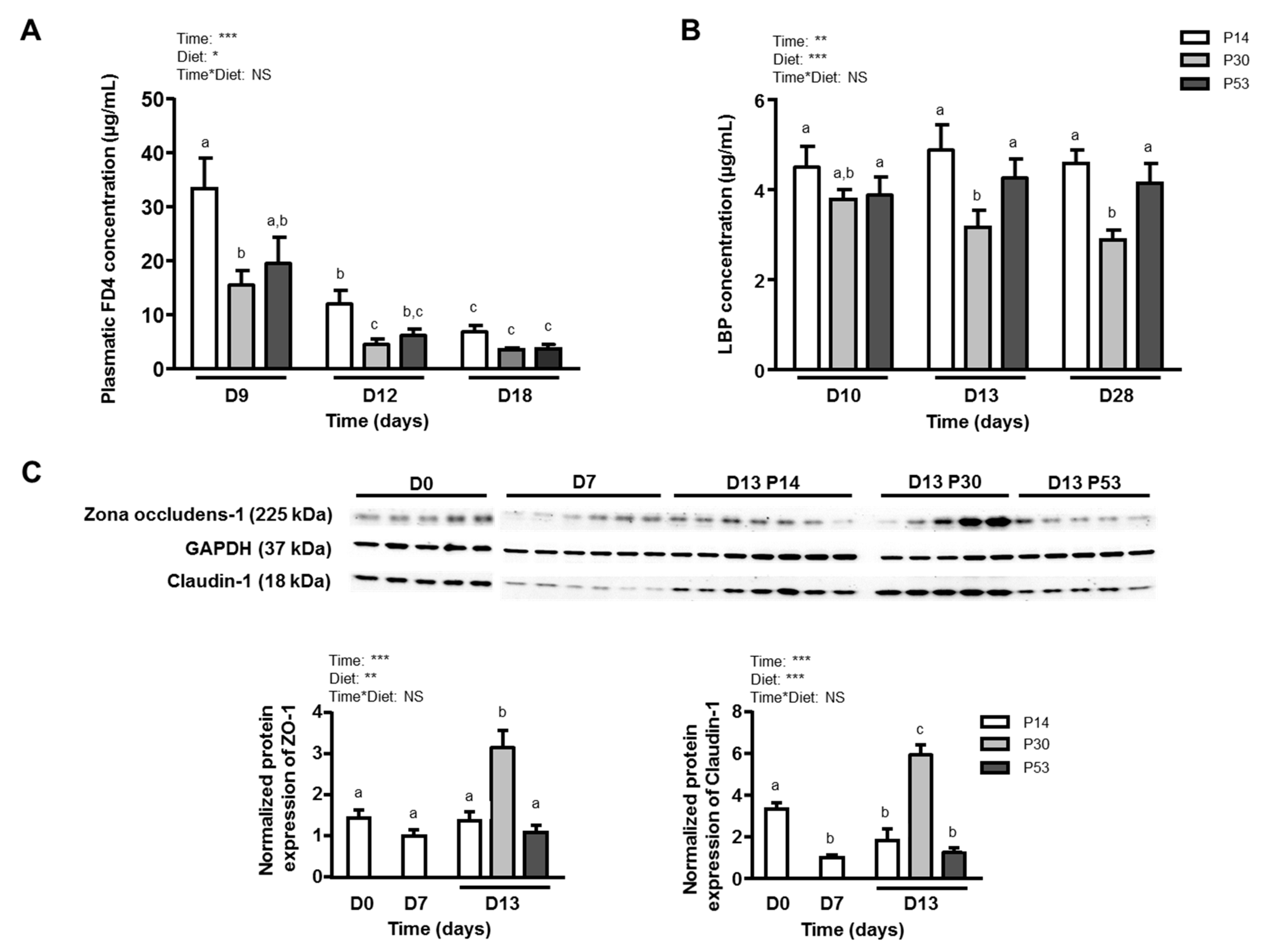
3.3. P30 Diet Improves Dss-Induced Barrier Function Alterations
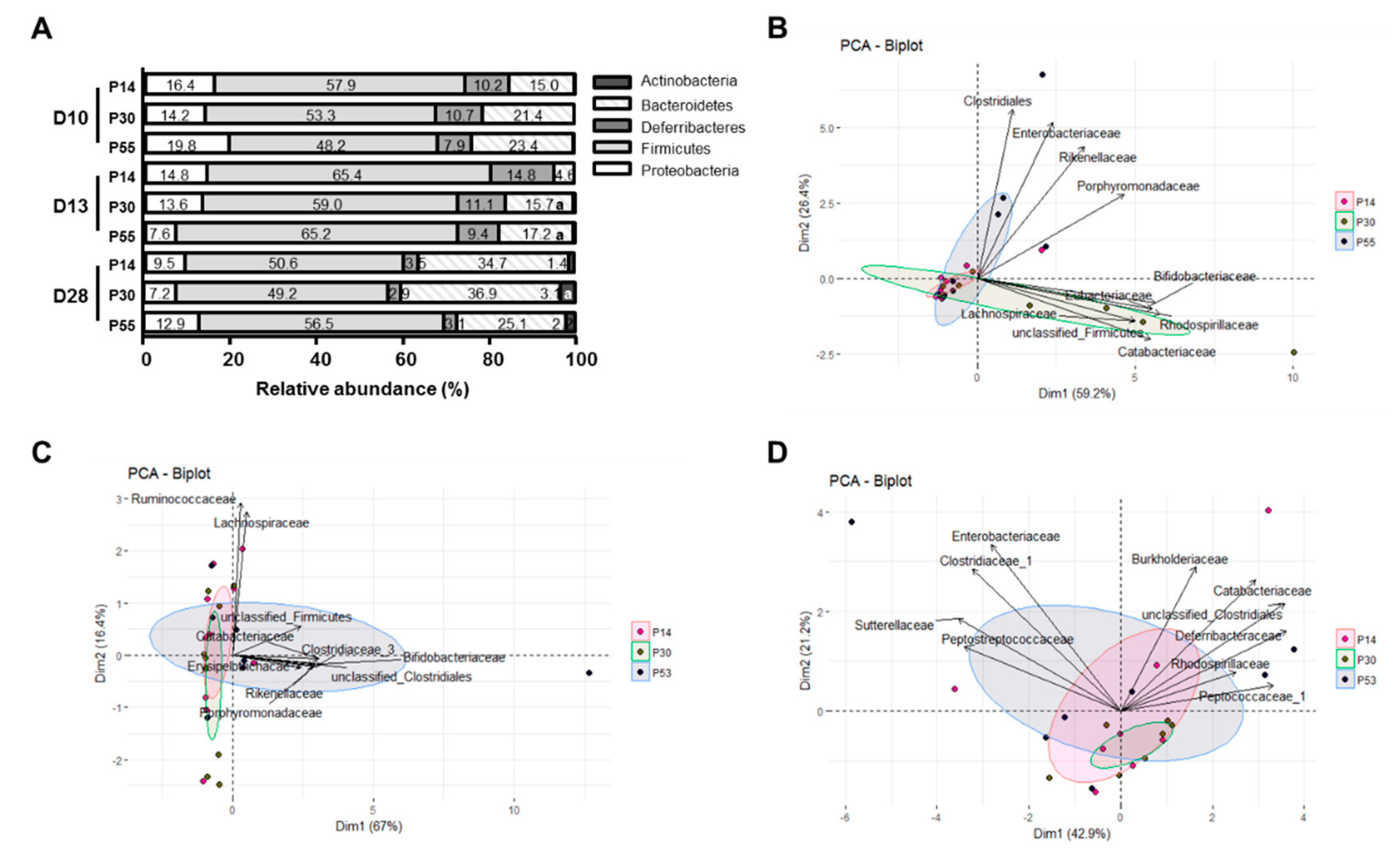
3.4. DSS-Induced Dysbiosis is not Alleviated by Diet, but P30 Diet Increases the Proportions of Butyrate-Producing Bacteria
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Danese, S.; Fiocchi, C. Ulcerative colitis. N. Engl. J. Med. 2011, 365, 1713–1725. [Google Scholar] [CrossRef] [PubMed]
- Neurath, M.F.; Travis, S.P.L. Mucosal healing in inflammatory bowel diseases: A systematic review. Gut 2012, 61, 1619–1635. [Google Scholar] [CrossRef] [PubMed]
- Papi, C.; Fascì-Spurio, F.; Rogai, F.; Settesoldi, A.; Margagnoni, G.; Annese, V. Mucosal healing in inflammatory bowel disease: Treatment efficacy and predictive factors. Dig. Liver Dis. 2013, 45, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Neurath, M.F. New targets for mucosal healing and therapy in inflammatory bowel diseases. Mucosal Immunol. 2014, 7, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A.; Dignass, A.U. Epithelial restitution and wound healing in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Forbes, A.; Goldesgeyme, E.; Paulon, E. Nutrition in inflammatory bowel disease. JPEN. J. Parenter. Enteral Nutr. 2011, 35, 571–580. [Google Scholar] [CrossRef]
- Forbes, A.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN guideline: Clinical nutrition in inflammatory bowel disease. Clin. Nutr. 2017, 36, 321–347. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Lletjós, S.; Beaumont, M.; Tomé, D.; Benamouzig, R.; Blachier, F.; Lan, A. Dietary Protein and Amino Acid Supplementation in Inflammatory Bowel Disease Course: What Impact on the Colonic Mucosa? Nutrients 2017, 9, 310. [Google Scholar] [CrossRef] [PubMed]
- Andriamihaja, M.; Davila, A.-M.; Eklou-Lawson, M.; Petit, N.; Delpal, S.; Allek, F.; Blais, A.; Delteil, C.; Tomé, D.; Blachier, F. Colon luminal content and epithelial cell morphology are markedly modified in rats fed with a high-protein diet. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G1030–G1037. [Google Scholar] [CrossRef] [PubMed]
- Blachier, F.; Beaumont, M.; Andriamihaja, M.; Davila, A.-M.; Lan, A.; Grauso, M.; Armand, L.; Benamouzig, R.; Tomé, D. Changes in the luminal environment of the colonic epithelial cells and physiopathological consequences. Am. J. Pathol. 2017, 187, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Geypens, B.; Claus, D.; Evenepoel, P.; Hiele, M.; Maes, B.; Peeters, M.; Rutgeerts, P.; Ghoos, Y. Influence of dietary protein supplements on the formation of bacterial metabolites in the colon. Gut 1997, 41, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Blouin, J.-M.; Santacruz, A.; Lan, A.; Andriamihaja, M.; Wilkanowicz, S.; Benetti, P.-H.; Tomé, D.; Sanz, Y.; Blachier, F.; et al. High-protein diet modifies colonic microbiota and luminal environment but not colonocyte metabolism in the rat model: The increased luminal bulk connection. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G459–G470. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Cerrudo, V.; Audebert, M.; Dumont, F.; Mancano, G.; Khodorova, N.; Andriamihaja, M.; et al. Quantity and source of dietary protein influence metabolite production by gut microbiota and rectal mucosa gene expression: A randomized, parallel, double-blind trial in overweight humans. Am. J. Clin. Nutr. 2017, 106, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Blachier, F.; Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Audebert, M.; Khodorova, N.; Andriamihaja, M.; Airinei, G.; Benamouzig, R.; et al. High-protein diets for weight management: Interactions with the intestinal microbiota and consequences for gut health. A position paper by the my new gut study group. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lan, A.; Blais, A.; Coelho, D.; Capron, J.; Maarouf, M.; Benamouzig, R.; Lancha, A.H.; Walker, F.; Tomé, D.; Blachier, F. Dual effects of a high-protein diet on DSS-treated mice during colitis resolution phase. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G624–G633. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, X.; Hu, C.-A. Therapeutic Potential of Amino Acids in Inflammatory Bowel Disease. Nutrients 2017, 9, 920. [Google Scholar] [CrossRef] [PubMed]
- Sprong, R.C.; Schonewille, A.J.; van der Meer, R. Dietary cheese whey protein protects rats against mild dextran sulfate sodium-induced colitis: Role of mucin and microbiota. J. Dairy Sci. 2010, 93, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Faure, M.; Moënnoz, D.; Montigon, F.; Mettraux, C.; Breuillé, D.; Ballèvre, O. Dietary threonine restriction specifically reduces intestinal mucin synthesis in rats. J. Nutr. 2005, 135, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Gaifem, J.; Gonçalves, L.G.; Dinis-Oliveira, R.J.; Cunha, C.; Carvalho, A.; Torrado, E.; Rodrigues, F.; Saraiva, M.; Castro, A.G.; Silvestre, R. L-Threonine Supplementation During Colitis Onset Delays Disease Recovery. Front. Physiol. 2018, 9, 1247. [Google Scholar] [CrossRef] [PubMed]
- Lan, A.; Blachier, F.; Benamouzig, R.; Beaumont, M.; Barrat, C.; Coelho, D.; Lancha, A.; Kong, X.; Yin, Y.; Marie, J.-C.; et al. Mucosal healing in inflammatory bowel diseases: Is there a place for nutritional supplementation? Inflamm. Bowel Dis. 2015, 21, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Chemouny, J.M.; Gleeson, P.J.; Abbad, L.; Lauriero, G.; Boedec, E.; Le Roux, K.; Monot, C.; Bredel, M.; Bex-Coudrat, J.; Sannier, A.; et al. Modulation of the microbiota by oral antibiotics treats immunoglobulin A nephropathy in humanized mice. Nephrol. Dial. Transplant. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dignass, A.; Lynch-devaney, K.; Kindon, H.; Thim, L.; Podolsky, D.K. Trefoil peptides promote epithelial migration through a transforming growth factor beta-independent pathway. J. Clin. Investig. 1994, 94, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Liu, J.; Wu, L.; Fan, Y.; Sun, L.; Qian, F.; Chen, D.; Ye, R.D. Elevated Expression of Serum Amyloid a 3 Protects Colon Epithelium against Acute Injury Through TLR2-Dependent Induction of Neutrophil IL-22 Expression in a Mouse Model of Colitis. Front. Immunol. 2018, 9, 1503. [Google Scholar] [CrossRef] [PubMed]
- Hiller, F.; Besselt, K.; Deubel, S.; Brigelius-Flohé, R.; Kipp, A.P. GPx2 induction is mediated through STAT transcription factors during acute colitis. Inflamm. Bowel Dis. 2015, 21, 2078–2089. [Google Scholar] [CrossRef] [PubMed]
- Sokol, H.; Seksik, P.; Furet, J.P.; Firmesse, O.; Nion-Larmurier, I.; Beaugerie, L.; Cosnes, J.; Corthier, G.; Marteau, P.; Doraé, J. Low counts of faecalibacterium prausnitzii in colitis microbiota. Inflamm. Bowel Dis. 2009, 15, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Machiels, K.; Joossens, M.; Sabino, J.; De Preter, V.; Arijs, I.; Eeckhaut, V.; Ballet, V.; Claes, K.; Van Immerseel, F.; Verbeke, K.; et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 2014, 63, 1275–1283. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, G.T.; Macfarlane, S. Bacteria, colonic fermentation, and gastrointestinal health. J. AOAC Int. 2012, 95, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.V.; Hao, L.; Offermanns, S.; Medzhitov, R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc. Natl. Acad. Sci. USA 2014, 111, 2247–2252. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.; Blachier, F.; Darcy-Vrillon, B.; de Coppet, P.; Bourreille, A.; Segain, J.-P.P. Butyrate utilization by the colonic mucosa in inflammatory bowel diseases: A transport deficiency. Inflamm. Bowel Dis. 2010, 16, 684–695. [Google Scholar] [CrossRef] [PubMed]
- Lucke, K.; Miehlke, S.; Jacobs, E.; Schuppler, M. Prevalence of Bacteroides and Prevotella spp. in ulcerative colitis. J. Med. Microbiol. 2006, 55, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.-Q.; Yu, T.-T.; Zhao, X.-J.; Zhang, Y.; Zhang, H.-J. Fecal microbial dysbiosis in Chinese patients with inflammatory bowel disease. World J. Gastroenterol. 2018, 24, 1464–1477. [Google Scholar] [CrossRef] [PubMed]
- Magee, E.A.; Richardson, C.J.; Hughes, R.; Cummings, J.H. Contribution of dietary protein to sulfide production in the large intestine: An in vitro and a controlled feeding study in humans. Am. J. Clin. Nutr. 2000, 72, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Andriamihaja, M.; Lan, A.; Beaumont, M.; Audebert, M.; Wong, X.; Yamada, K.; Yin, Y.; Tomé, D.; Carrasco-Pozo, C.; Gotteland, M.; et al. The deleterious metabolic and genotoxic effects of the bacterial metabolite p-cresol on colonic epithelial cells. Free Radic. Biol. Med. 2015, 85, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Andriamihaja, M.; Lan, A.; Khodorova, N.; Audebert, M.; Blouin, J.M.; Grauso, M.; Lancha, L.; Benetti, P.H.; Benamouzig, R.; et al. Detrimental effects for colonocytes of an increased exposure to luminal hydrogen sulfide: The adaptive response. Free Radic. Biol. Med. 2016, 93, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Portune, K.J.; Beaumont, M.; Davila, A.-M.; Tomé, D.; Blachier, F. Gut microbiota role in dietary protein metabolism and health-related outcomes: The two sides of the coin. Trends Food Sci. Technol. 2016, 57, 213–232. [Google Scholar] [CrossRef]
- Llewellyn, S.R.; Britton, G.J.; Contijoch, E.J.; Vennaro, O.H.; Mortha, A.; Colombel, J.F.; Grinspan, A.; Clemente, J.C.; Merad, M.; Faith, J.J. Interactions Between Diet and the Intestinal Microbiota Alter Intestinal Permeability and Colitis Severity in Mice. Gastroenterology 2018, 154, 1037–1046.e2. [Google Scholar] [CrossRef] [PubMed]
- Jantchou, P.; Morois, S.; Clavel-Chapelon, F.; Boutron-Ruault, M.-C.; Carbonnel, F. Animal protein intake and risk of inflammatory bowel disease: The E3N prospective study. Am. J. Gastroenterol. 2010, 105, 2195–2201. [Google Scholar] [CrossRef] [PubMed]
- Jowett, S.L.; Seal, C.J.; Pearce, M.S.; Phillips, E.; Gregory, W.; Barton, J.R.; Welfare, M.R. Influence of dietary factors on the clinical course of ulcerative colitis: A prospective cohort study. Gut 2004, 53, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Spooren, C.E.G.M.; Pierik, M.J.; Zeegers, M.P.; Feskens, E.J.M.; Masclee, A.A.M.; Jonkers, D.M.A.E. Review article: The association of diet with onset and relapse in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2013, 38, 1172–1187. [Google Scholar] [CrossRef] [PubMed]
- Racine, A.; Carbonnel, F.; Chan, S.S.M.; Hart, A.R.; Bas Bueno-De-Mesquita, H.; Oldenburg, B.; Van Schaik, F.D.M.; Tjønneland, A.; Olsen, A.; Dahm, C.C.; et al. Dietary Patterns and Risk of Inflammatory Bowel Disease in Europe: Results from the EPIC Study. Inflamm. Bowel Dis. 2016, 22, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Kominsky, D.J.; Campbell, E.L.; Colgan, S.P. Metabolic shifts in immunity and inflammation. J. Immunol. 2010, 184, 4062–4068. [Google Scholar] [CrossRef] [PubMed]
| P14 | P30 | P53 | |
|---|---|---|---|
| Weight content (g/kg) | |||
| Acid casein (Armor Protéines®, ref. #139860) | 112 | 232 | 424 |
| Whey protein (Armor Protéines®, Protarmor 80, ref. #139805) | 28 | 58 | 96 |
| Corn starch | 622.4 | 493.2 | 287 |
| Sucrose | 100.3 | 79.5 | 45.7 |
| Cellulose | 50 | 50 | 50 |
| Soybean oil | 40 | 40 | 40 |
| Mineral Mixture (AIN 93-M) | 35 | 35 | 35 |
| Vitamin Mixture (AIN 93-V) | 10 | 10 | 10 |
| Choline | 2.3 | 2.3 | 2.3 |
| Energy content (%) | |||
| Protein | 14 | 30 | 55 |
| Carbohydrate | 76 | 60 | 35 |
| Fat | 10 | 10 | 10 |
| Energy density (kJ/g) | 14.5 | 14.5 | 14.5 |
| Day 10 | Day 13 | Day 28 | Statistical Effect | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P14 | P30 | P53 | P14 | P30 | P53 | P14 | P30 | P53 | Time | Diet | Interaction | |
| Epithelial Repair | ||||||||||||
| Saa | 1.05 ± 0.16 | 0.92 ± 0.21 | 1.33 ± 0.28 | 3.40 ± 0.40 | 4.61 ± 0.55 a | 4.01 ± 0.68 | 3.18 ± 0.30 | 2.97 ± 0.36 | 2.45 ± 0.38 | *** | *** | NS |
| Il-13 | 0.82 ± 0.08 | 1.15 ± 0.20 | 0.98 ± 0.16 | 0.78 ± 0.10 | 0.97 ± 0.13 | 0.91 ± 0.11 | 2.53 ± 0.39 | 4.42 ± 0.77 a | 4.31 ± 0.84 a | *** | * | NS |
| Il-15 | 1.67 ± 0.18 | 2.23 ± 0.42 | 3.70 ± 0.83 | 3.61 ± 0.91 | 6.78 ± 2.06 a | 4.59 ± 1.28 | 4.73 ± 0.86 | 3.89 ± 0.67 | 2.37 ± 0.19 | ** | NS | * |
| Il-22 | 0.36 ± 0.08 | 0.61 ± 0.24 | 0.38 ± 0.08 | 0.47 ± 0.14 | 0.51 ± 0.15 | 0.57 ± 0.11 | 0.18 ± 0.04 | 0.20 ± 0.04 | 0.19 ± 0.04 | *** | NS | NS |
| Il-33 | 0.60 ± 0.11 | 0.75 ± 0.10 | 0.44 ± 0.11 | 0.43 ± 0.05 | 0.37 ± 0.07 | 0.30 ± 0.05 | 0.26 ± 0.06 | 0.18 ± 0.04 | 0.27 ± 0.07 | *** | NS | NS |
| Tgf-1ß | 0.72 ± 0.07 | 1.01 ± 0.10 ab | 0.60 ± 0.07 | 0.51 ± 0.07 | 0.64 ± 0.07 b | 0.46 ± 0.05 | 0.43 ± 0.04 | 0.44 ± 0.05 | 0.47 ± 0.07 | *** | * | NS |
| Tgf-ß3 | 0.72 ± 0.10 | 0.72 ± 0.11 | 0.67 ± 0.09 | 0.93 ± 0.12 | 0.73 ± 0.06 | 0.85 ± 0.09 | 1.16 ± 0.16 | 1.04 ± 0.12 | 1.08 ± 0.17 | ** | NS | NS |
| Tff3 | 1.05 ± 0.12 | 1.63 ± 0.36 | 1.14 ± 0.17 | 1.62 ± 0.20 | 2.34 ± 0.35 a | 1.70 ± 0.11 | 1.87 ± 0.13 | 2.40 ± 0.44 | 1.63 ± 0.23 | * | ** | NS |
| Mucin genes | ||||||||||||
| Muc1 | 0.58 ± 0.09 | 0.67 ± 0.21 | 0.70 ± 0.11 | 1.03 ± 0.14 | 1.41 ± 0.10 | 1.34 ± 0.10 | 0.90 ± 0.11 | 1.34 ± 0.17 | 0.75 ± 0.13 | *** | NS | NS |
| Muc2 | 1.72 ± 0.22 | 2.03 ± 0.21 | 2.37 ± 0.25 | 3.03 ± 0.18 | 3.80 ± 0.44 | 4.34 ± 0.37 a | 3.38 ± 0.21 | 4.72 ± 0.43 a | 5.27 ± 0.50 a | *** | *** | NS |
| Muc3 | 1.25 ± 0.11 | 1.53 ± 0.14 | 1.70 ± 0.15 | 1.73 ± 0.10 | 2.62 ± 0.44 a | 2.65 ± 0.27 a | 2.07 ± 0.27 | 1.94 ± 0.11 | 1.61 ± 0.08 | *** | * | * |
| Muc4 | 1.66 ± 0.15 | 2.06 ± 0.33 | 1.79 ± 0.18 | 2.32 ± 0.17 | 2.73 ± 0.34 | 3.10 ± 0.28 | 3.43 ± 0.41 | 3.36 ± 0.36 | 3.02 ± 0.32 | ** | NS | NS |
| Tight-Junction Proteins | ||||||||||||
| Cldn1 | 0.64 ± 0.10 | 0.84 ± 0.12 b | 0.53 ± 0.06 | 0.39 ± 0.04 | 0.85 ± 0.17 ab | 0.50 ± 0.11 | 0.38 ± 0.02 | 0.49 ± 0.07 | 0.45 ± 0.04 | *** | * | NS |
| Cldn2 | 2.77 ± 0.15 | 2.69 ± 0.26 | 3.14 ± 0.25 | 2.45 ± 0.33 | 2.34 ± 0.17 | 2.71 ± 0.23 | 2.42 ± 0.26 | 2.13 ± 0.21 | 2.69 ± 0.13 | *** | NS | NS |
| Tjp1 | 1.17 ± 0.10 | 1.34 ± 0.08 | 1.10 ± 0.07 | 1.30 ± 0.06 | 1.30 ± 0.05 | 1.28 ± 0.07 | 1.29 ± 0.06 | 1.09 ± 0.08 | 1.18 ± 0.11 | ** | NS | NS |
| Ocln | 1.36 ± 0.11 | 1.52 ± 0.15 | 1.78 ± 0.44 | 1.96 ± 0.18 | 2.61 ± 0.25 b | 1.89 ± 0.32 | 2.21 ± 0.30 | 2.70 ± 0.32 b | 1.54 ± 0.12 | *** | * | NS |
| Day 10 | Day 13 | Day 28 | Statistical Effect | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P14 | P30 | P53 | P14 | P30 | P53 | P14 | P30 | P53 | Time | Diet | Interaction | |
| Diversity and richness features | ||||||||||||
| Shannon Index | 4.56 ± 0.28 | 5.28 ± 0.33 | 4.53 ± 0.36 | 4.44 ± 0.26 | 4.23 ± 0.40 | 4.96 ± 0.29 | 5.02 ± 0.29 | 5.21 ± 0.19 | 5.31 ± 0.26 | * | NS | NS |
| Simpson Index | 0.89 ± 0.02 | 0.94 ± 0.01 ab | 0.88 ± 0.03 | 0.88 ± 0.03 | 0.85 ± 0.04 | 0.92 ± 0.01 | 0.92 ± 0.02 | 0.93 ± 0.01 | 0.94 ± 0.01 | *** | * | NS |
| Chao1 Index | 593 ± 31.1 | 678 ± 26.0 | 673 ± 41.1 | 635 ± 34.4 | 566 ± 25.5 | 642 ± 57.7 | 606 ± 51.7 | 648 ± 55.0 | 605 ± 39.7 | ** | NS | NS |
| Observed OTUs | 286 ± 36.9 | 350 ± 30.1 | 306 ± 32.7 | 293 ± 24.6 | 335 ± 29.1 b | 261 ± 34.8 | 285 ± 21.6 | 342 ± 30.6 | 305 ± 22.5 | *** | * | NS |
| Relative genera composition (%) | ||||||||||||
| Alloprevotella | 0.008 ± 0.006 | 0.000 ± 0.000 b | 0.079 ± 0.014 a | 0.013 ± 0.006 | 0.000 ± 0.000 | 0.025 ± 0.025 | 0.003 ± 0.003 | 0.002 ± 0.002 | 0.000 ± 0.000 | * | * | NS |
| Bacteroides | 11.55 ± 2.771 | 7.391 ± 1.788 a | 15.83 ± 4.937 b | 3.082 ± 1.079 | 14.04 ± 4.541 a | 11.92 ± 4.152 | 20.11 ± 4.979 | 21.92 ± 4.357 b | 7.166 ± 1.920 a | * | * | ** |
| Clostridium_XIVa | 5.555 ± 2.733 | 2.861 ± 0.517 | 2.950 ± 0.763 | 8.545 ± 2.801 | 10.61 ± 3.758 b | 3.653 ± 1.709 | 4.008 ± 1.635 | 4.212 ± 1.211 | 5.191 ± 1.197 | * | * | NS |
| Desulfovibrio | 3.062 ± 0.643 | 4.376 ± 0.870 b | 1.402 ± 0.272 a | 3.643 ± 0.946 | 3.892 ± 1.142 b | 1.565 ± 0.406 | 1.298 ± 0.306 | 1.699 ± 0.581 | 0.939 ± 0.207 | ** | ** | NS |
| Enterobacter | 0.021 ± 0.010 | 0.013 ± 0.008 | 0.025 ± 0.019 | 0.049 ± 0.020 | 0.023 ± 0.019 | 0.025 ± 0.013 | 0.031 ± 0.015 | 0.012 ± 0.005 | 0.015 ± 0.008 | NS | NS | NS |
| Enterorhabdus | 0.010 ± 0.005 | 0.009 ± 0.004 | 0.006 ± 0.003 | 0.004 ± 0.003 | 0.028 ± 0.016 | 0.034 ± 0.012 a | 0.030 ± 0.014 | 0.023 ± 0.008 | 0.014 ± 0.008 | * | * | NS |
| Ethanoligenens | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 b | 0.013 ± 0.013 a | NS | * | NS |
| Faecalibacterium | 0.000 ± 0.000 | 0.002 ± 0.002 | 0.000 ± 0.000 | 0.004 ± 0.004 | 0.007 ± 0.002 b | 0.001 ± 0.001 | 0.014 ± 0.014 | 0.048 ± 0.008 b | 0.000 ± 0.000 | ** | * | NS |
| Haemophilus | 0.000 ± 0.000 | 0.004 ± 0.004 b | 0.017 ± 0.003 a | 0.000 ± 0.000 | 0.013 ± 0.007 | 0.025 ± 0.012 a | 0.002 ± 0.002 | 0.004 ± 0.004 b | 0.043 ± 0.029 a | NS | * | NS |
| Klebsiella | 0.004 ± 0.005 | 0.000 ± 0.000 b | 0.005 ± 0.001 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.002 ± 0.002 | 0.000 ± 0.000 b | 0.005 ± 0.001 | * | * | NS |
| Porphyromonas | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 | 0.000 ± 0.000 b | 0.012 ± 0.012 a | ** | ** | NS |
| Roseburia | 0.005 ± 0.005 | 0.009 ± 0.004 | 0.010 ± 0.005 | 0.011 ± 0.006 | 0.006 ± 0.003 | 0.011 ± 0.008 | 0.009 ± 0.006 | 0.029±0.004 ab | 0.011 ± 0.001 | *** | *** | NS |
| Caecal SCFA (µmol/g) | ||||||||||||
| Total | 0.06 ± 0.04 | 2.05 ± 1.56 a | 0.38 ± 0.27 | 0.14 ± 0.07 | 1.32 ± 0.44 a | 0.99 ± 0.46 | 4.88 ± 1.08 | 6.85 ± 1.65 b | 12.32 ± 3.65 a | *** | * | NS |
| Acetate | 0.04 ± 0.04 | 1.40 ± 1.04 a | 0.28 ± 0.18 | 0.14 ± 0.07 | 1.20 ± 0.43 a | 1.00 ± 0.38 | 3.40 ± 0.71 | 4.66 ± 1.13 | 8.38 ± 2.42 | *** | * | NS |
| Propionate | 0.00 ± 0.00 | 0.44 ± 0.36 a | 0.07 ± 0.07 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.13 ± 0.13 | 0.90 ± 0.26 | 1.31 ± 0.35 | 2.55 ± 0.65 | *** | * | NS |
| Butyrate | 0.01 ± 0.01 | 0.21 ± 0.16 a | 0.03 ± 0.03 | 0.00 ± 0.00 | 0.12 ± 0.05 | 0.10 ± 0.07 | 0.59 ± 0.13 | 0.88 ± 0.26 | 1.40 ± 0.39 | *** | * | NS |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidal-Lletjós, S.; Andriamihaja, M.; Blais, A.; Grauso, M.; Lepage, P.; Davila, A.-M.; Viel, R.; Gaudichon, C.; Leclerc, M.; Blachier, F.; et al. Dietary Protein Intake Level Modulates Mucosal Healing and Mucosa-Adherent Microbiota in Mouse Model of Colitis. Nutrients 2019, 11, 514. https://doi.org/10.3390/nu11030514
Vidal-Lletjós S, Andriamihaja M, Blais A, Grauso M, Lepage P, Davila A-M, Viel R, Gaudichon C, Leclerc M, Blachier F, et al. Dietary Protein Intake Level Modulates Mucosal Healing and Mucosa-Adherent Microbiota in Mouse Model of Colitis. Nutrients. 2019; 11(3):514. https://doi.org/10.3390/nu11030514
Chicago/Turabian StyleVidal-Lletjós, Sandra, Mireille Andriamihaja, Anne Blais, Marta Grauso, Patricia Lepage, Anne-Marie Davila, Roselyne Viel, Claire Gaudichon, Marion Leclerc, François Blachier, and et al. 2019. "Dietary Protein Intake Level Modulates Mucosal Healing and Mucosa-Adherent Microbiota in Mouse Model of Colitis" Nutrients 11, no. 3: 514. https://doi.org/10.3390/nu11030514
APA StyleVidal-Lletjós, S., Andriamihaja, M., Blais, A., Grauso, M., Lepage, P., Davila, A.-M., Viel, R., Gaudichon, C., Leclerc, M., Blachier, F., & Lan, A. (2019). Dietary Protein Intake Level Modulates Mucosal Healing and Mucosa-Adherent Microbiota in Mouse Model of Colitis. Nutrients, 11(3), 514. https://doi.org/10.3390/nu11030514






