A Plant-Based Meal Stimulates Incretin and Insulin Secretion More Than an Energy- and Macronutrient-Matched Standard Meal in Type 2 Diabetes: A Randomized Crossover Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
2.2. Participants and Eligibility Criteria
2.3. Randomization and Masking
2.4. Interventions
2.5. Measurements
2.5.1. Anthropometric Measures and Blood Pressure
2.5.2. Metabolic Parameters
2.5.3. Gastrointestinal and Appetite Hormones
2.5.4. Beta-cell Function and Insulin Resistance
2.6. Statistical Analysis
3. Results
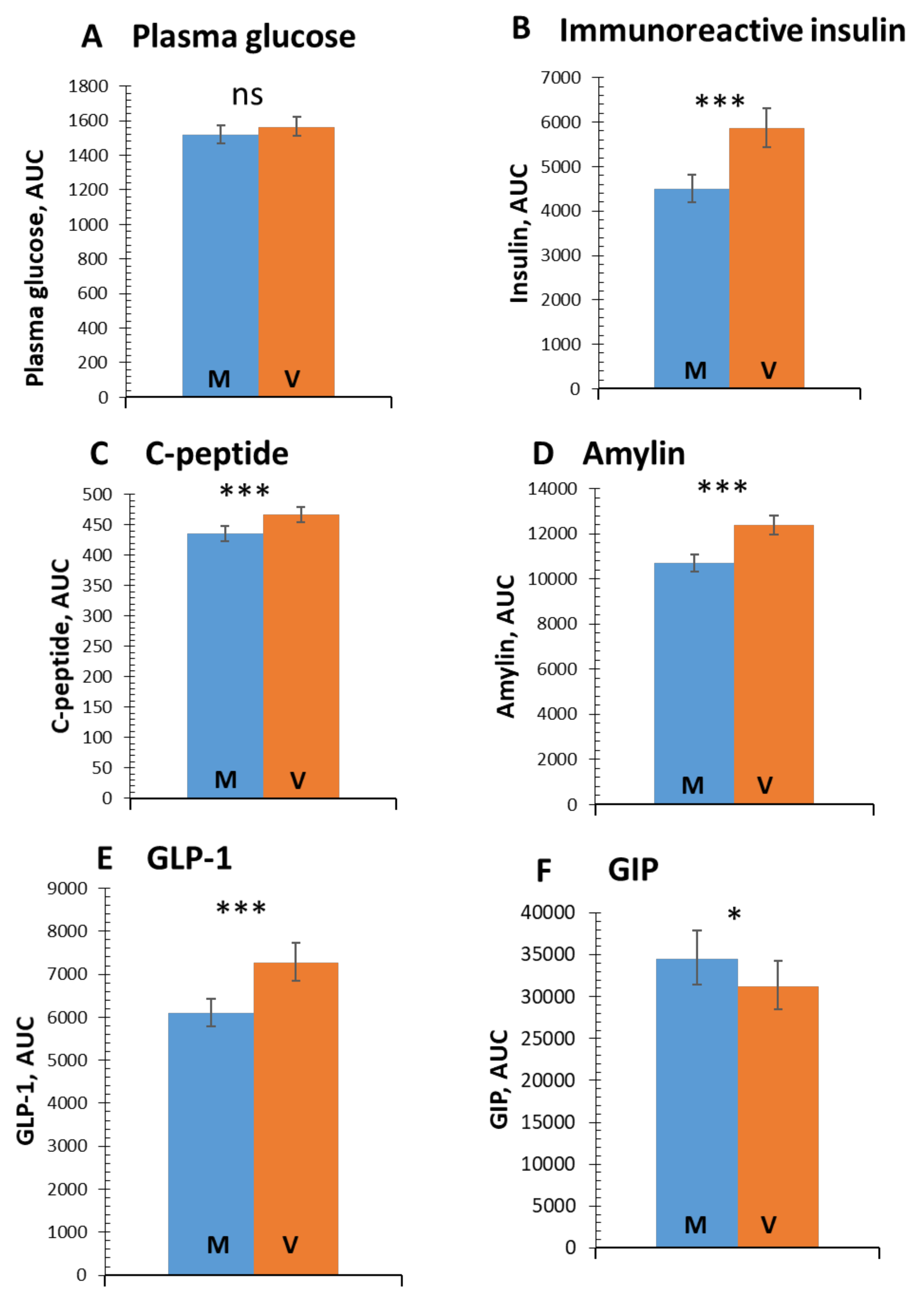
3.1. Postprandial Glucose and Insulin Response
3.2. Incretins
3.3. Beta-cell Function and Insulin Resistance
3.4. Correlations of Changes in Gastrointestinal Hormones with Glucose Metabolism
4. Discussion
4.1. Insulin Secretion and Beta-cell Function
4.2. Incretins
4.3. Amylin
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| HbA1c | glycated haemoglobin |
| GLP-1 | glucagon-like peptide -1 |
| GIP | gastric inhibitory peptide |
References
- Feskens, E.J.; Virtanen, S.M.; Räsänen, L.; Tuomilehto, J.; Stengård, J.; Pekkanen, J.; Nissinen, A.; Kromhout, D. Dietary factors determining diabetes and impaired glucose tolerance. A 20-year follow-up of the Finnish and Dutch cohorts of the Seven Countries Study. Diabetes Care 1995, 18, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Fizelova, M.; Jauhiainen, R.; Stančáková, A.; Kuusisto, J.; Laakso, M. Finnish Diabetes Risk Score Is Associated with Impaired Insulin Secretion and Insulin Sensitivity, Drug-Treated Hypertension and Cardiovascular Disease: A Follow-Up Study of the METSIM Cohort. PLoS ONE 2016, 11, e0166584. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.I.; De Leeuw, I.; Hermansen, K.; Karamanos, B.; Karlström, B.; Katsilambros, N.; Riccardi, G.; Rivellese, A.A.; Rizkalla, S.; Slama, G.; et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2004, 14, 373–394. [Google Scholar] [CrossRef]
- Perry, B.; Wang, Y. Appetite regulation and weight control: The role of gut hormones. Nutr. Diabetes 2012, 2, e26. [Google Scholar] [CrossRef] [PubMed]
- Meier, J.J. The contribution of incretin hormones to the pathogenesis of type 2 diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Verdich, C.; Flint, A.; Gutzwiller, J.P.; Näslund, E.; Beglinger, C.; Hellström, P.M.; Long, S.J.; Morgan, L.M.; Holst, J.J.; Astrup, A. A meta-analysis of the effect of glucagon-like peptide-1 (7–36) amide on ad libitum energy intake in humans. J. Clin. Endocrinol. Metab. 2001, 86, 4382–4389. [Google Scholar] [CrossRef] [PubMed]
- Lutz, T.A. The interaction of amylin with other hormones in the control of eating. Diabetes Obes. Metab. 2013, 15, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Belinova, L.; Kahleova, H.; Malinska, H.; Topolcan, O.; Vrzalova, J.; Oliyarnyk, O.; Kazdova, L.; Hill, M.; Pelikanova, T. Differential acute postprandial effects of processed meat and isocaloric vegan meals on the gastrointestinal hormone response in subjects suffering from type 2 diabetes and healthy controls: A randomized crossover study. PLoS ONE 2014, 9, e107561. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Ursin, G.; Veierød, M.B. Meat consumption and the risk of type 2 diabetes: A systematic review and meta-analysis of cohort studies. Diabetologia 2009, 52, 2277–2287. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Sun, Q.; Bernstein, A.M.; Schulze, M.B.; Manson, J.E.; Willett, W.C.; Hu, F.B. Red meat consumption and risk of type 2 diabetes: 3 cohorts of US adults and an updated meta-analysis. Am. J. Clin. Nutr. 2011, 94, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Vang, A.; Singh, P.N.; Lee, J.W.; Haddad, E.H.; Brinegar, C.H. Meats, processed meats, obesity, weight gain and occurrence of diabetes among adults: Findings from Adventist Health Studies. Ann. Nutr. Metab. 2008, 52, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Maron, D.J.; Fair, J.M.; Haskell, W.L. Saturated fat intake and insulin resistance in men with coronary artery disease. The Stanford Coronary Risk Intervention Project Investigators and Staff. Circulation 1991, 84, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Zong, G.; Li, Y.; Wanders, A.J.; Alssema, M.; Zock, P.L.; Willett, W.C.; Hu, F.B.; Sun, Q. Intake of individual saturated fatty acids and risk of coronary heart disease in US men and women: Two prospective longitudinal cohort studies. BMJ 2016, 355, i5796. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Butler, T.; Yan, R.; Fraser, G.E. Type of Vegetarian Diet, Body Weight, and Prevalence of Type 2 Diabetes. Diabetes Care 2009, 32, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Kahleova, H.; Matoulek, M.; Malinska, H.; Oliyarnik, O.; Kazdova, L.; Neskudla, T.; Skoch, A.; Hajek, M.; Hill, M.; Kahle, M.; et al. Vegetarian diet improves insulin resistance and oxidative stress markers more than conventional diet in subjects with Type 2 diabetes. Diabetes Med. 2011, 28, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Barnard, N.D.; Cohen, J.; Jenkins, D.J.A.; Turner-McGrievy, G.; Gloede, L.; Jaster, B.; Seidl, K.; Green, A.A.; Talpers, S. A low-fat vegan diet improves glycemic control and cardiovascular risk factors in a randomized clinical trial in individuals with type 2 diabetes. Diabetes Care 2006, 29, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Van Cauter, E.; Mestrez, F.; Sturis, J.; Polonsky, K.S. Estimation of insulin secretion rates from C-peptide levels. Comparison of individual and standard kinetic parameters for C-peptide clearance. Diabetes 1992, 41, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Mari, A.; Tura, A.; Gastaldelli, A.; Ferrannini, E. Assessing insulin secretion by modeling in multiple-meal tests: Role of potentiation. Diabetes 2002, 51 (Suppl. 1), S221–S226. [Google Scholar] [CrossRef]
- Mari, A.; Schmitz, O.; Gastaldelli, A.; Oestergaard, T.; Nyholm, B.; Ferrannini, E. Meal and oral glucose tests for assessment of beta-cell function: Modeling analysis in normal subjects. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E1159–E1166. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Tura, A.; Chemello, G.; Szendroedi, J.; Göbl, C.; Færch, K.; Vrbíková, J.; Pacini, G.; Ferrannini, E.; Roden, M. Prediction of clamp-derived insulin sensitivity from the oral glucose insulin sensitivity index. Diabetologia 2018, 61, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Ismail-Beigi, F. Clinical practice. Glycemic management of type 2 diabetes mellitus. N. Engl. J. Med. 2012, 366, 1319–1327. [Google Scholar] [CrossRef] [PubMed]
- Page, K.A.; Reisman, T. Interventions to Preserve Beta-Cell Function in the Management and Prevention of Type 2 Diabetes. Curr. Diabetes Rep. 2013, 13, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Kahleova, H.; Tura, A.; Hill, M.; Holubkov, R.; Barnard, N.D. A Plant-Based Dietary Intervention Improves Beta-Cell Function and Insulin Resistance in Overweight Adults: A 16-Week Randomized Clinical Trial. Nutrients 2018, 10, 189. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Homberger, E.; Siegel, E.G.; Allen, R.C.; Eaton, R.P.; Ebert, R.; Creutzfeldt, W. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J. Clin. Endocrinol. Metab. 1986, 63, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Knop, F.K.; Vilsbøll, T.; Højberg, P.V.; Larsen, S.; Madsbad, S.; Vølund, A.; Holst, J.J.; Krarup, T. Reduced incretin effect in type 2 diabetes: Cause or consequence of the diabetic state? Diabetes 2007, 56, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
- Calanna, S.; Christensen, M.; Holst, J.J.; Laferrère, B.; Gluud, L.L.; Vilsbøll, T.; Knop, F.K. Secretion of glucose-dependent insulinotropic polypeptide in patients with type 2 diabetes: Systematic review and meta-analysis of clinical studies. Diabetes Care 2013, 36, 3346–3352. [Google Scholar] [CrossRef] [PubMed]
- Kjems, L.L.; Holst, J.J.; Vølund, A.; Madsbad, S. The influence of GLP-1 on glucose-stimulated insulin secretion: Effects on beta-cell sensitivity in type 2 and nondiabetic subjects. Diabetes 2003, 52, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Kang, Z.F.; Deng, Y.; Zhou, Y.; Fan, R.R.; Chan, J.C.N.; Laybutt, D.R.; Luzuriaga, J.; Xu, G. Pharmacological reduction of NEFA restores the efficacy of incretin-based therapies through GLP-1 receptor signalling in the beta cell in mouse models of diabetes. Diabetologia 2013, 56, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, A.F.H.; Keyhani-Nejad, F. High Glycemic Index Metabolic Damage—A Pivotal Role of GIP and GLP-1. Trends Endocrinol. Metab. 2018, 29, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.K.; Kullman, E.L.; Scelsi, A.R.; Godin, J.-P.; Ross, A.B.; Kirwan, J.P. A Whole-Grain Diet Increases Glucose-Stimulated Insulin Secretion Independent of Gut Hormones in Adults at Risk for Type 2 Diabetes. Mol. Nutr. Food Res. 2019, e1800967. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-Y.; Kanthimathi, M.S.; Tan, A.T.-B.; Nesaretnam, K.; Teng, K.-T. The amount and types of fatty acids acutely affect insulin, glycemic and gastrointestinal peptide responses but not satiety in metabolic syndrome subjects. Eur. J. Nutr. 2018, 57, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Chalvon-Demersay, T.; Azzout-Marniche, D.; Arfsten, J.; Egli, L.; Gaudichon, C.; Karagounis, L.G.; Tomé, D. A Systematic Review of the Effects of Plant Compared with Animal Protein Sources on Features of Metabolic Syndrome. J. Nutr. 2017, 147, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Viguiliouk, E.; Stewart, S.E.; Jayalath, V.H.; Ng, A.P.; Mirrahimi, A.; de Souza, R.J.; Hanley, A.J.; Bazinet, R.P.; Blanco Mejia, S.; Leiter, L.A.; et al. Effect of Replacing Animal Protein with Plant Protein on Glycemic Control in Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2015, 7, 9804–9824. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Ke, M.-Y.; Li, W.-H.; Zhang, S.-Q.; Fang, X.-C. The impact of soluble dietary fibre on gastric emptying, postprandial blood glucose and insulin in patients with type 2 diabetes. Asia Pac. J. Clin. Nutr. 2014, 23, 210–218. [Google Scholar] [PubMed]
- Post, R.E.; Mainous, A.G.; King, D.E.; Simpson, K.N. Dietary fiber for the treatment of type 2 diabetes mellitus: A meta-analysis. J. Am. Board Fam. Med. 2012, 25, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Fukunaka, A.; Fujitani, Y. Role of Zinc Homeostasis in the Pathogenesis of Diabetes and Obesity. Int. J. Mol. Sci. 2018, 19, 476. [Google Scholar] [CrossRef] [PubMed]
- Domínguez Avila, J.A.; Rodrigo García, J.; González Aguilar, G.A.; de la Rosa, L.A. The Antidiabetic Mechanisms of Polyphenols Related to Increased Glucagon-Like Peptide-1 (GLP1) and Insulin Signaling. Molecules 2017, 22, 903. [Google Scholar] [CrossRef] [PubMed]
- McClean, P.L.; Irwin, N.; Cassidy, R.S.; Holst, J.J.; Gault, V.A.; Flatt, P.R. GIP receptor antagonism reverses obesity, insulin resistance, and associated metabolic disturbances induced in mice by prolonged consumption of high-fat diet. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1746–E1755. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Peshavaria, M.; Monga, N.; Jetton, T.L.; Leahy, J.L. Physiologic and pharmacologic modulation of glucose-dependent insulinotropic polypeptide (GIP) receptor expression in beta-cells by peroxisome proliferator-activated receptor (PPAR)-gamma signaling: Possible mechanism for the GIP resistance in type 2 diabetes. Diabetes 2010, 59, 1445–1450. [Google Scholar] [PubMed]
- Jirapinyo, P.; Haas, A.V.; Thompson, C.C. Effect of the Duodenal-Jejunal Bypass Liner on Glycemic Control in Patients With Type 2 Diabetes With Obesity: A Meta-analysis With Secondary Analysis on Weight Loss and Hormonal Changes. Diabetes Care 2018, 41, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; D’Alessio, D.A.; Fradkin, J.; Kernan, W.N.; Mathieu, C.; Mingrone, G.; Rossing, P.; Tsapas, A.; Wexler, D.J.; Buse, J.B. Management of Hyperglycemia in Type 2 Diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018, 41, 2669–2701. [Google Scholar] [CrossRef] [PubMed]
- Zander, M.; Madsbad, S.; Madsen, J.L.; Holst, J.J. Effect of 6-week course of glucagon-like peptide 1 on glycaemic control, insulin sensitivity, and beta-cell function in type 2 diabetes: A parallel-group study. Lancet 2002, 359, 824–830. [Google Scholar] [CrossRef]
- Lutz, T.A. Amylinergic control of food intake. Physiol. Behav. 2006, 89, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Young, A. Inhibition of food intake. Adv. Pharmacol. 2005, 52, 79–98. [Google Scholar] [PubMed]
- Kondo, Y.; Goto, A.; Noma, H.; Iso, H.; Hayashi, K.; Noda, M. Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis. Nutrients 2018, 11, 48. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Patients with T2D (n = 20) |
|---|---|
| Age (years) | 47.8 ± 8.2 |
| Weight (kg) | 108.2 ± 11.9 |
| Body mass index (kg∙m−2) | 34.5 ± 3.4 |
| Waist circumference (cm) | 106.9 ± 23.6 |
| HbA1c (IFCC; mmol/mol) | 48.5 ± 8.1 |
| HbA1C (DCCT; %) | 6.6 ± 1.0 |
| Fasting plasma glucose (mmol/L) | 7.2 ± 1.5 |
| Triglycerides (mmol/L) | 2.1 ± 1.1 |
| Total cholesterol (mmol/L) | 4.5 ± 0.9 |
| LDL-cholesterol (mmol/L) | 2.6 ± 0.1 |
| HDL-cholesterol (mmol/L) | 1.0 ± 0.2 |
| Systolic blood pressure (mm Hg) | 144.4 ± 13.4 |
| Diastolic blood pressure (mm Hg) | 96.2 ± 8.8 |
| Duration of diabetes (years) | 4.25 ± 3.25 |
| Meal | M-meal | V-meal |
|---|---|---|
| Total weight (g) | 200 | 200 |
| Energy content (kCal) | 513.6 | 514.9 |
| Carbohydrates (g) (%) | 55 (44.8%) | 54.2 (44.0%) |
| Sugar (g) (%) | 21 (17%) | 4 (3%) |
| Proteins (g) (%) | 20.5 (16.7%) | 19.9 (16.2%) |
| Lipids (g) (%) | 22 (38.6%) | 22.8 (39.8%) |
| Saturated fatty acids (g) | 8.6 | 2.2 |
| Fiber (g) | 2.2 | 7.8 |
| M-meal | V-meal | p-value | |
|---|---|---|---|
| Insulin secretion/ Beta-cell function | |||
| Basal insulin secretion (pmol min−1 m−2) | 151.9 (146.6–157.2) | 153.9 (148.6–159.3) | 0.72 |
| Total insulin secretion (nmol m−2) | 62.2 (60.2–64.3) | 68.2 (66.1–70.4) | <0.001 |
| Insulin secretion at a fixed glucose value (5 mM) (pmol min−1 m−2) | 99.0 (84.3–114.3) | 110.3 (95.2–126.1) | 0.04 |
| Insulin secretion at a fixed glucose value (6 mM) (pmol min−1 m−2) | 166.2 (146.3–186.8) | 174.6 (154.5–195.6) | 0.08 |
| Insulin secretion at a fixed glucose value (7 mM) (pmol min−1 m−2) | 234.3 (209.9–260.3) | 234.0 (209.7–260.1) | 0.89 |
| Glucose sensitivity (pmol min−1 m−2 mM−1) | 75.3 (65.7–85.8) | 72.0 (62.6–82.2) | 0.33 |
| Rate sensitivity (pmol m−2 mM−1) | 202.4 (100.5–300.1) | 313.7 (167.7–448.6) | 0.04 |
| Potentiation factor ratio (dimensionless) | 1.5 (1.4–1.6) | 1.7 (1.6–1.8) | 0.02 |
| Insulin sensitivity/resistance | |||
| HOMA-IR (dimensionless) | 4.2 (3.9–4.5) | 4.2 (4.0–4.5) | 0.90 |
| PREDIM (mg min−1 kg−1) | 2.7 (2.6–2.8) | 2.6 (2.5–2.7) | 0.73 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahleova, H.; Tura, A.; Klementova, M.; Thieme, L.; Haluzik, M.; Pavlovicova, R.; Hill, M.; Pelikanova, T. A Plant-Based Meal Stimulates Incretin and Insulin Secretion More Than an Energy- and Macronutrient-Matched Standard Meal in Type 2 Diabetes: A Randomized Crossover Study. Nutrients 2019, 11, 486. https://doi.org/10.3390/nu11030486
Kahleova H, Tura A, Klementova M, Thieme L, Haluzik M, Pavlovicova R, Hill M, Pelikanova T. A Plant-Based Meal Stimulates Incretin and Insulin Secretion More Than an Energy- and Macronutrient-Matched Standard Meal in Type 2 Diabetes: A Randomized Crossover Study. Nutrients. 2019; 11(3):486. https://doi.org/10.3390/nu11030486
Chicago/Turabian StyleKahleova, Hana, Andrea Tura, Marta Klementova, Lenka Thieme, Martin Haluzik, Renata Pavlovicova, Martin Hill, and Terezie Pelikanova. 2019. "A Plant-Based Meal Stimulates Incretin and Insulin Secretion More Than an Energy- and Macronutrient-Matched Standard Meal in Type 2 Diabetes: A Randomized Crossover Study" Nutrients 11, no. 3: 486. https://doi.org/10.3390/nu11030486
APA StyleKahleova, H., Tura, A., Klementova, M., Thieme, L., Haluzik, M., Pavlovicova, R., Hill, M., & Pelikanova, T. (2019). A Plant-Based Meal Stimulates Incretin and Insulin Secretion More Than an Energy- and Macronutrient-Matched Standard Meal in Type 2 Diabetes: A Randomized Crossover Study. Nutrients, 11(3), 486. https://doi.org/10.3390/nu11030486







