Time Trends in Age at Menarche and Related Non-Communicable Disease Risk during the 20th Century in Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Study Population
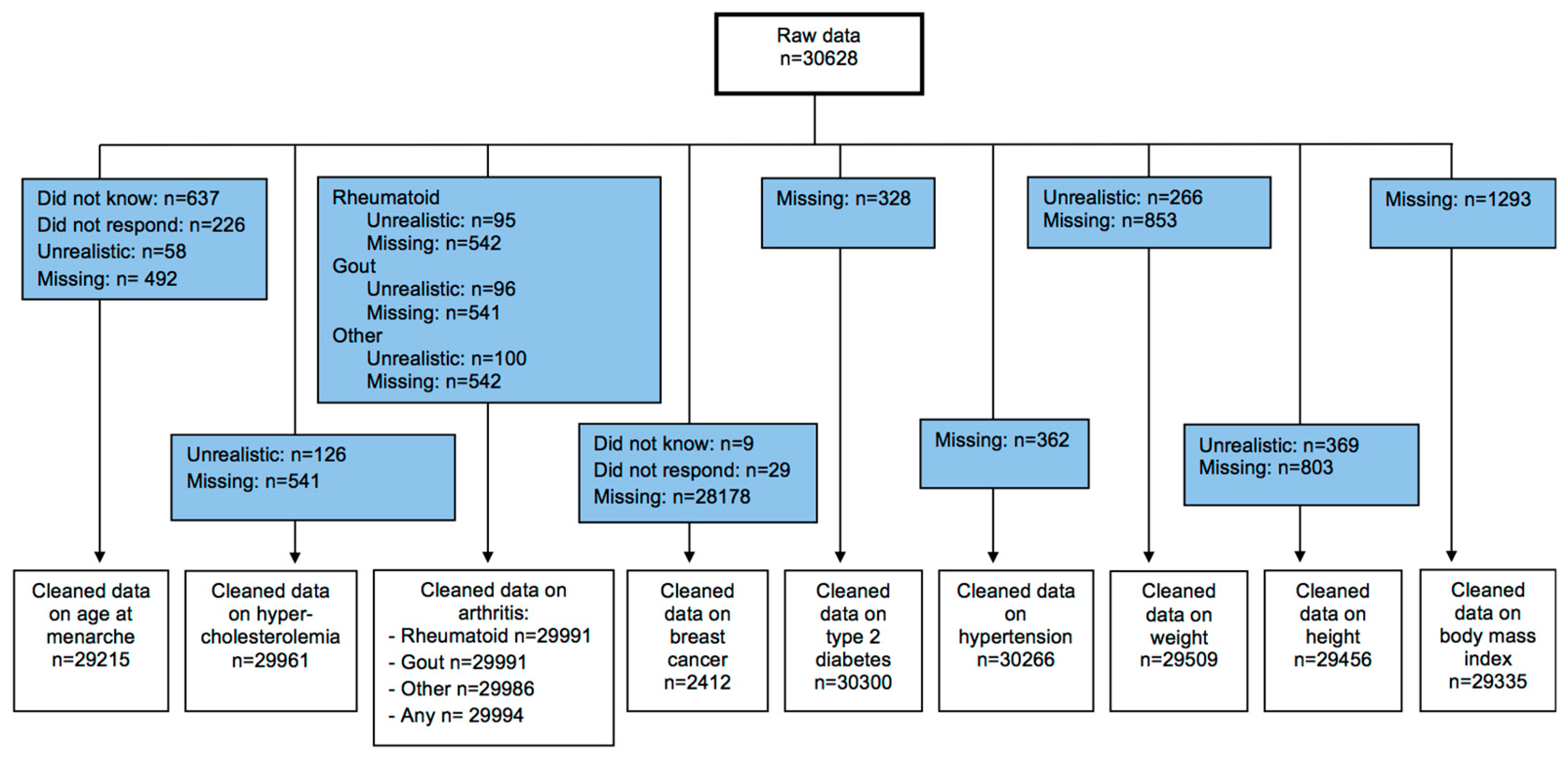
2.2. Data Collection and Variable Construction
2.3. Statistical Analysis
3. Results
3.1. Sample Characteristics
3.2. Association of Age at Menarche and Nutritional Status
3.3. Association of Age at Menarche and Disease Status
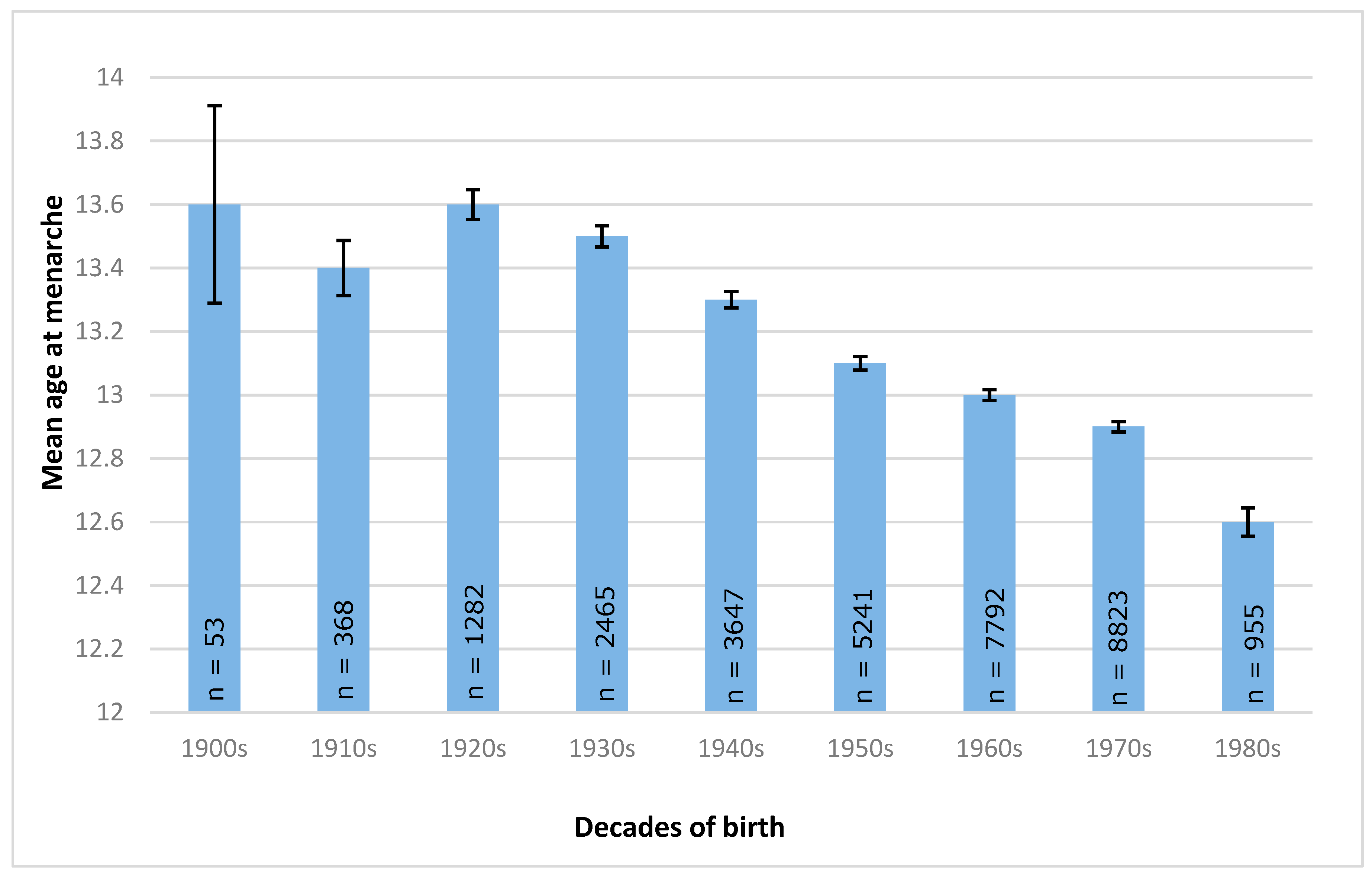
3.4. Time Trend of Age at Menarche
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alberga, A.S.; Sigal, R.J.; Goldfield, G.; Prud’homme, D.; Kenny, G.P. Overweight and obese teenagers: Why is adolescence a critical period? Pediatr. Obes. 2012, 7, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Demerath, E.W.; Cameron, N. Critical periods in human growth and their relationship to diseases of aging. Am. J. Phys. Anthropol. 2002, 119, 159–184. [Google Scholar]
- Dietz, W.H. Periods of risk in childhood for the development of adult obesity—What do we need to learn? J. Nutr. 1997, 127, 1884–1886. [Google Scholar] [CrossRef] [PubMed]
- Reinehr, T.; Roth, C.L. Is there a causal relationship between obesity and puberty? Lancet Child Adolesc. Health 2019, 3, 44–54. [Google Scholar] [CrossRef]
- Martos-Moreno, G.; Chowen, J.; Argente, J. Metabolic signals in human puberty: Effects of over and undernutrition. Mol. Cell. Endocrinol. 2010, 324, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.-M.; Oh, I.-H.; Yoon, T.-Y.; Choi, J.-M.; Choi, K.-S.; Choe, B.-K. Relationship Between Body Mass Index and Early Menarche of Adolescent Girls in Seoul. J. Prev. Med. Public Health 2012, 45, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Garrido, M.A.; Tena-Sempere, M. Metabolic control of puberty: Roles of leptin and kisspeptins. Horm. Behav. 2013, 64, 187–194. [Google Scholar] [CrossRef]
- Hollins-Martin, C.J.; Van den Akker, O.B.; Martin, C.R.; Preedy, V.R. (Eds.) Handbook of Diet and Nutrition in the Menstrual Cycle, Periconception and Fertility; Wageningen Academic Publishers: Wageningen, The Netherlands, 2014. [Google Scholar]
- Aksglaede, L.; Sorensen, K.; Petersen, J.H.; Skakkebaek, N.E.; Juul, A. Recent decline in age at breast development: The Copenhagen Puberty Study. Pediatrics 2009, 123, 932–939. [Google Scholar] [CrossRef]
- Jansen, E.C.; Herrán, O.F.; Villamor, E. Trends and correlates of age at menarche in Colombia: Results from a nationally representative survey. Econ. Hum. Boil. 2015, 19, 138–144. [Google Scholar] [CrossRef]
- Jaruratanasirikul, S.; Sriplung, H. Secular trends of growth and pubertal maturation of school children in Southern Thailand. Ann. Hum. Boil. 2015, 42, 447–454. [Google Scholar] [CrossRef]
- Song, Y.; Ma, J.; Wang, H.-J.; Wang, Z.; Hu, P.; Zhang, B.; Agardh, A. Trends of Age at Menarche and Association with Body Mass Index in Chinese School-Aged Girls, 1985–2010. J. Pediatrics 2014, 165, 1172–1177. [Google Scholar] [CrossRef] [PubMed]
- Manfredi-Lozano, M.; Roa, J.; Tena-Sempere, M. Connecting metabolism and gonadal function: Novel central neuropeptide pathways involved in the metabolic control of puberty and fertility. Front. Neuroendocrinol. 2018, 48, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.A.; Derraik, J.G.B.; Rowe, D.L.; Hofman, P.L.; Cutfield, W.S. Earlier Menarche Is Associated with Lower Insulin Sensitivity and Increased Adiposity in Young Adult Women. PLoS ONE 2015, 10, e0128427. [Google Scholar] [CrossRef] [PubMed]
- Okasha, P.M.M. Age at menarche: secular trends and association with adult anthropometric measures. Ann. Hum. Boil. 2001, 28, 68–78. [Google Scholar]
- Prentice, P.; Viner, R.M. Pubertal timing and adult obesity and cardiometabolic risk in women and men: a systematic review and meta-analysis. Int. J. Obes. 2012, 37, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Zhang, C.; Hunter, D.J.; Hankinson, S.E.; Buck Louis, G.M.; Hediger, M.L.; Hu, F.B. Age at menarche and risk of type 2 diabetes: Results from 2 large prospective cohort studies. Am. J. Epidemiol. 2010, 171, 334–344. [Google Scholar] [CrossRef]
- Yousefi, M.; Karmaus, W.; Zhang, H.; Roberts, G.; Matthews, S.; Clayton, B.; Arshad, S.H. Relationships between age of puberty onset and height at age 18 years in girls and boys. World J. Pediatr. 2013, 9, 230–238. [Google Scholar] [CrossRef]
- Svanes, C.; Bjørge, L.; Omenaas, E.R.; Real, F.G. Respiratory health in women: From menarche to menopause AU-Macsali, Ferenc. Expert Rev. Respir. Med. 2012, 6, 187–202. [Google Scholar]
- Varraso, R.; Siroux, V.; Maccario, J.; Pin, I.; Kauffmann, F. Asthma Severity Is Associated with Body Mass Index and Early Menarche in Women. Am. J. Respir. Crit. Care Med. 2005, 171, 334–339. [Google Scholar] [CrossRef]
- Akseer, N.; Al-Gashm, S.; Mehta, S.; Mokdad, A.; Bhutta, Z.A. Global and regional trends in the nutritional status of young people: a critical and neglected age group. Ann. N. Y. Acad. Sci. 2017, 1393, 3–20. [Google Scholar] [CrossRef]
- Bourgeois, B.; Watts, K.; Thomas, D.M.; Carmichael, O.; Hu, F.B.; Heo, M.; Hall, J.E.; Heymsfield, S.B. Associations between height and blood pressure in the United States population. Medicine 2017, 96, e9233. [Google Scholar] [CrossRef] [PubMed]
- Janghorbani, M.; Momeni, F.; Dehghani, M. Hip circumference, height and risk of type 2 diabetes: systematic review and meta-analysis. Obes. Rev. 2012, 13, 1172–1181. [Google Scholar] [CrossRef] [PubMed]
- Menarche, menopause, and breast cancer risk: individual participant meta-analysis, including 118 964 women with breast cancer from 117 epidemiological studies. Lancet Oncol. 2012, 13, 1141–1151. [CrossRef]
- Karlson, E.W.; Mandl, L.A.; Hankinson, S.E.; Grodstein, F. Do breast-feeding and other reproductive factors influence future risk of rheumatoid arthritis? Results from the Nurses’ Health Study. Arthritis Rheum. 2004, 50, 3458–3467. [Google Scholar] [CrossRef] [PubMed]
- Charalampopoulos, D.; McLoughlin, A.; Elks, C.E.; Ong, K.K. Age at menarche and risks of all-cause and cardiovascular death: A systematic review and meta-analysis. Am. J. Epidemiol. 2014, 180, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Lakshman, R.; Forouhi, N.G.; Sharp, S.J.; Luben, R.; Bingham, S.A.; Khaw, K.T.; Wareham, N.J.; Ong, K.K. Early age at menarche associated with cardiovascular disease and mortality. J. Clin. Endocrinol. Metab. 2009, 94, 4953–4960. [Google Scholar] [CrossRef] [PubMed]
- Stöckl, D.; Döring, A.; Peters, A.; Thorand, B.; Heier, M.; Huth, C.; Stöckl, H.; Rathmann, W.; Kowall, B.; Meisinger, C. Age at menarche is associated with prediabetes and diabetes in women (aged 32–81 years) from the general population: the KORA F4 Study. Diabetologia 2011, 55, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.S.; Ephross, S.A.; Sandler, D.P. Menstrual patterns and risk of adult-onset diabetes mellitus. J. Clin. Epidemiol. 2000, 53, 1170–1173. [Google Scholar] [CrossRef]
- Heys, M.; Schooling, C.M.; Jiang, C.; Cowling, B.J.; Lao, X.Q.; Zhang, W.; Cheng, K.K.; Adab, P.; Thomas, G.N.; Lam, T.H.; et al. Age of Menarche and the Metabolic Syndrome in China. Epidemiology 2007, 18, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Marván, M.L.; Catillo-López, R.L.; Alcalá-Herrera, V.; del Callejo, D. The decreasing age at menarche in Mexico. J. Pediatric Adolesc. Gynecol. 2016, 29, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Malina, R.M.; Pena Reyes, M.E.; Tan, S.K.; Little, B.B. Secular change in age at menarche in rural Oaxaca, southern Mexico: 1968–2000. Ann. Hum. Biol. 2004, 31, 634–646. [Google Scholar] [CrossRef] [PubMed]
- Valdespino, V.L.; Olaiz, G.; López-Barajas, M.P.; Mendoza, L.; Palma, O.; Velázquez, O.; Tapia, R.; Sepúlveda, J. Encuesta Nacional de Salud. 2000. Available online: https://ensanut.insp.mx/informes/ENSA_tomo1.pdf (accessed on 6 November 2017).
- Hernández-Cordero, S.; Cuevas-Nasu, L.; Morán-Ruán, M.C.; Méndez-Gómez Humarán, I.; Ávila-Arcos, M.A.; Rivera-Dommarco, J.A. Overweight and obesity in Mexican children and adolescents during the last 25 years. Nutr. Diabetes 2017, 7, 247. [Google Scholar] [CrossRef] [PubMed]
- Remsberg, K.E.; Demerath, E.W.; Schubert, C.M.; Chumlea, W.C.; Sun, S.S.; Siervogel, R.M. Early menarche and the development of cardiovascular disease risk factors in adolescent girls: The Fels Longitudinal Study. J. Clin. Endocrinol. Metab. 2005, 90, 2718–2724. [Google Scholar] [CrossRef] [PubMed]
- Britt, K. Menarche, menopause, and breast cancer risk. Lancet Oncol. 2012, 13, 1071–1072. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics Using IBM SPSS Statistics; SAGE Publications Ltd: London, UK, 2013. [Google Scholar]
- Barquera, S.; Campos-Nonato, I.; Hernández-Barrera, L.; Pedroza-Tobías, A.; Rivera-Dommarco, J.A. Prevalencia de obesidad en adultos mexicanos, ENSANUT 2012. Salud Pública de México 2013, 55, 151–160. [Google Scholar] [CrossRef]
- Coneval. Anexo Estadístico de Pobreza en México. Medición de la Pobreza. 2012. Available online: https://www.coneval.org.mx/Medicion/MP/Paginas/Anexo-estad%C3%ADstico-pobreza-2012.aspx (accessed on 11 January 2017).
- Onland-Moret, N.C.; Peeters, P.H.; van Gils, C.H.; Clavel-Chapelon, F.; Key, T.; Tjonneland, A.; Kaaks, R.; Manjer, J.; Panico, S.; Palli, D. Age at menarche in relation to adult height: The EPIC study. Am. J. Epidemiol. 2005, 162, 623–632. [Google Scholar] [CrossRef]
- Das, J.K.; Salam, R.A.; Thornburg, K.L.; Prentice, A.M.; Campisi, S.; Lassi, Z.S.; Koletzko, B.; Bhutta, Z.A. Nutrition in adolescents: Physiology, metabolism, and nutritional needs. Ann. N. Y. Acad. Sci. 2017, 1393, 21–33. [Google Scholar] [CrossRef]
- Ahmed, T.; Hossain, M.; Sanin, K.I. Global burden of maternal and child undernutrition and micronutrient deficiencies. Ann. Nutr. Metab. 2012, 61 (Suppl. 1), 8–17. [Google Scholar] [CrossRef]
- Macsali, F.; Real, F.G.; Plana, E.; Sunyer, J.; Antó, J.; Dratva, J.; Janson, C.; Jarvis, D.; Omenaas, E.R.; Zemp, E.; et al. Early Age at Menarche, Lung Function, and Adult Asthma. Am. J. Respi.r Crit. Care Med. 2011, 183, 8–14. [Google Scholar] [CrossRef]
- Pierce, M.B.; Leon, D.A. Age at menarche and adult BMI in the Aberdeen children of the 1950s cohort study. Am. J. Clin. Nutr. 2005, 82, 733–739. [Google Scholar] [CrossRef]
- Guh, D.P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C.L.; Anis, A.H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 2009, 9, 888. [Google Scholar] [CrossRef] [PubMed]
- Lakshman, R.; Forouhi, N.; Luben, R.; Bingham, S.; Khaw, K.; Wareham, N.; Ong, K.K. Association between age at menarche and risk of diabetes in adults: Results from the EPIC-Norfolk cohort study. Diabetologia 2008, 51, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Frontini, M.G.; RSrinivasan, S.; Berenson, G. Longitudinal changes in risk variables underlying metabolic Syndrome X from childhood to young adulthood in female subjects with a history of early menarche: The Bogalusa Heart Study. Int. J. Obes. 2003, 27, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Won, J.C.; Hong, J.W.; Noh, J.H.; Kim, D.J. Association Between Age at Menarche and Risk Factors for Cardiovascular Diseases in Korean Women: The 2010 to 2013 Korea National Health and Nutrition Examination Survey. Medicine 2016, 95, 3580. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Yang, Y.; Huang, W.; Zhang, N.; Zhang, F.; Li, G.; Lei, H. Association of age at menarche with obesity and hypertension among southwestern Chinese women: A new finding. Menopause 2018, 25, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Linos, A.; Worthington, J.W.; O’Fallon, W.M.; Kurland, L.T. Case-control study of rheumatoid arthritis and prior use of oral contraceptives. Lancet 1983, 321, 1299–1300. [Google Scholar] [CrossRef]
- Merlino, L.A.; Cerhan, J.R.; Criswell, L.A.; Mikuls, T.R.; Saag, K.G. Estrogen and other female reproductive risk factors are not strongly associated with the development of rheumatoid arthritis in elderly women. Semin. Arthritis Rheum. 2003, 33, 72–82. [Google Scholar] [CrossRef]
- Pedersen, M.; Jacobsen, S.; Klarlund, M.; Pedersen, B.V.; Wiik, A.; Wohlfahrt, J.; Frisch, M. Environmental risk factors differ between rheumatoid arthritis with and without auto-antibodies against cyclic citrullinated peptides. Arthritis Res. Ther. 2006, 8, 133. [Google Scholar] [CrossRef]
- Shishavan, N.G.; Pirouzpanah, S.; Hajialilo, M.; Khabbazi, A.; Jafarpour, F.; Mirtaheri, E.; Farrin, N.; Ebrahimzadeh, S.; Baban, N.; Shakiba, Z.; et al. Risk factors of rheumatoid arthritis development among females in north-west of iran: A case-control study. Iran. Red Crescent Med. J. 2016, 18, e26874. [Google Scholar]
- Dinsa, G.D.; Goryakin, Y.; Fumagalli, E.; Suhrcke, M. Obesity and socioeconomic status in developing countries: A systematic review. Obes. Rev. 2012, 13, 1067–1079. [Google Scholar] [CrossRef]
- Fisher-Hoch, S.P.; Rentfro, A.R.; Wilson, J.G.; Salinas, J.J.; Reininger, B.M.; Restrepo, B.I.; McCormick, J.B.; Pérez, A.; Brown, H.S.; Hossain, M.M.; et al. Peer Reviewed: Socioeconomic Status and Prevalence of Obesity and Diabetes in a Mexican American Community, Cameron County, Texas, 2004–2007. Prev. Chronic Dis. 2010, 7, A53. [Google Scholar] [PubMed]
- WHO. A Global Brief on Hypertension. 2013. Available online: http://apps.who.int/iris/bitstream/10665/79059/1/WHO_DCO_WHD_2013.2_eng.pdf?ua=1 (accessed on 16 January 2017).
- WHO. Obesity and Overweight—Fact Sheet. 2018. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 16 January 2017).
- James-Todd, T.; Tehranifar, P.; Rich-Edwards, J.; Titievsky, L.; Terry, M.B. The impact of socioeconomic status across early life on age at menarche among a racially diverse population of girls. Ann. Epidemiol. 2010, 20, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Cho, G.J.; Park, H.T.; Shin, J.H.; Hur, J.Y.; Kim, Y.T.; Kim, S.H.; Lee, K.W.; Kim, T. Age at menarche in a Korean population: secular trends and influencing factors. Eur. J. Pediatr. 2009, 169, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.H.; Jones, M.E.; Schoemaker, M.J.; Ashworth, A.; Swerdlow, A.J. Secular trends in age at menarche in women in the UK born 1908-93: results from the Breakthrough Generations Study. Paediatr. Perinat Epidemiol. 2011, 25, 394–400. [Google Scholar] [CrossRef] [PubMed]
| Age at Menarche | |||
|---|---|---|---|
| Early (<12 years) | Normal (12–14 years) | Late (>14 years) | |
| Observed n | 4238 | 21333 | 5055 |
| Observed % | 14 | 70 | 16 |
| Age (years) § | 37 (14.0) a | 40 (15.1) b | 45 (16.3) c |
| Height (cm) § | 153.3 (7.0) a | 152.9 (7.0) b | 153.1 (7.2) ab |
| Weight (kg) § | 67.6 (14.1) a | 64.9 (13.4) b | 64.2 (13.8) c |
| BMI (kg/m2) § | 28.8 (5.7) a | 27.7 (5.3) b | 27.3 (5.3) c |
| Hypertension † | 732 (18%) a | 3587 (18%) a | 1030 (22%) b |
| Diabetes † | 337 (8%) ab | 1559 (8%) a | 432 (9%) ab |
| Arthritis † | 201 (5%) a | 1122 (6%) a | 319 (7%) b |
| Hypercholesterolemia † | 385 (10%) a | 1454 (7%) b | 374 (8%) b |
| Breast cancer † | 15 (4%) a | 70 (4%) a | 12 (3%) a |
| Outcome Variables | β Estimates | CI | R2 |
|---|---|---|---|
| Crude model: age at menarche | |||
| BMI | −1.00 * | −1.005, −1.004 | 0.007 |
| Height | −0.02 * | −0.077, −0.034 | 0.000 |
| Weight | −1.01 * | −1.005, −1.004 | 0.006 |
| Adjusted model: age at menarche + current age | |||
| BMI | −1.01 * | −1.007, −1.006 | 0.040 |
| Height | 0.18 * | −0.112, −0.231 | 0.063 |
| Weight | −1.01 * | −1.006, -1.004 | 0.009 |
| Disease Outcome | RR 1 | 95% CI 1 |
|---|---|---|
| Diabetes | 0.95 * | 0.93, 0.98 |
| Hypertension | 1.00 | 0.98, 1.02 |
| Hypercholesterolemia | 0.93 * | 0.90, 0.95 |
| Breast cancer § | 0.95 | 0.98, 1.02 |
| Arthritis | 1.00 | 0.97, 1.03 |
| Gout arthritis | 1.05 | 0.94, 1.18 |
| Rheumatoid arthritis | 1.00 | 0.99, 1.02 |
| Other arthritis | 0.93 | 0.93, 1.07 |
| Decade | 1900s | 1910s | 1920s | 1930s | 1940s | 1950s | 1960s | 1970s | 1980s |
|---|---|---|---|---|---|---|---|---|---|
| 1900s | 1 | 0.10 | 0.14 | −0.04 | −0.20 | −0.40 | −0.49 | −0.69 * | −0.86 * |
| 1910s | 1 | 0.24 | −0.13 | −0.11 | −0.30 * | −0.39 ** | −0.59 ** | −0.76 ** | |
| 1920s | 1 | −0.10 | −0.34 ** | −0.53 ** | −0.63 ** | −0.83 ** | −0.99 ** | ||
| 1930s | 1 | −0.24 ** | −0.43 ** | −0.53 ** | −0.72 ** | −0.89 ** | |||
| 1940s | 1 | −0.19 ** | −0.29 ** | −0.49 ** | −0.65 ** | ||||
| 1950s | 1 | −0.10 * | −0.29 ** | −0.46 ** | |||||
| 1960s | 1 | −0.20 ** | −0.37 * | ||||||
| 1970s | 1 | −0.17 * | |||||||
| 1980s | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petersohn, I.; Zarate-Ortiz, A.G.; Cepeda-Lopez, A.C.; Melse-Boonstra, A. Time Trends in Age at Menarche and Related Non-Communicable Disease Risk during the 20th Century in Mexico. Nutrients 2019, 11, 394. https://doi.org/10.3390/nu11020394
Petersohn I, Zarate-Ortiz AG, Cepeda-Lopez AC, Melse-Boonstra A. Time Trends in Age at Menarche and Related Non-Communicable Disease Risk during the 20th Century in Mexico. Nutrients. 2019; 11(2):394. https://doi.org/10.3390/nu11020394
Chicago/Turabian StylePetersohn, Inga, Arli G. Zarate-Ortiz, Ana C. Cepeda-Lopez, and Alida Melse-Boonstra. 2019. "Time Trends in Age at Menarche and Related Non-Communicable Disease Risk during the 20th Century in Mexico" Nutrients 11, no. 2: 394. https://doi.org/10.3390/nu11020394
APA StylePetersohn, I., Zarate-Ortiz, A. G., Cepeda-Lopez, A. C., & Melse-Boonstra, A. (2019). Time Trends in Age at Menarche and Related Non-Communicable Disease Risk during the 20th Century in Mexico. Nutrients, 11(2), 394. https://doi.org/10.3390/nu11020394





