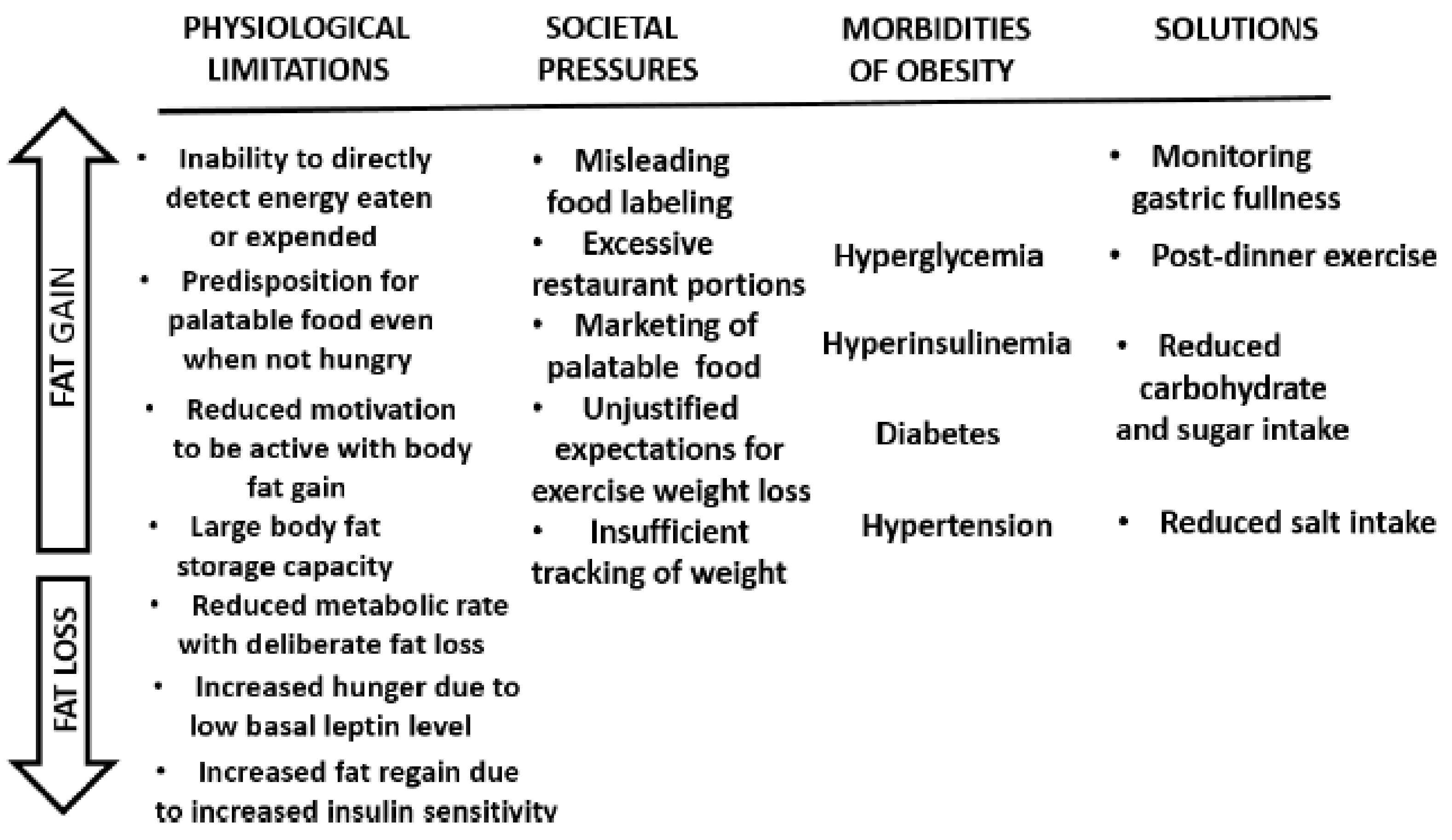
Understanding Human Physiological Limitations and Societal Pressures in Favor of Overeating Helps to Avoid Obesity
Abstract
1. Introduction
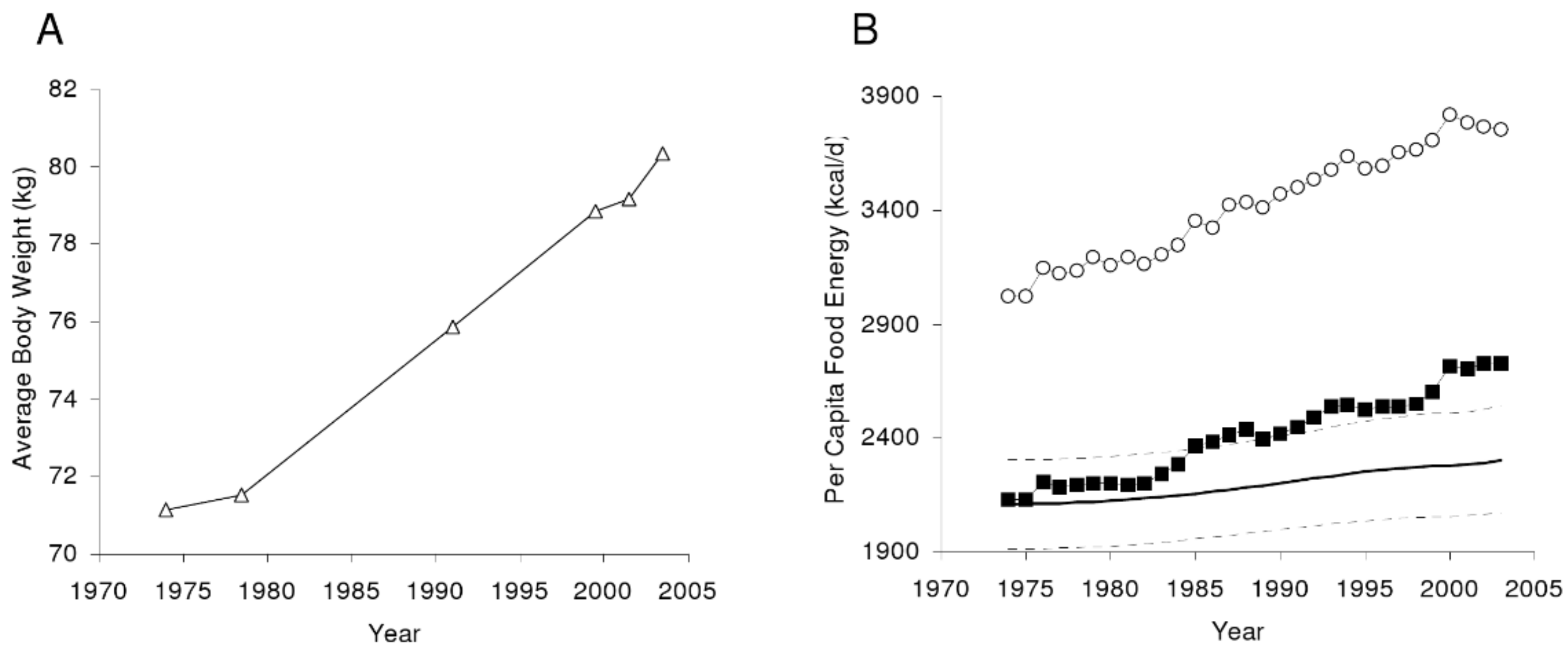
2. Sustained Rise of Obesity Is Associated with Pathology and Economic Burdens
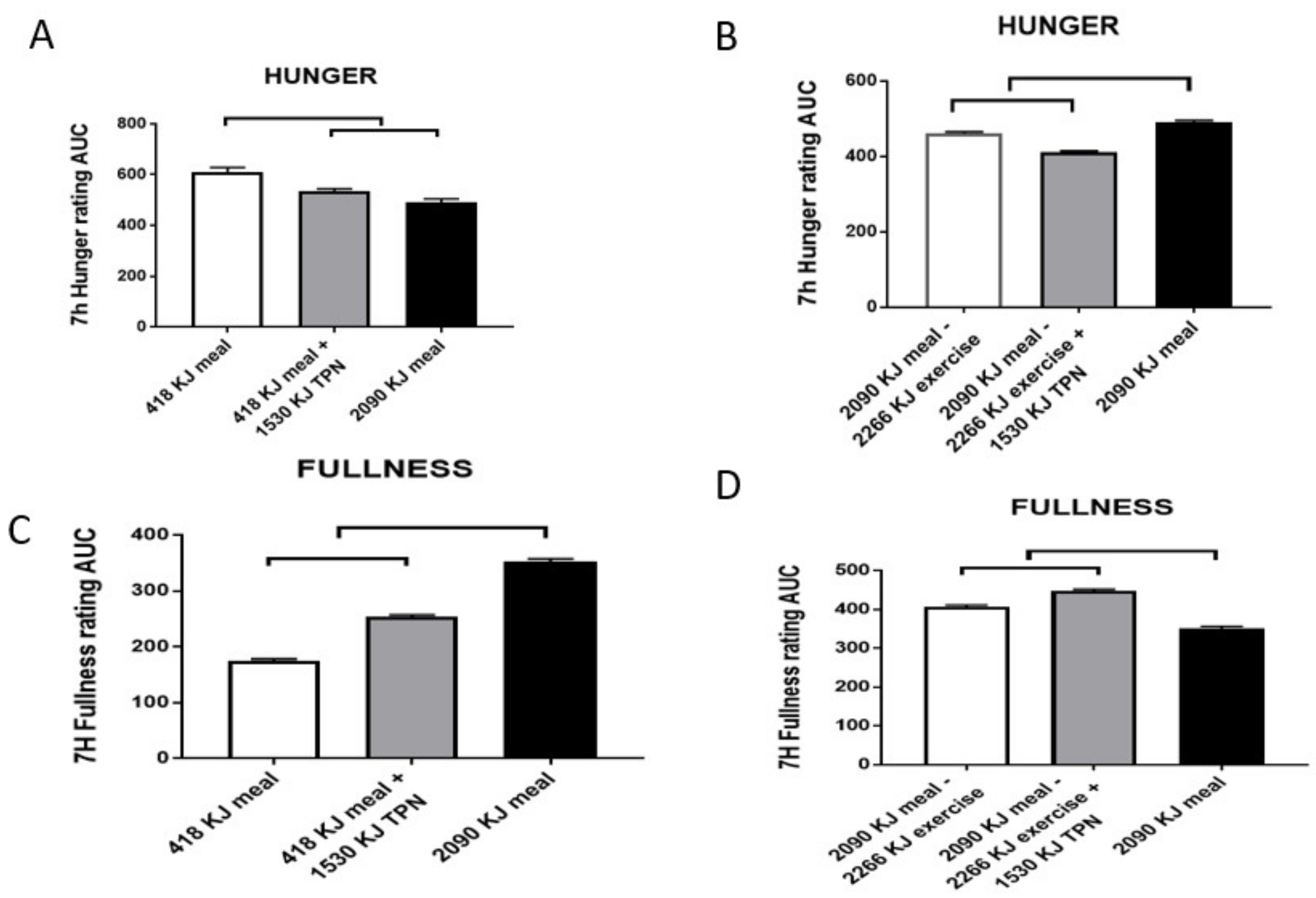
3. Strong Preference for Intake of Palatable Foods Even When Not Hungry
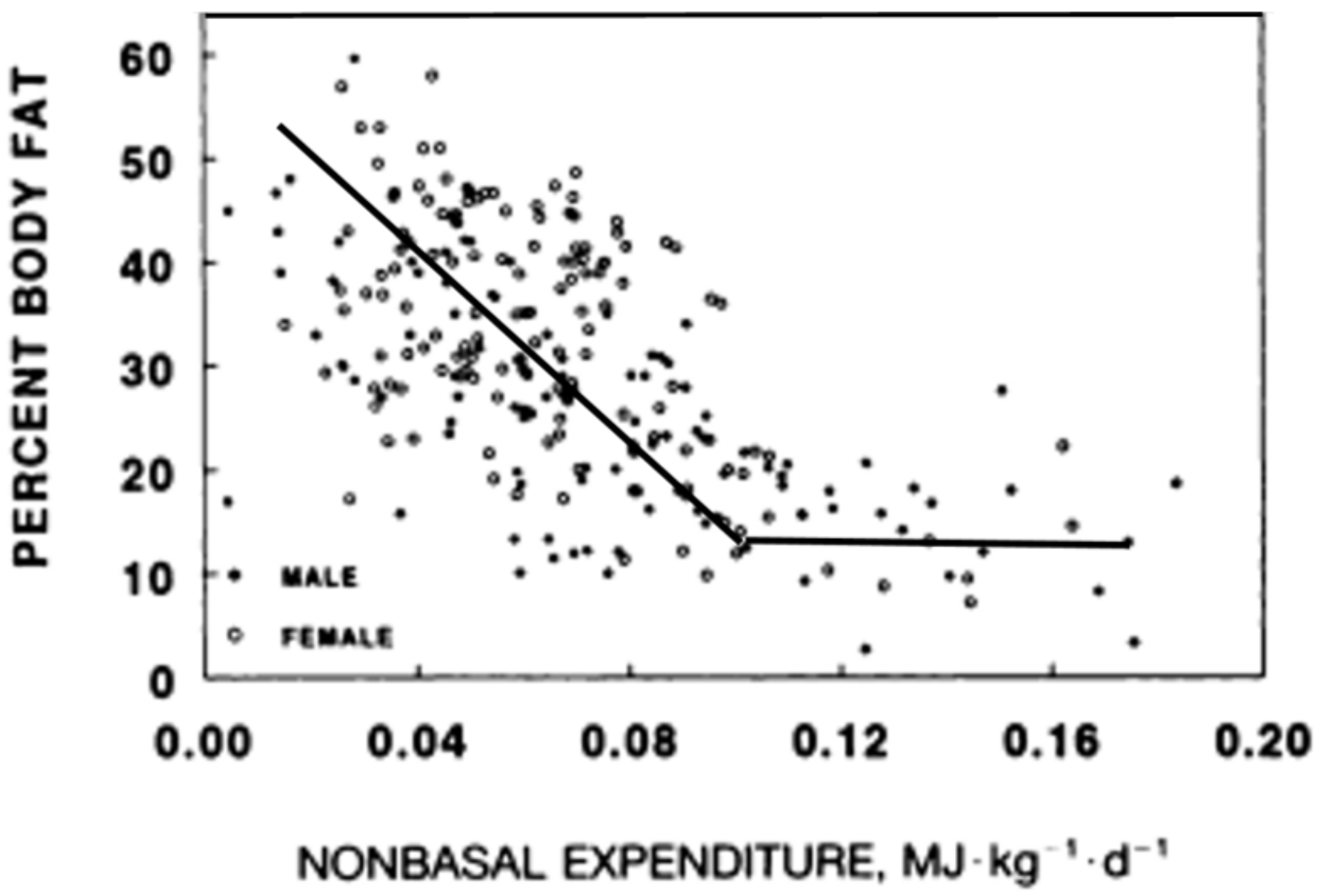
4. Inability to Detect Energy Eaten or Expended in Physical Activity Represents a Core Human Physiological Handicap for Successful Weight Management
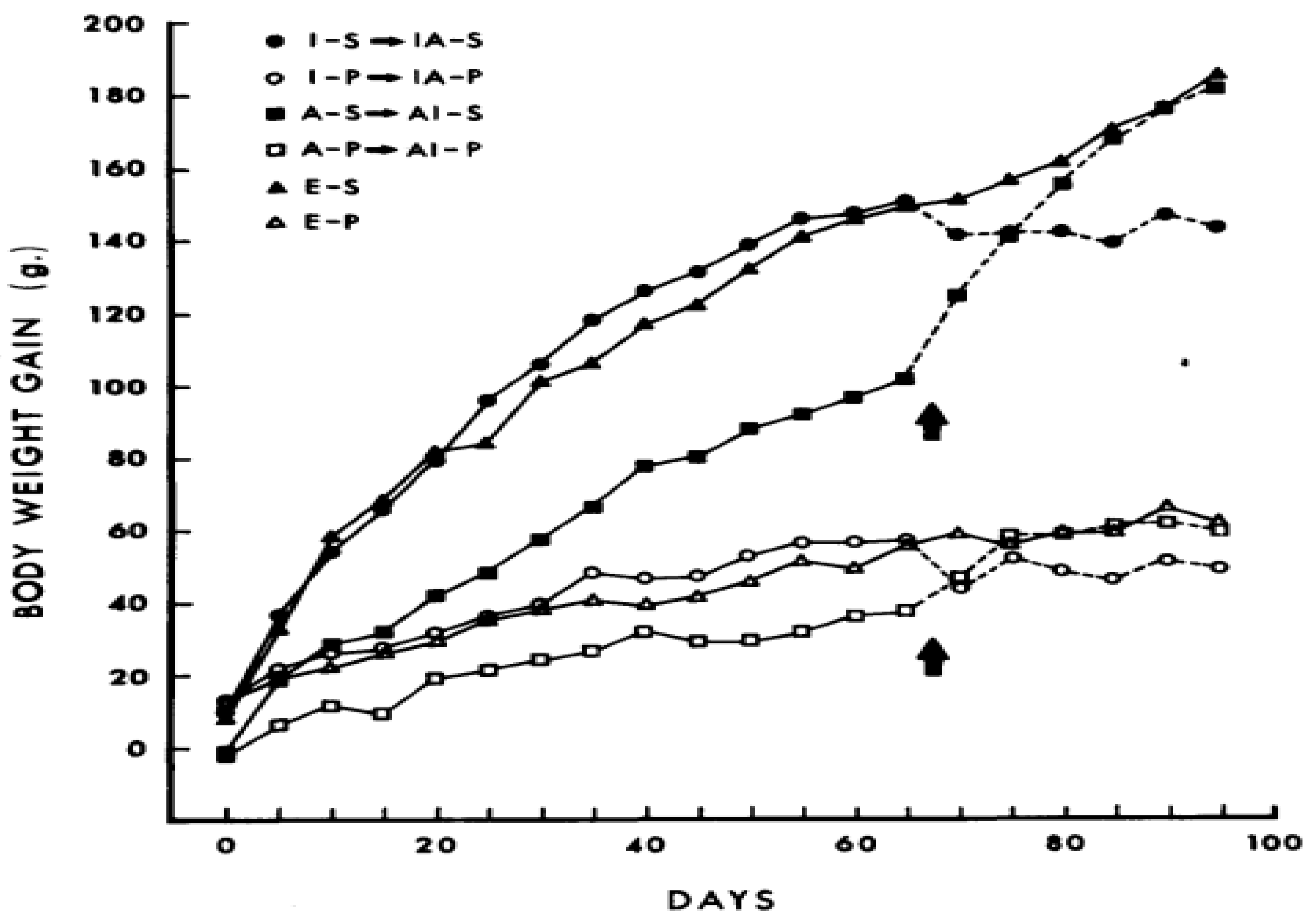
5. Human Genetic Predisposition for Obesity and Vigorous Physiological Defenses against Weight Loss Bias Our Physiology toward Fat Gain and Retention
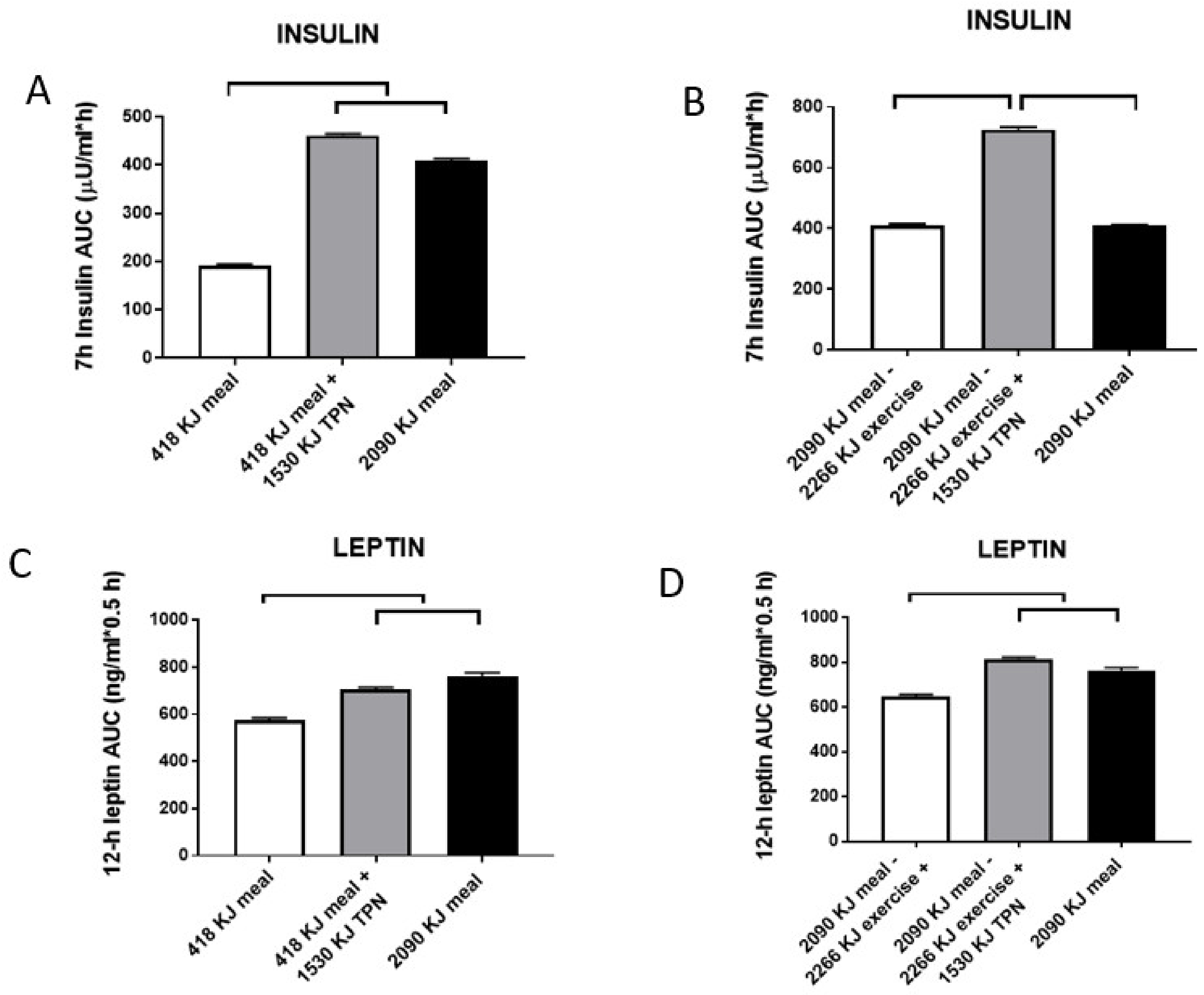
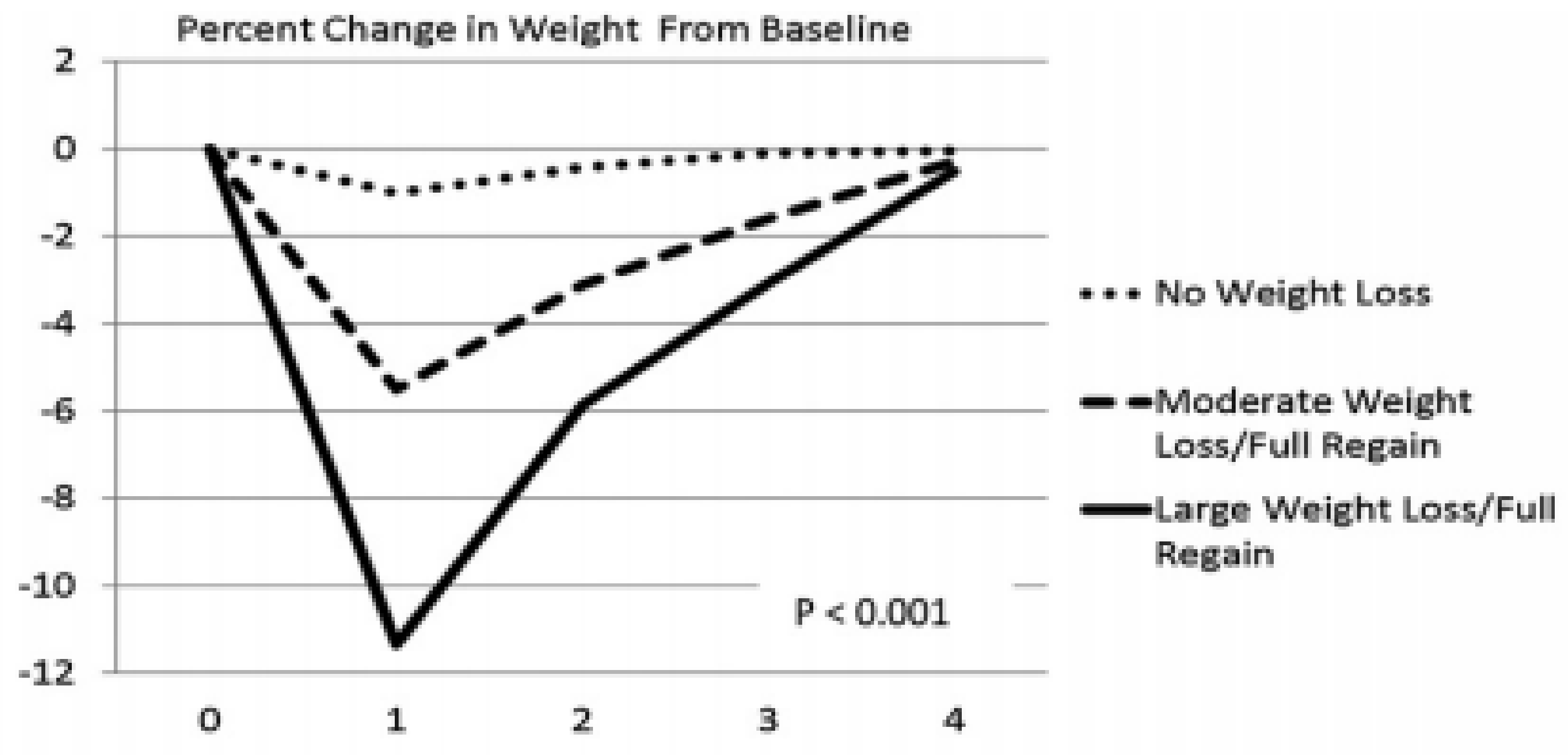
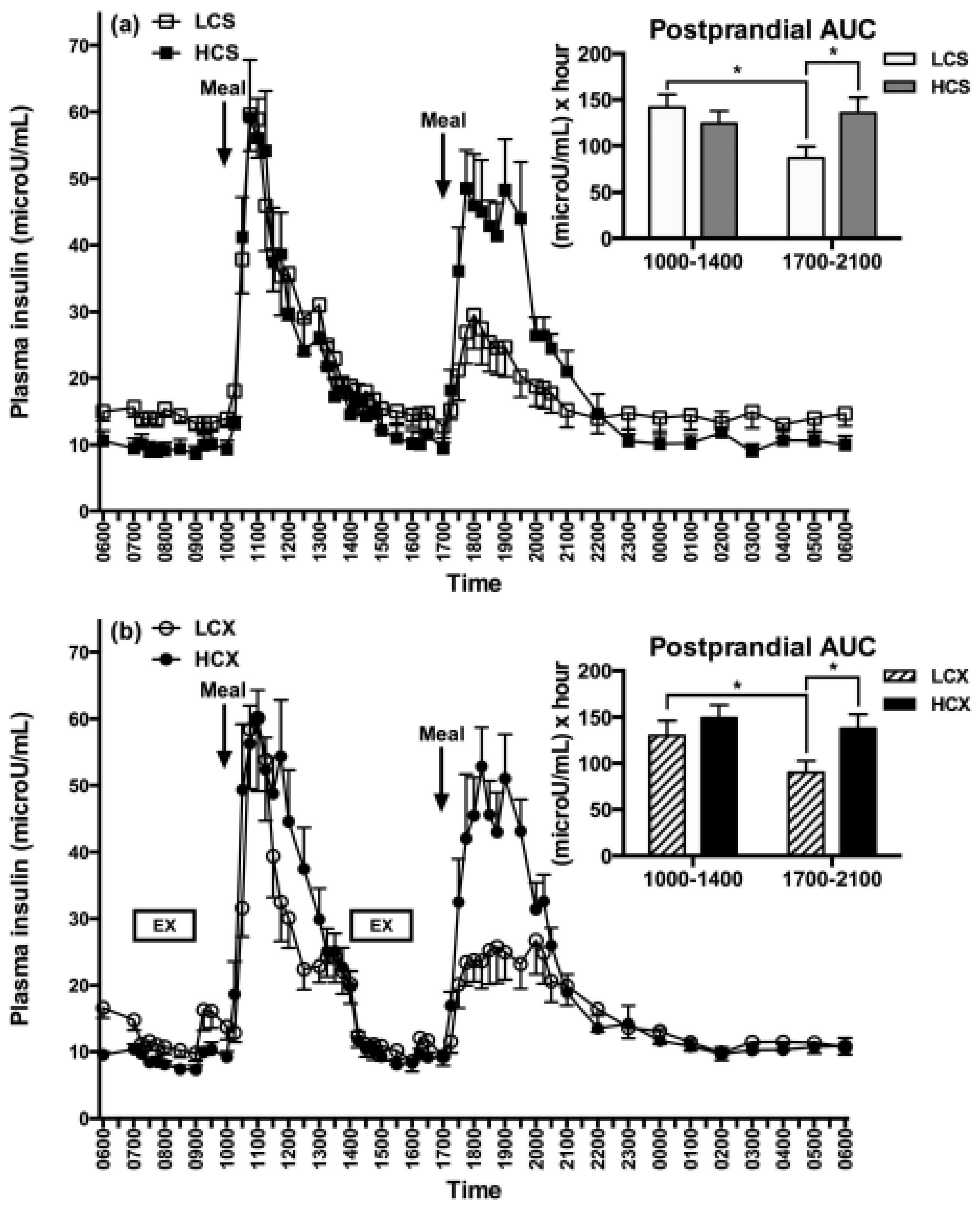
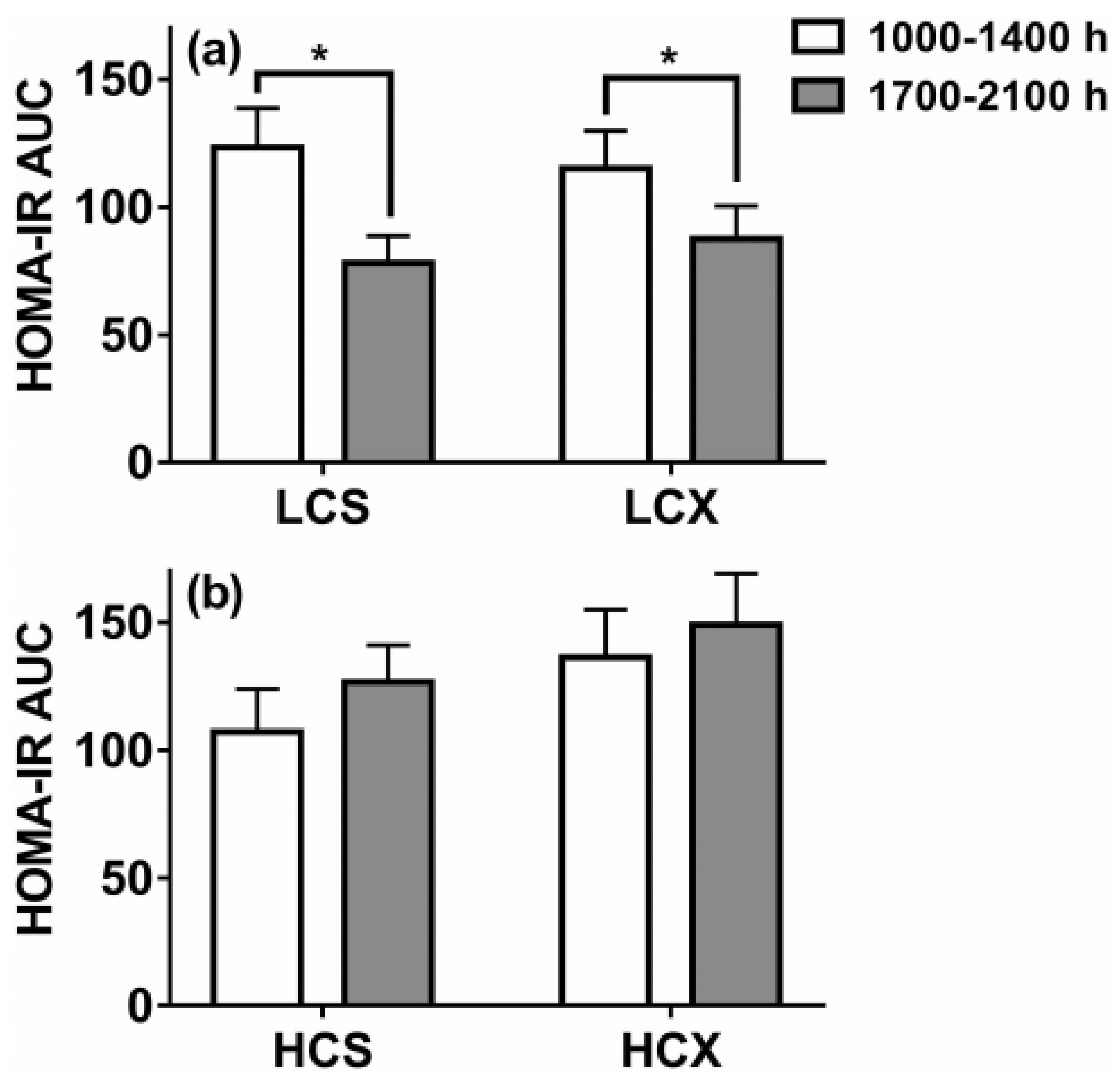
6. Right Nutrients Matter in Protecting Human Health as Does Timing of Meals and Exercise
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- NCD Risk Factor Collaboration. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- Hales, C.M.; Carrol, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of obesity among adults and youth: United States, 2015-2016. NCHS Data Briefs 2017, 288, 1–8. [Google Scholar]
- Finkelstein, E.A.; Trogdon, J.G.; Cohen, J.W.; Dietz, W. Annual medical spending attributable to obesity: Payer-and service-specific estimates. Health Affairs 2009, 28, W822–W831. [Google Scholar] [CrossRef]
- Muller-Riemenschneider, F.; Reinhold, T.; Berghofer, A.; Willich, S.N. Health-economic burden of obesity in Europe. Eur. J. Epidemiol. 2008, 23, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Vaněčková, I.; Maletínská, L.; Behuliak, M.; Nagelová, V.; Zicha, J.; Kuneš, J. Obesity-related hypertension: Possible pathophysiological mechanisms. J. Endocrinol. 2014, 223, R63–R78. [Google Scholar] [CrossRef] [PubMed]
- Lovren, F.; Teoh, H.; Verma, S. Obesity and atherosclerosis: Mechanistic insights. Can. J. Cardiol. 2015, 31, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Samad, F.; Ruf, W. Inflammation, obesity, and thrombosis. Blood 2013, 122, 3415–3422. [Google Scholar] [CrossRef] [PubMed]
- Iantorno, M.; Campia, U.; Di Daniele, N.; Nistico, S.; Forleo, G.B.; Cardillo, C.; Tesauro, M. Obesity, inflammation and endothelial dysfunction. J. Biol. Regul. Homeost. Agents 2014, 28, 169–176. [Google Scholar]
- Rankinen, T.; Sarzynski, M.A.; Ghosh, S.; Bouchard, C. Are there genetic paths common to obesity, cardiovascular disease outcomes, and cardiovascular risk factors? Circ. Res. 2015, 116, 909–922. [Google Scholar] [CrossRef]
- Ritz, E.; Rychlik, I.; Locatelli, F.; Halimi, S. End-stage renal failure in type 2 diabetes: A medical catastrophe of worldwide dimensions. Am. J. Kidney Dis. 1999, 34, 795–808. [Google Scholar] [CrossRef]
- Després, J.P.; Lamarche, B.; Mauriège, P.; Cantin, B.; Dagenais, G.R.; Moorjani, S.; Lupien, P.J. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N. Engl. J. Med. 1996, 334, 952–957. [Google Scholar] [CrossRef] [PubMed]
- Field, A.E.; Coakley, E.H.; Must, A.; Spadano, J.L.; Laird, N.; Dietz, W.H.; Rimm, E.; Colditz, G.A. Impact of overweight on the risk of developing common chronic diseases during a 10-year period. Arch. Intern. Med. 2001, 161, 1581–1586. [Google Scholar] [CrossRef]
- Farag, Y.M.; Gaballa, M.R. Diabesity: An overview of a rising epidemic. Nephrol. Dial. Transplant. 2011, 26, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.F.; Schatzkin, A.; Harris, T.B.; Kipnis, V.; Mouw, T.; Ballard-Barbash, R.; Hollenbeck, A.; Leitzmann, M.F. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N. Engl. J. Med. 2006, 355, 763–778. [Google Scholar] [CrossRef] [PubMed]
- Borer, K.T. How effective is exercise in producing fat loss? Kinesiology 2008, 40, 126–137. [Google Scholar]
- Catenacci, V.A.; Wyatt, H.R. The role of physical activity in producing and maintaining weight loss. Nat. Clin. Pract. Endocrinol. Metab. 2007, 3, 518–529. [Google Scholar] [CrossRef] [PubMed]
- NCBI, National Institute of Biotechnology Information, NIH. 2012. Available online: https://www-ncbi-nlm-nih-gov.proxy.lib.umich.edu/books/NBK201136/table/tab_2-5/?report=objectonly (accessed on 25 October 2018).
- King, N.A.; Burley, V.J.; Blundell, J.E. Exercise-induced suppression of appetite: Effects on food intake and implications for energy balance. Eur. J. Clin. Nutr. 1994, 48, 715–724. [Google Scholar] [PubMed]
- Borer, K.T.; Potter, C.D.; Fileccia, N. Basis for hypoactivity that accompanies rapid weight gain in hamsters. Physiol. Behav. 1983, 30, 389–397. [Google Scholar] [CrossRef]
- Borer, K.T. Nonhomeostatic control of human appetite and physical activity in regulation of energy balance. Exerc. Sport Sci. Rev. 2010, 38, 114–121. [Google Scholar] [CrossRef]
- Rising, R.; Harper, I.T.; Fontvielle, A.M.; Ferraro, R.T.; Spraul, M.; Ravussin, E. Determinants of total daily energy expenditure: Variability in physical activity. Am. J. Clin. Nutr. 1994, 59, 800–804. [Google Scholar] [CrossRef]
- Schulz, L.O.; Schoeller, D.A. A compilation of total daily energy expenditures and body weights in healthy adults. Am. J. Clin. Nutr. 1994, 60, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Fothergill, E.; Guo, J.; Howard, L.; Kerns, J.C.; Knuth, N.D.; Brychta, R.; Chen, K.Y.; Skarulis, M.C.; Walter, M.; Walter, P.J.; et al. Persistent metabolic adaptation 6 years after “The Biggest Loser” competition. Obesity 2016, 24, 1612–1619. [Google Scholar] [CrossRef] [PubMed]
- Knuth, N.D.; Johannsen, D.L.; Tamboli, R.A.; Marks-Shulman, P.A.; Huizenga, R.; Chen, K.Y.; Abumrad, N.N.; Ravussin, E.; Hall, K.D. Metabolic adaptation following massive weight loss is related to the degree of energy imbalance and changes in circulating leptin. Obesity 2014, 22, 2563–2569. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, M.V.; Booth, F.W. Eating, exercise, and “thrifty” genotypes: Connecting the dots toward an evolutionary understanding of modern chronic diseases. J. Appl. Physiol. 2004, 96, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Rosenstein, D.; Oster, H. Differential facial responses to four basic tastes in newborns. Child Dev. 1988, 59, 1555–1568. [Google Scholar] [CrossRef] [PubMed]
- Berridge, K.C. ‘Liking’ and ‘wanting’ food rewards: Brain substrates and roles in eating disorders. Physiol. Behav. 2009, 97, 537–550. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Krolewski, D.M.; Moore, S.; Kumar, V.; Li, F.; Martin, B.; Tomer, R.; Murphy, G.G.; Deisseroth, K.; Watson, S.J., Jr.; et al. Uneven balance of power between hypothalamic peptidergic neurons in the control of feeding. Proc. Natl. Acad. Sci. USA 2018, 115, E9489–E9498. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, A.; Springer, D. Dietary obesity in adult rats: Similarities to hypothalamic and human obesity syndromes. Physiol. Behav. 1976, 17, 461–471. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Hall, K.D.; McPherson, K.; Finegood, D.T.; Moodie, M.L.; Gortmaker, S.L. The global obesity pandemic: Shaped by global drivers and local environments. Lancet 2011, 378, 804–814. [Google Scholar] [CrossRef]
- Schwartz, M.W.; Woods, S.C.; Porte, D., Jr.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Bagdade, J.D.; Bierman, E.L.; Porte, D., Jr. The significance of basal insulin levels in the evaluation of the insulin response to glucose in diabetic and nondiabetic subjects. J. Clin. Investig. 1967, 46, 1549–1557. [Google Scholar] [CrossRef] [PubMed]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, I.S.; Jebb, S.A.; Langmack, G.; Lawrence, E.; Cheetham, C.H.; Prentice, A.M.; Hughes, I.A.; McCamish, M.A.; O’Rahilly, S. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N. Engl. J. Med. 1999, 341, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Pelleymounter, M.A.; Cullen, M.J.; Baker, M.B.; Hecht, R.; Winters, D.; Boone, T.; Collins, F. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 1995, 269, 540–543. [Google Scholar] [CrossRef] [PubMed]
- Seeley, R.J.; van Dijk, G.; Campfield, L.A.; Smith, F.J.; Burn, P.; Nelligan, J.A.; Bell, S.M.; Baskin, D.G.; Woods, S.C.; Schwartz, M.W. Intraventricular leptin reduces food intake and body weight of lean rats but not obese Zucker rats. Horm. Metab. Res. 1996, 28, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; Greenberg, A.S.; Fujioka, K.; Dixon, R.M.; Kushner, R.; Hunt, T.; Lubina, J.A.; Patane, J.; Self, B.; Hunt, P.; et al. Recombinant leptin for weight loss in obese and lean adults: A randomized, controlled, dose-escalation trial. JAMA 1999, 282, 1568–1575. [Google Scholar] [CrossRef] [PubMed]
- Ahrén, B.; Månsson, S.; Gingerich, R.L.; Havel, P.J. Regulation of plasma leptin in mice: Influence of age, high-fat diet, and fasting. Am. J. Physiol. 1997, 273 Pt 2, R113–R120. [Google Scholar] [CrossRef]
- Maffei, M.; Halaas, J.; Ravussin, E.; Pratley, R.E.; Lee, G.H.; Zhang, Y.; Fei, H.; Kim, S.; Lallone, R.; Ranganathan, S.; et al. Leptin levels in human and rodent: Measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat. Med. 1995, 1, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Samuel, V.T.; Petersen, K.F.; Shulman, G.I. Lipid-induced insulin resistance: Unravelling the mechanism. Lancet 2010, 375, 2267–2277. [Google Scholar] [CrossRef]
- Shulman, G.I. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N. Engl. J. Med. 2014, 371, 2237–2238. [Google Scholar] [CrossRef] [PubMed]
- Levitsky, D.A. The non-regulation of food intake in humans: Hope for reversing the epidemic of obesity. Physiol. Behav. 2005, 86, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Kendall, A.; Levitsky, D.A.; Strupp, B.J.; Lissner, L. Weight loss on a low-fat diet: Consequence of the imprecision of the control of food intake in humans. Am. J. Clin. Nutr. 1991, 53, 1124–1129. [Google Scholar] [CrossRef]
- Borer, K.T.; Wuorinen, E.; Chao, C.; Burant, C. Exercise energy expenditure is not consciously detected due to oro-gastric, not metabolic basis of hunger sensation. Appetite 2005, 45, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Borer, K.T.; Wuorinen, E.; Ku, K.; Burant, C. Appetite responds to changes in meal content, whereas ghrelin, leptin, and insulin track changes in energy availability. J. Clin. Endocrinol. Metab. 2009, 94, 2290–2298. [Google Scholar] [CrossRef]
- Golzarand, M.; Toolabi, K.; Djafarian, K. Changes in body composition, dietary intake, and substrate oxidation in patients underwent laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy: A comparative prospective study. Obes. Surg. 2018. [Google Scholar] [CrossRef] [PubMed]
- Borer, K.T. Counterregulation of insulin by leptin as key component of autonomic regulation of body weight. World J. Diabet. 2014, 5, 606–629. [Google Scholar] [CrossRef]
- Van Aggel-Leijssen, D.P.; van Baak, M.A.; Tenenbaum, R.; Campfield, L.A.; Saris, W.H. Regulation of average 24h human plasma leptin level; the influence of exercise and physiological changes in energy balance. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 151–158. [Google Scholar] [CrossRef]
- Bado, A.; Levasseur, S.; Attoub, S.; Kermorgant, S.; Laigneau, J.P.; Bortoluzzi, M.N.; Moizo, L.; Lehy, T.; Guerre-Millo, M.; Le Marchand-Brustel, Y.; et al. The stomach is a source of leptin. Nature 1998, 394, 790–793. [Google Scholar] [CrossRef]
- Cammisotto, P.; Bendayan, M. A review on gastric leptin: The exocrine secretion of a gastric hormone. Anat. Cell Biol. 2012, 45, 1–16. [Google Scholar] [CrossRef]
- Kissileff, H.R.; Carretta, J.C.; Geliebter, A.; Pi-Sunyer, F.X. Cholecystokinin and stomach distension combine to reduce food intake in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 285, R992–R998. [Google Scholar] [CrossRef]
- Smith, G.P.; Gibbs, J. Cholecystokinin and the satiating effect of fat. Gastroenterology 1992, 102, 1801–1803. [Google Scholar]
- Ruff, C.B.; Trinkaus, E.; Holliday, T.W. Body mass and encephalization in Pleistocene Homo. Nature 1997, 387, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Isler, K.; van Schaik, C.P. The Expensive Brain: A framework for explaining evolutionary changes in brain size. J. Hum. Evol. 2009, 57, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Leonard, W.R.; Snodgrass, J.J.; Robertson, M.L. Effects of brain evolution on human nutrition and metabolism. Annu. Rev. Nutr. 2007, 27, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Leonard, W.R.; Robertson, M.L.; Snodgrass, J.J.; Kuzawa, C.W. Metabolic correlates of hominid brain evolution. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2003, 136, 5–15. [Google Scholar] [CrossRef]
- Zihlman, A.L.; McFarland, R.K.; Underwood, C.E. Functional anatomy and adaptation of male gorillas (Gorilla gorilla gorilla) with comparison to male orangutans (Pongo pygmaeus). Anat. Rec. 2011, 294, 1842–1855. [Google Scholar] [CrossRef]
- Pond, C.M.; Mattacks, C.A. The anatomy of adipose tissue in captive Macaca monkeys and its implications for human biology. Folia Primatol 1987, 48, 164–185. [Google Scholar] [CrossRef]
- Stini, W. Body composition and nutrient reserves in evolutionary perspective. World Rev. Nutr. Diet. 1981, 37, 55–83. [Google Scholar]
- Leonard, W.R.; Snodgrass, J.J.; Robertson, M.L. Evolutionary perspectives on fat ingestion and metabolism in humans. In Fat Detection: Taste, Texture, and Post Ingestive Effects; Montmayeur, J.P., le Coutre, J., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2010; Chapter 1. [Google Scholar]
- Kuzawa, C.W. Adipose tissue in human infancy and childhood: An evolutionary perspective. Am. J. Phys. Anthropol. 1998, 107, 177–209. [Google Scholar] [CrossRef]
- Robbins, M.M.; Bermejo, M.; Cipolletta, C.; Magljocca, F.; Parnell, R.J.; Stokes, E. Social structure and life history patterns in western gorillas (Gorilla gorilla gorilla). Am. J. Primatol. 2004, 64, 145–159. [Google Scholar] [CrossRef]
- Nishida, T.; Corp, N.; Hamai, M.; Hasegawa, T.; Hiraiwa-Hasegawa, M.; Hosaka, K.; Hunt, K.D.; Itoh, N.; Kawanaka, K.; Matsumoto-Oda, A.; et al. Demography, female life history, and reproductive profiles among the chimpanzees of Mahale. Am. J. Primatol. 2003, 59, 99–121. [Google Scholar] [CrossRef] [PubMed]
- Hube, F.; Lietz, U.; Igel, M.; Jensen, P.B.; Tornqvist, H.; Joost, H.G.; Hauner, H. Difference in leptin mRNA levels between omental and subcutaneous abdominal adipose tissue from obese humans. Horm. Metab. Res. 1996, 28, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Leibel, R.L. 20 years of leptin: Role of leptin in energy homeostasis in humans. J. Endocrinol. 2014, 223, T83–T96. [Google Scholar] [CrossRef] [PubMed]
- Kissileff, H.R.; Thornton, J.C.; Torres, M.I.; Pavlovich, K.; Mayer, L.S.; Kalari, V.; Leibel, R.L.; Rosenbaum, M. Leptin reverses declines in satiation in weight-reduced obese humans. Am. J. Clin. Nutr. 2012, 95, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Espeland, M.A.; Clark, J.M.; Hazuda, H.P.; Knowler, W.C.; Pownall, H.J.; Unick, J.; Wadden, T.; Wagenknecht, L. Association of Weight Loss Maintenance and Weight Regain on 4-Year Changes in CVD Risk Factors: The Action for Health in Diabetes (Look AHEAD) Clinical Trial. Diabetes Care 2016, 39, 1345–1355. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.T.; Wing, R.R.; Klem, M.L.; Hill, J.O. Behavioral strategies of individuals who have maintained long-term weight losses. Obes. Res. 1999, 7, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Falhammar, H.; Frisén, L.; Norrby, C.; Hirschberg, A.L.; Almqvist, C.; Nordenskjöld, A.; Nördenstrom, A. Increased mortality in patients with congenital adrenal hyperplasia due to 21-hyroxylase deficiency. J. Clin. Endocrinol. Metab. 2014, 99, E2715–E2721. [Google Scholar] [CrossRef]
- Takamata, A.; Mack, G.W.; Gillen, C.M.; Nadel, E.R. Sodium appetite, thirst, and body fluid regulation in humans during rehydration without sodium replacement. Am. J. Physiol. 1995, 266 Pt 2, R1493–R1502. [Google Scholar] [CrossRef]
- Rust, P.; Ekmekcioglu, C. Impact of salt intake on the pathogenesis and treatment of hypertension. Adv. Exp. Med. Biol. 2017, 956, 61–84. [Google Scholar] [CrossRef] [PubMed]
- Rocchini, A.P. Obesity hypertension, salt sensitivity and insulin resistance. Nutr. Metab. Cardiovasc. Dis. 2000, 10, 287–294. [Google Scholar] [PubMed]
- Powell, E.S.; Smith-Taillie, L.P.; Popkin, B.M. Added sugars intake across the distribution of US children and adult consumers: 1977-2012. J. Acad. Nutr. Diet. 2016, 116, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.K.; Appel, L.J.; Brands, M.; Howard, B.V.; Lefevre, M.; Lustig, R.H.; Sacks, F.; Steffen, L.M.; Wylie-Rosett, J. Dietary sugars intake and cardiovascular health. A scientific statement from the American Heart Association. Circulation 2009, 120, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.-J.; Borer, K.T. Third exposure to a reduced carbohydrate meal lowers evening postprandial insulin and GIP responses and HOMA-IR estimate of insulin resistance. PLoS ONE 2016, 11, e0165378. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture and US Department of Health and Human Services. Dietary Guidelines for Americans, 2010, 7th ed.; U.S. Government Printing Office: Washington, DC, USA, 2010.
- Keys, A.; Anderson, J.T.; Grande, F. Serum cholesterol response to changes in the diet. IV. Particular saturated fatty acids in the diet. Metabolism 1965, 14, 776–787. [Google Scholar] [CrossRef]
- Keys, A.; Taylor, H.L.; Blackburn, H.; Brozek, J.; Anderson, J.T.; Simonson, E. Mortality and coronary heart disease among men studied for 23 years. Arch. Intern. Med. 1971, 128, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.; Cragg, M.; deFonseka, J.; Hite, A.; Rosenberg, M.; Zhou, B. Statistical review of US macronutrient consumption data, 1965–2011: Americans have been following dietary guidelines, coincident with the rise in obesity. Nutrition 2015, 31, 727–732. [Google Scholar] [CrossRef]
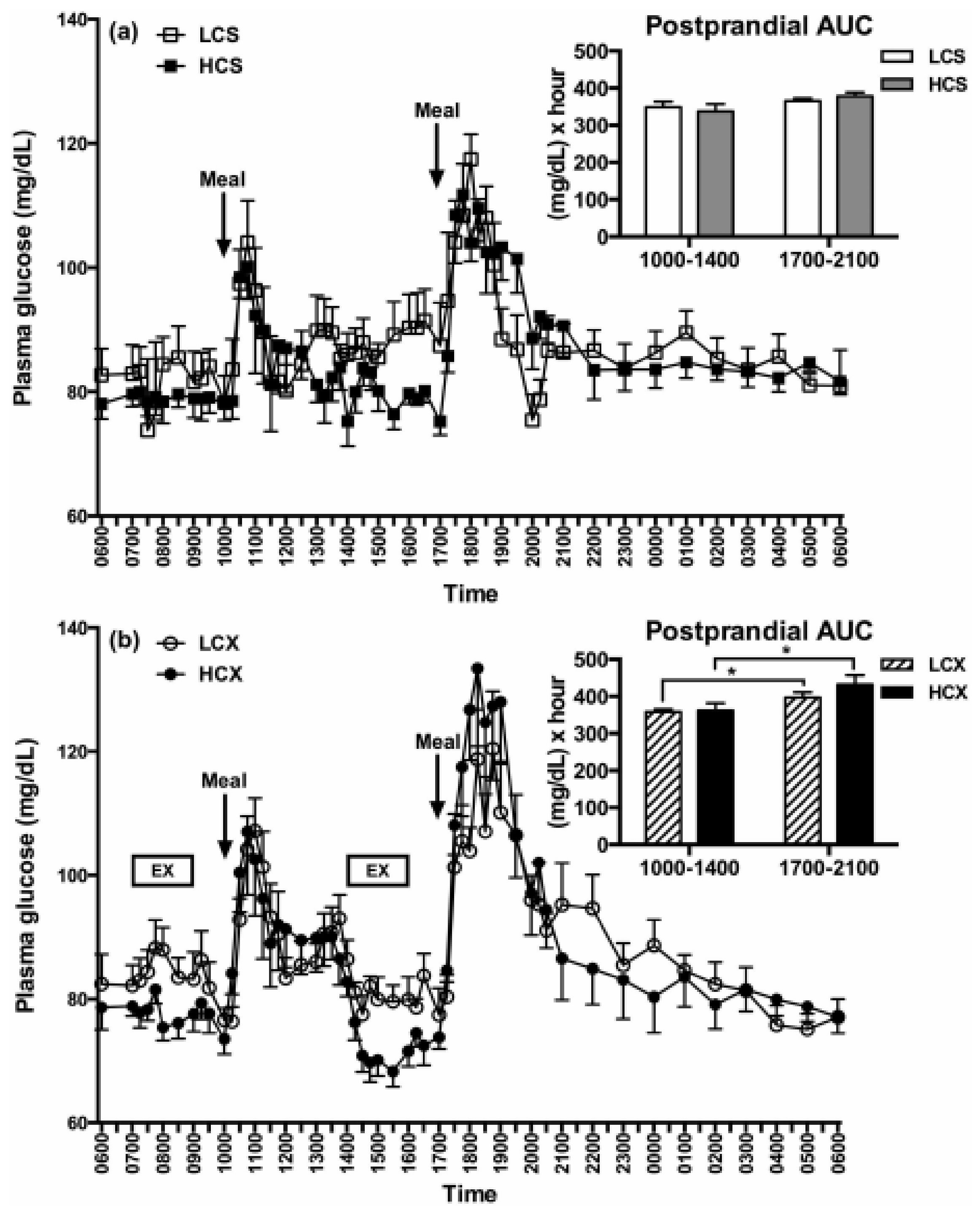
- Heden, T.D.; Winn, N.C.; Mari, A.; Booth, F.W.; Rector, R.S.; Thyfault, J.P.; Kanaley, J.A. Postdinner resistance exercise improves postprandial risk factors more effectively than predinner resistance exercise in patients with type 2 diabetes. J. Appl. Physiol. 2015, 118, 624–634. [Google Scholar] [CrossRef]

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borer, K.T. Understanding Human Physiological Limitations and Societal Pressures in Favor of Overeating Helps to Avoid Obesity. Nutrients 2019, 11, 227. https://doi.org/10.3390/nu11020227
Borer KT. Understanding Human Physiological Limitations and Societal Pressures in Favor of Overeating Helps to Avoid Obesity. Nutrients. 2019; 11(2):227. https://doi.org/10.3390/nu11020227
Chicago/Turabian StyleBorer, Katarina T. 2019. "Understanding Human Physiological Limitations and Societal Pressures in Favor of Overeating Helps to Avoid Obesity" Nutrients 11, no. 2: 227. https://doi.org/10.3390/nu11020227
APA StyleBorer, K. T. (2019). Understanding Human Physiological Limitations and Societal Pressures in Favor of Overeating Helps to Avoid Obesity. Nutrients, 11(2), 227. https://doi.org/10.3390/nu11020227



