Day-Time Patterns of Carbohydrate Intake in Adults by Non-Parametric Multi-Level Latent Class Analysis—Results from the UK National Diet and Nutrition Survey (2008/09–2015/16)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Diet Diary and Definition of CH Intake
2.3. Statistical Analysis
2.4. Characteristics and Associations between CH Intake Classes and T2D
3. Results
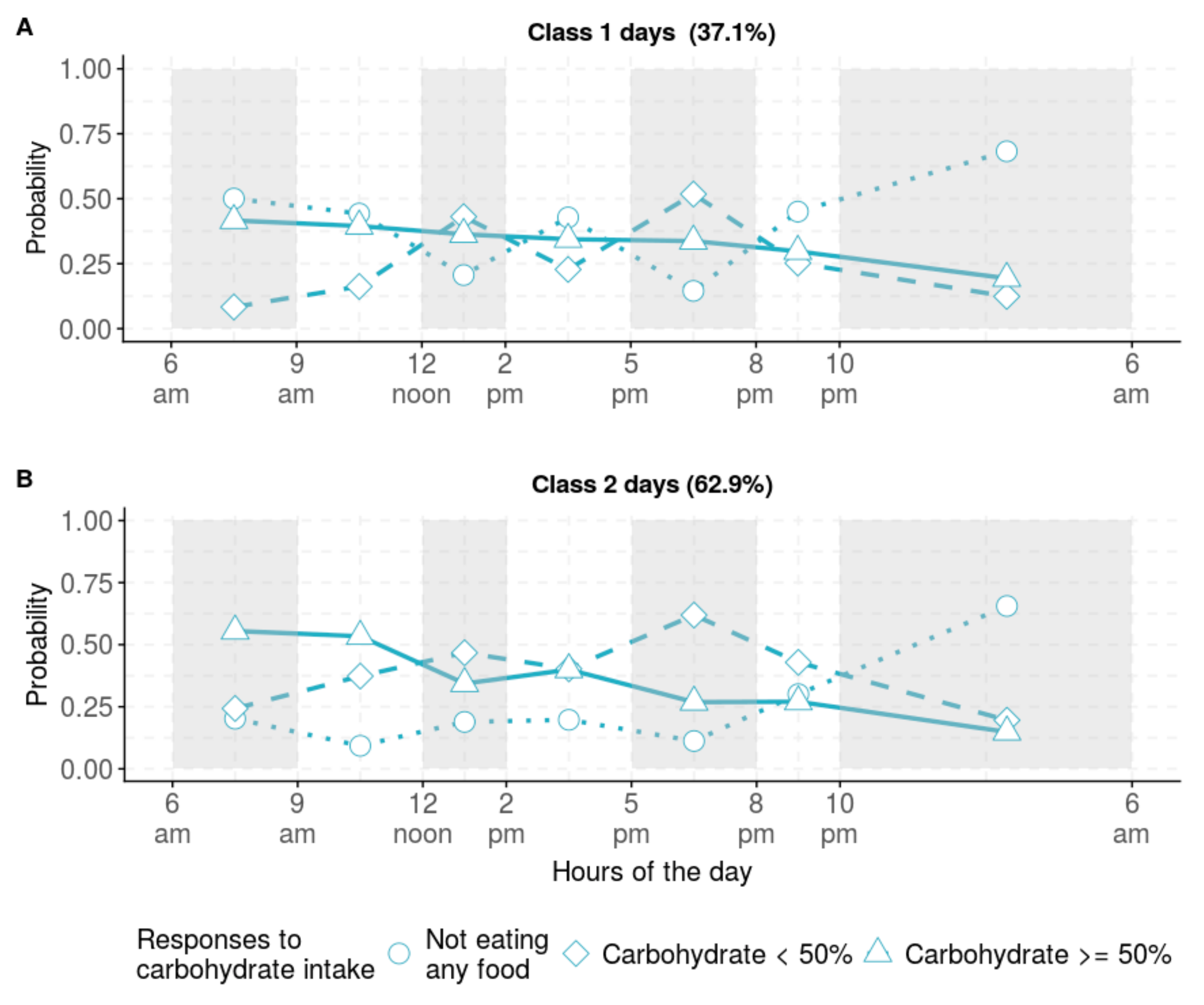
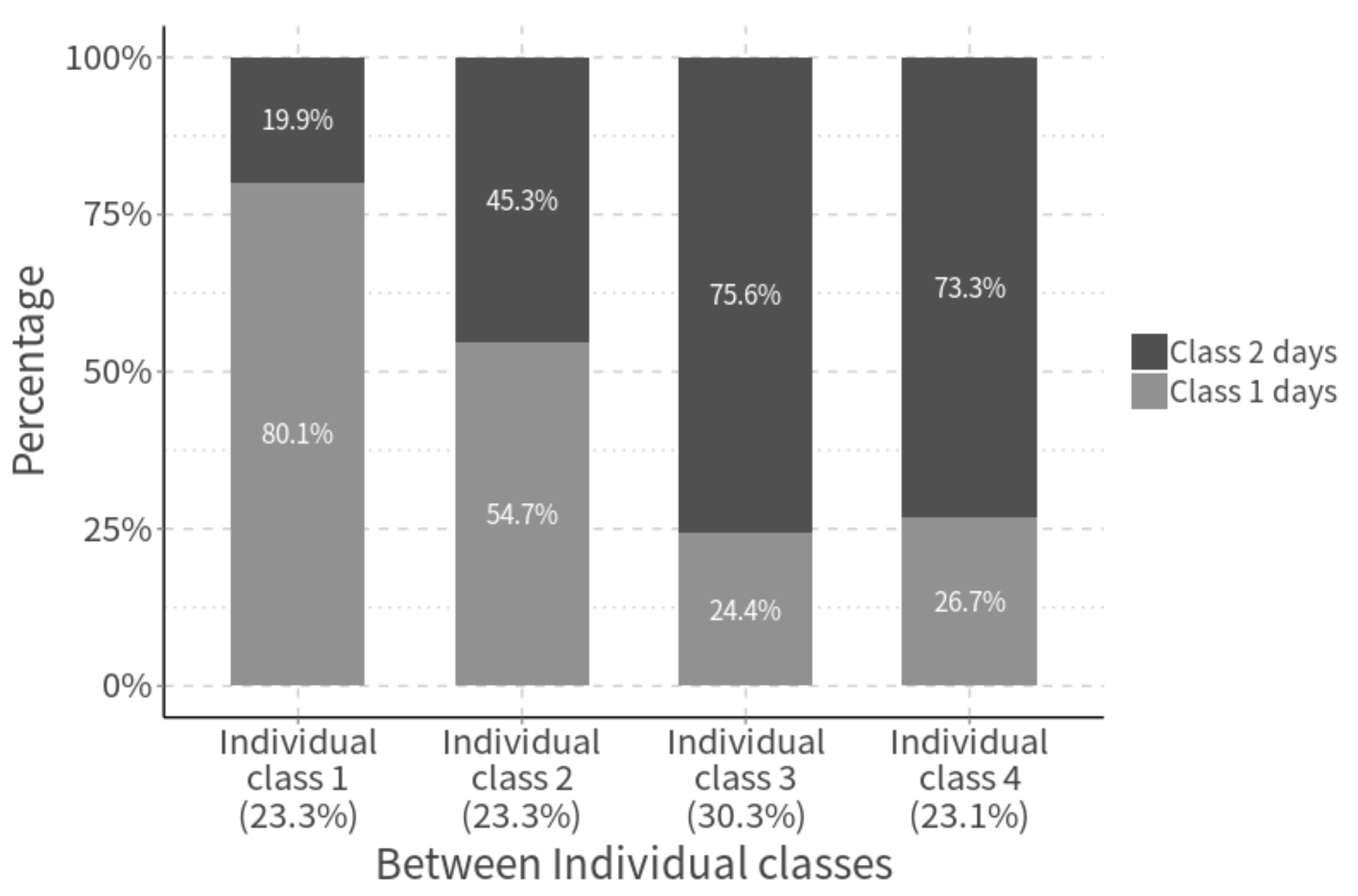
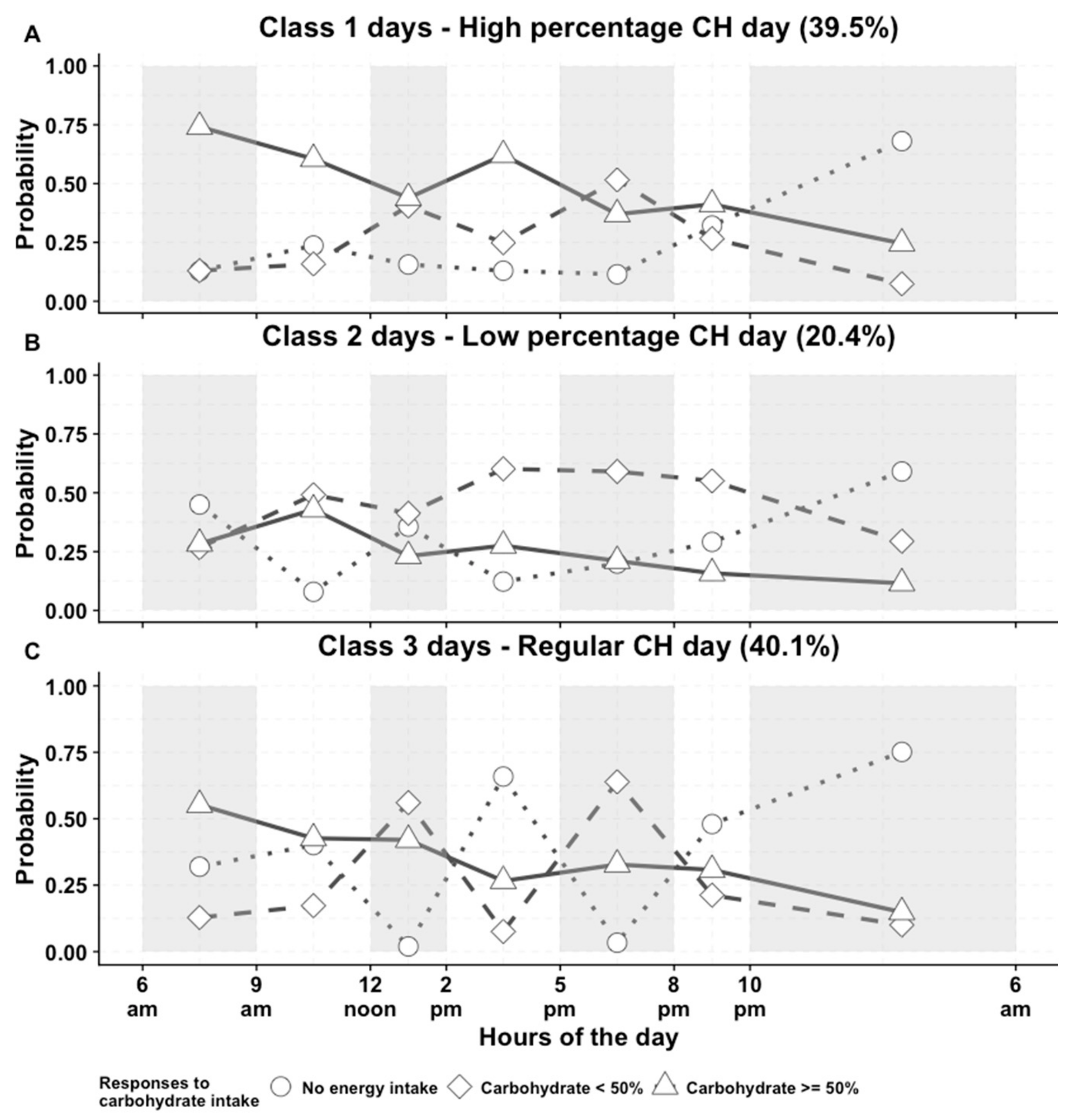
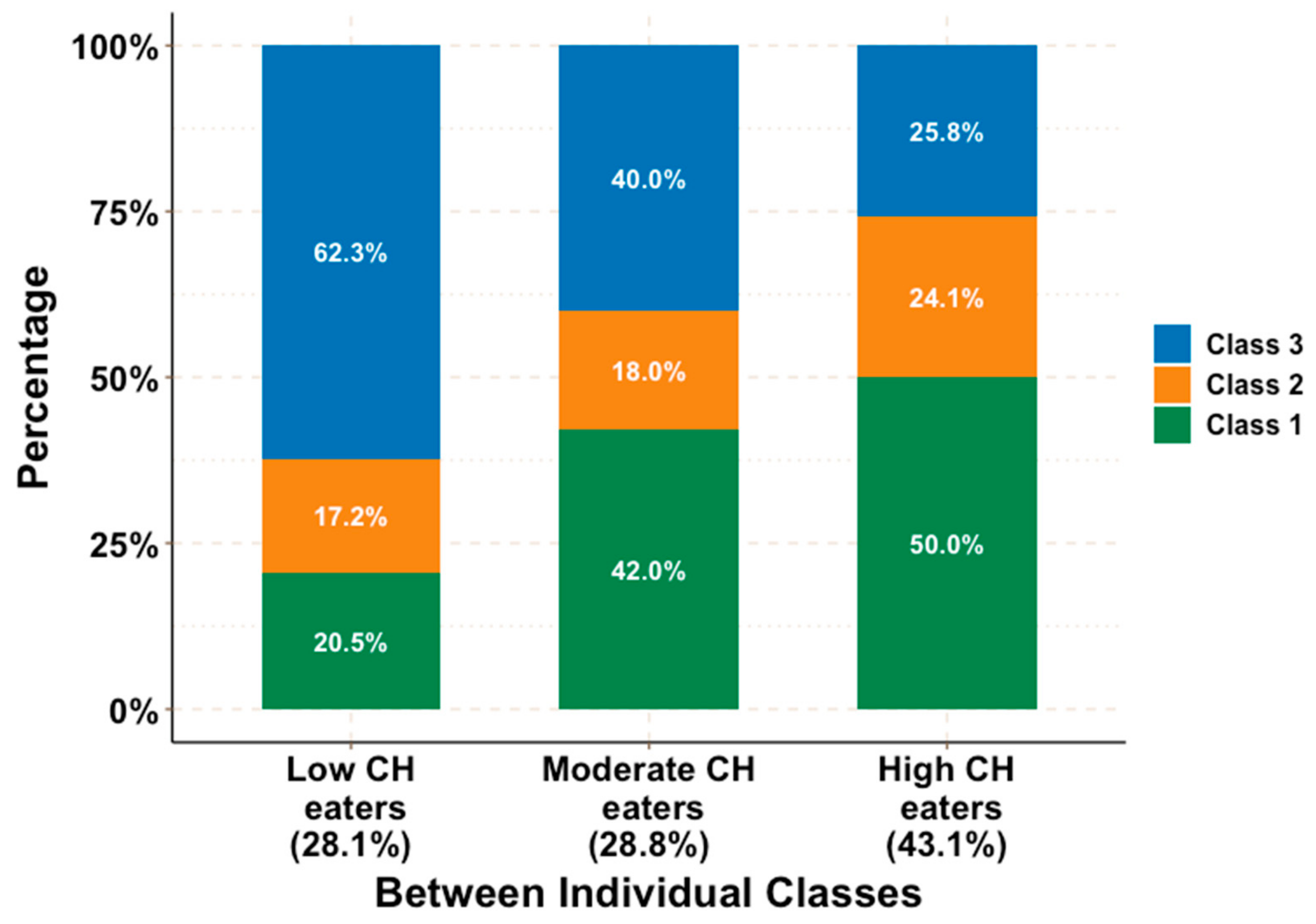
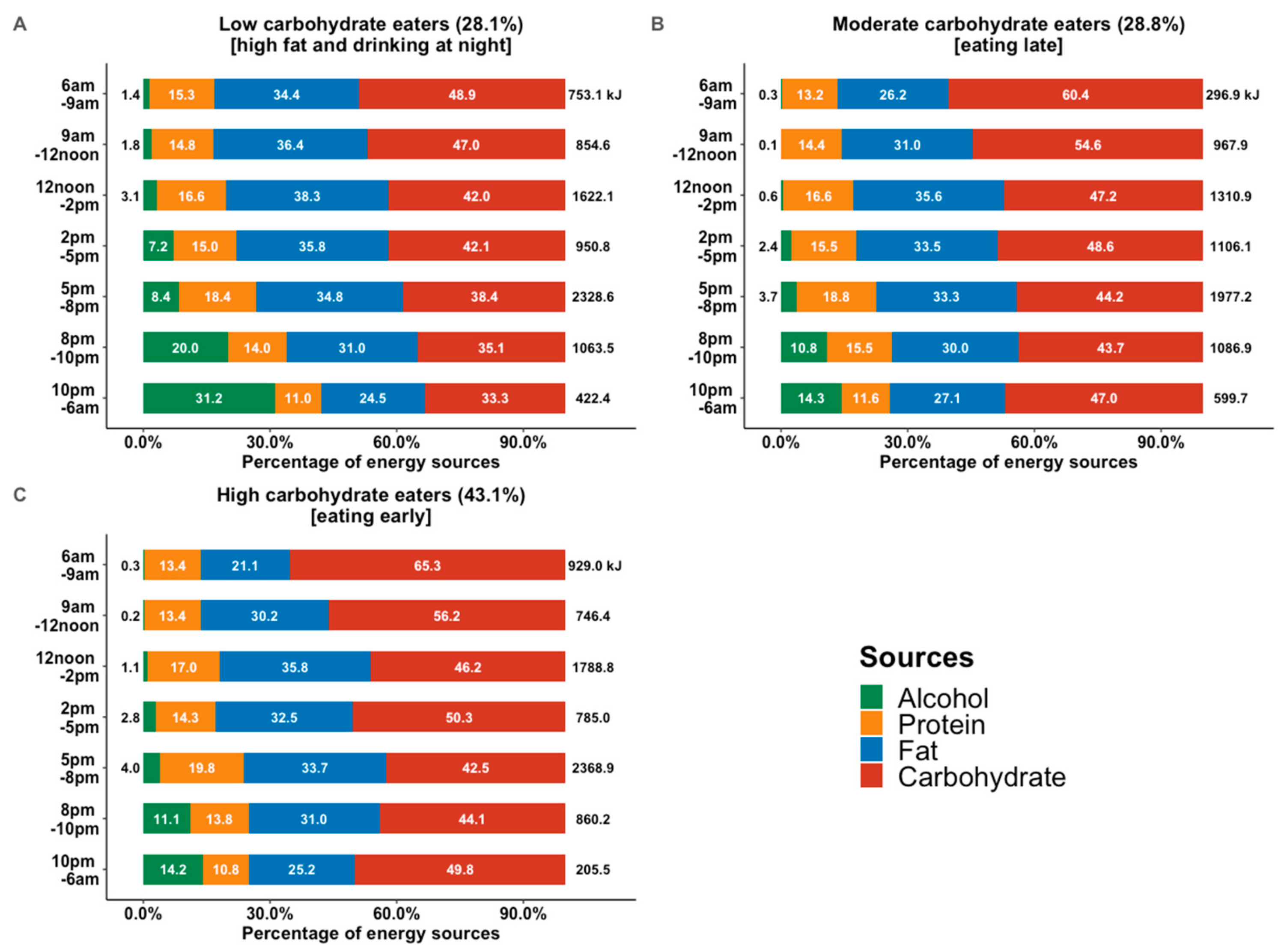
3.1. CH Eating Patterns in Day and Individual Levels
3.2. Characteristics of the three types of CH eaters
3.3. Association between the CH Eating Patterns and T2D
4. Discussion
Limitations and Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Mplus Example Codes for Multilevel Latent Class Models
| Mplus VERSION 8.3 (Linux) ! there are also Windows and Mac versions of the same program |
| MUTHEN & MUTHEN |
| 05/06/2019 5:09 PM |
| INPUT INSTRUCTIONS |
| TITLE: 3-class at level 1 (CW), 3-classes at level 2 (CB) random effects model - non-parametric approach ordered polytomous variables for CH intake at each time slot over four days of NDNS survey 2008/09 - 2015/16 |
| variable 0 = no energy intake |
| 1 = eating & carb provided <50% calorie |
| 2 = eating & carb provided ≥50% calorie |
| DATA: File is “../NDNS_Tslots.dat”; ! you will need to specify the path to your data |
| VARIABLE: NAMES = id id_dy Age Sex H6_9 H9_12 H12_14 H14_17 H17_20 |
| H20_22 H22_6; |
| USEVAR = H6_9 H9_12 H12_14 H14_17 H17_20 |
| H20_22 H22_6; |
| auxiliary = Age Sex; |
| CATEGORICAL = H6_9 H9_12 H12_14 H14_17 H17_20 |
| H20_22 H22_6; |
| CLUSTER = id; !days nested within individuals |
| IDVARIABLE = id_dy;! Id with day number |
| BETWEEN = CB; |
| WITHIN = H6_9 H9_12 H12_14 H14_17 H17_20 |
| H20_22 H22_6; ! time slot indicators for day level |
| CLASSES = CB(3) CW(3); |
| MISSING are .; |
| ANALYSIS: |
| type = twolevel mixture; |
| starts = 200 100; ! when increasing the n of classes, these numbers may need to be changed to larger ones |
| process = 8(starts); ! number of processors on your machine |
| MODEL: |
| %within% |
| %overall% |
| %between% |
| %overall% |
| CW ON CB; |
| Savedata: |
| file is “../NDNSslot_CW3CB3.txt”; ! you will need to specify the location to store the predicted latent class membership |
| save is cprob; |
| format is free; |
Appendix B
| Models | Number of Day Level Classes | ||||
|---|---|---|---|---|---|
| 1 Class | 2 Classes | 3 Classes | 4 Classes | 5 Classes | |
| Fixed Effects Model | |||||
| No. of free parameters | 14 | 29 | 44 | 59 | 74 |
| Log-likelihood | −173,793.306 | −172,669.771 | −172,039.204 | −171,633.941 | −171,377.292 |
| AIC | 347,614.612 | 345,397.542 | 344,166.407 | 343,385.883 | 342,902.585 |
| BIC | 347,728.092 | 345,632.608 | 344,523.06 | 343,864.121 | 343,502.409 |
| aBIC | 347,683.601 | 345,540.447 | 344,383.229 | 343,676.621 | 343,267.239 |
| Lo-Mendell-Rubun LRT | -- | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Entropy | 1 | 0.31 | 0.392 | 0.51 | 0.481 |
| Random Effects Model | |||||
| 2 individual level classes | |||||
| No. of free parameters | 59 | 89 | 119 | ||
| Log-likelihood | −169,331.132 | −168,700.96 | −168,366.193 | ||
| BIC | 339,258.502 | 338,301.338 | 337,934.968 | ||
| Entropy | 0.581 | 0.569 | 0.555 | ||
| 3 individual level classes | |||||
| No. of free parameters | 89 | 134 | 179 | ||
| Log-likelihood | −166,936.279 | −166,348.815 | −166,062.761 | ||
| BIC | 334,771.968 | 334,051.799 | 333,934.448 | ||
| Entropy | 0.677 | 0.63 | 0.644 | ||
| 4 individual level classes | |||||
| No. of free parameters | 119 | 179 | |||
| Log-likelihood | −165,441.731 | −164,845.696 | |||
| BIC | 332,086.045 | 331,500.318 | |||
| Entropy | 0.729 | 0.659 | |||
References
- Almoosawi, S.; Vingeliene, S.; Gachon, F.; Voortman, T.; Palla, L.; Johnston, J.D.; Van Dam, R.M.; Darimont, C.; Karagounis, L.G. Chronotype: Implications for Epidemiologic Studies on Chrono-Nutrition and Cardiometabolic Health. Adv. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- De Bacquer, D.; Van Risseghem, M.; Clays, E.; Kittel, F.; De Backer, G.; Braeckman, L. Rotating shift work and the metabolic syndrome: A prospective study. Int. J. Epidemiol. 2009, 38, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Schernhammer, E.S.; Sun, Q.; Hu, F.B. Rotating night shift work and risk of type 2 diabetes: Two prospective cohort studies in women. PLoS Med. 2011, 8, e1001141. [Google Scholar] [CrossRef] [PubMed]
- Leech, R.M.; Worsley, A.; Timperio, A.; McNaughton, S.A. Temporal eating patterns: A latent class analysis approach. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D. Physiological responses to food intake throughout the day. Nutr. Res. Rev. 2014, 27, 107–118. [Google Scholar] [CrossRef]
- Furutani, A.; Ikeda, Y.; Itokawa, M.; Nagahama, H.; Ohtsu, T.; Furutani, N.; Kamagata, M.; Yang, Z.H.; Hirasawa, A.; Tahara, Y.; et al. Fish Oil Accelerates Diet-Induced Entrainment of the Mouse Peripheral Clock via GPR120. PLoS ONE 2015, 10, e0132472. [Google Scholar] [CrossRef]
- Oike, H. Modulation of circadian clocks by nutrients and food factors. Biosci. Biotechnol. Biochem. 2017, 81, 863–870. [Google Scholar] [CrossRef]
- Almoosawi, S.; Prynne, C.J.; Hardy, R.; Stephen, A.M. Time-of-day of energy intake: Association with hypertension and blood pressure 10 years later in the 1946 British Birth Cohort. J. Hypertens. 2013, 31, 882–892. [Google Scholar] [CrossRef]
- Morgan, L.M.; Shi, J.W.; Hampton, S.M.; Frost, G. Effect of meal timing and glycaemic index on glucose control and insulin secretion in healthy volunteers. Br. J. Nutr. 2012, 108, 1286–1291. [Google Scholar] [CrossRef]
- Chang, C.R.; Francois, M.E.; Little, J.P. Restricting carbohydrates at breakfast is sufficient to reduce 24-hour exposure to postprandial hyperglycemia and improve glycemic variability. Am. J. Clin. Nutr. 2019, 109, 1302–1309. [Google Scholar] [CrossRef]
- Tsuchida, Y.; Hata, S.; Sone, Y. Effects of a late supper on digestion and the absorption of dietary carbohydrates in the following morning. J. Physiol. Anthropol. 2013, 32, 9. [Google Scholar] [CrossRef] [PubMed]
- MRC Elsie Widdowson Laboratory and NatCen Social Research. National Diet and Nutrition Survey Years 1–8, 2008/09–2015/16 [Data Collection], 11th ed.; UK Data Service: Colchester, UK, 2018. [Google Scholar]
- Bates, B.; Lennox, A.; Prentice, A.; Bates, C.J.; Page, P.; Nicholson, S.; Swan, G. National Diet and Nutrition Survey: Results from Years 1, 2, 3 and 4 (Combined) of the Rolling Programme (2008/2009–2011/2012): A Survey Carried Out on Behalf of Public Health England and the Food Standards Agency; Public Health England: London, UK, 2014. [Google Scholar]
- Roberts, C.; Steer, T.; Maplethorpe, N.; Cox, L.; Meadows, S.; Nicholson, S.; Page, P.; Swan, G. National Diet and Nutrition Survey: Results from Years 7 and 8 (Combined) of the Rolling Programme (2014/2015–2015/2016); Public Health England: London, UK, 2018. [Google Scholar]
- Fitt, E.; Cole, D.; Ziauddeen, N.; Pell, D.; Stickley, E.; Harvey, A.; Stephen, A.M. DINO (Diet In Nutrients Out)—An integrated dietary assessment system. Public Health Nutr. 2015, 18, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Smithers, G. MAFF’s nutrient databank. Nutr. Food. Sci. 1993, 93, 16–19. [Google Scholar] [CrossRef]
- Gregory, J.; Foster, K.; Tyler, H.; Wiseman, M. The Dietary and Nutritional Survey of British Adults; HMSO Publications Centre: London, UK, 1990. [Google Scholar]
- Laboratory, M.E.W.; Research, N.S. National Diet and Nutrition Survey Years 1–9, 2008/09–2016/17, 13th ed.; UK Data Service: Colchester, UK, 2019. [Google Scholar]
- SACN. Carbohydrates and Health Report; Public Health England: London, UK, 2015. [Google Scholar]
- Collins, L.M.; Lanza, S.T. Latent Class and Latent Transition Analysis: With Applications in the Social, Behavioral, and Health Sciences; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Finch, W.H.; Bolin, J.E. Multilevel Modeling Using Mplus; Chapman and Hall/CRC: Boca Raton, FL, USA, 2017. [Google Scholar]
- Henry, K.L.; Muthen, B. Multilevel Latent Class Analysis: An Application of Adolescent Smoking Typologies with Individual and Contextual Predictors. Struct. Equ. Model. 2010, 17, 193–215. [Google Scholar] [CrossRef] [PubMed]
- Muthen, B.; Asparouhov, T. Multilevel regression mixture analysis. J. R. Stat. Soc. 2009, 172, 639–657. [Google Scholar] [CrossRef]
- Finch, W.H.; French, B.F. Multilevel Latent Class Analysis: Parametric and Nonparametric Models. J. Exp. Educ. 2014, 82, 307–333. [Google Scholar] [CrossRef]
- Lo, Y.; Mendell, N.R.; Rubin, D.B. Testing the number of components in a normal mixture. Biometrika 2001, 88, 767–778. [Google Scholar] [CrossRef]
- Nylund, K.L.; Asparoutiov, T.; Muthen, B.O. Deciding on the number of classes in latent class analysis and growth mixture modeling: A Monte Carlo simulation study. Struct. Equ. Model. 2007, 14, 535–569. [Google Scholar] [CrossRef]
- Muthén, L.K.; Muthén, B.O. Mplus: Statistical Analysis with Latent Variables: User’s Guide; Muthén & Muthén: Los Angeles, CA, USA, 2017. [Google Scholar]
- Besson, H.; Brage, S.; Jakes, R.W.; Ekelund, U.; Wareham, N.J. Estimating physical activity energy expenditure, sedentary time, and physical activity intensity by self-report in adults. Am. J. Clin. Nutr. 2010, 91, 106–114. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 15; StataCorp LLC: College Station, TX, USA, 2017. [Google Scholar]
- Mansukhani, R.; Palla, L. Investigating eating time patterns in UK adults from The 2008–2012 National Diet and Nutrition Survey. Proc. Nutr. Soc. 2018, 77. [Google Scholar] [CrossRef]
- Noakes, T.D. Low-carbohydrate and high-fat intake can manage obesity and associated conditions: Occasional survey. S. Afr. Med. J. 2013, 103, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Naude, C.E.; Schoonees, A.; Senekal, M.; Young, T.; Garner, P.; Volmink, J. Low carbohydrate versus isoenergetic balanced diets for reducing weight and cardiovascular risk: A systematic review and meta-analysis. PLoS ONE 2014, 9, e100652. [Google Scholar] [CrossRef] [PubMed]
- Seidelmann, S.B.; Claggett, B.; Cheng, S.; Henglin, M.; Shah, A.; Steffen, L.M.; Folsom, A.R.; Rimm, E.B.; Willett, W.C.; Solomon, S.D. Dietary carbohydrate intake and mortality: A prospective cohort study and meta-analysis. Lancet Public Health 2018, 3, e419–e428. [Google Scholar] [CrossRef]
- Noto, H.; Goto, A.; Tsujimoto, T.; Noda, M. Low-carbohydrate diets and all-cause mortality: A systematic review and meta-analysis of observational studies. PLoS ONE 2013, 8, e55030. [Google Scholar] [CrossRef] [PubMed]
- Ala-Korpela, M. Data-driven subgrouping in epidemiology and medicine. Int. J. Epidemiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Andruff, H.; Carraro, N.; Thompson, A.; Gaudreau, P.; Louvet, B. Latent Class Growth Modelling: A Tutorial. Tutor. Quant. Methods. Psychol. 2009, 5, 11–24. [Google Scholar] [CrossRef]
- Davidian, M.; Fitzmaurice, G.; Molenberghs, G.; Verbeke, G. Growth mixture modeling: Analysis with non-Gaussian random effects. In Longitudinal Data Analysis; Chapman and Hall/CRC: Boca Raton, FL, USA, 2008; pp. 157–180. [Google Scholar]
- Jung, T.; Wickrama, K.A.S. An introduction to latent class growth analysis and growth mixture modeling. Soc. Personal. Psychol. Compass 2008, 2, 302–317. [Google Scholar] [CrossRef]
- Nagin, D.S. Group-Based Modeling of Development; Harvard University Press: Cambridge, MA, USA, 2005. [Google Scholar]
- Goodman, L.A. On the Assignment of Individuals to Latent Classes. Sociol. Methodol. 2007, 37, 1–22. [Google Scholar] [CrossRef]
- Bray, B.C.; Lanza, S.T.; Tan, X. Eliminating Bias in Classify-Analyze Approaches for Latent Class Analysis. Struct. Equ. Model. 2015, 22, 1–11. [Google Scholar] [CrossRef]
- Wang, C.P.; Brown, C.H.; Bandeen-Roche, K. Residual diagnostics for growth mixture models: Examining the impact of a preventive intervention on multiple trajectories of aggressive behavior. J. Am. Stat. Assoc. 2005, 100, 1054–1076. [Google Scholar] [CrossRef]
- Itokawa, M.; Hirao, A.; Nagahama, H.; Otsuka, M.; Ohtsu, T.; Furutani, N.; Hirao, K.; Hatta, T.; Shibata, S. Time-restricted feeding of rapidly digested starches causes stronger entrainment of the liver clock in PER2: LUCIFERASE knock-in mice. Nutr. Res. 2013, 33, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Previdelli, A.N.; de Andrade, S.C.; Fisberg, R.M.; Marchioni, D.M. Using Two Different Approaches to Assess Dietary Patterns: Hypothesis-Driven and Data-Driven Analysis. Nutrients 2016, 8, 593. [Google Scholar] [CrossRef] [PubMed]
| Total | LCE | MCE | HCE | |
|---|---|---|---|---|
| n | 6155 | 1730 | 1772 | 2653 |
| Total energy (kJ) | 7668.9 (7583.1, 7754.7) | 7985.8 (7823.3, 8146.3) | 7341.8 (7825.3, 8146.3) | 7677.8 (7555.8, 7799.8) |
| Protein (g) | 74.2 (73.3, 75.1) | 79.9 (77.9, 81.8) | 69.3 (67.6, 71.0) | 73.7 (72.5, 74.8) |
| Protein-% | 16.6 (16.5, 16.8) | 17.2 (16.9, 17.5) | 16.3 (16.0, 16.6) | 16.5 (16.3, 16.6) |
| Fat (g) | 67.7 (66.8, 68.6) | 74.7 (73.1, 76.4) | 63.8 (62.1, 65.5) | 65.7 (64.4, 67.0) |
| Fat- % | 33.1 (32.9, 33.4) | 35.4 (34.9, 35.8) | 32.5 (32.1, 32.9) | 32.0 (31.7, 32.3) |
| Alcohol (g) | 12.8 (11.9, 13.7) | 20.8 (18.3, 23.2) | 10.7 (9.4, 11.9) | 8.9 (8.1, 9.8) |
| Alcohol- % | 4.4 (4.1, 4.6) | 6.8 (6.3, 7.4) | 3.8 (3.4, 4.3) | 3.2 (2.8, 3.4) |
| Carbohydrate (g) | 220.7 (218.2, 223.2) | 203.8 (199.8, 207.8) | 218.3 (212.9, 223.7) | 233.4 (229.6, 237.2) |
| Carbohydrate- % | 45.8 (45.6, 46.0) | 40.6 (40.2, 41.0) | 47.3 (46.8, 47.8) | 48.3 (47.9, 48.6) |
| Total fiber (g) | 13.8 (13.6, 13.9) | 13.7 (13.4, 14.0) | 12.5 (12.1, 12.9) | 14.7 (14.4, 14.9) |
| Total sugar (g) | 95.2 (93.6, 96.7) | 83.4 (81.1, 85.7) | 92.2 (88.9, 95.6) | 104.9 (102.5, 107.3) |
| Total NME sugar (g) | 56.9 (55.5, 58.3) | 46.1 (44.1, 48.0) | 60.7 (57.5, 63.8) | 61.5 (59.4, 63.6) |
| Total starch (g) | 125.4 (123.9, 126.9) | 120.3 (117.7, 122.9) | 125.9 (122.8, 129.2) | 128.4 (126.2, 130.6) |
| 6 am–9 a.m. | 26.1 (25.3. 26.9) | 23.0 (21.8, 24.3) | 11.2 (10.0, 12.3) | 37.9 (36.8, 39.2) |
| Fiber (g) | 1.4 (1.3, 1.5) | 1.4 (1.3, 1.5) | 0.6 (0.5, 0.7) | 2.0 (1.9, 2.2) |
| Sugar (g) | 12.9 (12.4, 13.3) | 10.2 (9.6, 10.9) | 5.3 (4.8, 5.8) | 19.7 (19.0, 20.4) |
| NME sugar (g) | 7.1 (6.8, 7.3) | 4.7 (4.3, 5.1) | 3.2 (2.9, 3.6) | 11.1 (10.7, 11.6) |
| Starch (g) | 13.2 (12.7, 13.6) | 12.8 (12.0, 13.5) | 5.9 (5.1, 6.6) | 18.3 (17.6, 19.1) |
| 9 am–12 noon | 27.8 (27.1, 28.6) | 25.1 (23.9, 26.3) | 33.0 (31.4, 34.6) | 26.2 (25.1, 27.2) |
| Fiber (g) | 1.4 (1.3, 1.5) | 1.5 (1.4, 1.6) | 1.6 (1.5, 1.7) | 1.3 (1.2, 1.3) |
| Sugar (g) | 13.9 (13.5, 14.3) | 11.6 (10.9, 12.3) | 15.7 (14.8, 16.6) | 14.2 (13.6, 14.8) |
| NME sugar (g) | 7.8 (7.5, 8.1) | 5.7 (5.2, 6.2) | 9.6 (8.9, 10.2) | 8.1 (7.7, 8.5) |
| Starch (g) | 13.9 (13.5, 14.4) | 13.5 (12.8, 14.3) | 17.3 (16.4, 18.3) | 11.9 (11.3, 12.6) |
| 12 noon–2 p.m. | 45.3 (44.4, 46.2) | 42.6 (40.9, 44.3) | 38.7 (37.0, 40.4) | 51.6 (50.2, 52.9) |
| Fiber (g) | 3.1 (3.0, 3.2) | 3.1 (2.9, 3.2) | 2.3 (2.2, 2.5) | 3.6 (3.5, 3.7) |
| Sugar (g) | 16.8 (16.3, 17.2) | 14.7 (14.0, 15.4) | 14.9 (14.0, 15.7) | 19.4 (18.7, 20.0) |
| NME sugar (g) | 9.1 (8.8, 9.4) | 7.3 (6.7, 7.8) | 9.1 (8.4, 9.8) | 10.3 (9.8, 10.8) |
| Starch (g) | 28.5 (27.9, 29.2) | 27.9 (26.6, 29.1) | 23,8 (22.6, 24.9) | 32.2 (31.2, 33.1) |
| 2 pm–5 p.m. | 27.3 (26.5, 28.2) | 25.0 (23.6, 26.4) | 33.6 (31.6, 35.6) | 24.7 (23.6, 25.7) |
| Fiber (g) | 1.5 (1.4, 1.6) | 1.6 (1.5, 1.7) | 1.9 (1.7, 2.0) | 1.3 (1.2, 1.4) |
| Sugar (g) | 13.3 (12.9, 13.7) | 11.9 (11.3, 12.7) | 14.5 (13.5, 15.5) | 13.4 (12.8, 13.9) |
| NME sugar (g) | 8.5 (8.2, 8.9) | 6.9 (6.4, 7.5) | 9.9 (9.0, 8.6) | 8.6 (8.2, 9.1) |
| Starch (g) | 14.0 (13.3, 14.6) | 13.1 (12.1, 13.9) | 19.1 (17.7, 20.4) | 11.3 (10.6, 11.9) |
| 5 p.m.–8 p.m. | 58.5 (57.4, 59.7) | 55.9 (54.1, 57.9) | 54.6 (52.1, 57.0) | 62.9 (61.3, 64.4) |
| Fiber (g) | 4.4 (4.3, 4.5) | 4.4 (4.2, 4.5) | 3.7 (3.5, 3.9) | 4.9 (4.7, 5.0) |
| Sugar (g) | 19.9 (19.5, 20.5) | 18.7 (17.9, 19.5) | 18.6 (17.6, 19.5) | 21.8 (20.9, 22.5) |
| NME sugar (g) | 11.4 (11.0, 11.9) | 10.2 (9.6, 10.8) | 11.8 (10.9, 12.6) | 12.1 (11.4, 12.7) |
| Starch (g) | 38.5 (37.7, 39.4) | 37.3 (35.8, 38.8) | 35.9 (34.1, 37.9) | 41.1 (39.9, 42.2) |
| 8 pm–10 p.m. | 25.3 (24.4, 26.2) | 23.3 (21.9, 24.6) | 29.7 (27.6, 31.7) | 23.7 (22.5, 24.9) |
| Fiber (g) | 1.4 (1.3, 1.5) | 1.4 (1.3, 1.6) | 1.6 (1.5, 1.8) | 1.3 (1.5, 1.8) |
| Sugar (g) | 12.2 (11.8, 12.6) | 10.9 (10.3, 11.5) | 13.2 (12.2, 14.2) | 12.4 (11.8, 13.0) |
| NME sugar (g) | 8.4 (7.9, 8.7) | 7.3 (6.8, 7.8) | 9.4 (8.5, 10.4) | 8.3 (7.8, 8.8) |
| Starch (g) | 13.1 (12.5. 13.7) | 12.3 (11.4, 13.3) | 16.4 (15.0, 17.8) | 11.3 (10.5, 12.1) |
| 10 p.m.–6 a.m. | 10.3 (9.5, 11.1) | 8.8 (7.7, 9.8) | 17.6 (15.2, 19.9) | 6.4 (5.8, 7.1) |
| Fiber (g) | 0.41 (0.37, 0.45) | 0.34 (0.29, 0.39) | 0.74 (0.63, 0.85) | 0.24 (0.21, 0.27) |
| Sugar (g) | 6.2 (5.7, 6.7) | 5.3 (4.6, 6.1) | 10.0 (8.6, 11.5) | 4.1 (3.7, 4.5) |
| NME sugar (g) | 4.6 (4.1, 5.0) | 3.9 (3.3, 4.6) | 7.7 (6.4, 8.9) | 2.9 (2.6, 3.3) |
| Starch (g) | 4.1 (3.7, 4.6) | 3.5 (2.9, 3.9) | 7.5 (6.3, 8.8) | 2.3 (1.9, 2.7) |
| LCE | MCE | HCE | ||
|---|---|---|---|---|
| (n = 1730) | (n = 1772) | (n = 2653) | p-Value 1 | |
| Total 2 (%) | 28.3 (26.9, 29.9) | 28.7 (27.1, 30.3) | 43.0 (41.3, 44.7) | |
| Age (years) | 51.0 (49.9, 52.1) | 40.3 (39.1, 41.6) | 51.7 (50.7, 52.7) | <0.001 |
| Sex (%) | 0.119 | |||
| Men | 50.0 (46.9, 53.1) | 50.2 (47.0, 53.5) | 46.6 (44.0, 49.1) | |
| Women | 50.0 (46.9, 53.1) | 49.8 (46.5, 53.0) | 53.4 (50.9, 56.0) | |
| Survey years (% in rows) | 0.015 | |||
| 1 | 32.5 (28.4, 36.9) | 26.3 (21.9, 31.2) | 41.2 (36.6, 46.0) | |
| 2 | 26.8 (22.6, 31.3) | 22.6 (18.6, 27.3) | 50.6 (45.8, 55.4) | |
| 3 | 22.6 (18.8, 26.9) | 33.7 (28.6, 39.2) | 43.6 (38.7, 48.7) | |
| 4 | 27.9 (24.1, 32.2) | 27.6 (23.8, 31.8) | 44.4 (40.2, 48.7) | |
| 5 | 27.9 (24.2, 32.0) | 28.7 (24.4, 33.5) | 43.3 (38.2, 48.6) | |
| 6 | 28.0 (24.0, 32.4) | 31.5 (26.9, 36.6) | 40.5 (35.8, 45.3) | |
| 7 | 29.1 (25.2, 33.4) | 29.0 (24.5, 34.0) | 41.8 (37.1, 46.7) | |
| 8 | 31.1 (27.3, 35.3) | 30.5 (25.9, 35.5) | 38.4 (34.1, 42.8) | |
| Paid employment 3, Yes (%) | 40.3 (37.0, 43.6) | 40.8 (37.1, 44.5) | 39.8 (37.1, 42.6) | 0.907 |
| Live with partner 4, Yes (%) | 56.9 (53.6, 60.1) | 38.4 (35.2, 41.8) | 61.3 (58.7, 63.7) | <0.001 |
| Household Income, GBP/year | 36,558.5 (34,800.2, 38316.8) | 27,180.8 (25,597.9, 28763.7) | 32,171.6 (31,024.9, 33318.2) | <0.001 |
| Ethnicity (%) | <0.001 | |||
| White | 94.2 (92.4, 95.6) | 79.5 (76.4, 82.3) | 91.9 (90.1, 93.4) | |
| Non-White | 5.8 (4.4, 7.6) | 20.5 (17.7, 23.6) | 8.1 (6.6, 9.9) | |
| Education (%) | 0.019 | |||
| Degree or higher | 29.0 (26.1, 32.1) | 23.3 (20.5, 26.3) | 26.2 (24.1, 28.5) | |
| Lower than degree | 71.0 (67.9, 73.9) | 76.7 (73.7, 79.5) | 73.8 (71.5, 75.9) | |
| BMI (kg/m2) | 27.8 (27.4, 28.2) | 27.2 (26.7, 27.7) | 27.3 (26.9, 27.6) | 0.006 |
| WC (cm) | ||||
| Men | 98.9 (97.4, 100.5) | 95.9 (94.1, 97.8) | 98.1 (96.9, 99.2) | 0.056 |
| Women | 89.9 (88.7, 91.3) | 88.7 (87.1, 90.3) | 87.2 (86.1, 88.2) | 0.004 |
| Physical Activity 5 (hours/day) | 0.73 (0.68, 0.79) | 0.62 (0.57, 0.68) | 0.73 (0.68, 0.77) | 0.005 |
| Smoking status (%) | <0.001 | |||
| Current | 20.4 (18.0, 23.0) | 27.8 (25.0, 30.9) | 17.1 (15.4, 19.0) | |
| Ex-smoker | 29.3 (26.5, 32.2) | 16.8 (14.6, 19.2) | 26.1 (24.9, 28.3) | |
| Never | 50.3 (47.2, 32.2) | 55.4 (52.2, 58.6) | 56.8 (54.3, 59.3) |
| Outcomes | LCE | MCE | HCE | ||
|---|---|---|---|---|---|
| (n = 1730) | p-Value 1 | (n = 1772) | p-Value 1 | (n = 2653) | |
| T2D (Self-reported, Yes)-Adjusted by Individual Weightings | |||||
| n | 85 | 55 | 72 | ||
| % 2 | 10.6 (8.1, 13.8) | 7.2 (5.1, 10.0) | 5.8 (4.3, 7.7) | ||
| Crude model | 1.94 (1.28, 2.95) | 0.002 | 1.26 (0.80, 2.00) | 0.321 | 1 |
| Adjusted Model 3 | 2.33 (1.48. 3.69) | < 0.001 | 1.40 (0.83, 2.34) | 0.203 | 1 |
| Adjusted Model 4 | 2.39 (1.51, 3.77) | < 0.001 | 1.42 (0.85, 2.37) | 0.181 | 1 |
| T2D (HbA1c ≥6.5% or Glucose ≥7 mmol/L, or Self-Reported)-Adjusted by Blood Weightings | |||||
| N | 107 | 77 | 121 | ||
| % 2 | 12.2 (9.5, 15.4) | 9.0 (6.6, 12.0) | 8.5 (6.7, 10.6) | ||
| Crude model | 1.49 (1.04, 2.15) | 0.030 | 1.06 (0.73, 1.60) | 0.780 | 1 |
| Adjusted Model 3 | 1.66 (1.09, 2.52) | 0.017 | 1.23 (0.78, 1.95) | 0.375 | 1 |
| Adjusted Model 4 | 1.67 (1.10, 2.52) | 0.016 | 1.22 (0.77, 1.93) | 0.407 | 1 |
| Undiagnosed T2D (HbA1c ≥6.5% or Glucose ≥7 mmol/L but Self-Reported No Diabetes)-Adjusted by Blood Weightings. Self-Reported T2D Cases Excluded. | |||||
| n | 22 | 22 | 49 | ||
| % 2 | 2.8 (1.8, 4.6) | 3.1 (1.7, 5.6) | 4.2 (2.9, 5.9) | ||
| Crude model | 0.71 (0.38, 1.33) | 0.280 | 0.75 (0.38, 1.33) | 0.420 | 1 |
| Adjusted Model 3 | 0.76 (0.39, 1.50) | 0.433 | 0.95 (0.45, 1.99) | 0.885 | 1 |
| Adjusted Model 4 | 0.69 (0.35, 1.35) | 0.280 | 0.85 (0.39, 1.86) | 0.688 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Almoosawi, S.; Palla, L. Day-Time Patterns of Carbohydrate Intake in Adults by Non-Parametric Multi-Level Latent Class Analysis—Results from the UK National Diet and Nutrition Survey (2008/09–2015/16). Nutrients 2019, 11, 2476. https://doi.org/10.3390/nu11102476
Wang C, Almoosawi S, Palla L. Day-Time Patterns of Carbohydrate Intake in Adults by Non-Parametric Multi-Level Latent Class Analysis—Results from the UK National Diet and Nutrition Survey (2008/09–2015/16). Nutrients. 2019; 11(10):2476. https://doi.org/10.3390/nu11102476
Chicago/Turabian StyleWang, Chaochen, Suzana Almoosawi, and Luigi Palla. 2019. "Day-Time Patterns of Carbohydrate Intake in Adults by Non-Parametric Multi-Level Latent Class Analysis—Results from the UK National Diet and Nutrition Survey (2008/09–2015/16)" Nutrients 11, no. 10: 2476. https://doi.org/10.3390/nu11102476
APA StyleWang, C., Almoosawi, S., & Palla, L. (2019). Day-Time Patterns of Carbohydrate Intake in Adults by Non-Parametric Multi-Level Latent Class Analysis—Results from the UK National Diet and Nutrition Survey (2008/09–2015/16). Nutrients, 11(10), 2476. https://doi.org/10.3390/nu11102476





