Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand
Abstract
1. Introduction
2. The Problem with the Available Supply of n-3 LC-PUFA
2.1. Gap between Supply and Demand
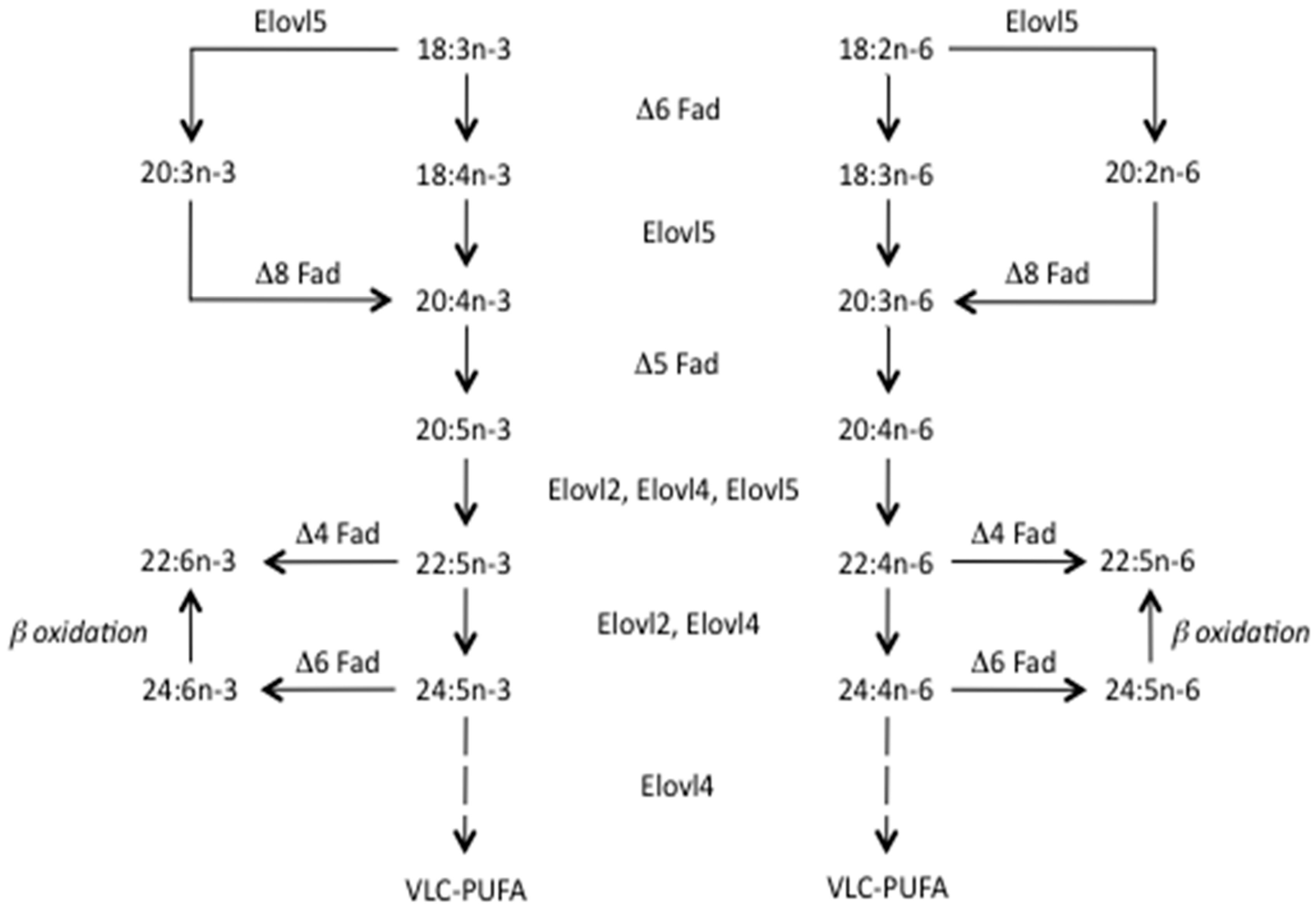
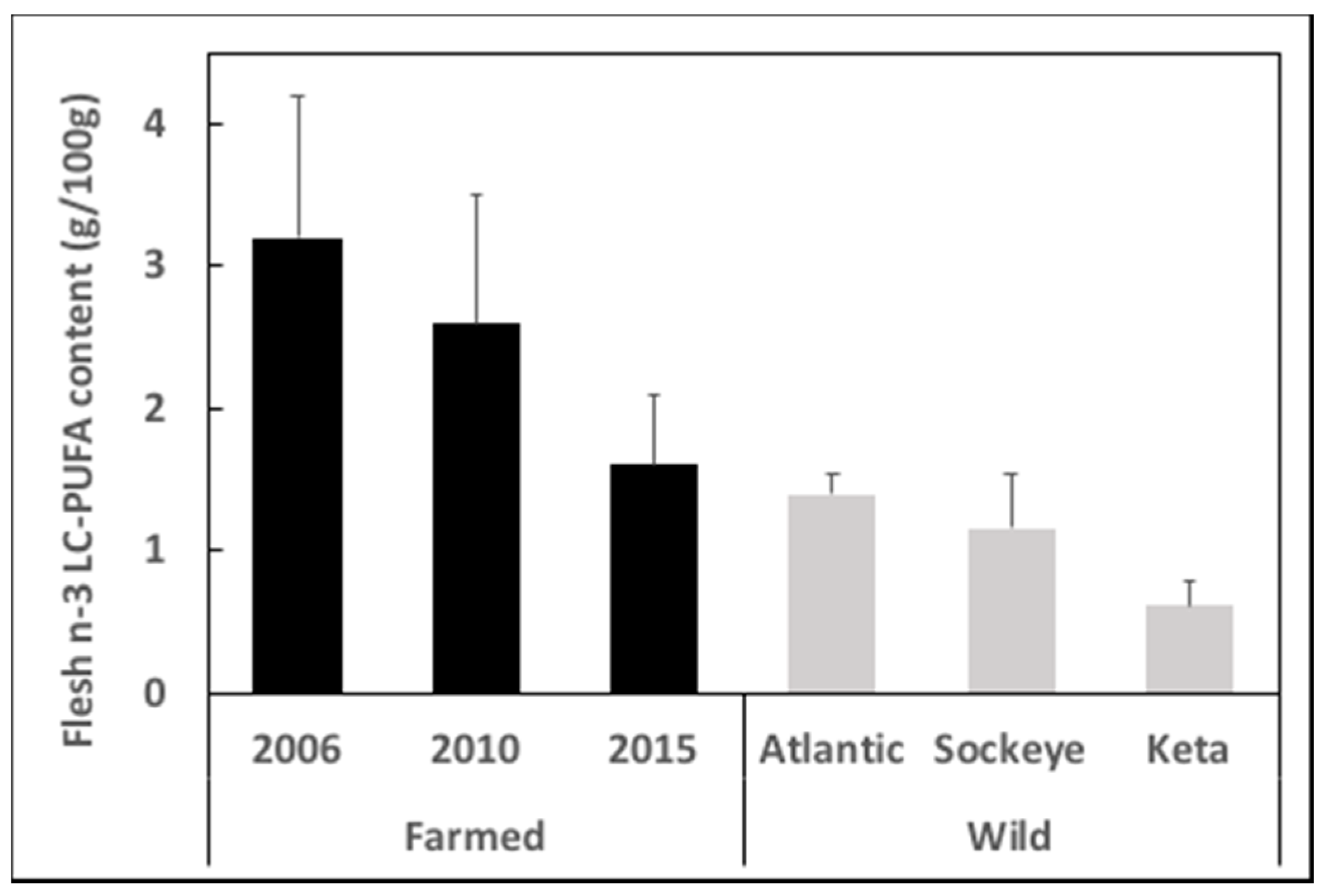
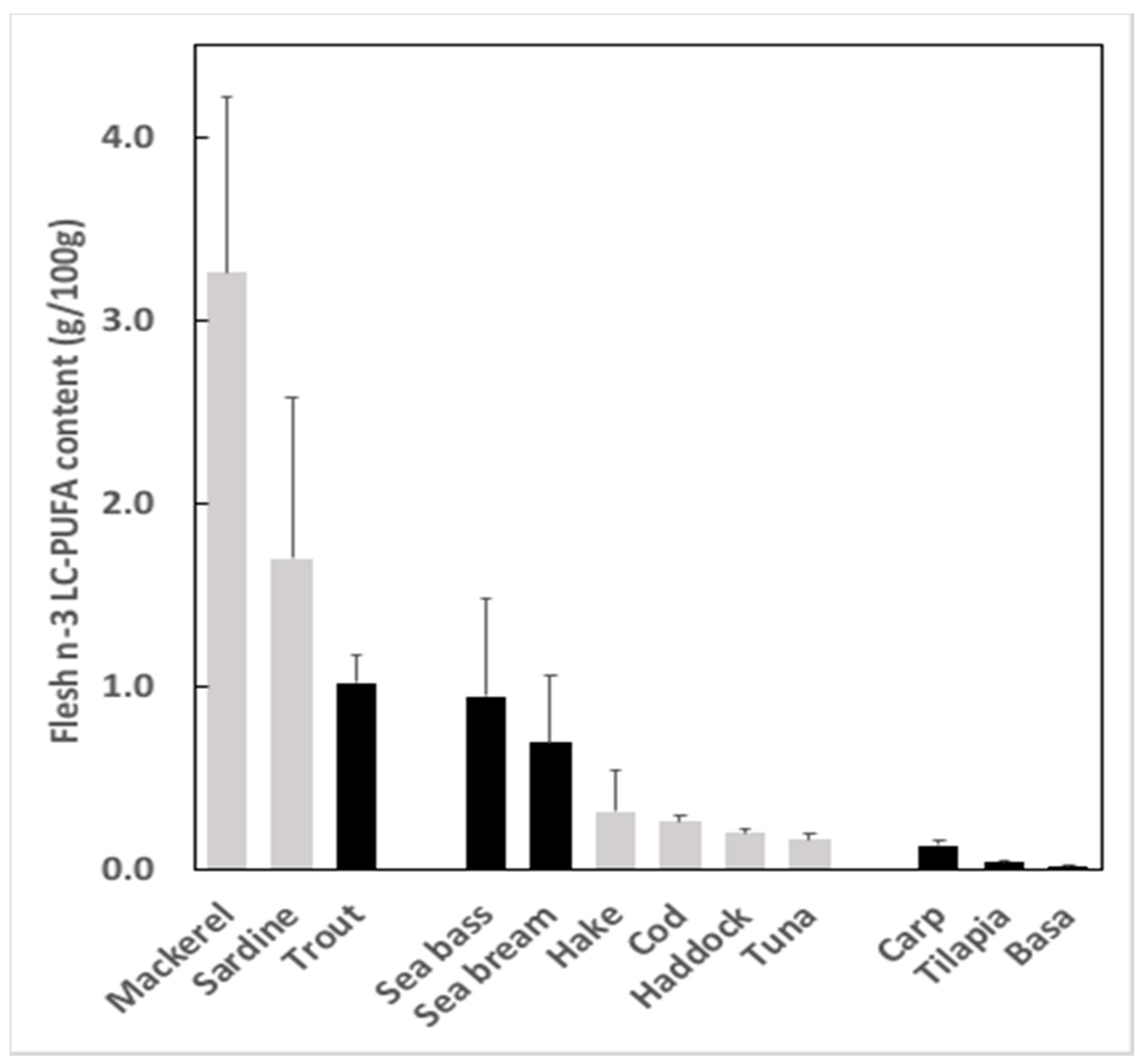
2.2. Why Is There a Gap and What Impact Does It Have?
3. The Roles of Aquaculture
4. The Fundamental Solution
5. Alternative New Sources of EPA and DHA
6. Can these Products be Alternatives to Fish Oil in Aquaculture?
7. Current Use in Commercial Aquafeeds and other Commercial Products
8. The Future
8.1. Use in Aquafeeds to Maintain Fish Growth and Health
8.2. Use in Aquafeeds to Support Human Health
8.3. Different Products, Different Niches?
8.4. Non-Aquaculture Uses
9. Conclusions
Funding
Conflicts of Interest
References
- Castro, L.F.C.; Tocher, D.R.; Monroig, Ó. Long-chain polyunsaturated fatty acid biosynthesis in chordates: Insights into the evolution of fads and elovl gene repertoire. Prog. Lipid Res. 2016, 62, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Tocher, D.R. Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 2015, 449, 94–107. [Google Scholar] [CrossRef]
- Innis, S.M. Dietary (n-3) fatty acids and brain development. J. Nutr. 2007, 137, 855–859. [Google Scholar] [CrossRef] [PubMed]
- Campoy, C.; Escolano-Margarit, V.; Anjos, T.; Szajewska, H.; Uauy, R. Omega 3 fatty acids on child growth, visual acuity and neurodevelopment. Br. J. Nutr. 2012, 107, S85–S106. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Lista, J.; Perez-Martinez, P.; Lopez-Miranda, J.; Perez-Jimenez, F. Long chain omega-3 fatty acids and cardiovascular disease: A systematic review. Br. J. Nutr. 2012, 107, S201–S213. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Very long chain omega-3 (n-3) fatty acids and human health. Eur. J. Lipid Sci. Technol. 2014, 116, 1280–1300. [Google Scholar] [CrossRef]
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018, 77, 52–72. [Google Scholar] [CrossRef] [PubMed]
- Naylor, R.L.; Hardy, R.W.; Bureau, D.P.; Chiu, A.; Elliot, M.; Farrell, A.P.; Forster, I.; Gatlin, D.M.; Goldburg, R.J.; Hua, K.; et al. Feeding aquaculture in an era of finite resources. Proc. Natl. Acad. Sci. USA 2009, 106, 15103–15110. [Google Scholar] [CrossRef]
- Sprague, M.; Betancor, M.B.; Tocher, D.R. Microbial and genetically engineered oils as replacements for fish oil in aquaculture feeds. Biotechnol. Lett. 2017, 39, 1599–1609. [Google Scholar] [CrossRef]
- Gil, A.; Serra-Majem, L.; Calder, P.C.; Uauy, R. Systematic reviews of the role of omega-3 fatty acids in the prevention and treatment of disease. Br. J. Nutr. 2012, 107, S1–S2. [Google Scholar] [CrossRef]
- Poudyal, H.; Panchal, S.K.; Diwan, V.; Brown, L. Omega-3 fatty acids and metabolic syndrome: Effects and emerging mechanisms of action. Prog. Lipid Res. 2011, 50, 372–387. [Google Scholar] [CrossRef] [PubMed]
- Aranceta, J.; Pérez-Rodrigo, C. Recommended dietary reference intakes, nutritional goals and dietary guidelines for fat and fatty acids: A systematic review. Br. J. Nutr. 2012, 107, S8–S22. [Google Scholar] [CrossRef]
- Richter, C.K.; Skulas-Ray, A.C.; Kris-Etherton, P.M. Recommended intake of fish and fish oils worldwide. In Fish and Fish Oil in Health and Disease Prevention; Raatz, S.K., Bibus, D.M., Eds.; Academic Press, Elsevier: New York, NY, USA, 2016; pp. 27–48. [Google Scholar]
- Global Recommendations for EPA and DHA Intake (Rev. 19 November 2014). Available online: www.goedomega3.com/index.php/files/download/304 (accessed on 30 September 2018).
- Salem, N., Jr.; Eggersdorfer, M. Is the world supply of omega-3 fatty acids adequate for optimal human nutrition? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Deckelbaum, R.J.; Calder, P.C. From where will all the omega-3 fatty acids come? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 111–112. [Google Scholar] [CrossRef] [PubMed]
- Kabeya, N.; Fonseca, M.M.; Ferrier, D.E.K.; Navarro, J.C.; Bay, L.K.; Francis, D.S.; Tocher, D.R.; Castro, L.F.C.; Monroig, Ó. Genes for de novo biosynthesis of omega-3 polyunsaturated fatty acids are widespread in animals. Sci. Adv. 2018, 4, eaar6849. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L.; Gushina, I.A. The versatility of algae and their lipid metabolism. Biochimie 2009, 91, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L. Recent advances in the biochemistry of plant fatty acids. Biochim. Biophys. Acta 1996, 1301, 7–56. [Google Scholar] [CrossRef]
- Givens, D.I.; Gibbs, R.A. Very long chain n-3 polyunsaturated fatty acids in the food chain in the UK and the potential of animal-derived foods to increase intake. Nutr. Bull. 2006, 31, 104–110. [Google Scholar] [CrossRef]
- Givens, D.I.; Gibbs, R.A. Current intakes of EPA and DHA in European populations and the potential of animal-derived foods to increase them. Proc. Nutr. Soc. 2008, 67, 273–280. [Google Scholar] [CrossRef]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef]
- Rosell, M.S.; Lloyd-Wright, Z.; Appleby, P.N.; Sanders, T.A.; Allen, N.E.; Key, T.J. Long-chain n-3 polyunsaturated fatty acids in plasma in British meat-eating, vegetarian, and vegan men. Am. J. Clin. Nutr. 2005, 82, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Tan, S.Y.; Henry, C.J. Long-chain n-3 PUFA in vegetarian women: A metabolic perspective. J. Nutr. Sci. 2017, 6, e58. [Google Scholar] [CrossRef]
- Micha, R.; Khatibzadeh, S.; Shi, P.; Fahimi, S.; Lim, S.; Andrews, K.G.; Engell, R.E.; Powles, J.; Ezzati, M.; Mozaffarian, D. Global, regional, and national consumption levels of dietary fats and oils in 1990 and 2010: A systematic analysis including 266 country-specific nutrition surveys. Br. Med. J. 2014, 348, g2272. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organisation (FAO). The State of the World Fisheries and Aquaculture 2016. Contributing to Food Security and Nutrition for All; FAO: Rome, Italy, 2016; 200p, ISBN 978-92-5-109185-2. [Google Scholar]
- The Continuing Importance of Fishmeal and Fish Oil in Aquafeeds. Available online: http://www.iffo.net/system/files/AquaFarm%20Feb18%20NA.pdf (accessed on 5 November 2018).
- Members of Global Salmon Initiative Announce Tender for Novel Oil Sources Rich in Omega-3. Available online: http://globalsalmoninitiative.org/en/news/members-of-global-salmon-initiative-announce-tender-for-novel-oil-sources-rich-in-omega-3/ (accessed on 30 September 2018).
- Turchini, G.M.; Ng, W.K.; Tocher, D.R. (Eds.) Fish Oil Replacement and Alternative Lipid Sources in Aquaculture Feeds; Taylor & Francis, CRC Press: Boca Raton, FL, USA, 2011; p. 533. ISBN 9781439808627. [Google Scholar]
- Ytrestøyl, T.; Aas, T.S.; Åsgård, T. Utilisation of feed resources in production of Atlantic salmon (Salmo salar). Aquaculture 2015, 448, 365–374. [Google Scholar] [CrossRef]
- Sprague, M.; Dick, J.R.; Tocher, D.R. Impact of sustainable feeds on omega-3 long-chain fatty acid levels in farmed Atlantic salmon, 2006–2015. Sci. Rep. 2016, 6, 21892. [Google Scholar] [CrossRef]
- De Roos, B.; Sneddon, A.A.; Sprague, M.; Horgan, G.W.; Brouwer, I.A. The potential impact of compositional changes in farmed fish on its health-giving properties: Is it time to reconsider current dietary recommendations? Public Health Nutr. 2017, 20, 2042–2049. [Google Scholar] [CrossRef] [PubMed]
- Sprague, M.; Betancor, M.B.; Dick, J.R.; Tocher, D.R. Nutritional evaluation of seafood, with respect to long-chain omega-3 fatty acids, available to UK consumers. Proc. Nutr. Soc. 2017, 76, E38. [Google Scholar] [CrossRef]
- Sprague, M.; Betancor, M.B.; Tocher, D.R. Nutritional evaluation of seafood available to consumers in the UK. In Proceedings of the 18th International Symposium on Fish Nutrition & Feeding (ISFNF), Las Palmas, Spain, 3–7 June 2018. Abstract P.1.04. [Google Scholar]
- Montero, D.; Izquierdo, M. Welfare and health of fish fed vegetable oil. In Fish Oil Replacement and Alternative Lipid Sources in Aquaculture Feeds; Turchini, G.M., Ng, W.-K., Tocher, D.R., Eds.; Taylor & Francis, CRC Press: Boca Raton, FL, USA, 2011; pp. 439–485. ISBN 9781439808627. [Google Scholar]
- Tocher, D.R.; Glencross, B.D. Lipids and fatty acids. In Dietary Nutrients, Additives, and Fish Health; Lee, C.-S., Lim, C., Webster, C., Gatlin, D.M., III, Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2015; pp. 47–94. ISBN 978-0-470-96288-6. [Google Scholar]
- Simopoulos, A.P. Evolutionary aspects of diet: The omega-6/omega-3 ratio and the brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef]
- Olsen, R.E.; Waagbø, R.; Melle, W.; Ringø, E.; Lall, S.P. Alternative marine sources. In Fish Oil Replacement and Alternative Lipid Sources in Aquaculture Feeds; Turchini, G.M., Ng, W.-K., Tocher, D.R., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 267–324. ISBN 9781439808627. [Google Scholar]
- Winwood, R.J. Recent developments in the commercial production of DHA and EPA rich oils from micro-algae. OCL 2013, 20, D604. [Google Scholar] [CrossRef]
- Haslam, R.P.; Usher, S.; Sayanova, O.; Napier, J.A.; Betancor, M.B.; Tocher, D.R. The supply of fish oil to aquaculture: A role for transgenic oilseed crops? World Agric. 2015, 5, 15–23. [Google Scholar]
- Napier, J.A.; Usher, S.; Haslam, R.P.; Ruiz-Lopez, N.; Sayanova, O. Transgenic plants as a sustainable, terrestrial source of fish oils. Eur. J. Lipid Sci. Technol. 2015, 117, 1317–1324. [Google Scholar] [CrossRef] [PubMed]
- Introducing AlgaPrime™ DHA: A Traceable, Sustainable, High-Quality Alternative to Marine-Based Omega-3s. Available online: http://algaprime.com/wp-content/uploads/2018/07/TVAN0618-005-Aquaculture_v2.pdf (accessed on 30 September 2018).
- DHAGold® for Companion Animals. Available online: https://www.dsm.com/content/dam/dsm/anh/en_US/documents/DHAgold_for_companion_animals.pdf (accessed on 19 November 2018).
- DHA Natur™. Available online: https://service.admani.com/portal/pls/portal/docs/1/8302766.PDF (accessed on 19 November 2018).
- Kousoulaki, K.; Mørkøre, T.; Nengas, I.; Berge, R.K.; Sweetman, J. Microalgae and organic minerals enhance lipid retention efficiency and fillet quality in Atlantic salmon (Salmo salar L.). Aquaculture 2016, 451, 47–57. [Google Scholar] [CrossRef]
- Nymega™ Optimisation, Efficiency and Choice. Available online: https://syndel.ca/downloads/dl/file/id/124/nymega_information_sheet.pdf (accessed on 19 November 2018).
- Application for the Authorization of DHA and EPA-Rich Algal Oil from Schizochytrium sp. Available online: https://acnfp.food.gov.uk/sites/default/files/mnt/drupal_data/sources/files/multimedia/pdfs/dhaoapplicdossier.pdf (accessed on 30 October 2018).
- Betancor, M.B.; Sprague, M.; Usher, S.; Sayanova, O.; Campbell, P.J.; Napier, J.A.; Tocher, D.R. A nutritionally-enhanced oil from transgenic Camelina sativa effectively replaces fish oil as a source of eicosapentaenoic acid for fish. Sci. Rep. 2015, 5, 8104. [Google Scholar] [CrossRef] [PubMed]
- Betancor, M.B.; Li, K.; Bucerzan, V.S.; Sprague, M.; Sayanova, O.; Usher, S.; Han, L.; Norambuena, F.; Torrissen, O.; Napier, J.A.; et al. Oil from transgenic Camelina sativa containing over 25% n-3 long-chain polyunsaturated fatty acids as the major lipid source in feed for Atlantic salmon (Salmo salar). Br. J. Nutr. 2018, 119, 1378–1392. [Google Scholar] [CrossRef] [PubMed]
- Petition for the Determination of Nonregulated Status for EPA+DHA Canola Event LBFLFK. Available online: https://www.aphis.usda.gov/brs/aphisdocs/17_32101p.pdf (accessed on 30 October 2018).
- Petition for Determination of Nonregulated Status for DHA Canola. Available online: https://www.aphis.usda.gov/brs/aphisdocs/17_23601p.pdf (accessed on 30 October 2018).
- Xie, D.; Jackson, E.N.; Zhu, Q. Sustainable source of omega-3 eicosapentaenoic acid from metabolically engineered Yarrowia lipolytica: From fundamental research to commercial production. Appl. Microbiol. Biotechnol. 2015, 99, 1599–1610. [Google Scholar] [CrossRef] [PubMed]
- Microalgae-Based Products for the Food and Feed Sector: An Outlook for Europe. JRC Scientific and Policy Report 85709. Joint Research Centre, Institute for Prospective Technological Studies: Seville, Spain. Available online: http://publications.jrc.ec.europa.eu/repository/bitstream/JRC85709/final%20version%20online%20ipts%20jrc%2085709.pdf (accessed on 19 November 2018).
- Harnessing Nature. Enabling Growth. Available online: https://www.veramaris.com/files/assets/downloads/publications/Imagebr_Veramaris_04-2018.pdf (accessed on 30 October 2018).
- Santigosa, E.; Verlhac-Trichet, V.; Olsen, R.E.; Figuereido-Silva, C. A microalgal oil containing EPA+DHA can be an effective source of omega 3 for Atlantic salmon post- smolts. In Proceedings of the 18th International Symposium on Fish Nutrition & Feeding (ISFNF), Las Palmas, Spain, 3–7 June 2018. Abstract 3.20. [Google Scholar]
- Kousalaki, K. Recent developments and future perspectives of using marine microalgae in fish farming. Aquac. Eur. 2015, 40, 5–11. [Google Scholar]
- Hatlen, B.; Berge, G.M.; Odom, J.M.; Mundheim, H.; Ruyter, B. Growth performance, feed utilisation and fatty acid deposition in Atlantic salmon, Salmo salar L., fed graded levels of high-lipid/high EPA Yarrowia lipolytica biomass. Aquaculture 2012, 364–365, 39–47. [Google Scholar] [CrossRef]
- Berge, G.M.; Hatlen, B.; Odom, J.M.; Ruyter, B. Physical treatment of high EPA Yarrowia lipolytica biomass increases the availability of n-3 highly unsaturated fatty acids when fed to Atlantic salmon. Aquac. Nutr. 2013, 19, 11–121. [Google Scholar] [CrossRef]
- Salunkhe, D.K.; Adsule, R.N.; Chavan, J.K.; Kadam, S.S. World Oilseeds: Chemistry, Technology and Utilization; Van Nostrand Reinhold: New York, NY, USA, 1992; 580p, ISBN 9780442001124. [Google Scholar]
- Gatlin, D.M.; Barrows, F.T.; Brown, P.; Dabrowski, K.; Gibson, G.T.; Hardy, R.W.; Elliot, H.; Hu, G.; Krogdahl, A.; Nelson, R.; et al. Expanding the utilization of sustainable plant products in aquafeeds: A review. Aquac. Res. 2007, 38, 551–579. [Google Scholar] [CrossRef]
- Hixson, S.M.; Parrish, C.C.; Anderson, D.M. Full substitution of fish oil with camelina (Camelina sativa) oil, with partial substitution of fish meal with camelina meal, in diets for farmed Atlantic salmon (Salmo salar) and its effect on tissue lipids and sensory quality. Food Chem. 2014, 157, 51–61. [Google Scholar] [CrossRef]
- Bullerwell, C.N.; Collins, S.A.; Lall, S.P.; Anderson, D.M. Growth performance, proximate and histological analysis of rainbow trout fed diets containing Camelina sativa seeds, meal (high-oil and solvent-extracted) and oil. Aquaculture 2016, 452, 342–350. [Google Scholar] [CrossRef]
- Betancor, M.B.; Li, K.; Sprague, M.; Bardal, T.; Sayanova, O.; Usher, S.; Han, L.; Måsøval, K.; Torrissen, O.; Napier, J.A.; et al. An oil containing EPA and DHA from transgenic Camelina sativa to replace marine fish oil in feeds for Atlantic salmon (Salmo salar L.): Effects on intestinal transcriptome, histology, tissue fatty acid profiles and plasma biochemistry. PLoS ONE 2017, 12, e0175415. [Google Scholar] [CrossRef]
- Napier, J.A.; Olsen, R.E.; Tocher, D.R. Update on GM canola crops as novel sources of omega-3 fish oils. Plant Biotechnol. 2018. [Google Scholar] [CrossRef]
- Bibus, D.M. Long chain omega-3s from forage fish provides value to farmed seafood. Lipid Technol. 2015, 27, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Tacon, A.G.; Metian, M. Global overview on the use of fishmeal and fish oil in industrially compounded aquafeeds: Trends and future prospects. Aquaculture 2008, 285, 146–158. [Google Scholar] [CrossRef]
- Ytrestøyl, T.; Aas, T.S.; Berge, G.M.; Hatlen, B.; Sorensen, M.; Ruyter, B.; Thomassen, M.; Skontorp Hognes, E.; Ziegler, F.; Sund, V.; et al. Resource Utilisation and Ecoefficiency of Norwegian Salmon Farming in 2010; NOFIMA Report No. 53/2011; The Norwegian Institute of Food, Fisheries, and Aquaculture Research (NOFIMA): Tromsø, Norway, 2011; 65p, ISBN 978-82-7251-945-1. [Google Scholar]
- Welch, A.; Hoenig, R.; Steiglitz, J.; Benetti, D.; Tacon, A.; Sims, N.; O’Hanlon, B. From fishing to the sustainable farming of carnivorous marine finfish. Rev. Fish. Sci. 2010, 18, 235–247. [Google Scholar] [CrossRef]
- Trushenski, J.; Schwarz, M.; Bergman, A.; Rombenso, A.; Delbos, B. DHA is essential, EPA appears largely expendable, in meeting the n−3 long-chain polyunsaturated fatty acid requirements of juvenile cobia Rachycentron canadum. Aquaculture 2012, 326–329, 81–89. [Google Scholar] [CrossRef]
- Sprague, M.; Walton, J.; Campbell, P.J.; Strachan, F.; Dick, J.R.; Bell, J.G. Replacement of fish oil with a DHA-rich algal meal derived from Schizochytrium sp. on the fatty acid and persistent organic pollutant levels in diets and flesh of Atlantic salmon (Salmo salar L.) post-smolts. Food Chem. 2015, 185, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Betiku, O.C.; Barrows, F.T.; Ross, C.; Sealey, W.M. The effect of total replacement of fish oil with DHA-Gold® and plant oils on growth and fillet quality of rainbow trout (Oncorhynchus mykiss) fed a plant-based diet. Aquac. Nutr. 2016, 22, 158–169. [Google Scholar] [CrossRef]
- Patnaik, S.; Samocha, T.M.; Davis, D.A.; Bullis, R.A.; Browdy, C.L. The use of HUFA-rich algal meals in diets for Litopenaeus vannamei. Aquac. Nutr. 2006, 12, 395–401. [Google Scholar] [CrossRef]
- Mata-Sotres, J.A.; Tinajero-Chavez, A.; Barreto-Curiel, F.; Pares-Sierra, G.; Del Rio-Zaragoza, O.B.; Viana, M.T.; Rombenso, A.N. DHA (22:6n-3) supplementation is valuable in Totoaba macdonaldi fish oil-free feeds containing poultry by-product meal and beef tallow. Aquaculture 2018, 497, 440–451. [Google Scholar] [CrossRef]
- Kousoulaki, K.; Østbye, T.-K.; Krasnov, A.; Torgersen, J.S.; Mørkøre, T.; Sweetman, J. Metabolism, health and fillet nutritional quality in Atlantic salmon (Salmo salar) fed diets containing n-3 rich microalgae. J. Nutr. Sci. 2015, 4, e24. [Google Scholar] [CrossRef]
- Wang, Y.; Li, M.; Filer, K.; Xue, Y.; Ai, Q.; Mai, K. Replacement of fish oil with a DHA-rich Schizochytrium meal on growth performance, activities of digestive enzyme and fatty acid profile of Pacific white shrimp (Liptopenaeus vannamei) larvae. Aquac. Nutr. 2016, 23, 1113–1120. [Google Scholar] [CrossRef]
- Kissinger, K.R.; García-Ortega, A.; Trushenski, J.T. Partial fish meal replacement by soy protein concentrate, squid and algal meals in low fish-oil diets containing Schizochytrium limacinum for longfin yellowtail Seriola rivoliana. Aquaculture 2016, 452, 37–44. [Google Scholar] [CrossRef]
- García-Ortega, A.; Kissinger, K.R.; Trushenski, J.T. Evaluation of fish meal and fish oil replacement by soybean protein and algal meal from Schizochytrium limacinum in diets for giant grouper Epinephelus lanceolatus. Aquaculture 2016, 452, 1–8. [Google Scholar] [CrossRef]
- Perez-Velazqueza, M.; Gatlin, D.M., III; González-Félix, M.L.; García-Ortega, A. Partial replacement of fishmeal and fish oil by algal meals in diets of red drum Sciaenops ocellatus. Aquaculture 2018, 487, 41–50. [Google Scholar] [CrossRef]
- Rosenlund, G.; Baardsen, G.K.; Stubhaug, I.; Holme, M.-H. Sensory quality of Atlantic salmon (S. salar) fed no fish meal–no fish oil diets. In Proceedings of the 18th International Symposium on Fish Nutrition & Feeding (ISFNF), Las Palmas, Spain, 3–7 June 2018. Abstract P.1.14. [Google Scholar]
- Betancor, M.B.; Sprague, M.; Sayanova, O.; Usher, S.; Campbell, P.J.; Napier, J.A.; Caballero, M.J.; Tocher, D.R. Evaluation of a high-EPA oil from transgenic Camelina sativa in feeds for Atlantic salmon (Salmo salar L.): Effects on tissue fatty acid composition, histology and gene expression. Aquaculture 2015, 444, 1–12. [Google Scholar] [CrossRef]
- Betancor, M.B.; Sprague, M.; Montero, D.; Usher, S.; Sayanova, O.; Campbell, P.J.; Napier, J.A.; Caballero, M.J.; Izquierdo, M.; Tocher, D.R. Replacement of marine fish oil with de novo omega-3 oils from transgenic Camelina sativa in feeds for Gilthead sea bream (Sparus aurata L.). Lipids 2016, 51, 1171–1191. [Google Scholar] [CrossRef]
- Betancor, M.B.; Sprague, M.; Sayanova, O.; Metochis, C.; Campbell, P.J.; Napier, J.A.; Tocher, D.R. Nutritional evaluation of an EPA-DHA oil from transgenic Camelina sativa in feeds for post-smolt Atlantic salmon (Salmo salar L.). PLoS ONE 2016, 11, e0159934. [Google Scholar] [CrossRef]
- Iassonova, D.; Arriagada, P.; Capdeville, A. An alternative source of long chain Omega-3 fatty acids from Novel Canola oil for salmonid farming. Feeding trial report. In Proceedings of the 18th International Symposium on Fish Nutrition & Feeding (ISFNF), Las Palmas, Spain, 3–7 June 2018. Abstract 3.04. [Google Scholar]
- Ruyter, B.; Sissener, N.; Ostbye, T.-K.; Mira, M.B.; Simon, C.; Berge, G.; Sanden, M.; Mørkøre, T.; Krasnov, A. Omega-3 Canola Oil effectively replaces fish oil as a dietary source of docosahexaenoic acid (DHA) in feed for Atlantic salmon in freshwater and seawater. In Proceedings of the 18th International Symposium on Fish Nutrition & Feeding (ISFNF), Las Palmas, Spain, 3–7 June 2018. Abstract 3.02. [Google Scholar]
- National Research Council (NRC). Nutrient Requirements of Fish and Shrimp; The National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-16338-5. [Google Scholar]
- Tocher, D.R. Metabolism and functions of lipids and fatty acids in teleost fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Sherratt, S.C.R.; Mason, R.P. Eicosapentaenoic acid and docosahexaenoic acid have distinct membrane locations and lipid interactions as determined by X-ray diffraction. Chem. Phys. Lipids 2018, 212, 73–79. [Google Scholar] [CrossRef]
- Farooqui, A.A. Beneficial Effects of Fish Oil on Human Brain; Springer: Heidelberg, Germany, 2009; 396p, ISBN 978-1-4419-0542-0. [Google Scholar]
- Wang, W.; Zhu, J.; Lyu, F.; Panigrahy, D.; Ferrara, K.W.; Hammock, B.; Zhang, G. ω-3 Polyunsaturated fatty acids-derived lipid metabolites on angiogenesis, inflammation and cancer. Prostaglandins Other Lipid Med. 2014, 113–115, 13–20. [Google Scholar] [CrossRef]
- Fard, G.S.; Wang, F.; Sinclair, A.J.; Elliot, G.; Turchini, G.M. How does high DHA fish oil affect health? A systematic review of evidence. Crit. Rev. Food Sci. Nutr. 2018, 58, 1–44. [Google Scholar] [CrossRef]
- Nanton, D.A.; Ruohonen, K.; Robb, D.H.F.; El-Mowafi, A.; Hartnell, G.F. Effect of soy oil containing stearidonic acid on growth performance and fillet fatty acid composition of Atlantic salmon. Aquac. Nutr. 2012, 18, 640–650. [Google Scholar] [CrossRef]
- Industrial Fish Feed. Available online: https://www.alltechcoppens.com/en/industrial (accessed on 19 November 2018).
- Alltech has not Exited Algae. Available online: https://www.feednavigator.com/Article/2018/08/29/Alltech-has-not-exited-algae#.W4avXpli0m4.twitter (accessed on 19 November 2018).
- Success of AlgaPrime™ DHA in Aquaculture Market Drives Increased Commercial Distribution. Available online: http://www.bunge.com/news/success-algaprimetm-dha-aquaculture-market-drives-increased-commercial-distribution (accessed on 30 September 2018).
- Ledesma-Amaro, R.; Nicaud, J.-M. Yarrowia lipolytica as a biotechnological chassis to produce usual and unusual fatty acids. Prog. Lipid Res. 2016, 61, 40–50. [Google Scholar] [CrossRef]
- Omega-3 Deficiency a Concern for Human Health and Our Oceans. Available online: http://www.nuseed.com/innovation/omega-3/ (accessed on 19 November 2018).
- Cargill Launches Latitude™, a Sustainable, Plant-Based Alternative Source of Omega-3 for Fish Feed Applications. Available online: https://www.cargill.com/2018/cargill-launches-latitude (accessed on 30 October 2018).
- Desaint, N.; Varbanova, M. The use and value of polling to determine public opinion on GMOs in Europe: Limitations and ways forward. GM Crops Food 2013, 4, 183–194. [Google Scholar] [CrossRef]
- Lucht, J.M. Public acceptance of plant biotechnology and GM crops. Viruses 2015, 7, 4254–4281. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Hemre, G.I.; Mommsen, T. Carbohydrates in fish nutrition: Digestion and absorption in postlarval stages. Aquac. Nutr. 2005, 11, 103–122. [Google Scholar] [CrossRef]
- Darley, W.M.; Porter, D.; Fuller, M.S. Cell wall composition and synthesis via Golgi-directed scale formation in the marine eucaryote, Schizochytrium aggregatum, with a note on Thraustochytrium sp. Arch. Mikrobiol. 1973, 90, 89–106. [Google Scholar] [CrossRef]
- Castillo, S.; Gatlin, D.M. Dietary supplementation of exogenous carbohydrase enzymes in fish nutrition: A review. Aquaculture 2015, 435, 286–292. [Google Scholar] [CrossRef]
- Sarker, P.K.; Kapuscinski, A.R.; Lanois, A.; Livesey, E.D.; Bernhard, K.P.; Coley, M.L. Towards sustainable aquafeeds: Complete substitution of fish oil with marine microalga Schizochytrium sp. improves growth and fatty acid deposition in juvenile Nile tilapia (Oreochromis niloticus). PLoS ONE 2016, 11, e0156684. [Google Scholar] [CrossRef]
- Turchini, G.M.; Torstensen, B.E.; Ng, W.K. Fish oil replacement in finfish nutrition. Rev. Aquac. 2009, 1, 10–57. [Google Scholar] [CrossRef]
- Shepherd, C.J.; Little, D.C. Aquaculture: Are the criticisms justified? II—Aquaculture’s environmental impact and use of resources, with special reference to farming Atlantic salmon. World Agric. 2014, 4, 37–52. [Google Scholar]
- Torrissen, O.; Olsen, R.E.; Toresen, R.; Hemre, G.I.; Tacon, A.G.J.; Asche, F.; Hardy, R.W.; Lall, S. Atlantic salmon (Salmo salar): The ‘super-chicken’ of the sea? Rev. Fish. Sci. 2011, 19, 257–278. [Google Scholar] [CrossRef]
- Howe, P.R.C. ω3-Enriched pork. In The Return of ω3 Fatty Acids into the Food Supply. I. Land-Based Animal Food Products and Their Health Effects; Simopoulos, A.P., Ed.; World Review of Nutrition and Dietetics; Karger: Basel, Switzerland, 1998; Volume 83, pp. 132–143. ISBN 978-3-8055-6694-0. [Google Scholar]
- Rymer, C.; Givens, D.I. n-3 fatty acid enrichment of edible tissue of poultry: A review. Lipids 2005, 40, 121–130. [Google Scholar] [CrossRef]
- Fraeye, I.; Bruneel, C.; Lemahieu, C.; Buyse, J.; Muylaert, K.; Foubert, I. Dietary enrichment of eggs with omega-3 fatty acids: A review. Food Res. Int. 2012, 48, 961–969. [Google Scholar] [CrossRef]
- Auchterlonie, N. Beyond protein and fat: The underestimated uses of fishmeal and fish oil in swine and poultry diets. Milling Grain 2017, 128, 62–64. [Google Scholar]
- Mandell, I.B.; Buchanan-Smith, J.G.; Holub, B.J. Enrichment of beef with ω3 fatty acids. In The Return of ω3 Fatty Acids into the Food Supply. I. Land-Based Animal Food Products and Their Health Effects; Simopoulos, A.P., Ed.; World Review of Nutrition and Dietetics; Karger: Basel, Switzerland, 1998; Volume 83, pp. 144–159. ISBN 978-3-8055-6694-0. [Google Scholar]
- Jacobsen, C.; Nielsen, N.S.; Horn, A.F.; Sørensen, A.D.M. (Eds.) Food Enrichment with Omega-3 Fatty Acids; Woodgate Publishing Limited, Elsevier: Amsterdam, The Netherlands, 2013; p. 464. ISBN 978-0-85709-428-5. [Google Scholar]
- Lane, K.E.; Derbyshire, E.J. Omega-3 fatty acids—A review of existing and innovative delivery methods. Crit. Rev. Food Sci. Nutr. 2018, 58, 62–69. [Google Scholar] [CrossRef]
- Newton, I.S. Global food fortification perspectives of long-chain ω3 fatty acids. In The Return of ω3 Fatty Acids into the Food Supply. I. Land-Based Animal Food Products and Their Health Effects; Simopoulos, A.P., Ed.; World Review of Nutrition and Dietetics; Karger: Basel, Switzerland, 1998; Volume 83, pp. 199–209. ISBN 978-3-8055-6694-0. [Google Scholar]
- Omega-3 Summit 2011, Consensus Statements (1) 4 March 2011. Available online: http://www.omega3summit.org/pdf/ConsensusStatements.pdf (accessed on 30 October 2018).
- Omega-3 Summit 2018, Global Summit on Nutrition, Health and Human Behaviour—Science, Market and Policy Update. Available online: http://www.omega3summit.org/program.html (accessed on 30 October 2018).
| Composition a | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Total n-3 LC-PUFA | |||||||||
| Product | Development Partners | Source | Type | Lipid Content b | EPA c | DHA c | % of TFA d | % of Product | Reference |
| AlgaPrime™ DHA | Corbion (TerraVia/Bunge) e | Microalgae | Algal biomass | 60 | 0 | 48 | 48 | 28 | [42] |
| DHAgold™ | DSM Nutritional Products | Microalgae | Algal biomass | 49 | 1.0 | 44.4 | 45.8 | 22.5 | [43] |
| DHA Natur™ | ADM Animal Nutrition | Microalgae | Algal biomass | 50–60 | 0.25 | 34 | 34.3 | 17.2–20.6 | [44] |
| ForPlus™ | Alltech Coppens f | Microalgae | Algal biomass | 61 | 0.3 | 29 | 29.3 | 17.9 | [45] |
| Nymega™ | Heliae Development g | Microalgae | Algal biomass | 65 | ~0.1 | 20 | ~31 | 21 | [46] |
| Veramaris® Oil | Veramaris h | Microalgae | Oil | 100 | ~16 | ~34 | ~54 | ~54 | [47] |
| Camelina sativa | Rothamsted Research/UoS | GM camelina | Oil | 100 | 20 | 0 | 24 | 24 | [48] |
| Camelina sativa | Rothamsted Research/UoS | GM camelina | Oil | 100 | 9 | 11 | 28 | 28 | [49] |
| Latitude™ | BASF/Cargill | GM canola | Oil | 100 | 7 | 1 | 12 | 12 | [50] |
| Aquaterra™/Nutriterra™ i | CSIRO/Nuseed/GRDC | GM canola | Oil | 100 | 0.5 | 10 | 12 | 12 | [51] |
| Yarrowia lipolyticaj | DuPont | GM yeast | Yeast biomass | ~50 | ~50 | 0 | 50 | 25 | [52] |
| Product | Species | Replacing | Inclusion Levels | Reference |
|---|---|---|---|---|
| AlgaPrime™ DHA biomass b | N/A | |||
| DHAgold™ biomass | Cobia (Rachycentron canadum) | FO and soybean oil | 1.5, 2.2, 3.0 and 4.3% of diet c | [69] |
| Atlantic salmon (Salmo salar) | FO, FM and plant meals | 5.5 and 11% of diet c | [70] | |
| Rainbow trout (Oncorhynchus mykiss) | Corn oil and wheat flour | 3, 6 and 9% of diet | [71] | |
| DHA Natur™ biomass | Shrimp (Litopenaeus vannamei) | FO and VO, soybean meal and wheat starch | 1.3 and 5.0% of diet | [72] |
| ForPlus™ biomass | Drum (Totoaba macdonaldi) | Corn starch | 0.2 and 0.6% of diet | [73] |
| Atlantic salmon | FO, horse beans, maize gluten | 1, 6 and 15% of diet | [74] | |
| Atlantic salmon | FO and FM | 2.5 and 5.0% of diet | [45] | |
| Shrimp (Litopenaeus vannamei) | FO and wheat flour | 0.6, 1.2, 1.8, 2.3 and 3.5% of diet | [75] | |
| Longfin yellowtail (Seriola rivoliana) | FM | 5% of diet | [76] | |
| Giant grouper (Epinephelus lanceolatus) | FO, FM and squid | 10, 15 and 18% of diet | [77] | |
| Red drum (Sciaenops ocellatus) | FO, SPC and FM | 1.3, 2.7, 4.0, 5.4, 6.7 and 9.9% of diet d | [78] | |
| Veramaris® oil | Atlantic salmon | FO | 25, 50, 75 and 100% of FO e | [55] |
| FO and FM | 2–4 and 8–15% of diet | [79] | ||
| Camelina sativa EPA oil | Atlantic salmon | FO and RO | 100% of added oil | [48,80] |
| Sea bream (Sparus aurata) | FO and RO | 100% of added oil | [81] | |
| Camelina sativa EPA+DHA oil | Atlantic salmon | FO and RO | 100% of added oil | [49,63,82] |
| Sea bream | FO and RO | 100% of added oil | [81] | |
| Latitude™ | Atlantic salmon | FO | 100% of added FO f | [83] |
| Aquaterra™ | Atlantic salmon | FO | [84] | |
| Yarrowia lipolytica biomass | Atlantic salmon | FM, wheat meal and RO | 10–30% of diet | [57,58] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tocher, D.R.; Betancor, M.B.; Sprague, M.; Olsen, R.E.; Napier, J.A. Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand. Nutrients 2019, 11, 89. https://doi.org/10.3390/nu11010089
Tocher DR, Betancor MB, Sprague M, Olsen RE, Napier JA. Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand. Nutrients. 2019; 11(1):89. https://doi.org/10.3390/nu11010089
Chicago/Turabian StyleTocher, Douglas R, Monica B Betancor, Matthew Sprague, Rolf E Olsen, and Johnathan A Napier. 2019. "Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand" Nutrients 11, no. 1: 89. https://doi.org/10.3390/nu11010089
APA StyleTocher, D. R., Betancor, M. B., Sprague, M., Olsen, R. E., & Napier, J. A. (2019). Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand. Nutrients, 11(1), 89. https://doi.org/10.3390/nu11010089





