Executive Functioning in Obesity, Food Addiction, and Binge-Eating Disorder
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Ethics and Procedure
2.3. Sample
2.4. Measures
2.4.1. Eating Disorder Examination
2.4.2. Yale Food Addiction Scale
2.4.3. Eating Disorder Examination-Questionnaire
2.4.4. Patient Health Questionnaire-Depression Scale
2.5. Neuropsychological Assessment
2.5.1. Iowa Gambling Task
2.5.2. Delay Discounting Task
2.5.3. Go/No Go
2.5.4. Wisconsin Card Sorting Test
2.5.5. Alertness
2.6. Statistical Procedure
3. Results
3.1. Sample
3.2. Neuropsychological Assessment
4. Discussion
4.1. Methodological Considerations
4.2. Implications and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization [WHO]. Obesity and Overweight: Fact Sheet. Available online: http://www.who.int/ mediacentre/factsheets/fs311/en/ (accessed on 30 July 2017).
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- Horstmann, A. It wasn’t me; it was my brain-Obesity-associated characteristics of brain circuits governing decision-making. Physiol. Behav. 2017, 176, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Makaronidis, J.M.; Batterham, R.L. Obesity, body weight regulation and the brain: Insights from fMRI. Br. J. Radiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Shields, G.S.; Guo, C.; Liu, Y. Executive function performance in obesity and overweight individuals: A meta-analysis and review. Neurosci. Biobehav. Rev. 2018, 84, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Shallice, T. From Neuropsychology to Mental Structure, 1st ed.; Cambridge University Press: Cambridge, England, 1988; ISBN 978-0-521-31360-5. [Google Scholar]
- Stuss, D.T.; Benson, D.F. The Frontal Lobes; Raven Press: New York, NY, USA, 1986; ISBN 978-0-88167-153-7. [Google Scholar]
- Wyckoff, E.P.; Evans, B.C.; Manasse, S.M.; Butryn, M.L.; Forman, E.M. Executive functioning and dietary intake: Neurocognitive correlates of fruit, vegetable, and saturated fat intake in adults with obesity. Appetite 2017, 111, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Brockmeyer, T.; Hamze Sinno, M.; Skunde, M.; Wu, M.; Woehning, A.; Rudofsky, G.; Friederich, H.-C. Inhibitory control and hedonic response towards food interactively predict success in a weight loss programme for adults with obesity. Obes. Facts 2016, 9, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Hardman, C.A.; Lawrence, N.; Field, M. Cognitive training as a potential treatment for overweight and obesity: A critical review of the evidence. Appetite 2017, 124, 50–67. [Google Scholar] [CrossRef] [PubMed]
- Michaud, A.; Vainik, U.; Garcia-Garcia, I.; Dagher, A. Overlapping neural endophenotypes in addiction and obesity. Front. Endocrinol. 2017, 8, 127. [Google Scholar] [CrossRef]
- Lindgren, E.; Gray, K.; Miller, G.; Tyler, R.; Wiers, C.E.; Volkow, N.D.; Wang, G.-J. Food addiction: A common neurobiological mechanism with drug abuse. Front. Biosci. Landmark Ed. 2018, 23, 811–836. [Google Scholar]
- Jauch-Chara, K.; Oltmanns, K.M. Obesity—A neuropsychological disease? Systematic review and neuropsychological model. Prog. Neurobiol. 2014, 114, 84–101. [Google Scholar] [CrossRef]
- Burrows, T.; Kay-Lambkin, F.; Pursey, K.; Skinner, J.; Dayas, C. Food addiction and associations with mental health symptoms: A systematic review with meta-analysis. J. Hum. Nutr. Diet. 2018, 31, 544–572. [Google Scholar] [CrossRef] [PubMed]
- Gordon, E.L.; Ariel-Donges, A.H.; Bauman, V.; Merlo, L.J. What is the evidence for “food addiction?” a systematic review. Nutrients 2018, 10, 477. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.G.; Robbins, T.W. The neurobiological underpinnings of obesity and binge eating: A rationale for adopting the food addiction model. Biol. Psychiatry 2013, 73, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Davis, C. A commentary on the associations among “food addiction”, binge eating disorder, and obesity: Overlapping conditions with idiosyncratic clinical features. Appetite 2017, 115, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Corbin, W.R.; Brownell, K.D. Preliminary validation of the yale food addiction scale. Appetite 2009, 52, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, E.; Tavares, H.; von Ranson, K.M. Moving beyond the “eating addiction” versus “food addiction” debate: Comment on Schulte et al. (2017). Appetite 2018, 130, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Schulte, E.M.; Potenza, M.N.; Gearhardt, A.N. A commentary on the “eating addiction” versus “food addiction” perspectives on addictive-like food consumption. Appetite 2017, 115, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Hilbert, A. Binge-Eating Disorder; Psychiatric Clinics of North America in Press: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Gearhardt, A.N.; Corbin, W.R.; Brownell, K.D. Development of the yale food addiction scale version 2.0. Psychol. Addict. Behav. 2016, 30, 113–121. [Google Scholar] [CrossRef]
- American Psychiatric Association. DSM-5: Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- Steward, T.; Mestre-Bach, G.; Vintró-Alcaraz, C.; Lozano-Madrid, M.; Agüera, Z.; Fernández-Formoso, J.A.; Granero, R.; Jiménez-Murcia, S.; Vilarrasa, N.; García-Ruiz-de-Gordejuela, A.; et al. Food addiction and impaired executive functions in women with obesity. Eur. Eat. Disord. Rev. 2018, 26, 574–584. [Google Scholar] [CrossRef]
- Rodrigue, C.; Ouellette, A.-S.; Lemieux, S.; Tchernof, A.; Biertho, L.; Bégin, C. Executive functioning and psychological symptoms in food addiction: A study among individuals with severe obesity. Eat. Weight Disord. 2018, 23, 469–478. [Google Scholar] [CrossRef]
- Cassin, S.E.; von Ranson, K.M. Is binge eating experienced as an addiction? Appetite 2007, 49, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Mackew, L.; Levitan, R.D.; Kaplan, A.S.; Carter, J.C.; Kennedy, J.L. Binge eating disorder (BED) in relation to addictive behaviors and personality risk factors. Front. Psychol. 2017, 8, 579. [Google Scholar] [CrossRef] [PubMed]
- Ricca, V.; Castellini, G.; Lo Sauro, C.; Ravaldi, C.; Lapi, F.; Mannucci, E.; Rotella, C.M.; Faravelli, C. Correlations between binge eating and emotional eating in a sample of overweight subjects. Appetite 2009, 53, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Córdova, M.E.; Schiavon, C.C.; Busnello, F.M.; Reppold, C.T. Nutritional and neuropsychological profile of the executive functions on binge eating disorder in obese adults. Nutr. Hosp. 2017, 34, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Giel, K.E.; Teufel, M.; Junne, F.; Zipfel, S.; Schag, K. Food-related impulsivity in obesity and binge eating disorder—A Systematic update of the evidence. Nutrients 2017, 9, 1170. [Google Scholar] [CrossRef] [PubMed]
- Kollei, I.; Rustemeier, M.; Schroeder, S.; Jongen, S.; Herpertz, S.; Loeber, S. Cognitive control functions in individuals with obesity with and without binge-eating disorder. Int. J. Eat. Disord. 2018, 51, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Leehr, E.J.; Schag, K.; Dresler, T.; Grosse-Wentrup, M.; Hautzinger, M.; Fallgatter, A.J.; Zipfel, S.; Giel, K.E.; Ehlis, A.-C. Food specific inhibitory control under negative mood in binge-eating disorder: Evidence from a multimethod approach. Int. J. Eat. Disord. 2018, 51, 112–123. [Google Scholar] [CrossRef]
- Manasse, S.M.; Forman, E.M.; Ruocco, A.C.; Butryn, M.L.; Juarascio, A.S.; Fitzpatrick, K.K. Do executive functioning deficits underpin binge eating disorder? A comparison of overweight women with and without binge eating pathology. Int. J. Eat. Disord. 2015, 48, 677–683. [Google Scholar] [CrossRef]
- Svaldi, J.; Naumann, E.; Biehl, S.; Schmitz, F. Impaired early-response inhibition in overweight females with and without binge eating disorder. PLoS ONE 2015, 10, e0133534. [Google Scholar] [CrossRef]
- Kessler, R.M.; Hutson, P.H.; Herman, B.K.; Potenza, M.N. The neurobiological basis of binge-eating disorder. Neurosci. Biobehav. Rev. 2016, 63, 223–238. [Google Scholar] [CrossRef]
- Schag, K.; Schönleber, J.; Teufel, M.; Zipfel, S.; Giel, K.E. Food-related impulsivity in obesity and binge eating disorder—A systematic review: Food-related impulsivity. Obes. Rev. 2013, 14, 477–495. [Google Scholar] [CrossRef]
- Balodis, I.M.; Grilo, C.M.; Potenza, M.N. Neurobiological features of binge eating disorder. CNS Spectr. 2015, 20, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Kittel, R.; Brauhardt, A.; Hilbert, A. Cognitive and emotional functioning in binge-eating disorder: A systematic review. Int. J. Eat. Disord. 2015, 48, 535–554. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Curtis, C.; Levitan, R.D.; Carter, J.C.; Kaplan, A.S.; Kennedy, J.L. Evidence that “food addiction” is a valid phenotype of obesity. Appetite 2011, 57, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; White, M.A.; Masheb, R.M.; Morgan, P.T.; Crosby, R.D.; Grilo, C.M. An examination of the food addiction construct in obese patients with binge eating disorder. Int. J. Eat. Disord. 2012, 45, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Hilbert, A.; Blume, M.; Petroff, D.; Neuhaus, P.; Smith, E.; Hay, P.J.; Hübner, C. Group cognitive remediation therapy for adults with obesity prior to behavioural weight loss treatment: Study protocol for a randomised controlled superiority study (CRT study). BMJ Open 2018, 8, e022616. [Google Scholar] [CrossRef] [PubMed]
- Blume, M.; Schmidt, R.; Schmidt, J.; Martin, A.; Hilbert, A. German Clinical Trials Register: EEG Neurofeedback for Adults with Binge-Eating Disorder. Available online: https://www.drks.de/drks_web/navigate.do?navigationId=trial.HTML&TRIAL_ID=DRKS00010496 (accessed on 7 August 2018).
- Meule, A.; Müller, A.; Gearhardt, A.N.; Blechert, J. German version of the Yale Food Addiction Scale 2.0: Prevalence and correlates of “food addiction” in students and obese individuals. Appetite 2017, 115, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G.; Cooper, Z.; O’Connor, M. Eating Disorder Examination (17.0D); Guilford Press: New York, NY, USA, 2014. [Google Scholar]
- Hilbert, A.; Tuschen-Caffier, B. Eating Disorder Examination. Deutschsprachige Übersetzung, 2nd ed.; Deutsche Gesellschaft für Verhaltensterapie: Tübingen, Germany, 2016; ISBN 978-3-87159-411-3. [Google Scholar]
- Berg, K.C.; Peterson, C.B.; Frazier, P.; Crow, S.J. Psychometric evaluation of the eating disorder examination and eating disorder examination-questionnaire: A systematic review of the literature. Int. J. Eat. Disord. 2012, 45, 428–438. [Google Scholar] [CrossRef]
- Kliem, S.; Mößle, T.; Zenger, M.; Strauß, B.; Brähler, E.; Hilbert, A. The eating disorder examination-questionnaire 8: A brief measure of eating disorder psychopathology (EDE-Q8). Int. J. Eat. Disord. 2016, 49, 613–616. [Google Scholar] [CrossRef]
- Gräfe, K.; Zipfel, S.; Herzog, W.; Löwe, B. Screening psychischer Störungen mit dem “Gesundheitsfragebogen für Patienten (PHQ-D)”. Diagnostica 2004, 50, 171–181. [Google Scholar] [CrossRef]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B. Validation and utility of a self-report version of PRIME-MD: The PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999, 282, 1737–1744. [Google Scholar] [CrossRef] [PubMed]
- Bechara, A.; Damasio, A.R.; Damasio, H.; Anderson, S.W. Insensitivity to future consequences following damage to human prefrontal cortex. Cognition 1994, 50, 7–15. [Google Scholar] [CrossRef]
- Richards, J.B.; Zhang, L.; Mitchell, S.H.; de Wit, H. Delay or probability discounting in a model of impulsive behavior: Effect of alcohol. J. Exp. Anal. Behav. 1999, 71, 121–143. [Google Scholar] [CrossRef] [PubMed]
- Millisecond. Inquisit (Version 4), Windows; Millisecond Software: San Francisco, CA, USA, 2015.
- Myerson, J.; Green, L.; Warusawitharana, M. Area under the curve as a measure of discounting. J. Exp. Anal. Behav. 2001, 76, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, S.; Aschenbrenner, S.; Pfüller, U.; Roesch-Ely, D.; Weisbrod, M. Wiener Testsystem: Response Inhibition, Schuhfried: Mödling, Germany, 2015.
- Schuhfried Gmbh. Vienna Test System, Windows; Schuhfried Gmbh: Mödling, Austria, 2015.
- Lezak, M.D.; Howieson, D.B.; Bigler, E.D.; Tranel, D. Neuropsychological Assessment; Oxford University Press: Oxford, NY, USA, 2012; ISBN 978-0-19-539552-5. [Google Scholar]
- Heaton, R.K.; Chelune, G.J.; Talley, J.L.; Kay, G.G.; Curtiss, G. Wisconsin Card Sorting Test Manual; Psychological Assessment Resources, Inc.: Odessa, FL, USA, 1993. [Google Scholar]
- Sturm, W. Wiener Testsystem: Wahrnehmungs- und Aufmerksamkeitsfunktionen, Schuhfried: Mödling, Austria, 2015.
- Lix, L.M.; Keselman, J.C.; Keselman, H.J. Consequences of assumption violations revisited: A quantitative review of alternatives to the one-way analysis of variance f test. Rev. Educ. Res. 1996, 66, 579–619. [Google Scholar] [CrossRef]
- Keppel, G. Design and Analysis: A Researcher’s Handbook, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1991. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: New York, NY, USA, 1988. [Google Scholar]
- Tamm, L.; Narad, M.E.; Antonini, T.N.; O’Brien, K.M.; Hawk, L.W.; Epstein, J.N. Reaction time variability in ADHD: A review. Neurotherapeutics 2012, 9, 500–508. [Google Scholar] [CrossRef]
- Cortese, S.; Bernardina, B.D.; Mouren, M.-C. Attention-deficit/hyperactivity disorder (adhd) and binge eating. Nutr. Rev. 2007, 65, 404–411. [Google Scholar] [CrossRef]
- Voon, V. Cognitive biases in binge eating disorder: The hijacking of decision making. CNS Spectr. 2015, 20, 566–573. [Google Scholar] [CrossRef]
- Restivo, M.R.; McKinnon, M.C.; Frey, B.N.; Hall, G.B.; Syed, W.; Taylor, V.H. The impact of obesity on neuropsychological functioning in adults with and without major depressive disorder. PLoS ONE 2017, 12, e0176898. [Google Scholar] [CrossRef]
- Schmidt, R.; Lüthold, P.; Kittel, R.; Tetzlaff, A.; Hilbert, A. Visual attentional bias for food in adolescents with binge-eating disorder. J. Psychiatr. Res. 2016, 80, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Sperling, I.; Baldofski, S.; Lüthold, P.; Hilbert, A. Cognitive food processing in binge-eating disorder: An eye-tracking study. Nutrients 2017, 9, 903. [Google Scholar] [CrossRef] [PubMed]
| Variable | Obesity Only n = 23 | Food Addiction n = 23 | Binge-Eating Disorder n = 19 | Food Addiction Plus Binge-Eating Disorder n = 23 | Test | Effect Size | ||
|---|---|---|---|---|---|---|---|---|
| M (SD) | M (SD) | M (SD) | M (SD) | F | df | p | Est. ω2 | |
| Age (years) | 40.48 (10.85) | 43.39 (10.35) | 38.84 (9.43) | 37.53 (10.12) | 1.38 | 3, 84 | 0.254 | 0.013 |
| Body mass index (kg/m2) | 42.84 (4.76) | 44.14 (6.59) | 41.92 (5.25) | 42.23 (6.13) | 0.64 | 3, 84 | 0.589 | −0.012 |
| n | n | n | n | Χ2 | df | p | ||
| Sex (female/male) | 17/6 | 18/5 | 14/5 | 16/7 | 0.45 | 3 | 0.930 | |
| Education (low/middle/high) 1 | 3/11/9 | 3/16/4 | 1/7/11 | 4/10/9 | 8.50 | 6 | 0.203 | |
| M (SD) | M (SD) | M (SD) | M (SD) | F | df | p | Est. ω2 | |
| Eating disorder psychopathology (EDE-Q8) | 3.59 (0.98) a | 4.20 (0.80) a | 3.55 (1.03) a | 4.36 (1.20) a | 3.92 | 3, 84 | 0.011 | 0.091 |
| Depression (PHQ-D) | 5.00 (3.47) a | 7.71 (3.80) a, b | 7.99 (5.27) a, b | 10.17 (4.10) b | 5.98 | 3, 84 | 0.001 | 0.145 |
| n | n | n | n | Χ2 | df | p | ||
| FA severity (mild/moderate/severe) | - | 9/10/4 | - | 0/6/17 | 18.05 | 2 | <0.001 | |
| BED severity (mild/moderate/severe) | - | - | 14/5/0 | 16/4/3 | 2.89 | 2 | 0.236 |
| Variable | Obesity Only n = 23 | Food Addiction n = 23 | Binge-Eating-Disorder n = 19 | Food Addiction plus Binge-Eating Disorder n = 23 | Welch ANOVA | Effect Size | |||
|---|---|---|---|---|---|---|---|---|---|
| M (SD) | M (SD) | M (SD) | M (SD) | F | df | p | Est. ω2 | ||
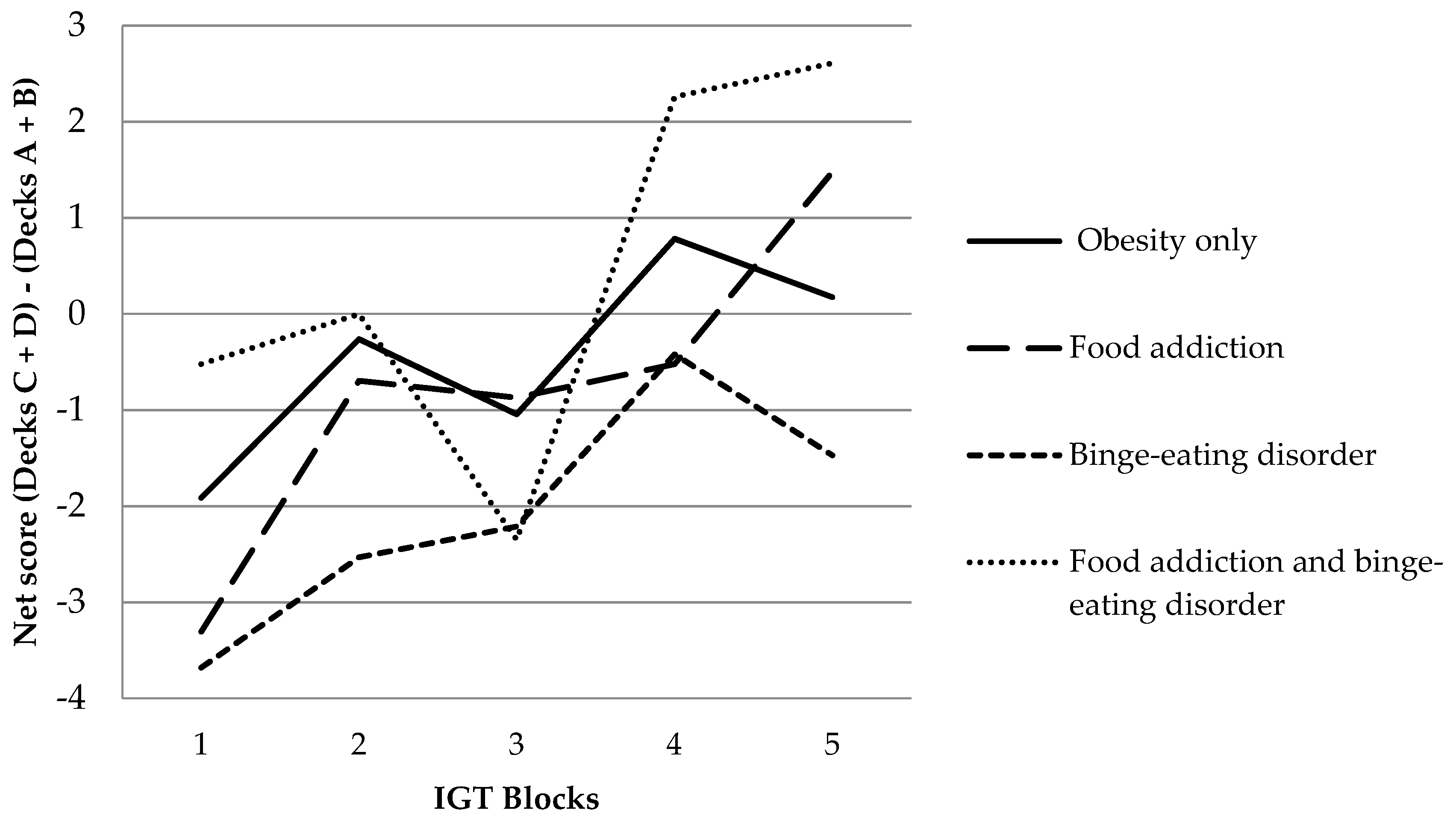
| IGT | Net score | −2.26 (32.78) | −3.91 (32.18) | −10.32 (39.36) | 2.00 (35.17) | 0.37 | 3, 46 | 0.773 | −0.022 |
| DDT | Area under the curve | 0.47 (0.32) | 0.31 (0.26) | 0.54 (0.26) | 0.46 (0.24) | 2.80 | 3, 46 | 0.050 | 0.058 |
| Go/No Go | Commission errors | 11.61 (5.98) | 11.39 (7.88) | 9.74 (6.06) | 12.52 (7.22) | 0.64 | 3, 46 | 0.591 | −0.012 |
| WCST | Perseverative errors | 17.70 (6.67) | 17.40 (8.38) | 14.31 (4.75) | 18.38 (8.28) | 2.02 | 3, 46 | 0.125 | 0.034 |
| Learning to learn 1 | 2.01 (4.22) a | 0.88 (6.60) a, b | −1.45 (3.90) b | 3.70 (10.08) a, b | 2.94 | 3, 40 | 0.045 | 0.069 | |
| WAFA | Mean reaction time intrinsic in ms | 254.22 (45.69) | 264.61 (45.02) | 265.37 (43.22) | 266.00 (72.05) | 0.30 | 3, 46 | 0.824 | −0.024 |
| Mean reaction time phasic in ms | 253.78 (57.09) | 242.04 (61.98) | 223.63 (88.57) | 249.00 (95.92) | 0.56 | 3, 45 | 0.642 | −0.015 | |
| Measure of dispersion intrinsic | 1.18 (0.67) | 1.24 (0.11) | 1.18 (0.06) | 1.28 (0.39) | 2.53 | 3, 45 | 0.069 | 0.050 | |
| Measure of dispersion phasic | 1.41 (0.18) a | 1.56 (0.50) a, b | 2.29 (1.15) b | 1.79 (1.02) a, b | 4.80 | 3, 37 | 0.006 | 0.115 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blume, M.; Schmidt, R.; Hilbert, A. Executive Functioning in Obesity, Food Addiction, and Binge-Eating Disorder. Nutrients 2019, 11, 54. https://doi.org/10.3390/nu11010054
Blume M, Schmidt R, Hilbert A. Executive Functioning in Obesity, Food Addiction, and Binge-Eating Disorder. Nutrients. 2019; 11(1):54. https://doi.org/10.3390/nu11010054
Chicago/Turabian StyleBlume, Marie, Ricarda Schmidt, and Anja Hilbert. 2019. "Executive Functioning in Obesity, Food Addiction, and Binge-Eating Disorder" Nutrients 11, no. 1: 54. https://doi.org/10.3390/nu11010054
APA StyleBlume, M., Schmidt, R., & Hilbert, A. (2019). Executive Functioning in Obesity, Food Addiction, and Binge-Eating Disorder. Nutrients, 11(1), 54. https://doi.org/10.3390/nu11010054






