Short-Term Fish Oil Treatment Changes the Composition of Phospholipids While Not Affecting the Expression of Mfsd2a Omega-3 Transporter in the Brain and Liver of the 5xFAD Mouse Model of Alzheimer’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Treatments
2.3. Lipids Extraction
2.4. Phospholipid Analysis
2.5. Western Blotting
2.6. Real Time Quantitative Polymerase Chain Reaction (RT-PCR)
2.7. Plaque Quantification
2.8. Statistics
3. Results
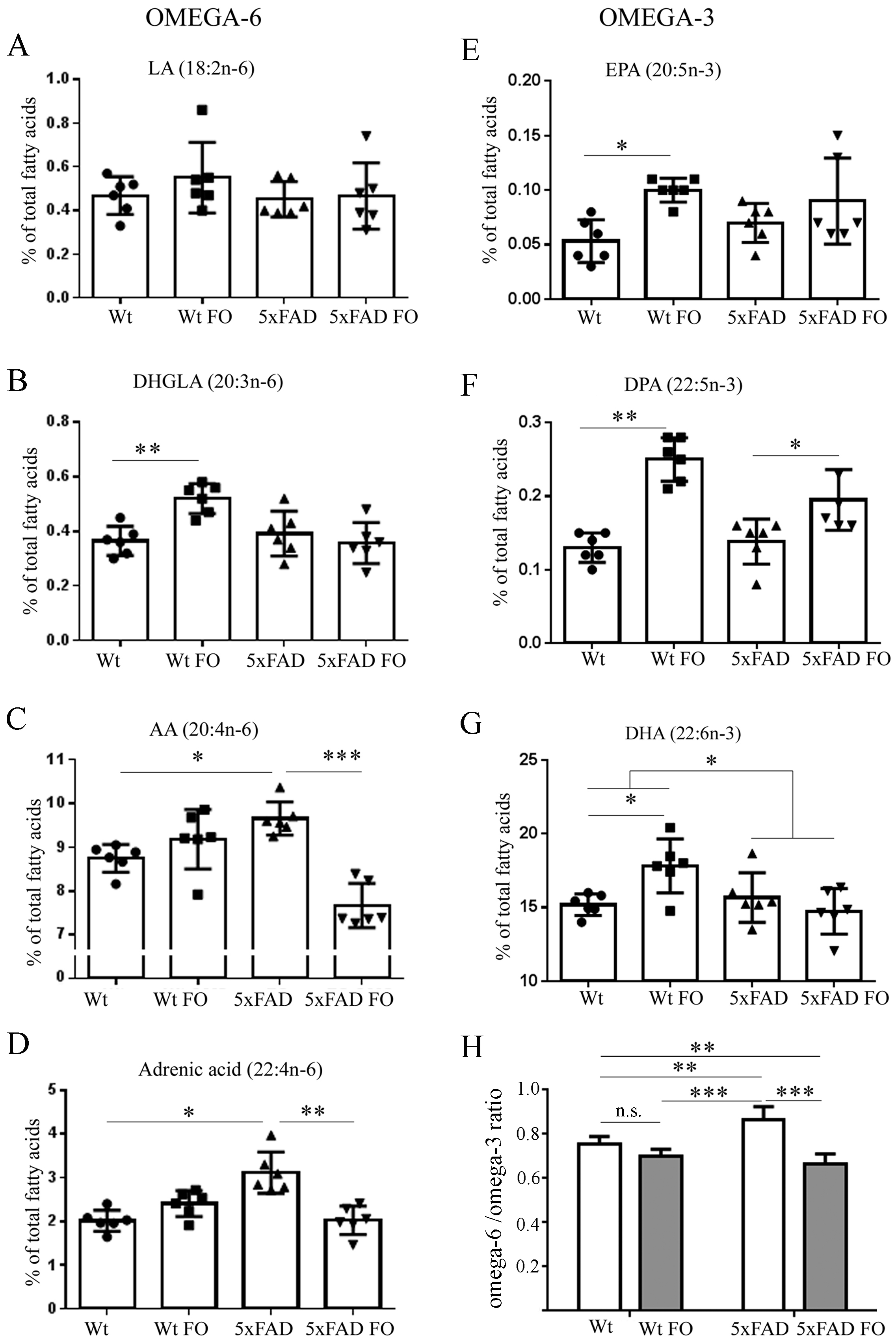
3.1. Fish Oil (FO) Differentially Affected n-3 and n-6 Polyunsaturated Fatty Acids (PUFA) Content in the Brains of the Wild-Type (WT) and 5xFAD Mice
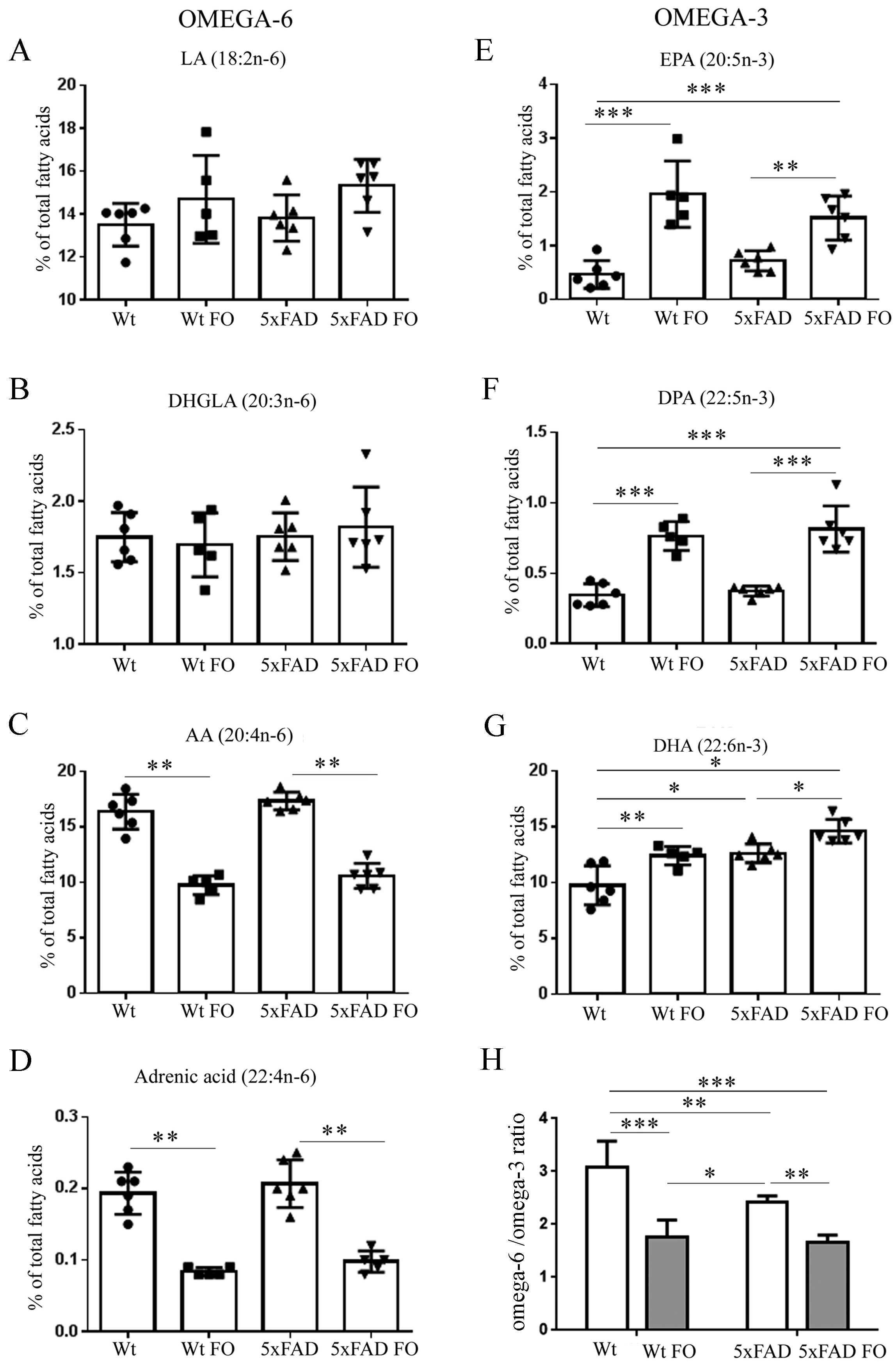
3.2. FO Induced a Similar Pattern of Changes in n-3 and n-6 PUFA Levels in the Livers of Both Genotypes
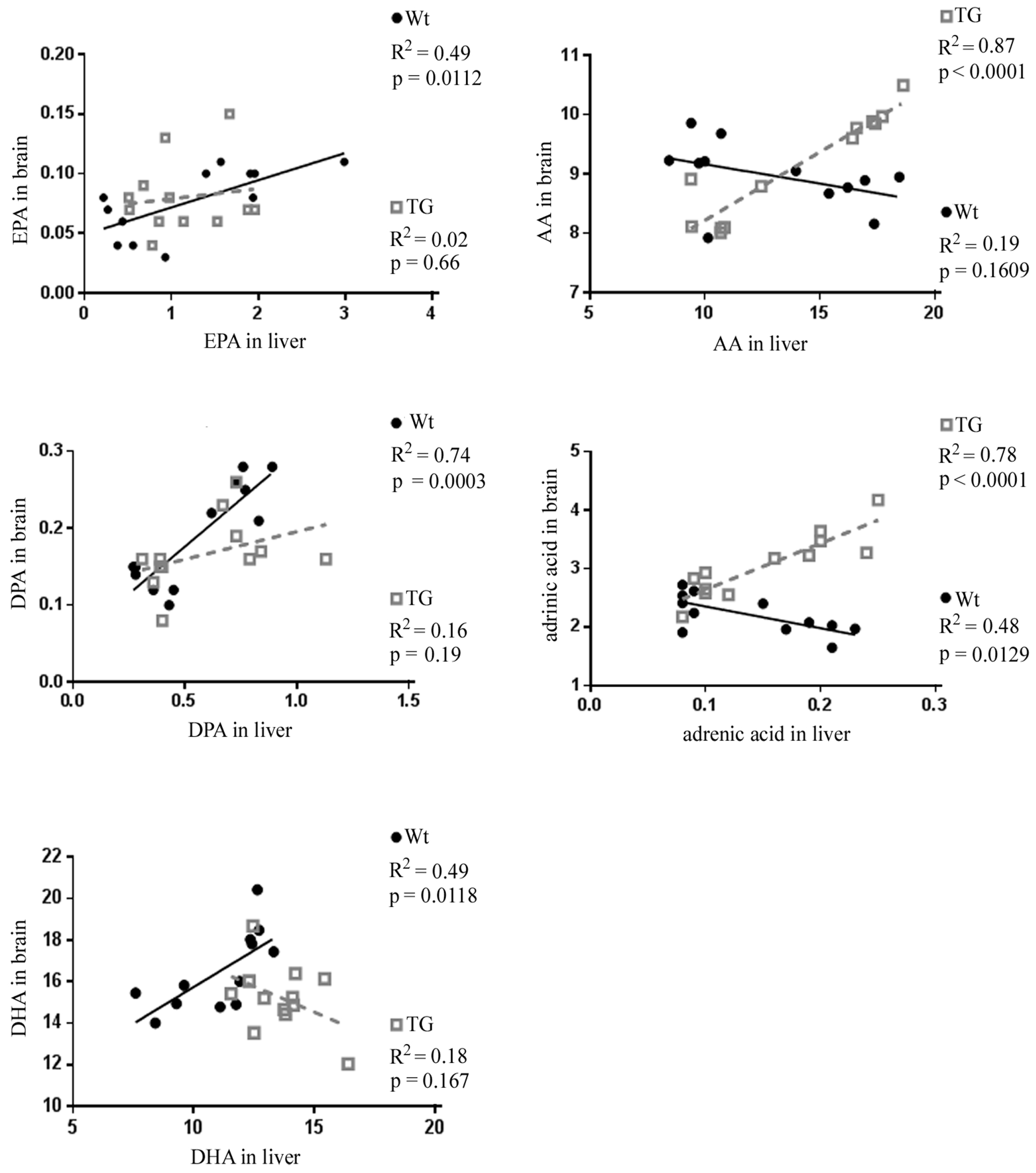
3.3. Correlation of Individual Omega-6 and Omega-3 Fatty Acids between Liver and Brain
3.4. Effects of FO Supplementation on Total PUFA, Mono-Unsaturated Fatty Acids (MUFA) and Saturated Fatty Acids (SFA) in the Brains and Livers of Wt and 5xFAD Mice
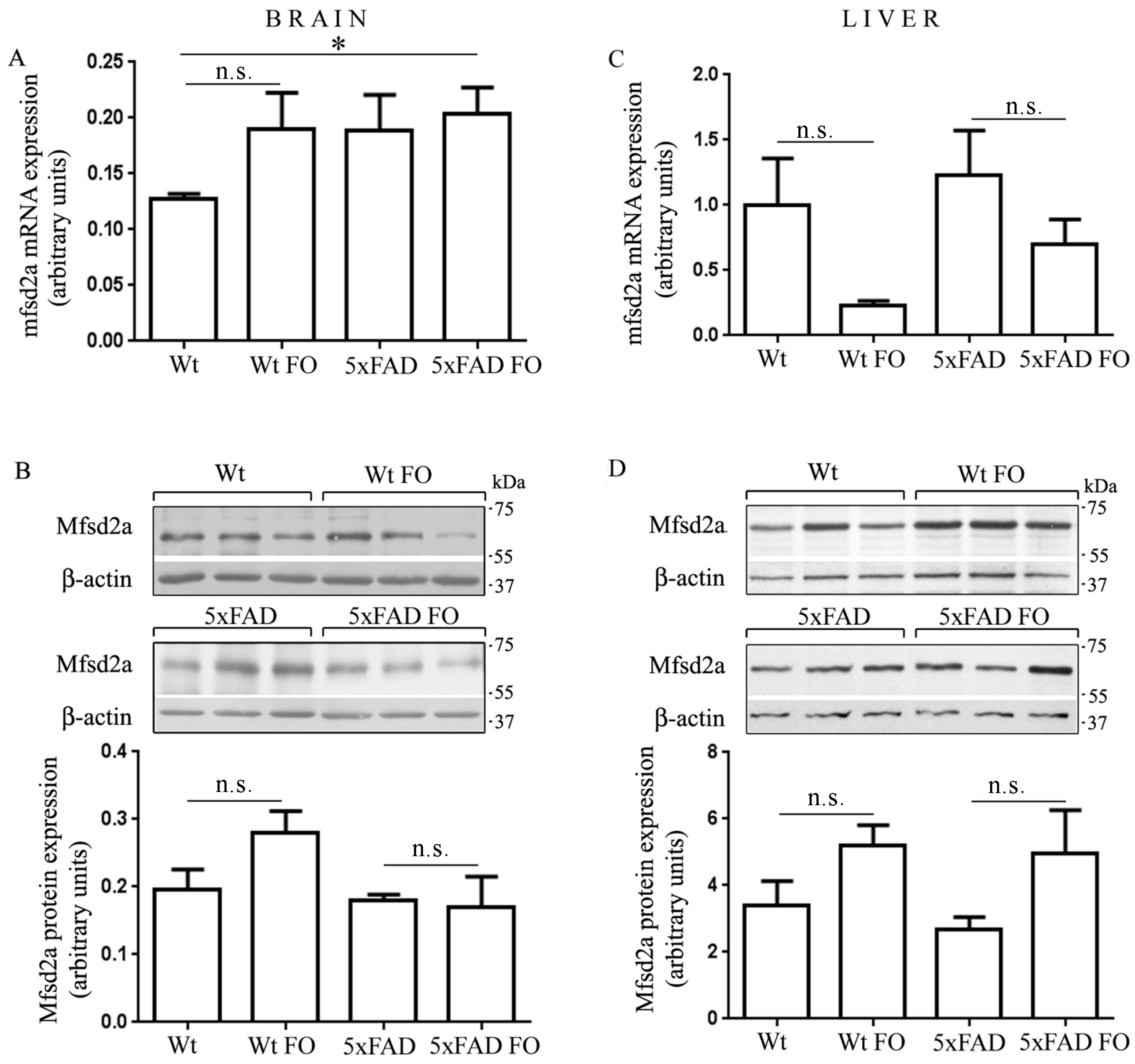
3.5. The Expression of Mfsd2a Transporter Remained Unchanged in the Livers and Brains of Wt and 5xFAD Mice Following FO Supplementation
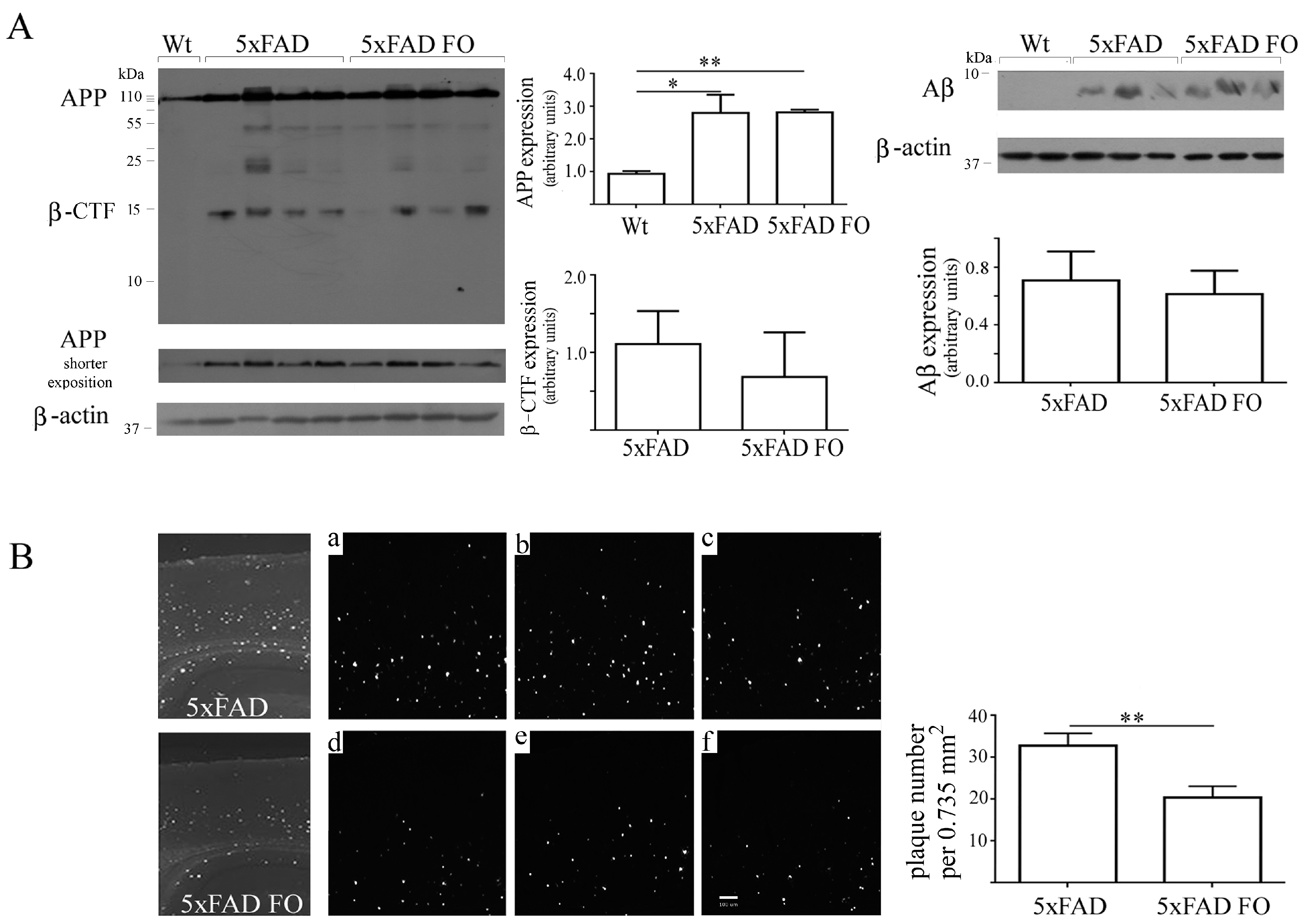
3.6. FO Supplementation Decreased the Number of Aβ Plaques in the Parietal Cortex of 5xFAD Mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bazinet, R.P.; Laye, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Zárate, R.; El Jaber-Vazdekis, N.; Tejera, N.; Pérez, J.A.; Rodríguez, C. Significance of long chain polyunsaturated fatty acids in human health. Clin. Trans. Med. 2017, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Teng, E.; Taylor, K.; Bilousova, T.; Weiland, D.; Pham, T.; Zuo, X.; Yang, F.; Chen, P.P.; Glabe, C.G.; Takacs, A.; et al. Dietary DHA supplementation in an APP/PS1 transgenetic rat model of AD reduces behavioral and Aβ pathology and modulates Aβ oligomerization. Neurobiol. Dis. 2015, 82, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Casali, B.T.; Corona, A.W.; Mariani, M.M.; Karlo, J.C.; Ghosal, K.; Landreth, G.E. Omega-3 fatty acids augment the actions of nuclear receptor agonists in a mouse model of Alzheimer’s disease. J. Neurosci. 2015, 35, 9173–9181. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, D.; Julien, C.; Tremblay, C.; Calon, F. DHA improves cognition and prevents dysfunction of entorhinal cortex neurons in 3xTg-AD mice. PLoS ONE 2011, 6, e17397. [Google Scholar] [CrossRef] [PubMed]
- Green, K.N.; Martinez-Coria, H.; Khashwji, H.; Hall, E.B.; Yurko-Mauro, K.A.; Ellis, L.; LaFerla, F.M. Dietary docosahexaenoic acid and docosapentaenoic acid ameliorate amyloid-â and tau pathology via a mechanism involving presenilin 1 levels. J. Neurosci. 2007, 27, 4385–4395. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Van der Zee, C.E.; Dederen, P.J.; Brouwer, K.M.; Reijmer, Y.D.; van Groen, T.; Broersen, L.M.; Lutjohann, D.; Heerschap, A.; Kiliaan, A.J. DHA and cholesterol containing diets influence Alzheimer-like pathology, cognition and cerebral vasculature in APPswe/PS1dE9 mice. Neurobiol. Dis. 2009, 33, 482–498. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.P.; Calon, F.; Morihara, T.; Yang, F.; Teter, B.; Ubeda, O.; Salem, N., Jr.; Frautschy, S.A.; Cole, G.M. A diet enriched with the omega-3 fatty acid docosahexaenoic acid reduces amyloid burden in an aged Alzheimer mouse model. J. Neurosci. 2005, 25, 3032–3040. [Google Scholar] [CrossRef] [PubMed]
- Oksman, M.; Iivonen, H.; Hogyes, E.; Amtul, Z.; Penke, B.; Leenders, I.; Broersen, L.; Lütjohann, D.; Hartmann, T.; Tanila, H. Impact of different saturated fatty acid, polyunsaturated fatty acid and cholesterol containing diets on beta-amyloid accumulation in APP/PS1 transgenic mice. Neurobiol. Dis. 2006, 23, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Perez, S.E.; Berg, B.M.; Moore, K.A.; He, B.; Counts, S.E.; Fritz, J.J.; Hu, Y.S.; Lazarov, O.; Lah, J.J.; Mufson, E.J. DHA diet reduces AD pathology in young APPswe/PS1 Delta E9 transgenic mice: Possible gender effects. J. Neurosci. Res. 2010, 88, 1026–1040. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.L.; Yang, F.; Rosario, E.R.; Ubeda, O.J.; Beech, W.; Gant, D.J.; Chen, P.P.; Hudspeth, B.; Chen, C.; Zhao, Y.; et al. Beta-amyloid oligomers induce phosphorylation of tau and inactivation of insulin receptor substrate via c-Jun N-terminal kinase signaling: Suppression by omega-3 fatty acids and curcumin. J. Neurosci. 2009, 29, 9078–9089. [Google Scholar] [CrossRef] [PubMed]
- Calon, F.; Lim, G.P.; Yang, F.; Morihara, T.; Teter, B.; Ubeda, O.; Rostaing, P.; Triller, A.; Salem, N.J.R.; Ashe, K.H.; et al. Docosahexaenoic acid protects from dendritic pathology in an Alzheimer’s disease mouse model. Neuron 2004, 43, 633–645. [Google Scholar] [CrossRef] [PubMed]
- Trépanier, M.O.; Hopperton, K.E.; Orr, S.K.; Bazinet, R.P. N-3 polyunsaturated fatty acids in animal models with neuroinflammation: An update. Eur. J. Pharmacol. 2016, 785, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Labrousse, V.F.; Nadjar, A.; Joffre, C.; Costes, L.; Aubert, A.; Grégoire, S.; Bretillon, L.; Layé, S. Short-term long chain omega3 diet protects from neuroinflammatory processes and memory impairment in aged mice. PLoS ONE 2012, 7, e36861. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Hossain, S.; Al Mamun, A.; Matsuzaki, K.; Arai, H. Docosahexaenoic acid: One molecule diverse functions. Crit. Rev. Biotechnol. 2017, 37, 579–597. [Google Scholar] [CrossRef] [PubMed]
- Wurtman, R.J. A nutrient combination that can affect synapse formation. Nutrients 2014, 6, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Lebbadi, M.; Julien, C.; Phivilay, A.; Tremblay, C.; Emond, V.; Kang, J.X.; Calon, F. Endogenous conversion of omega-6 into omega-3 fatty acids improves neuropathology in an animal model of Alzheimer’s disease. J. Alzheimers Dis. 2011, 27, 853–869. [Google Scholar] [CrossRef] [PubMed]
- Mohaibes, R.J.; Fiol-deRoque, M.A.; Torres, M.; Ordinas, M.; López, D.J.; Castro, J.A.; Escribá, P.V.; Busquets, X. The hydroxylated form of docosahexaenoic acid (DHA-H) modifies the brain lipid composition in a model of Alzheimer’s disease, improving behavioral motor function and survival. Biochim. Biophys. Acta 2017, 1859, 1596–1603. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Marcilla-Etxenike, A.; Fiol-deRoque, M.A.; Escribá, P.V.; Busquets, X. The unfolded protein response in the therapeutic effect of hydroxy-DHA against Alzheimer’s disease. Apoptosis 2015, 20, 712–724. [Google Scholar] [CrossRef] [PubMed]
- Fiol-Deroque, M.A.; Gutierrez-Lanza, R.; Terés, S.; Torres, M.; Aarceló, P.; Rial, R.V.; Escriba, P.V.; Busquets, X.; Rodriguez, J.J. Cognitive recovery and restoration of cell proliferation in the dentate gyrus in the XFAD transgenic mice model of Alzheimer’s disease following-hydroxy-DHA treatment. Biogerontology 2013, 14, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Huang, X.; Zhen, J.; Van Halm-Lutterodt, N.; Wang, J.; Zhou, C.; Yuan, L. Dietary vitamin E status dictates oxidative stress outcomes by modulating effects of fish oil supplementation in Alzheimer disease model APPswe/PS1dE9 mice. Mol. Neurobiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hooperton, K.E.; Trépanier, M.O.; James, N.C.E.; Chouinard-Watkins, R.; Bazinet, R.P. Fish oil feeding attenuates neuroinflammatory gene expression without concomitant changes in brain eicosanoids and docosanoids in a mouse model of Alzheimer’s disease. Brain Behav. Immun. 2018, 69, 74–90. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Busquets, X.; Escriba, P.V. Brain Lipids in the pathophysiology and treatment of Alzheimer’s disease. In Update of Dementia; Moretti, D., Ed.; Elsevier-Harvard: Chichago, IL, USA, 2016; pp. 127–167. [Google Scholar]
- Zhang, W.; Chen, R.; Yang, T.; Xu, N.; Chen, J.; Gao, Y.; Stetler, R.A. Fatty acid transporting proteins: Roles in brain development, aging, and stroke. Prostaglandins Leukot. Essent. Fatty Acids 2017. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.N.; Ma, D.; Shui, G.; Wong, P.; Cazenave-Gassiot, A.; Zhang, X.; Wenk, M.R.; Goh, E.L.; Silver, D.L. Mfsd2a is a transporter for the essentialomega-3 fatty aciddocosahexaenoic acid. Nature 2014, 509, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Ben-Zvi, A.; Lacoste, B.; Kur, E.; Andreone, B.J.; Mayshar, Y.; Yan, H.; Gu, C. Mfsd2a is critical for the formation and function of the blood-brain barrier. Nature 2014, 509, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Kahle, M.; Horsch, M.; Fridrich, B.; Seelig, A.; Schultheiß, J.; Leonhardt, J.; Irmler, M.; Beckers, J.; Rathkolb, B.; Wolf, E.; et al. Phenotypic omparison of common mouse strains developing high-fat diet-induced hepatosteatosis. Mol. Metab. 2013, 2, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.H.; Charron, M.J.; Silver, D.L. Major facilitator superfamily domain-containing protein 2a (MFSD2A) has roles in body growth, motor function, and lipid metabolism. PLoS ONE 2012, 7, e50629. [Google Scholar] [CrossRef] [PubMed]
- Astarita, G.; Jung, K.M.; Berchtpld, N.C.; Nguyen, V.Q.; Gillen, D.L.; Head, E.; Cotman, C.W.; Piomelli, D. Deficient liver biosynthesis of docosahexaenoic acid correlates with cognitive impairment in Alzheimer’s disease. PLoS ONE 2010, 5, e12538. [Google Scholar] [CrossRef] [PubMed]
- Bascoul-Colombo, C.; Guschina, I.A.; Maskrey, B.H.; Good, M.; O’Donnell, V.B.; Harwood, J.L. Dietary DHA supplementation causes selective changes in phospholipids from different brain regions in both wild type mice and the Tg2576 mouse model of Alzheimer’s disease. Biochim. Biophys. Acta 2016, 1861, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Price, S.L.; Fiol-Deroque, M.A.; Marcilla-Etxenike, A.; Ahyayauch, H.; AarcelóCoblijn, G.; Barceló-Coblijn, G.; Terés, S.; Katsouri, L.; Ordinas, M.; et al. Membrane lipid modifications and therapeutic effects mediated by hydroxydocosahexaenoic acid on Alzheimer’s disease. Biochim. Biophys. Acta 2014, 1838, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Begg, D.P.; Barr, D.; Garg, M.; Cameron-Smith, D.; Sinclair, A.J. Short-term docosapentaenoic acid (22:5n-3) supplementation increases tissue docosapentaenoic acid, DHA and EPA concentrations in rats. Br. J. Nutr. 2010, 103, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Ibarguren, M.; López, D.J.; Escribá, P.V. The effect of natural and synthetic fatty acids on membrane structure, microdomain organization, cellular functions and human health. Biochim. Biophys. Acta 2014, 1838, 1518–1528. [Google Scholar] [CrossRef] [PubMed]
- Ibarguren, M.; López, D.J.; Encinar, J.A.; González-Ros, J.M.; Busquets, X.; Escribá, P.V. Partitioning of liquid-ordered/liquid-disordered membrane microdomains induced by the fluidifying effect of -hydroxylated fatty acid derivatives. Biochim. Biophys. Acta 2013, 1828, 2553–2563. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.W.; Kuchenbecker, J.; Grosgen, S.; Burg, V.K.; Hundsdorfer, B.; Rothhaar, T.L.; Friess, P.; De Wilde, M.C.; Broersen, L.M.; Penke, B.; et al. Docosahexaenoic acid reduces amyloid β production via multiple pleiotropic mechanisms. J. Biol. Chem. 2011, 286, 14028–14039. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.W.; Mett, J.; Grimm, H.S.; Hartmann, T. APP Function and Lipids: A Bidirectional Link. Front. Mol. Neurosci. 2017, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Oakley, H.; Cole, S.L.; Logan, S.; Maus, E.; Shao, P.; Craft, J.; Guillozet-Bongaarts, A.; Ohno, M.; Disterhoft, J.; Van Eldik, L.; et al. Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: Potential factors in amyloid plaque formation. J. Neurosci. 2006, 26, 10129–10140. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, S.; Navarro, V.; Moyano, J.; Sanchez-Mico, M.; Torres, M.; Davila, J.C.; Vizuete, M.; Gutierrez, A.; Vitorica, J. Disruption of amyloid plaques integrity affects the soluble oligomers content from Alzheimer disease brains. PLoS ONE 2014, 9, e114041. [Google Scholar] [CrossRef] [PubMed]
- Wagner, L.K.; Gilling, K.E.; Schormann, E.; Kloetzel, P.M.; Heppner, F.L.; Krüger, E.; Prokop, S. Immunoproteasome deficiency alters microglial cytokine response and improves cognitive deficits in Alzheimer’s disease-like APPPS1 mice. Acta Neuropathol. Commun. 2017, 5–52. [Google Scholar] [CrossRef] [PubMed]
- Joly, S.; Lamoureux, S.; Pernet, V. Nonamyloidogenic processing of amyloid beta precursor protein is associated with retinal function improvement in aging male APPswe/PS1ΔE9 mice. Neurobiol. Aging 2017, 53, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Mejia, O.R.; Mucke, L. Phospholipase A2 and arachidonic acid in Alzheimer’s disease. Biochim. Biophys. Acta 2010, 1801, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.H.; Pelleieux, S.; Vitale, N.; Olivier, J.L. Dietary arachidonic acid as a risk factor for age-associated neurodegenerative diseases: Potential mechanisms. Biochimie 2016, 130, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Arendash, G.W.; Jensenn, M.T.; Salem, N.J.R.; Husein, N.; Cracchiolo, J.; Dickson, A.; Leighty, R.; Potter, H. A diet high in omega-3 fatty acids does not improve or protect cognitive performance in Alzheimer’s transgenic mice. Neuroscience 2007, 149, 286–3027. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, T.; Loewke, J.; Garrison, M.; Catalan, J.N.; Salem, N.J.R. Reversal of docosahexaenoic acid deficiency in the rat brain, retina, liver, and serum. J. Lipid Res. 2001, 42, 419–427. [Google Scholar] [PubMed]
- Chouinard-Watkins, R.; Pincon, A.; Coulombe, J.D.; Spencer, R.; Massenavette, L.; Plourde, M. A diet rich in docosahexaenoic acid restores liver arachidonic acid and docosahexaenoic acid concentrations in mice homozygous for the human apolipoprotein E ε4 allele. J. Nutr. 2016, 146, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Vauzour, D.; Martinsen, A.; Laye, S. Neuroinflmmatory processes in cognitive disorders: Is there a role for flavonoids and n-3 polyunsaturated fatty acids in counteracting their detrimental effects? Neurochem. Int. 2015, 89, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Mas, E.L.; Croft, K.D.; Zahra, P.; Barden, A.; Mori, T.A. Resolvins D1, D2, and other mediators of self-limited resolution of inflammation in human blood following n-3 fatty acid supplementation. Clin. Chem. 2012, 58, 1476–1484. [Google Scholar] [CrossRef] [PubMed]
- Lukiw, L.; Cui, J.G.; Marcheselli, V.L.; Bodker, M.; Botkjaer, A.; Gotlinger, K.; Serhan, C.N.; Bazan, N.G. JCI25420A role for docosahexaenoic acid–derived neuroprotectin D1 in neural cell survival and Alzheimer disease. J. Clin. Investig. 2005, 115, 2774–2783. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Pasker-de Jong, P.; de Vries, R.; Ritskes-Hoitinga, M. The effect of long term omega-3 fatty acid supplementation on cognition and Alzheimer’s pathology in animal models of Alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimers Dis. 2012, 28, 191–209. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, H.I.L.; Van Boxtel, M.P.J.; Jolles, J.; Verhey, F.R.J.; Uylings, H.B.M. Parietal cortex matters in Alzheimer’s disease: An overview of structural, functional and metabolic findings. Neurosci. Biobehav. Rev. 2012, 36, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, H.; Grimm, M.O.; Rothhaar, T.L.; Berkecz, R.; Lutjohann, D.D.; Giniatullina, R.; Takalo, M.; Miettinen, P.O.; Lahtinen, H.M.; Giniatullin, R.; et al. Special lipid-based diets alleviate cognitive deficits in the APPswe/PS1dE9 transgenetic mouse model of Alzheimer’s disease independent of brain amyloid deposition. J. Nutr. Biochem. 2014, 25, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Landel, V.; Baranger, K.; Virard, I.; Loriod, B.; Khrestchatisky, M. Temporal gene profiling of the 5XFAD transgenic mouse model highlights the importance of microglial activation in Alzheimer’s disease. Mol. Neurodegener. 2014, 9, 33. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Carson, M.J.; El Khoury, J.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L.; Jacobs, A.H.; Wyss-Coray, T.; Vitorica, J.; Ransohoff, R.M.; et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef]
- Condello, C.; Yuan, P.; Schain, A.; Grutzendler, J. Microglia constitute a barrier that prevents neurotoxic protofibrillar Ab42 hotspots around plaques. Nat. Commun. 2015, 6, 6176. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Luehmann, M.; Spires-Jones, T.L.; Prada, C.; Garcia-Alloza, M.; De Calignon, A.; Rozkalne, A.; Koenigsknecht-Talboo, J.; Holtzman, D.M.; Bacskai, B.J.; Hyman, B.T. Rapid appearance and local toxicity of amyloid-β plaques in a mouse model of Alzheimer’s disease. Nature 2008, 451, 720–724. [Google Scholar] [CrossRef] [PubMed]
| Content | Percentage |
|---|---|
| Protein | 17.2% |
| Carbohydrate | 60.9% |
| Fat | 3.7% |
| PUFA/SFA | 1.3 |
| n-3/n-6 PUFA | 0.05 |
| fiber | 5.6% |
| ash | 7.6% |
| adequate amount of vitamins and minerals | |
| Animals | 0 Week | 1 Week | 2 Week | 3 Week |
|---|---|---|---|---|
| Wt | 20.22 ± 0.529 | 20.53 ± 0.629 | 20.88 ± 0.419 | 20.80 ± 0.394 |
| Wt FO | 20.98 ± 0.627 | 21.62 ± 0.517 | 21.40 ± 0.494 | 21.32 ± 0.346 |
| 5xFAD | 21.56 ± 0.871 | 21.96 ± 1.352 | 21.85 ± 1.155 | 22.46 ± 1.256 |
| 5xFAD FO | 21.09 ± 0.681 | 21.07 ± 0.802 | 21.03 ± 0.664 | 21.26 ± 0.969 |
| FA Saturation | Number of Carbons | Common Name | Percentage |
|---|---|---|---|
| SFA | 16:0n | palmitic | 22.90 |
| 18:0n | stearic | 2.23 | |
| MUFA | 16:1n-7 | palmitoleic | 11.90 |
| 18:1n-9 | oleic | 10.25 | |
| 18:1n-7 | vaccenic | 4.54 | |
| n-6 | 18:2n-6 | linoleic | 1.67 |
| 20:3n-6 | dihomo-gama-linolenic | 0.29 | |
| 20:4n-6 | arachidonic | 1.62 | |
| 22:4n-6 | adrenic | 1.78 | |
| n-3 | 20:5n-3 | EPA | 25.51 |
| 22:5n-3 | DPA | 1.82 | |
| 22:6n-3 | DHA | 15.49 |
| FA (%) | Wt | Wt FO | 5xFAD | 5xFAD FO |
|---|---|---|---|---|
| Palmitic acid (16:0) | 22.33 ± 1.48 | 26.56 ± 1.47 a2 | 20.92 ± 0.89 | 24.87 ± 2.10 a,b1 |
| Stearic acid (18:0) | 22.67 ± 2.61 | 21.57 ± 1.57 | 21.57 ± 1.43 | 21.00 ± 1.59 |
| SFA | 45.01 ± 3.09 | 48.13 ± 1.73 | 42.48 ± 1.74 | 45.87 ± 2.63 b |
| Palmitoleic acid (16:1n-7) | 1.04 ± 0.30 | 0.83 ± 0.08 | 0.74 ± 0.16 | 0.66 ± 0.08 a |
| Oleic acid (18:1n-9) | 8.40 ± 0.48 | 7.47 ± 0.59 a | 7.33 ± 0.46 a1 | 6.56 ± 0.72 a2 |
| Vaccenic acid (18:1n-7) | 3.15 ± 0.54 | 2.21 ± 0.19 a1 | 2.58 ± 0.51 | 2.01 ± 0.43 a1 |
| MUFA | 12.59 ± 1.12 | 10.52 ± 0.67 a1 | 10.66 ±1.07 a | 9.23 ± 1.13 a2 |
| n-6 | 31.84 ± 1.81 | 26.21 ± 2.79 a1 | 33.13 ± 1.41 | 27.95 ± 1.12 a2,b2 |
| n-3 | 10.56 ± 1.89 | 15.14 ± 1.35 a2 | 13.73 ± 0.73 a1 | 16.95 ± 1.48 a2,b2 |
| PUFA | 42.40 ± 3.23 | 41.35 ± 2.05 | 46.86 ± 2.00 a | 44.90 ± 1.99 |
| FA (%) | Wt | Wt FO | 5xFAD | 5xFAD FO |
|---|---|---|---|---|
| Palmitic acid (16:0) | 28.49 ± 1.46 | 25.83 ± 2.57 a | 27.00 ± 1.85 | 29.11 ± 3.17 |
| Stearic acid (18:0) | 25.92 ± 3.0 | 22.94 ± 0.99 | 24.29 ± 2.20 | 26.89 ± 2.53 |
| SFA | 54.41 ± 22.69 | 48.77 ± 4.36 a1 | 51.29 ± 1.64 a | 56.01 ± 4.36 b |
| Palmitoleic acid (16:1n-7) | 0.46 ± 0.15 | 0.49 ± 0.13 | 0.37 ± 0.09 | 0.47 ± 0.10 |
| Oleic acid (18:1n-9) | 14.32 ± 1.52 | 15.02 ± 0.75 | 14.64 ± 1.67 | 14.05 ± 2.38 |
| Vaccenic acid (18:1n-7) | 3.85 ± 0.26 | 4.71 ± 0.54 a | 4.19 ± 0.77 | 3.93 ± 0.61 |
| MUFA | 18.62 ± 1.87 | 20.40 ± 0.31 a | 19.21 ± 2.33 | 18.45 ± 2.86 |
| n-6 | 11.60 ± 0.49 | 12.66 ± 0.99 a | 13.62 ± 0.85 a2 | 10.51 ± 0.66 a1,b2 |
| n-3 | 15.26 ± 0.71 | 18.17 ± 1.84 a1 | 15.88 ± 1.72 | 15.03 ± 1.56 |
| PUFA | 26.97 ± 1.06 | 30.83 ± 2.75 a1 | 29.50 ± 2.46 a | 25.54 ± 2.16 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milanovic, D.; Petrovic, S.; Brkic, M.; Avramovic, V.; Perovic, M.; Ivkovic, S.; Glibetic, M.; Kanazir, S. Short-Term Fish Oil Treatment Changes the Composition of Phospholipids While Not Affecting the Expression of Mfsd2a Omega-3 Transporter in the Brain and Liver of the 5xFAD Mouse Model of Alzheimer’s Disease. Nutrients 2018, 10, 1250. https://doi.org/10.3390/nu10091250
Milanovic D, Petrovic S, Brkic M, Avramovic V, Perovic M, Ivkovic S, Glibetic M, Kanazir S. Short-Term Fish Oil Treatment Changes the Composition of Phospholipids While Not Affecting the Expression of Mfsd2a Omega-3 Transporter in the Brain and Liver of the 5xFAD Mouse Model of Alzheimer’s Disease. Nutrients. 2018; 10(9):1250. https://doi.org/10.3390/nu10091250
Chicago/Turabian StyleMilanovic, Desanka, Snjezana Petrovic, Marjana Brkic, Vladimir Avramovic, Milka Perovic, Sanja Ivkovic, Marija Glibetic, and Selma Kanazir. 2018. "Short-Term Fish Oil Treatment Changes the Composition of Phospholipids While Not Affecting the Expression of Mfsd2a Omega-3 Transporter in the Brain and Liver of the 5xFAD Mouse Model of Alzheimer’s Disease" Nutrients 10, no. 9: 1250. https://doi.org/10.3390/nu10091250
APA StyleMilanovic, D., Petrovic, S., Brkic, M., Avramovic, V., Perovic, M., Ivkovic, S., Glibetic, M., & Kanazir, S. (2018). Short-Term Fish Oil Treatment Changes the Composition of Phospholipids While Not Affecting the Expression of Mfsd2a Omega-3 Transporter in the Brain and Liver of the 5xFAD Mouse Model of Alzheimer’s Disease. Nutrients, 10(9), 1250. https://doi.org/10.3390/nu10091250





