Contaminants in Grain—A Major Risk for Whole Grain Safety?
Abstract
1. Introduction
2. The Role of Whole Grains for Health
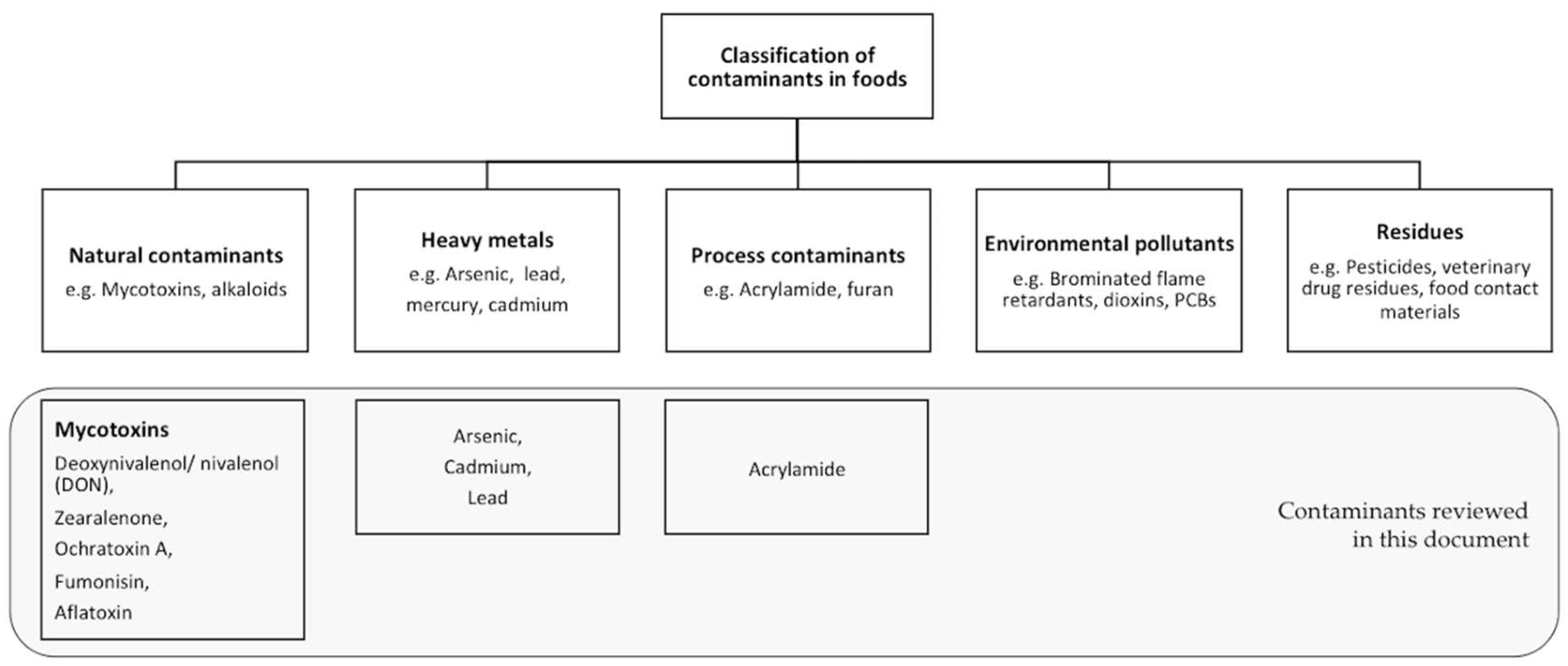
3. Types and Nature of Contaminants
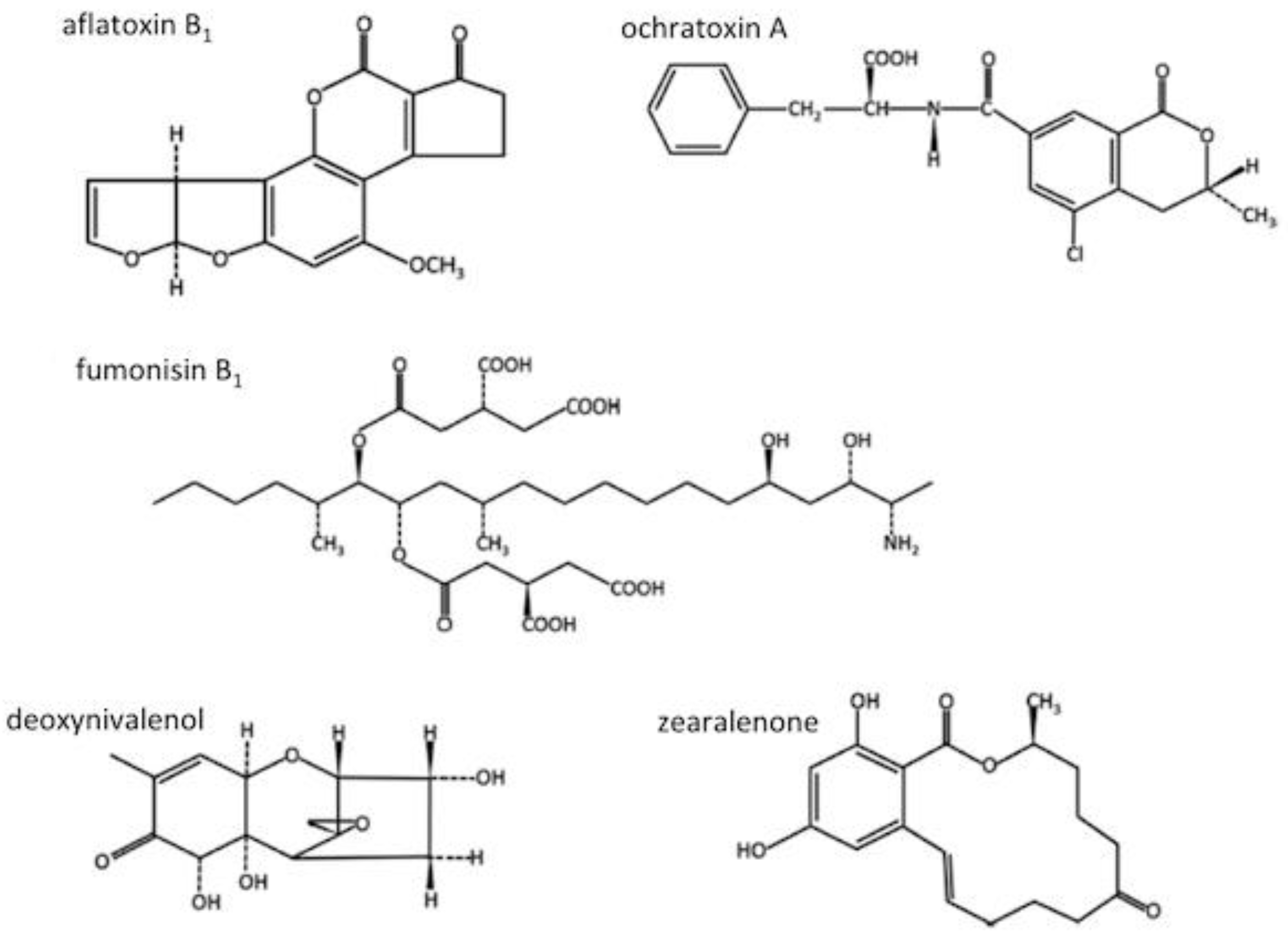
3.1. Mycotoxins
3.1.1. Deoxynivalenol/Nivalenol
3.1.2. Zearalenone
3.1.3. Ochratoxin A
3.1.4. Fumonisin B1
3.1.5. Aflatoxin
3.2. Metals as Contaminants
3.2.1. Cadmium
3.2.2. Arsenic
3.2.3. Lead
3.2.4. Mercury
3.3. Acrylamide as Process Contaminant
4. Magnitude of Mycotoxin Contribution from (Whole) Grains
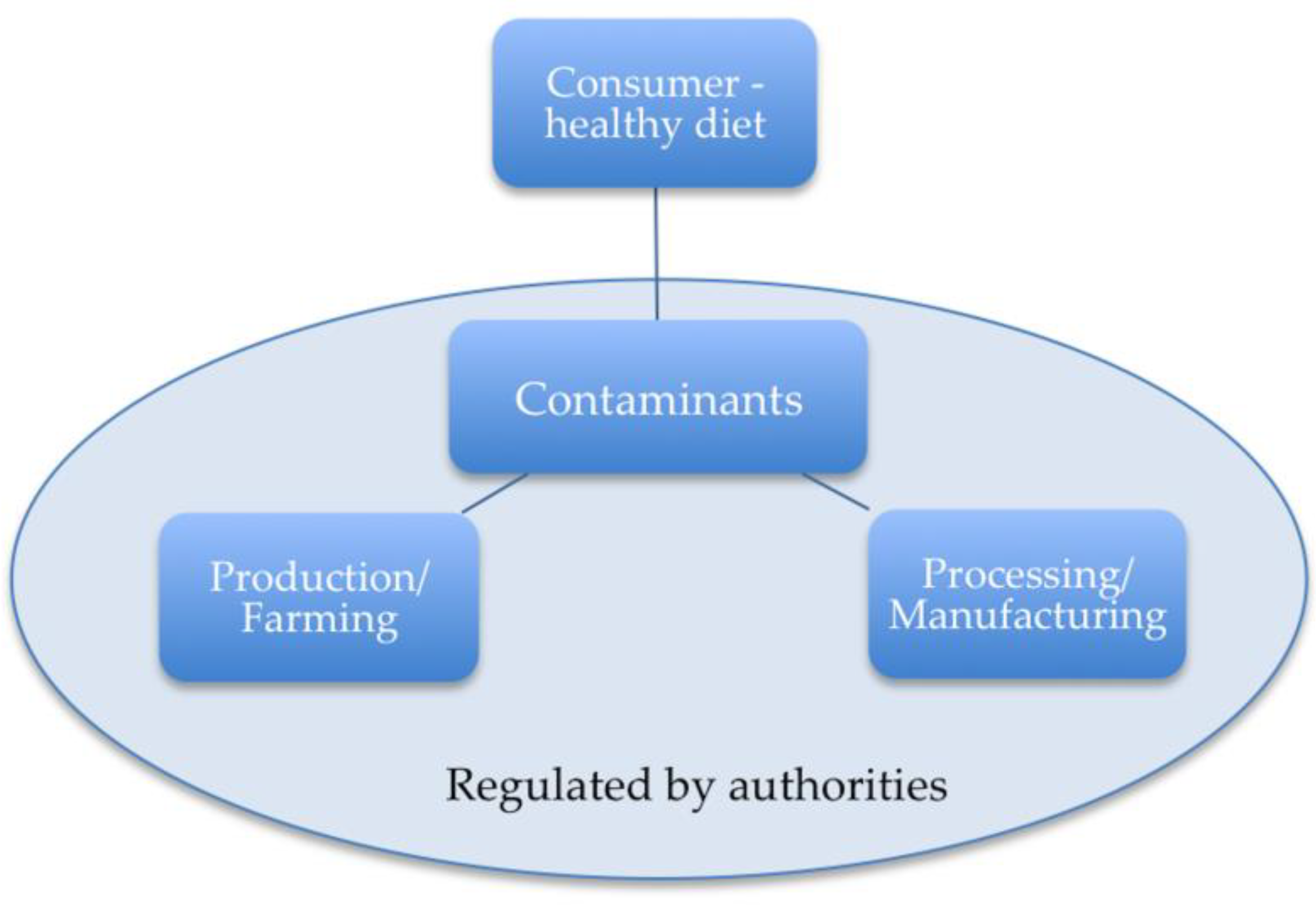
5. Strategies to Mitigate the Risk of Contaminants for Human Health
5.1. Regulations
5.2. Farming/Production
Organic vs. Conventional Farming
5.3. Processing/Manufacturing
Treatment
5.4. The Role of Whole Grains as Part of A Healthy Diet for the Mitigation of Mycotoxins and Toxic Metals
5.4.1. Nutritional Content of Whole Grain vs. Refined Grain
5.4.2. Mycotoxins
5.4.3. Toxic Metals
6. Final Considerations
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- FAO. Staple Foods: What Do People Eat? Available online: http://www.fao.org/docrep/u8480e/u8480e07.htm (accessed on 13 July 2018).
- Mercader, J. Mozambican grass seed consumption during the middle stone age. Science 2009, 326, 1680–1683. [Google Scholar] [CrossRef] [PubMed]
- FAO. World Food Situation. Available online: http://www.fao.org/worldfoodsituation/csdb/en/ (accessed on 13 August 2018).
- EC. Laying Down Community Procedures for Contaminants in Food; Offcial Journal of the European Communities: Brussels, Belgium, 1993. [Google Scholar]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Van der Kamp, J.W.; Poutanen, K.; Seal, C.J.; Richardson, D.P. The healthgrain definition of ‘whole grain’. Food Nutr. Res. 2014, 58, 22100. [Google Scholar] [CrossRef] [PubMed]
- AACC International. Whole Grains. Available online: http://www.aaccnet.org/initiatives/definitions/Pages/WholeGrain.aspx (accessed on 4 August 2018).
- Zong, G.; Gao, A.; Hu, F.B.; Sun, Q. Whole grain intake and mortality from all causes, cardiovascular disease, and cancer: A meta-analysis of prospective cohort studies. Circulation 2016, 133, 2370–2380. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Flint, A.J.; Qi, Q.; van Dam, R.M.; Sampson, L.A.; Rimm, E.B.; Holmes, M.D.; Willett, W.C.; Hu, F.B.; Sun, Q. Association between dietary whole grain intake and risk of mortality: Two large prospective studies in us men and women. JAMA Intern. Med. 2015, 175, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Benisi-Kohansal, S.; Saneei, P.; Salehi-Marzijarani, M.; Larijani, B.; Esmaillzadeh, A. Whole-grain intake and mortality from all causes, cardiovascular disease, and cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Adv. Nutr. 2016, 7, 1052–1065. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J.; Nugent, A.P.; Tee, E.S.; Thielecke, F. Whole-grain dietary recommendations: The need for a unified global approach. Br. J. Nutr. 2016, 115, 2031–2038. [Google Scholar] [CrossRef] [PubMed]
- Euromonitor. Food Trends for 2018. Available online: http://go.euromonitor.com/rs/805-KOK-719/images/8_Food_Trends_for_2018.pdf (accessed on 27 July 2018).
- Luis, S.; Andréia, B. From field to table: A review on the microbiological quality and safety of wheat-based products. Cereal Chem. 2016, 93, 105–115. [Google Scholar]
- Thorning, T.K.; Bertram, H.C.; Bonjour, J.P.; de Groot, L.; Dupont, D.; Feeney, E.; Ipsen, R.; Lecerf, J.M.; Mackie, A.; McKinley, M.C.; et al. Whole dairy matrix or single nutrients in assessment of health effects: Current evidence and knowledge gaps. Am. J. Clin. Nutr. 2017, 105, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Bertoia, M.L.; Triche, E.W.; Michaud, D.S.; Baylin, A.; Hogan, J.W.; Neuhouser, M.L.; Tinker, L.F.; Van Horn, L.; Waring, M.E.; Li, W.; et al. Mediterranean and dietary approaches to stop hypertension dietary patterns and risk of sudden cardiac death in postmenopausal women. Am. J. Clin. Nutr. 2014, 99, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J.; Brownlee, I.A. Whole-grain foods and chronic disease: Evidence from epidemiological and intervention studies. Proc. Nutr. Soc. 2015, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Albertson, A.M.; Reicks, M.; Joshi, N.; Gugger, C.K. Whole grain consumption trends and associations with body weight measures in the united states: Results from the cross sectional national health and nutrition examination survey 2001–2012. Nutr. J. 2016, 15, 8. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Lau, R.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dietary fibre, whole grains, and risk of colorectal cancer: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2011, 343, d6617. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Xu, M.; Lee, A.; Cho, S.; Qi, L. Consumption of whole grains and cereal fiber and total and cause-specific mortality: Prospective analysis of 367,442 individuals. BMC Med. 2015, 13, 59. [Google Scholar]
- Giacco, R.; Della Pepa, G.; Luongo, D.; Riccardi, G. Whole grain intake in relation to body weight: From epidemiological evidence to clinical trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Kassebaum, N.J.; Barber, R.M.; Bhutta, Z.A.; Dandona, L.; Gething, P.W.; Hay, S.I.; Kinfu, Y.; Larson, H.J.; Liang, X.; Lim, S.S.; et al. Global, regional, and national levels of maternal mortality, 1990–2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1775–1812. [Google Scholar] [CrossRef]
- FDA. Health Claim Notification for Whole Grain Foods. Available online: https://www.fda.gov/food/labelingnutrition/ucm073639.htm (accessed on 2 August 2018).
- Ross, A.B.; van der Kamp, J.W.; King, R.; Le, K.A.; Mejborn, H.; Seal, C.J.; Thielecke, F.; Healthgrain, F. Perspective: A definition for whole-grain food products-recommendations from the healthgrain forum. Adv. Nutr. 2017, 8, 525–531. [Google Scholar] [PubMed]
- Whole Grain Stamp. Available online: https://wholegrainscouncil.org/whole-grain-stamp (accessed on 2 August 2018).
- Contaminants in Food and Feed. Available online: https://www.efsa.europa.eu/en/topics/topic/contaminants-food-and-feed (accessed on 2 July 2018).
- EFSA. The 2016 european union report on pesticide residues in food. EFSA J. 2016, 16. [Google Scholar] [CrossRef]
- Alshannaq, A.; Yu, J.H. Occurrence, toxicity, and analysis of major mycotoxins in food. Int. J. Environ. Res. Public Health 2017, 14, 632. [Google Scholar] [CrossRef] [PubMed]
- Scudamore, K.A.; Patel, S. Fusarium mycotoxins in milling streams from the commercial milling of maize imported to the UK, and relevance to current legislation. Food Addit Contam Part A Chem. Anal. Control Expo. Risk Assess. 2009, 26, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.W.; Bentley, R. Pride and Prejudice: The Story of Ergot. Perspect. Biol. Med. 1999, 42, 333–355. [Google Scholar] [CrossRef]
- Lee, H.J.; Ryu, D. Worldwide occurrence of mycotoxins in cereals and cereal-derived food products: Public health perspectives of their co-occurrence. J. Agric. Food Chem. 2017, 65, 7034–7051. [Google Scholar] [CrossRef] [PubMed]
- Moretti, A.; Logrieco, A.F.; Susca, A. Mycotoxins: An underhand food problem. Methods Mol. Biol. 2017, 1542, 3–12. [Google Scholar] [PubMed]
- Van der Fels-Klerx, H.J.; van Asselt, E.D.; Madsen, M.S.; Olesen, J.E. Impact of climate change effects on contamination of cereal grains with deoxynivalenol. PLoS ONE 2013, 8, e73602. [Google Scholar] [CrossRef] [PubMed]
- Marvin, H.J.P. Emtox: Climate change impacts on natural toxins in marine and primary plant production system in north west europe by 2040. Food Addit. Contam. Part A 2012, 29, 1501. [Google Scholar] [CrossRef] [PubMed]
- Battilani, P.; Toscano, P.; Van der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin b1 contamination in maize in europe increases due to climate change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef] [PubMed]
- IARC. Some Industrial Chemicals; International Agency for Research on Cancer: Lyon, France, 1994; p. 435. [Google Scholar]
- CTA. Mycotoxins in Grain; Technical Centre for Agricultural and Rural Cooperation ACP-EU: Wageningen, The Netherlands, 1997. [Google Scholar]
- Sobrova, P.; Adam, V.; Vasatkova, A.; Beklova, M.; Zeman, L.; Kizek, R. Deoxynivalenol and its toxicity. Interdiscip. Toxicol. 2010, 3, 94–99. [Google Scholar] [CrossRef] [PubMed]
- FDA. Guidance for Industry and FDA: Advisory Levels for Deoxynivalenol (don) in Finished Wheat Products for Human Consumption and Grains and Grain by-Products Used for Animal Feed. Available online: https://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/ChemicalContaminantsMetalsNaturalToxinsPesticides/ucm120184.htm (accessed on 20 July 2018).
- Kowalska, K.; Habrowska-Gorczynska, D.E.; Piastowska-Ciesielska, A.W. Zearalenone as an endocrine disruptor in humans. Environ. Toxicol. Pharmacol. 2016, 48, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Bui-Klimke, T.R.; Wu, F. Ochratoxin a and human health risk: A review of the evidence. Crit. Rev. Food Sci. Nutr. 2015, 55, 1860–1869. [Google Scholar] [CrossRef] [PubMed]
- FDA. Guidance for Industry: Fumonisin Levels in Human Foods and Animal Feeds. Available online: https://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/ucm109231.htm (accessed on 1 August 2018).
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed—Focus on europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbiol. 2016, 7, 2170. [Google Scholar] [CrossRef] [PubMed]
- Clemens, S.; Ma, J.F. Toxic heavy metal and metalloid accumulation in crop plants and foods. Annu. Rev. Plant Biol. 2016, 67, 489–512. [Google Scholar] [CrossRef] [PubMed]
- Andrade, V.M.; Aschner, M.; Dos Santos, A.P.M. Neurotoxicity of metal mixtures. Adv. Neurobiol. 2017, 18, 227–265. [Google Scholar] [PubMed]
- Kosek-Hoehne, K.; Panocha, B.; Śliwa, A. Heavy metals—A silent threat to health. J. Educ. Health Sport 2017, 7, 121–132. [Google Scholar]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metals toxicity and the environment. Mol. Clin. Environ. Toxicol. 2012, 101, 133–164. [Google Scholar]
- Srikanth, R.; Ramana, D.; Rao, V. Role of rice and cereal products in dietary cadmium and lead intake among different socio-economic groups in south India. Food Addit. Contam. 1995, 12, 695–701. [Google Scholar] [CrossRef] [PubMed]
- WHO. Chemiclas of Public Health Concern and Their Management in the African Region. Available online: https://www.afro.who.int/sites/default/files/2017-06/9789290232810.pdf (accessed on 4 August 2018).
- Huo, J.; Huang, Z.; Li, R.; Song, Y.; Lan, Z.; Ma, S.; Wu, Y.; Chen, J.; Zhang, L. Dietary cadmium exposure assessment in rural areas of southwest China. PLoS ONE 2018, 13, e0201454. [Google Scholar] [CrossRef] [PubMed]
- BNF. Arsenic in Rice—Is It a Cause for Concern? Available online: https://www.nutrition.org.uk/nutritioninthenews/headlines/arsenicinrice.html (accessed on 26 July 2018).
- Greenerchoices. Report: Analysis of Arsenic in Rice and Other Grains. Executive Summary. Available online: http://greenerchoices.org/2014/11/01/test-results-arsenic-rice-grains/ (accessed on 13 July 2018).
- EFSA. Scientific opinion on arsenic in food. EFSA J. 2009, 7, 1351. [Google Scholar] [CrossRef]
- EFSA. Dietary exposure to inorganic arsenic in the european population. EFSA J. 2014, 12, 3597. [Google Scholar]
- EC. Commission Recommendation (Eu) 2015/1381 of 10 August 2015 on the Monitoring of Arsenic in Food; L 213/10; European Commission: Brussels, Belgium, 2015. [Google Scholar]
- McKevith, B. Nutritional aspects of cereals. Nutr. Bull. 2004, 29, 111–142. [Google Scholar] [CrossRef]
- Hershko, C. Lead poisoning by contaminated flour: An unfinished story. Harefuah 2005, 144, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Panariti, E.; Berxholi, K. Lead toxicity in humans from contaminated flour in Albania. Vet. Hum. Toxicol. 1998, 40, 91–92. [Google Scholar] [PubMed]
- Semla, M.; Goc, Z.; Martiniakova, M.; Omelka, R.; Formicki, G. Acrylamide: A common food toxin related to physiological functions and health. Physiol. Res. 2017, 66, 205–217. [Google Scholar] [PubMed]
- EC. Commission Regulation 2017/2158 Establishing Mitigation Measures and Benchmark Levels for the Reduction of the Presence of Acrylamide in Food; Official Journal of the European Union: Brussels, Belgium, 2017. [Google Scholar]
- FDA. Guidance for Industry Acrylamide in Foods. Available online: https://www.fda.gov/downloads/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/ChemicalContaminantsMetalsNaturalToxinsPesticides/UCM374534.pdf (accessed on 1 August 2018).
- Mycotoxins. Available online: https://www.world-grain.com/search?exclude_datatypes%5B%5D=blog&page=1&q=mycotoxins (accessed on 29 August 2018).
- Public Health England. NDNS: Results from Years 7 and 8 (Combined). Available online: https://www.gov.uk/government/statistics/ndns-results-from-years-7-and-8-combined (accessed on 27 August 2018).
- EFSA. Cadmium Dietary Exposure in the European European Food Safety Authority. Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2012.2551 (accessed on 5 August 2018).
- Public Health England. New Eatwell Guide Illustrates a Healthy, Balanced Diet. Available online: https://www.gov.uk/government/news/new-eatwell-guide-illustrates-a-healthy-balanced-diet (accessed on 3 August 2018).
- Veldman, A.; Meijs, J.A.C.; Borggreve, G.J.; Heeres-van der Tol, J.J. Carry-over of aflatoxin from cows’ food to milk. Anim. Sci. 1992, 55, 163–168. [Google Scholar] [CrossRef]
- Pulina, G.; Battacone, G.; Brambilla, G.; Cheli, F.; Danieli, P.P.; Masoero, F.; Pietri, A.; Ronchi, B. An update on the safety of foods of animal origin and feeds. Ital. J. Anim. Sci. 2014, 13, 3571. [Google Scholar] [CrossRef]
- FDA. Dough’s a Raw Deal and Could Make You Sick. Available online: https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm508450.htm (accessed on 30 August 2018).
- FAO; WHO. Codex General Standard for Contaminants and Toxins in Food and Feed; Codex Alimentarius: Rome, Italy, 1995. [Google Scholar]
- FAO; WHO. Code of Practice for the Prevention and Deduction of Mycotoxin Contamination in Cereals; Codex Alimentarius: Rome, Italy, 2003. [Google Scholar]
- Code of Practise for the Prevention and Reduction of Lead Contamination in Foods. Available online: www.fao.org/input/download/standards/10099/CXP_056e.pdf (accessed on 30 August 2018).
- EC. Setting Maximum Levels for Certain Contaminants in Foodstuffs and Amendments; European Commission: Brussels, Belgium, 2006. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- EC. Commission Regulation (EC) no 401/2006 of 23 February 2006 Laying down the Methods of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02006R0401-20100313&qid=1398850616000&from=EN (accessed on 1 August 2018).
- EC. Commission Regulation (EC) no 401/2006 of 23 February 2006 Laying down the Methods of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs; Offcial Journal of the European Communities: Brussels, Belgium, 2006. [Google Scholar]
- EC. Setting Maximum Levels for Certain Contaminants in Foodstuffs as Regards Fusarium Toxins in Maize and Maize Products; Offcial Journal of the European Communities: Brussels, Belgium, 2007. [Google Scholar]
- EC. Amending Regulation (EC) no 1881/2006 as Regards Maximum Levels of Inorganic Arsenic in Foodstuffs; Offcial Journal of the European Communities: Brussels, Belgium, 2015. [Google Scholar]
- FDA. CPG Sec. 555.400 Foods—Adulteration with Aflatoxin. Available online: https://www.fda.gov/ICECI/ComplianceManuals/CompliancePolicyGuidanceManual/ucm074555.htm (accessed on 1 August 2018).
- FDA. Inorganic Arsenic in Rice Cereals for Infants: Action Level Guidance for Industry. Draft Guidance. Available online: https://www.fda.gov/downloads/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/UCM493152.pdf (accessed on 1 August 2018).
- FDA. Chemical Contaminants, Metals, Natural Toxins & Pesticides Guidance Documents & Regulations. Available online: https://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/ChemicalContaminantsMetalsNaturalToxinsPesticides/default.htm (accessed on 1 August 2018).
- Zhao, F.J.; Ma, Y.; Zhu, Y.G.; Tang, Z.; McGrath, S.P. Soil contamination in China: Current status and mitigation strategies. Environ. Sci. Technol. 2015, 49, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Curtis, T.Y.; Halford, N.G. Reducing the acrylamide-forming potential of wheat. Food Energy Secur. 2016, 5, 153–164. [Google Scholar] [CrossRef]
- Lairon, D. Nutritional quality and safety of organic food. A review. Agron. Sustain. Dev. 2010, 30, 33–41. [Google Scholar] [CrossRef]
- Magkos, F.; Arvaniti, F.; Zampelas, A. Organic food: Buying more safety or just peace of mind? A critical review of the literature. Crit. Rev. Food Sci. Nutr. 2006, 46, 23–56. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Bertuzzi, T.; Comizzoli, S.; Turconi, G.; Roggi, C.; Pagani, M.; Cravedi, P.; Pietri, A. Preliminary survey on composition and quality of conventional and organic wheat. Ital. J. Food Sci. 2006, 18, 355–366. [Google Scholar]
- Akhandaf, Y.; Van Klaveren, J.; De Henauw, S.; Van Donkersgoed, G.; Van Gorcum, T.; Papadopoulos, A.; Sirot, V.; Kennedy, M.; Pinchen, H.; Ruprich, J.; et al. Exposure assessment within a total diet study: A comparison of the use of the pan-european classification system foodex-1 with national food classification systems. Food Chem. Toxicol. 2015, 78, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Brodal, G.; Hofgaard, I.S.; Eriksen, G.S.; Bernhoft, A.; Sundheim, L. Mycotoxins in organically versus conventionally produced cereal grains and some other crops in temperate regions. World Mycotoxin J. 2016, 9, 755–770. [Google Scholar] [CrossRef]
- Brera, C.; Catano, C.; de Santis, B.; Debegnach, F.; de Giacomo, M.; Pannunzi, E.; Miraglia, M. Effect of industrial processing on the distribution of aflatoxins and zearalenone in corn-milling fractions. J. Agric. Food Chem. 2006, 54, 5014–5019. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, T.; Ritieni, A.; Visone, M.; Cocchieri, R.A. Evaluation of conventional and organic Italian foodstuffs for deoxynivalenol and fumonisins b(1) and b(2). J. Agric. Food Chem. 2003, 51, 8128–8131. [Google Scholar] [CrossRef] [PubMed]
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions. LWT-Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Hocking, A.D. Responses of Fungi to Modified Atmospheres. In Proceedings of the International Conference, Singapore, 14–18 February 1989; IEEE: Singapore, 1989. [Google Scholar]
- Krishnakumar, T.; Visvanathan, R. Acrylamide in food products: A review. J. Food Process. Technol. 2014, 5, 1. [Google Scholar]
- Public Health England. Sacn Salt and Health Report: Recommendations on Salt in Diet; Scientific Advisory Committee on Nutrition: London, UK, 2003. [Google Scholar]
- Surdyk, N.; Rosen, J.; Andersson, R.; Aman, P. Effects of asparagine, fructose, and baking conditions on acrylamide content in yeast-leavened wheat bread. J. Agric. Food Chem. 2004, 52, 2047–2051. [Google Scholar] [CrossRef] [PubMed]
- Bednar, G.E.; Patil, A.R.; Murray, S.M.; Grieshop, C.M.; Merchen, N.R.; Fahey, G.C., Jr. Starch and fiber fractions in selected food and feed ingredients affect their small intestinal digestibility and fermentability and their large bowel fermentability in vitro in a canine model. J. Nutr. 2001, 131, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Cubadda, F.; Raggi, A.; Zanasi, F.; Carcea, M. From durum wheat to pasta: Effect of technological processing on the levels of arsenic, cadmium, lead and nickel—A pilot study. Food Addit. Contam. 2003, 20, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Ragaee, S.; Seetharaman, K.; Abdel-Aal el, S.M. The impact of milling and thermal processing on phenolic compounds in cereal grains. Crit. Rev. Food Sci. Nutr. 2014, 54, 837–849. [Google Scholar] [CrossRef] [PubMed]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Guttieri, M.J.; Seabourn, B.W.; Liu, C.; Baenziger, P.S.; Waters, B.M. Distribution of cadmium, iron, and zinc in millstreams of hard winter wheat (Triticum aestivum L.). J. Agric. Food Chem. 2015, 63, 10681–10688. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, Y.; Fulgoni, V.L. Grain foods are contributors of nutrient density for American adults and help close nutrient recommendation gaps: Data from the national health and nutrition examination survey, 2009–2012. Nutrients 2017, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- EUFIC. Whole Grains (Updated 2015). Available online: https://www.eufic.org/en/whats-in-food/article/whole-grains-updated-2015 (accessed on 2 July 2018).
- Galvano, F.; Piva, A.; Ritieni, A.; Galvano, G. Dietary strategies to counteract the effects of mycotoxins: A review. J. Food Prot. 2001, 64, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Arnao, M.B.; Williamson, G.; Garcia-Conesa, M.-T. Superoxide scavenging by polyphenols: Effect of conjugation and dimerization. Redox Rep. 2002, 7, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Atroshi, F.; Rizzo, A.; Westermarck, T.; Ali-Vehmas, T. Antioxidant nutrients and mycotoxins. Toxicology 2002, 180, 151–167. [Google Scholar] [CrossRef]
- Gross-Steinmeyer, K.; Eaton, D.L. Dietary modulation of the biotransformation and genotoxicity of aflatoxin b(1). Toxicology 2012, 299, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Wochner, K.F.; Becker-Algeri, T.A.; Colla, E.; Badiale-Furlong, E.; Drunkler, D.A. The action of probiotic microorganisms on chemical contaminants in milk. Crit. Rev. Microbiol. 2018, 44, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Taheur, F.B.; Fedhila, K.; Chaieb, K.; Kouidhi, B.; Bakhrouf, A.; Abrunhosa, L. Adsorption of aflatoxin b1, zearalenone and ochratoxin a by microorganisms isolated from kefir grains. Int. J. Food Microbiol. 2017, 251, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, H.; Lee, S.; Lee, J.; Ha, J.; Choi, Y.; Yoon, Y.; Choi, K.H. Invited review: Microbe-mediated aflatoxin decontamination of dairy products and feeds. J. Dairy Sci. 2017, 100, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Elsenhans, B.; Schümann, K.; Forth, W. Toxic metals: Interactions with essential metals. In Nutrition, Toxicity, and Cancer; Rowlan, I.R., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 223–258. [Google Scholar]
- Djukic-Cosic, D.; Ninkovic, M.; Malicevic, Z.; Plamenac-Bulat, Z.; Matovic, V. Effect of supplemental magnesium on the kidney levels of cadmium, zinc, and copper of mice exposed to toxic levels of cadmium. Biol. Trace Elem. Res. 2006, 114, 281–291. [Google Scholar] [CrossRef]
- Reeves, P.G.; Chaney, R.L. Nutritional status affects the absorption and whole-body and organ retention of cadmium in rats fed rice-based diets. Environ. Sci. Technol. 2002, 36, 2684–2692. [Google Scholar] [CrossRef] [PubMed]
- Van Barneveld, A.A.; Van den Hamer, C.J. Influence of Ca and Mg on the uptake and deposition of Pb and Cd in mice. Toxicol. Appl. Pharmacol. 1985, 79, 1–10. [Google Scholar] [CrossRef]
- Ou, S.; Gao, K.; Li, Y. An in vitro study of wheat bran binding capacity for Hg, Cd, and Pb. J. Agric. Food Chem. 1999, 47, 4714–4717. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, K.; Shen, J.; Liu, Y. Wheat bran intake can attenuate chronic cadmium toxicity in mice gut microbiota. Food Funct. 2016, 7, 3524–3530. [Google Scholar] [CrossRef] [PubMed]
- Grosicki, A.; Małagocki, P.; Kycko, A.; Monkiewicz, J.; Korol, W. Magnesium supplements affect selected cadmium toxic actions and uptake of repeated doses of cadmium. Bull. Vet. Inst. Pulawy 2015, 59, 541. [Google Scholar] [CrossRef]
- Shabb, J.B.; Muhonen, W.W.; Mehus, A.A. Quantitation of human metallothionein isoforms in cells, tissues, and cerebrospinal fluid by mass spectrometry. Methods Enzymol. 2017, 586, 413–431. [Google Scholar] [PubMed]
- McCarty, M.F. Zinc and multi-mineral supplementation should mitigate the pathogenic impact of cadmium exposure. Med. Hypotheses 2012, 79, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Peraza, M.A.; Ayala-Fierro, F.; Barber, D.S.; Casarez, E.; Rael, L.T. Effects of micronutrients on metal toxicity. Environ. Health Perspect. 1998, 106 (Suppl. 1), 203–216. [Google Scholar] [CrossRef]
- Kordas, K. Iron, lead, and children’s behavior and cognition. Annu. Rev. Nutr. 2010, 30, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Wright, R.O.; Tsaih, S.-W.; Schwartz, J.; Wright, R.J.; Hu, H. Association between iron deficiency and blood lead level in a longitudinal analysis of children followed in an urban primary care clinic. J. Pediatr. 2003, 142, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, A.A.; Zaher, M.M.; Abd el-hafez, M.A.; Morsy, A.A.; Saleh, R.A. Relation between anemia and blood levels of lead, copper, zinc and iron among children. BMC Res. Notes 2010, 3, 133. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.T.; Chang, W.T.; Huang, C.W.; Chou, S.S.; Lin, C.T.; Liau, S.J.; Wang, R.T. Studies on the concentrations of arsenic, selenium, copper, zinc and iron in the hair of blackfoot disease patients in different clinical stages. Eur. J. Clin. Chom. Clin. Biochem. 1994, 32, 107–111. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Gartside, P.S.; Glueck, C.J. Blood lead levels and dietary calcium intake in 1- to 11-year-old children: The second national health and nutrition examination survey, 1976 to 1980. Pediatrics 1986, 78, 257–262. [Google Scholar] [PubMed]
- Raab, A.; Feldmann, J.; Meharg, A.A. Levels of Arsenic in Rice: The Effects of Cooking; Food Standards Agency: London, UK, 2009. [Google Scholar]
- Gray, P.J.; Conklin, S.D.; Todorov, T.I.; Kasko, S.M. Cooking rice in excess water reduces both arsenic and enriched vitamins in the cooked grain. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2016, 33, 78–85. [Google Scholar] [CrossRef] [PubMed]
- EFSA. EFSA Awards €1m to Innovative Research Projects. Available online: https://www.efsa.europa.eu/en/press/news/170223-0 (accessed on 6 August 2018).
- Pellegrino, E.; Bedini, S.; Nuti, M.; Ercoli, L. Impact of genetically engineered maize on agronomic, environmental and toxicological traits: A meta-analysis of 21 years of field data. Sci. Rep. 2018, 8, 3113. [Google Scholar] [CrossRef] [PubMed]
| Mycotoxin | Fungal source(s) | Effects of ingestion for humans | Commodity |
|---|---|---|---|
| Deoxynivalenol/nivalenol | Fusarium graminearum | Human toxicoses e.g. nausea, vomiting, diarrhoea, headache, fever | Wheat, maize, barley |
| Fusarium crookwellense | |||
| Fusarium culmorum | |||
| Zearalenone | F. graminearum | Identified by the International Agency for Research on Cancer (IARC) [36] as a possible human carcinogen. | Maize, wheat |
| F. culmorum | |||
| F. crookwellense | |||
| Ochratoxin A | Aspergillus ochraceus | Suspected by IARC as human carcinogen. | Barley, wheat, and many other commodities |
| Penicillium verrucosum | |||
| Fumonisin B1 | Fusarium moniliforme plus several less common species | Suspected by IARC as human carcinogen. | Maize |
| Aflatoxin B1, B2 | Aspergillus flavus | Identified as potent human carcinogens by IARC. | Maize, peanuts, and many other commodities |
| Aflatoxin B1, B2, G1, G2 | Aspergillus parasiticus | Maize, peanuts |
| Food Group | Energy | Protein | Fat | Carbohydrate | Fibre |
|---|---|---|---|---|---|
| All Cereal and cereal products | 32 | 23 | 21 | 46 | 38 |
| Meat and meat products | 17 | 37 | 24 | 6 | 12 |
| Milk and milk products | 9 | 13 | 12 | 5 | 1 |
| Fruit, vegetables & salad vegetables | 9 | 7 | 6 | 11 | 28 |
| Contaminant | EU | US | ||
|---|---|---|---|---|
| Food Category | Maximum Level ** (ppb) | Food Category | Maximum Level ** (ppb) | |
| Mycotoxins | ||||
| Aflatoxin | All cereals (inc. maize and rice) for direct human consumption | 4 | All foods except milk | 20 |
| Baby foods and processed cereal based foods for infants and young children | 0.1 for aflatoxin B1 | |||
| Deoxynivalenol | Cereal flour, maize flour, maize, grits and maize meal, dry pasta | 750 | Finished wheat products for human consumption | 1000 |
| Bread, biscuits, pastries, cereal snacks and breakfast cereals | 500 | |||
| Processed cereal based baby and infant food | 200 | |||
| Fumonisin | Maize and maize based foods intended for direct human consumption | 1000 | Degermed dry milled corn products (e.g. corn meal or corn flour with fat content < 2.25%, dry weight basis) | 2000 |
| Maize based breakfast cereals and maize based snacks | 800 | Cleaned corn intended for popcorn | 3000 | |
| Processed maize based foods and baby foods for infants and young children | 200 | Whole or partially degermed dry milled corn products dry milled corn bran; cleaned corn intended for mass production | 4000 | |
| Ochratoxin A | Cereal products and cereal grains intended for direct human consumption | 3 | None identified | |
| Baby foods and processed cereal based foods for infants & young children | 0.5 | |||
| Zearalenone | Cereals for direct human consumption (e.g., cereal flour, bran) | 75 | None identified | |
| Maize for direct human consumption, maize based snacks & breakfast cereals | 100 | |||
| Bread, pastries, biscuits, cereal snacks, breakfast cereals | 50 | |||
| Processed cereal & maize based foods and baby foods for infants & young children | 20 | |||
| Metals | ||||
| Cadmium | Cereal grains excluding wheat & rice | 100 | None identified | |
| Wheat and rice grains, wheat bran & wheat germ for direct consumption | 200 | |||
| Arsenic | Parboiled rice and husked rice | 250 | Infant rice cereals *** | 100 |
| Rice waffles, rice wafers, rice crackers and rice cakes | 300 | |||
| Rice destined for production of foods for infants and young children | 100 | |||
| Lead | Cereals (pulses & legumes) | 200 | None identified | |
| Process Contaminant | ||||
| Acrylamide **** | Soft bread–wheat | 50 | None identified | |
| Non-wheat based soft bread | 100 | |||
| Breakfast cereals, excluding porridge: | ||||
| (i) Bran and whole grain cereal, gun-puffed grain, wheat and rye based products | 300 | |||
| (ii) Maize, oats, spelt, rye barley and rice-based products | 150 | |||
| Processed cereal foods for infants and young children | 40 | |||
| Nutrient | Whole wheat flour | White, wheat flour, 75% extraction | Rye flour | Rye flour, 60% extraction | Brown rice (raw) | White rice (raw) | Barley (whole grain raw) | Pearl barley |
|---|---|---|---|---|---|---|---|---|
| Carbohydrates, g (% of energy) | 62 (75.6) | 71 (80.6) | 55 (71.4) | 73 (85) | 73.5 (82.4) | 78 (87) | 60.8 (72.8) | 67 (79) |
| Protein, g (% of energy) | 10 (12.2) | 12.6 (14.3) | 10 (13) | 8 (9.3) | 8.3 (9.3) | 7 (8) | 10.6 (12.7) | 9 (10.6) |
| Fat, g (% of energy) | 2 (5.5) | 1.1 (2.8) | 2 (5.8) | 1 (2.6) | 2.6 (6.6) | 1 (2.6) | 2.1 (5.7) | 2 (5.3) |
| Dietary fibre, g | 11 | 4 | 15 | 5 | 3 | 1.3 | 14.8 | 8.6 |
| Vitamin B1 (Thiamine), µg | 0.4 | 0.07 | 0.4 | 0.15 | 0.34 | 0.04 | 0.31 | 0.03 |
| Vitamin B2 (Riboflavin), µg | 0.35 | 0.04 | 0.2 | 0.07 | 0.03 | 0.03 | 0.10 | 0.03 |
| Vitamin B3 (Niacin), mg | 5.7 | 1 | 1.7 | 1 | 6.1 | 1 | 5.2 | 3 |
| Vitamin B6 (pyridoxine), µg | 0.35 | 0.12 | 0.22 | 0.23 | 0.25 | 0.12 | 0.56 | 0.25 |
| Vitamin B9 (Folate), µg | 37 | 22 | 78 | 28 | 49 | 20 | 50 | 20 |
| Iron, mg | 4 | 0.8 | 4 | 1.5 | 1.3 | 0.4 | 6.0 | 2 |
| Zinc, mg | 2.9 | 0.64 | 3 | 1.3 | 0.8 | 1.8 | 3.3 | 2 |
| Magnesium, mg | 124 | 20 | 20 | 92 | 51 | 157 | 13 | 44 |
| Sodium (salt), mg | 5 | 2 | 5 | 10 | 1 | 2 | 4 | 5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thielecke, F.; Nugent, A.P. Contaminants in Grain—A Major Risk for Whole Grain Safety? Nutrients 2018, 10, 1213. https://doi.org/10.3390/nu10091213
Thielecke F, Nugent AP. Contaminants in Grain—A Major Risk for Whole Grain Safety? Nutrients. 2018; 10(9):1213. https://doi.org/10.3390/nu10091213
Chicago/Turabian StyleThielecke, Frank, and Anne P Nugent. 2018. "Contaminants in Grain—A Major Risk for Whole Grain Safety?" Nutrients 10, no. 9: 1213. https://doi.org/10.3390/nu10091213
APA StyleThielecke, F., & Nugent, A. P. (2018). Contaminants in Grain—A Major Risk for Whole Grain Safety? Nutrients, 10(9), 1213. https://doi.org/10.3390/nu10091213





