Challenges in the Diagnosis of Magnesium Status
Abstract
1. Introduction
Causes of Magnesium Deficiency
2. Magnesium Absorption
2.1. Anatomic Considerations
2.2. Absorption
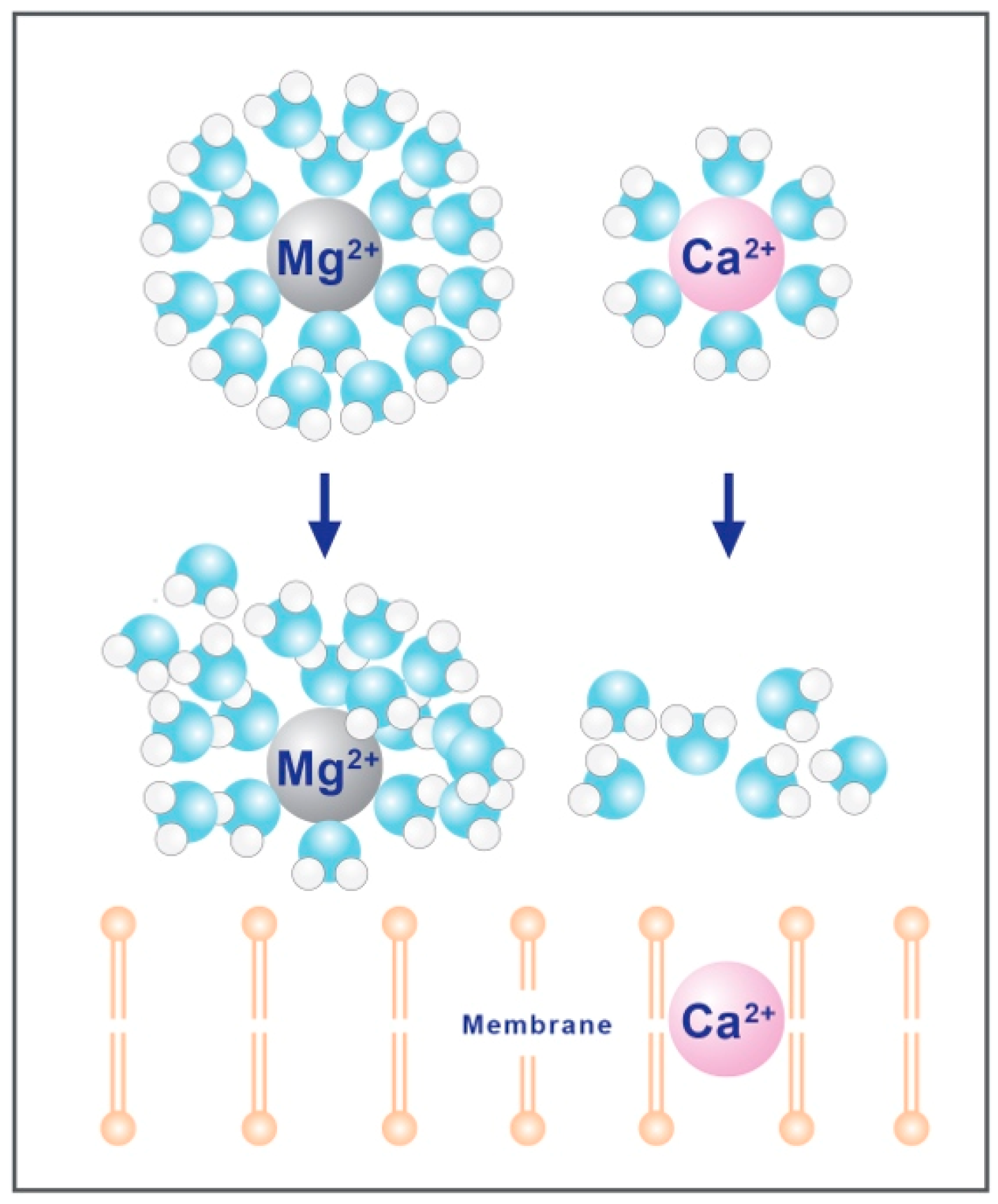
2.3. Hydration Shell
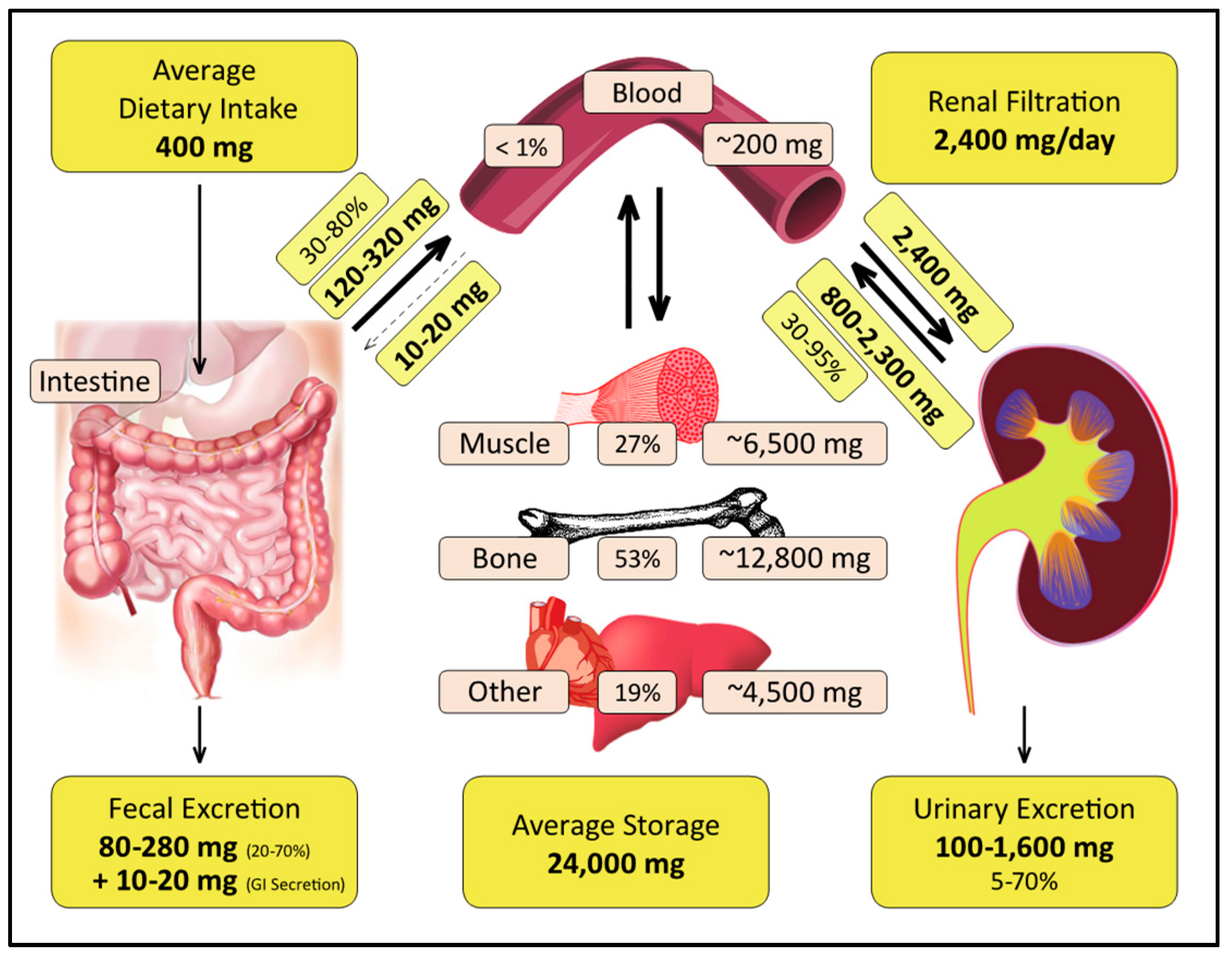
2.4. Distribution in the Human Body
2.5. Factors That Influence Magnesium Absorption
2.6. Factors That Affect Magnesium Status
3. Analytical Challenges in Establishing Magnesium Status
3.1. Blood Levels
3.2. Urine Levels
3.3. Oral Sampling
3.4. Magnesium Isotopes
4. Conclusions
Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geiger, H.; Wanner, C. Magnesium in disease. Clin. Kidney J. 2012, 5, i25–i38. [Google Scholar] [CrossRef] [PubMed]
- Volpe, S.L. Magnesium in Disease Prevention and Overall Health. Adv. Nutr. 2013, 4, 378S–383S. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, J.T.; Kumar, R. Atlas of Diseases of the Kidney: Divalent Cation: Magnesium; Current Medicine: Philadelphia, PA, USA, 1999. [Google Scholar]
- De Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Magnesium in Man: Implications for Health and Disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L.; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, Fortificants, and Supplements: Where Do Americans Get Their Nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The Case for an Evidence-Based Reference Interval for Serum Magnesium: The Time Has Come12345. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef] [PubMed]
- Rosenstein, D.L.; Ryschon, T.W.; Niemela, J.E.; Elin, R.J.; Balaban, R.S.; Rubinow, D.R. Skeletal muscle intracellular ionized magnesium measured by 31P-NMR spectroscopy across the menstrual cycle. J. Am. Coll. Nutr. 1995, 14, 486–490. [Google Scholar] [CrossRef] [PubMed]
- What We Eat in America, NHANES 2011–2012, Day 1 Food and Supplement Intake Data. Available online: https://www.ars.usda.gov/ARSUserFiles/80400530/pdf/1112/Table_37_SUP_GEN_11.pdf (accessed on 27 April 2018).
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press (US): Washington, DC, USA, 1997. [Google Scholar]
- Magnesium. Office of Dietary Supplements: National Institutes of Health. 2018. Available online: http://ods.od.nih.gov/factsheets/folate (accessed on 23 April 2018).
- Fardet, A. Food and Nutrition Sciences—Open Special Issues: Public Health Nutrition Initiatives. Food Nutr. Sci. 2013, 4, 1. [Google Scholar] [CrossRef]
- Davis, D.R. Declining Fruit and Vegetable Nutrient Composition: What Is the Evidence? HortScience 2009, 44, 15–19. [Google Scholar]
- Guo, W.; Nazim, H.; Liang, Z.; Yang, D. Magnesium deficiency in plants: An urgent problem. Crop J. 2016, 4, 83–91. [Google Scholar] [CrossRef]
- Senate Document 264, 74th Congress, 2nd Session, 5 June 1936. Available online: https://www.prismnet.com/~lenb/centurynutrition/senate264.htm (accessed on 23 April 2018).
- Current Eating Patterns in the United States—2015–2020 Dietary Guidelines. Available online: https://health.gov/dietaryguidelines/2015/guidelines/chapter-2/current-eating-patterns-in-the-united-states/ (accessed on 23 April 2018).
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Bowman, S.A.; Gortmaker, S.L.; Ebbeling, C.B.; Pereira, M.A.; Ludwig, D.S. Effects of Fast-Food Consumption on Energy Intake and Diet Quality Among Children in a National Household Survey. Pediatrics 2004, 113, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Paeratakul, S.; Ferdinand, D.P.; Champagne, C.M.; Ryan, D.H.; Bray, G.A. Fast-food consumption among US adults and children: Dietary and nutrient intake profile. J. Am. Diet. Assoc. 2003, 10, 1332–1338. [Google Scholar] [CrossRef]
- Steele, E.M.; Popkin, B.M.; Swinburn, B.; Monteiro, C.A. The share of ultra-processed foods and the overall nutritional quality of diets in the US: Evidence from a nationally representative cross-sectional study. Popul. Health Metr. 2017, 15, 6. [Google Scholar] [CrossRef] [PubMed]
- de Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Regulation of magnesium balance: Lessons learned from human genetic disease. Clin. Kidney J. 2012, 5, i15–i24. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghamdi, S.M.G.; Cameron, E.C.; Sutton, R.A.L. Magnesium Deficiency: Pathophysiologic and Clinical Overview. Am. J. Kidney Dis. 1994, 24, 737–752. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Wilson, W. Subclinical magnesium deficiency: A principal driver of cardiovascular disease and a public health crisis. Open Heart 2018, 5, e000668. [Google Scholar] [CrossRef] [PubMed]
- Stritt, S.; Nurden, P.; Favier, R.; Favier, M.; Ferioli, S.; Gotru, S.K.; van Eeuwijk, J.M.M.; Schulze, H.; Nurden, A.T.; Lambert, M.P.; et al. Defects in TRPM7 channel function deregulate thrombopoiesis through altered cellular Mg2+ homeostasis and cytoskeletal architecture. Nat. Commun. 2016, 7, 11097. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.; Cacoub, P.; Macdougall, I.C.; Peyrin-Biroulet, L. Iron deficiency anaemia. Lancet 2016, 387, 907–916. [Google Scholar] [CrossRef]
- Weaver, C.M.; Peacock, M. Calcium. Adv. Nutr. 2011, 2, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Nordin, B.E.C. Osteomalacia, Osteoporosis and Calcium Deficiency. Clin. Orthop. Relat. Res. 1960, 17, 235–258. [Google Scholar]
- Reinhart, R.A. Magnesium Metabolism: A Review with Special Reference to the Relationship Between Intracellular Content and Serum Levels. Arch. Intern. Med. 1988, 148, 2415–2420. [Google Scholar] [CrossRef] [PubMed]
- Di Silvestro, R. Current Research on Comparative Utility of Magnesium Supplements. Nat. Prod. Insider 2013, 18, 9. [Google Scholar]
- King, D.E.; Mainous, A.G.; Geesey, M.E.; Woolson, R.F. Dietary magnesium and C-reactive protein levels. J. Am. Coll. Nutr. 2005, 24, 166–171. [Google Scholar] [CrossRef] [PubMed]
- USDA, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 28. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/usda-national-nutrient-database-for-standard-reference/ (accessed on 1 June 2018).
- Schulze-Rettmer, R. The Simultaneous Chemical Precipitation of Ammonium and Phosphate in the form of Magnesium-Ammonium-Phosphate. Water Sci. Technol. 1991, 23, 659–667. [Google Scholar] [CrossRef]
- Davis, D.R.; Epp, M.D.; Riordan, H.D. Changes in USDA food composition data for 43 garden crops, 1950 to 1999. J. Am. Coll. Nutr. 2004, 23, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A. Historical changes in the mineral content of fruits and vegetables. Br. Food J. 1997, 99, 207–211. [Google Scholar] [CrossRef]
- Beeson, K.C. The Mineral Composition of Crops with Particular Reference to the Soils in Which They Were Grown: A Review and Compilation; U.S. Department of Agriculture: Washington, DC, USA, 1941. [Google Scholar]
- Firman, B. Ash and Mineral Cation Content of Vegetables. Soil Sci. Soc. Am. Proc. 1948, 13, 380–384. [Google Scholar]
- Lindlahr, H. Nature Cure; Philosophy and Practice Based on the Unity of Disease and Cure; The Nature Cure Series; The Nature Cure Publishing Co.: Chicago, IL, USA, 1914; Volume I. [Google Scholar]
- USDA, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 13. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/usda-national-nutrient-database-for-standard-reference/ (accessed on 1 June 2018).
- Steele, E.M.; Baraldi, L.G.; da Costa Louzada, M.L.; Moubarac, J.-C.; Mozaffarian, D.; Monteiro, C.A. Ultra-processed foods and added sugars in the US diet: Evidence from a nationally representative cross-sectional study. BMJ Open 2016, 6, e009892. [Google Scholar] [CrossRef] [PubMed]
- Devika, S.J.; Tanumihardjo, S.A. Effects of Different Processing Methods on the Micronutrient and Phytochemical Contents of Maize: From A to Z. Compr. Rev. Food Sci. Food Saf. 2016, 15, 912–926. [Google Scholar] [CrossRef]
- Bohn, T. Dietary Factors Influencing Magnesium Absorption in Humans. Curr. Nutr. Food Sci. 2008, 4, 53–72. [Google Scholar] [CrossRef]
- Philipp Schuchardt, J.; Hahn, A. Intestinal Absorption and Factors Influencing Bioavailability of Magnesium—An Update. Curr. Nutr. Food Sci. 2017, 13, 260–278. [Google Scholar] [CrossRef]
- Schwartz, R.; Walker, G.; Linz, M.D.; MacKellar, I. Metabolic responses of adolescent boys to two levels of dietary magnesium and protein. I. Magnesium and nitrogen retention. Am. J. Clin. Nutr. 1973, 26, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Davidsson, L.; Walczyk, T.; Hurrell, R.F. Phytic acid added to white-wheat bread inhibits fractional apparent magnesium absorption in humans. Am. J. Clin. Nutr. 2004, 79, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Neutra, R.R. Magnesium in drinking water and ischemic heart disease. Epidemiol. Rev. 1997, 19, 258–272. [Google Scholar] [CrossRef] [PubMed]
- Rosborg, I.; Kozisek, F.; Ferrrante, M. Health Effects of Demineralization Drinking Water. In Drinking Water Minerals and Mineral Balance; Springer: Cham, Switzerland, 2015; pp. 119–123. ISBN 978-3-319-09592-9. [Google Scholar]
- Barker, L.K.; Duchon, K.K.; Lesaja, S.; Robison, V.A.; Presson, S.M. Adjusted Fluoride Concentrations and Control Ranges in 34 States: 2006–2010 and 2015. J. Am. Water Works Assoc. 2017, 109, E331. [Google Scholar] [CrossRef]
- Machoy-Mokrzynska, A. Fluoride-Magnesium Interaction. J. Int. Soc. Fluoride Res. 1995, 28, 175–177. [Google Scholar]
- Ersoy, I.H.; Koroglu, B.K.; Varol, S.; Ersoy, S.; Varol, E.; Aylak, F.; Tamer, M.N. Serum copper, zinc, and magnesium levels in patients with chronic fluorosis. Biol. Trace Elem. Res. 2011, 143, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Rylander, R.; Mégevand, Y.; Lasserre, B.; Amstutz, W.; Granbom, S. Moderate alcohol consumption and urinary excretion of magnesium and calcium. Scand. J. Clin. Lab. Investig. 2001, 61, 401–405. [Google Scholar] [CrossRef]
- Kynast-Gales, S.A.; Massey, L.K. Effect of caffeine on circadian excretion of urinary calcium and magnesium. J. Am. Coll. Nutr. 1994, 13, 467–472. [Google Scholar] [CrossRef] [PubMed]
- William, J.H.; Danziger, J. Magnesium Deficiency and Proton-Pump Inhibitor Use: A Clinical Review. J. Clin. Pharmacol. 2016, 56, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Begley, J.; Smith, T.; Barnett, K.; Strike, P.; Azim, A.; Spake, C.; Richardson, T. Proton pump inhibitor associated hypomagnesaemia—A cause for concern? Br. J. Clin. Pharmacol. 2016, 81, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Polk, R.E. Drug-drug interactions with ciprofloxacin and other fluoroquinolones. Am. J. Med. 1989, 87, S76–S81. [Google Scholar] [CrossRef]
- Dante, G.; Vaiarelli, A.; Facchinetti, F. Vitamin and mineral needs during the oral contraceptive therapy: A systematic review. Int. J. Reprod. Contracept. Obstet. Gynecol. 2016, 3, 1–10. [Google Scholar] [CrossRef]
- Akinloye, O.; Adebayo, T.O.; Oguntibeju, O.O.; Oparinde, D.P.; Ogunyemi, E.O. Effects of contraceptives on serum trace elements, calcium and phosphorus levels. West Indian Med. J. 2011, 60, 308–315. [Google Scholar] [PubMed]
- Dørup, I. Magnesium and potassium deficiency. Its diagnosis, occurrence and treatment in diuretic therapy and its consequences for growth, protein synthesis and growth factors. Acta Physiol. Scand. Suppl. 1994, 618, 1–55. [Google Scholar] [PubMed]
- Lim, P.; Jacob, E. Magnesium Deficiency in Patients on Long-Term Diuretic Therapy for Heart Failure. Br. Med. J. 1972, 3, 620–622. [Google Scholar] [CrossRef] [PubMed]
- Hardwick, L.L.; Jones, M.R.; Brautbar, N.; Lee, D.B. Site and mechanism of intestinal magnesium absorption. Miner. Electrolyte Metab. 1990, 16, 174–180. [Google Scholar] [PubMed]
- Behar, J. Magnesium absorption by the rat ileum and colon. Am. J. Physiol. Leg. Content 1974, 227, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Kiela, P.R.; Ghishan, F.K. Molecular Mechanisms of Intestinal Transport of Calcium, Phosphate, and Magnesium. In Physiology of the Gastrointestinal Tract, 6th ed.; Said, H.M., Ed.; Academic Press: Cambridge, MA, USA, 2018; Chapter 59; pp. 1405–1449. ISBN 978-0-12-809954-4. [Google Scholar]
- Schlingmann, K.P.; Gudermann, T. A critical role of TRPM channel-kinase for human magnesium transport. J. Physiol. 2005, 566, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Walder, R.Y.; Landau, D.; Meyer, P.; Shalev, H.; Tsolia, M.; Borochowitz, Z.; Boettger, M.B.; Beck, G.E.; Englehardt, R.K.; Carmi, R.; et al. Mutation of TRPM6 causes familial hypomagnesemia with secondary hypocalcemia. Nat. Genet. 2002, 31, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Chubanov, V.; Gudermann, T.; Schlingmann, K.P. Essential role for TRPM6 in epithelial magnesium transport and body magnesium homeostasis. Pflüg. Arch. 2005, 451, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, K.P.; Waldegger, S.; Konrad, M.; Chubanov, V.; Gudermann, T. TRPM6 and TRPM7—Gatekeepers of human magnesium metabolism. Biochim. Biophys. Acta 2007, 1772, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, C.; Perraud, A.-L.; Johnson, C.O.; Inabe, K.; Smith, M.K.; Penner, R.; Kurosaki, T.; Fleig, A.; Scharenberg, A.M. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell 2003, 114, 191–200. [Google Scholar] [CrossRef]
- Ryazanova, L.V.; Rondon, L.J.; Zierler, S.; Hu, Z.; Galli, J.; Yamaguchi, T.P.; Mazur, A.; Fleig, A.; Ryazanov, A.G. TRPM7 is essential for Mg(2+) homeostasis in mammals. Nat. Commun. 2010, 1, 109. [Google Scholar] [CrossRef] [PubMed]
- Ryazanova, L.V.; Dorovkov, M.V.; Ansari, A.; Ryazanov, A.G. Characterization of the protein kinase activity of TRPM7/ChaK1, a protein kinase fused to the transient receptor potential ion channel. J. Biol. Chem. 2004, 279, 3708–3716. [Google Scholar] [CrossRef] [PubMed]
- Kayne, L.H.; Lee, D.B. Intestinal magnesium absorption. Miner. Electrolyte Metab. 1993, 19, 210–217. [Google Scholar] [PubMed]
- Schwartz, R.; Spencer, H.; Welsh, J.J. Magnesium absorption in human subjects from leafy vegetables, intrinsically labeled with stable 26Mg. Am. J. Clin. Nutr. 1984, 39, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Graham, L.A.; Caesar, J.J.; Buegen, A.S.V. Gastrointestinal absorption and excretion of Mg28 in man. Metabolism 1960, 9, 646–659. [Google Scholar] [PubMed]
- Fine, K.D.; Santa Ana, C.A.; Porter, J.L.; Fordtran, J.S. Intestinal absorption of magnesium from food and supplements. J. Clin. Investig. 1991, 88, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Brannan, P.G.; Vergne-Marini, P.; Pak, C.Y.; Hull, A.R.; Fordtran, J.S. Magnesium absorption in the human small intestine. Results in normal subjects, patients with chronic renal disease, and patients with absorptive hypercalciuria. J. Clin. Investig. 1976, 57, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Karbach, U. Cellular-mediated and diffusive magnesium transport across the descending colon of the rat. Gastroenterology 1989, 96, 1282–1289. [Google Scholar] [CrossRef]
- Lu, Z.; Ding, L.; Lu, Q.; Chen, Y.-H. Claudins in intestines. Tissue Barriers 2013, 1, e24978. [Google Scholar] [CrossRef] [PubMed]
- Amasheh, S.; Fromm, M.; Günzel, D. Claudins of intestine and nephron—A correlation of molecular tight junction structure and barrier function. Acta Physiol. 2010, 201, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Günzel, D.; Yu, A.S.L. Claudins and the Modulation of Tight Junction Permeability. Physiol. Rev. 2013, 93, 525–569. [Google Scholar] [CrossRef] [PubMed]
- Capaldo, C.T.; Nusrat, A. Claudin switching: Physiological plasticity of the Tight Junction. Semin. Cell Dev. Biol. 2015, 42, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Thongon, N.; Krishnamra, N. Omeprazole decreases magnesium transport across Caco-2 monolayers. World J. Gastroenterol. 2011, 17, 1574–1583. [Google Scholar] [CrossRef] [PubMed]
- Thongon, N.; Krishnamra, N. Apical acidity decreases inhibitory effect of omeprazole on Mg2+ absorption and claudin-7 and -12 expression in Caco-2 monolayers. Exp. Mol. Med. 2012, 44, 684. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Renigunta, A.; Gomes, A.S.; Hou, M.; Paul, D.L.; Waldegger, S.; Goodenough, D.A. Claudin-16 and claudin-19 interaction is required for their assembly into tight junctions and for renal reabsorption of magnesium. Proc. Natl. Acad. Sci. USA 2009, 106, 15350–15355. [Google Scholar] [CrossRef] [PubMed]
- Thongon, N.; Ketkeaw, P.; Nuekchob, C. The roles of acid-sensing ion channel 1a and ovarian cancer G protein-coupled receptor 1 on passive Mg2+ transport across intestinal epithelium-like Caco-2 monolayers. J. Physiol. Sci. 2014, 64, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Krause, G.; Winkler, L.; Mueller, S.L.; Haseloff, R.F.; Piontek, J.; Blasig, I.E. Structure and function of claudins. Biochim. Biophys. Acta BBA Biomembr. 2008, 1778, 631–645. [Google Scholar] [CrossRef] [PubMed]
- Wolf, F.I.; Cittadini, A. Chemistry and biochemistry of magnesium. Mol. Asp. Med. 2003, 24, 3–9. [Google Scholar] [CrossRef]
- Maguire, M.E.; Cowan, J.A. Magnesium chemistry and biochemistry. Biometals 2002, 15, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Tommaso, D.D.; Leeuw, N.H. de Structure and dynamics of the hydrated magnesium ion and of the solvated magnesium carbonates: Insights from first principles simulations. Phys. Chem. Chem. Phys. 2010, 12, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Moomaw, A.S.; Maguire, M.E. The Unique Nature of Mg2+ Channels. Physiology 2008, 23, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, M.D.; Jeansonne, B.G.; Renegar, R.H.; Tatum, R.; Chen, Y.H. The first extracellular domain of claudin-7 affects paracellular Cl- permeability. Biochem. Biophys. Res. Commun. 2007, 357, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Sugimoto, K.; Inatomi, S.; Maeda, T.; Osanai, M.; Uchiyama, Y.; Yamamoto, Y.; Wada, T.; Kojima, T.; Yokozaki, H.; et al. Tight Junction Proteins Claudin-2 and -12 Are Critical for Vitamin D-dependent Ca2+ Absorption between Enterocytes. Mol. Biol. Cell 2008, 19, 1912–1921. [Google Scholar] [CrossRef] [PubMed]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney J. 2012, 5, i3–i14. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, R. Magnesium Metabolism and its Disorders. Clin. Biochem. Rev. 2003, 24, 47–66. [Google Scholar] [PubMed]
- Vormann, J. Magnesium: Nutrition and metabolism. Mol. Asp. Med. 2003, 24, 27–37. [Google Scholar] [CrossRef]
- Wallach, S. Availability of body magnesium during magnesium deficiency. Magnesium 1988, 7, 262–270. [Google Scholar] [PubMed]
- Elin, R.J. Assessment of magnesium status. Clin. Chem. 1987, 33, 1965–1970. [Google Scholar] [PubMed]
- Blaine, J.; Chonchol, M.; Levi, M. Renal Control of Calcium, Phosphate, and Magnesium Homeostasis. Clin. J. Am. Soc. Nephrol. 2015, 10, 1257–1272. [Google Scholar] [CrossRef] [PubMed]
- Wacker, W.E.; Parisi, A.F. Magnesium metabolism. N. Engl. J. Med. 1968, 278, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Heijnen, A.M.; Brink, E.J.; Lemmens, A.G.; Beynen, A.C. Ileal pH and apparent absorption of magnesium in rats fed on diets containing either lactose or lactulose. Br. J. Nutr. 1993, 70, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Coudray, C.; Rambeau, M.; Feillet-Coudray, C.; Gueux, E.; Tressol, J.C.; Mazur, A.; Rayssiguier, Y. Study of magnesium bioavailability from ten organic and inorganic Mg salts in Mg-depleted rats using a stable isotope approach. Magnes. Res. 2005, 18, 215–223. [Google Scholar] [PubMed]
- van der Heijden, A.; van den Berg, G.J.; Lemmens, A.G.; Beynen, A.C. Dietary fructose v. glucose in rats raises urinary excretion, true absorption and ileal solubility of magnesium but decreases magnesium retention. Br. J. Nutr. 1994, 72, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Coudray, C.; Demigné, C.; Rayssiguier, Y. Effects of Dietary Fibers on Magnesium Absorption in Animals and Humans. J. Nutr. 2003, 133, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sabatier, M.; Arnaud, M.J.; Kastenmayer, P.; Rytz, A.; Barclay, D.V. Meal effect on magnesium bioavailability from mineral water in healthy women. Am. J. Clin. Nutr. 2002, 75, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Behall, K.M.; Scholfield, D.J.; Lee, K.; Powell, A.S.; Moser, P.B. Mineral balance in adult men: Effect of four refined fibers. Am. J. Clin. Nutr. 1987, 46, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Drews, L.M.; Kies, C.; Fox, H.M. Effect of dietary fiber on copper, zinc, and magnesium utilization by adolescent boys. Am. J. Clin. Nutr. 1979, 32, 1893–1897. [Google Scholar] [CrossRef] [PubMed]
- Coudray, C.; Bellanger, J.; Castiglia-Delavaud, C.; Rémésy, C.; Vermorel, M.; Rayssignuier, Y. Effect of soluble or partly soluble dietary fibres supplementation on absorption and balance of calcium, magnesium, iron and zinc in healthy young men. Eur. J. Clin. Nutr. 1997, 51, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Cole, D.E.; Quamme, G.A. Inherited disorders of renal magnesium handling. J. Am. Soc. Nephrol. 2000, 11, 1937–1947. [Google Scholar] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Relationship between serum magnesium levels and C-reactive protein concentration, in non-diabetic, non-hypertensive obese subjects. Int. J. Obes. 2002, 26, 469–474. [Google Scholar] [CrossRef]
- Vetter, T.; Lohse, M.J. Magnesium and the parathyroid. Curr. Opin. Nephrol. Hypertens. 2002, 11, 403–410. [Google Scholar] [CrossRef] [PubMed]
- de Rouffignac, C.; Quamme, G. Renal magnesium handling and its hormonal control. Physiol. Rev. 1994, 74, 305–322. [Google Scholar] [CrossRef] [PubMed]
- Oun, R.; Moussa, Y.E.; Wheate, N.J. The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018. [Google Scholar] [CrossRef]
- Dirks, J.H.; Wong, N.L.M. Renal Magnesium Wasting Disorders. In Phosphate and Mineral Homeostasis; Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 1986; pp. 193–197. ISBN 978-1-4684-5208-2. [Google Scholar]
- Seelig, M.S. Interrelationship of magnesium and estrogen in cardiovascular and bone disorders, eclampsia, migraine and premenstrual syndrome. J. Am. Coll. Nutr. 1993, 12, 442–458. [Google Scholar] [CrossRef] [PubMed]
- Jankunas, R.; Driziene, Z.; Stakisaitis, D.; Kuliesiene, I. Gender-dependent Magnesium Urinary Excretion in Healthy Adolescents and Adults. Acta Medica Lituanica 2001, 8, 167–172. [Google Scholar]
- De, I.L.; Vansant, G.; Van, L.G. Magnesium and obesity: Influence of gender, glucose tolerance, and body fat distribution on circulating magnesium concentrations. Magnes. Res. 1992, 5, 183–187. [Google Scholar]
- Muneyyirci-Delale, O.; Nacharaju, V.L.; Dalloul, M.; Altura, B.M.; Altura, B.T. Serum ionized magnesium and calcium in women after menopause: Inverse relation of estrogen with ionized magnesium. Fertil. Steril. 1999, 71, 869–872. [Google Scholar] [CrossRef]
- Grossi, E.; Castiglioni, S.; Moscheni, C.; Antonazzo, P.; Cetin, I.; Savasi, V.M. Serum magnesium and calcium levels in infertile women during a cycle of reproductive assistance. Magnes. Res. 2017, 30, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Palmery, M.; Saraceno, A.; Vaiarelli, A.; Carlomagno, G. Oral Contraceptives and Changes in Nutritional Requirements. Eur. Rev. 2013, 17, 1804–1813. [Google Scholar]
- YAKINCI, G.; PAÇ, A.; KüÇüKBAY, F.Z.; TAYFUN, M.; GüL, A. Serum zinc, copper, and magnesium levels in obese children. Pediatr. Int. 2011, 39, 339–341. [Google Scholar] [CrossRef]
- Hassan, S.A.; Ahmed, I.; Nasrullah, A.; Haq, S.; Ghazanfar, H.; Sheikh, A.B.; Zafar, R.; Askar, G.; Hamid, Z.; Khushdil, A.; et al. Comparison of Serum Magnesium Levels in Overweight and Obese Children and Normal Weight Children. Cureus 2017, 9, e1607. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Morán, M.; Guerrero-Romero, F. Elevated concentrations of TNF-alpha are related to low serum magnesium levels in obese subjects. Magnes. Res. 2004, 17, 189–196. [Google Scholar] [PubMed]
- Agarwal, S.; Reider, C.; Brooks, J.R.; Fulgoni, V.L., III. Comparison of Prevalence of Inadequate Nutrient Intake Based on Body Weight Status of Adults in the United States: An Analysis of NHANES 2001–2008. J. Am. Coll. Nutr. 2015, 34, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Lowenstein, F.W.; Stanton, M.F. Serum magnesium levels in the United States, 1971–1974. J. Am. Coll. Nutr. 1986, 5, 399–414. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.H.; Elin, R.J. Relationships between magnesium and protein concentrations in serum. Clin. Chem. 1985, 31, 244–246. [Google Scholar] [PubMed]
- Lim, P.; Jacob, E.; Dong, S.; Khoo, O.T. Values for tissue magnesium as a guide in detecting magnesium deficiency. J. Clin. Pathol. 1969, 22, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, M.; Hubert, J.; Mazur, A. Methods of assessment of magnesium status in humans: A systematic review. Magnes. Res. 2012, 24, 163–180. [Google Scholar] [CrossRef]
- Elin, R.J. Assessment of magnesium status for diagnosis and therapy. Magnes. Res. 2010, 23, 194–198. [Google Scholar] [CrossRef]
- Fairley, J.; Glassford, N.J.; Zhang, L.; Bellomo, R. Magnesium status and magnesium therapy in critically ill patients: A systematic review. J. Crit. Care 2015, 30, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.C.; Fernández-Lázaro, D.; Mielgo-Ayuso, J.; Calvo, J.S.; García, A.C. Effect of magnesium supplementation on muscular damage markers in basketball players during a full season. Magnes. Res. 2017, 30, 61–70. [Google Scholar] [CrossRef]
- Yeh, D.D.; Chokengarmwong, N.; Chang, Y.; Yu, L.; Arsenault, C.; Rudolf, J.; Lee-Lewandrowski, E.; Lewandrowski, K. Total and ionized magnesium testing in the surgical intensive care unit—Opportunities for improved laboratory and pharmacy utilization. J. Crit. Care 2017, 42, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, M.J. Update on the assessment of magnesium status. Br. J. Nutr. 2008, 99, S24–S36. [Google Scholar] [CrossRef] [PubMed]
- Basso, L.E.; Ubbink, J.B.; Delport, R. Erythrocyte magnesium concentration as an index of magnesium status: A perspective from a magnesium supplementation study. Clin. Chim. Acta Int. J. Clin. Chem. 2000, 291, 1–8. [Google Scholar] [CrossRef]
- Zhang, X.; Del Gobbo, L.C.; Hruby, A.; Rosanoff, A.; He, K.; Dai, Q.; Costello, R.B.; Zhang, W.; Song, Y. The Circulating Concentration and 24-h Urine Excretion of Magnesium Dose- and Time-Dependently Respond to Oral Magnesium Supplementation in a Meta-Analysis of Randomized Controlled Trials. J. Nutr. 2016, 146, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Zemel, P.C.; Zemel, M.B.; Urberg, M.; Douglas, F.L.; Geiser, R.; Sowers, J.R. Metabolic and hemodynamic effects of magnesium supplementation in patients with essential hypertension. Am. J. Clin. Nutr. 1990, 51, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Borella, P.; Sances, G.; Fioroni, L.; Nappi, R.E.; Genazzani, A.R. Oral magnesium successfully relieves premenstrual mood changes. Obstet. Gynecol. 1991, 78, 177–181. [Google Scholar] [PubMed]
- Desbiens, N.A.; Marx, J.J.; Haas, R.G.; Reinhart, R.A. Can the magnesium content of mononuclear blood cells be altered by oral magnesium supplementation? Clin. Biochem. 1992, 25, 289–292. [Google Scholar] [CrossRef]
- Ferrara, L.A.; Iannuzzi, R.; Castaldo, A.; Iannuzzi, A.; Russo, D.; Mancini, M. Long-Term Magnesium Supplementation in Essential Hypertension. Cardiology 1992, 81, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Bashir, Y.; Sneddon, J.F.; Staunton, H.A.; Haywood, G.A.; Simpson, I.A.; McKenna, W.J.; Camm, A.J. Effects of long-term oral magnesium chloride replacement in congestive heart failure secondary to coronary artery disease. Am. J. Cardiol. 1993, 72, 1156–1162. [Google Scholar] [CrossRef]
- Plum-Wirell, M.; Stegmayr, B.G.; Wester, P.O. Nutritional magnesium supplementation does not change blood pressure nor serum or muscle potassium and magnesium in untreated hypertension. A double-blind crossover study. Magnes. Res. 1994, 7, 277–283. [Google Scholar] [PubMed]
- Witteman, J.C.; Grobbee, D.E.; Derkx, F.H.; Bouillon, R.; de Bruijn, A.M.; Bruijn, D.A.; Hofman, A. Reduction of blood pressure with oral magnesium supplementation in women with mild to moderate hypertension. Am. J. Clin. Nutr. 1994, 60, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Eibl, N.L.; Kopp, H.P.; Nowak, H.R.; Schnack, C.J.; Hopmeier, P.G.; Schernthaner, G. Hypomagnesemia in type II diabetes: Effect of a 3-month replacement therapy. Diabetes Care 1995, 18, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.; Kohvakka, A. Magnesium and Ascorbic Acid Supplementation in Diabetes mellitus. Ann. Nutr. Metab. 1995, 39, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Kawasaka, T.; Nakamura, M. The effects of high oral magnesium supplementation on blood pressure, serum lipids and related variables in apparently healthy Japanese subjects. Br. J. Nutr. 1997, 78, 737–750. [Google Scholar] [CrossRef] [PubMed]
- Sanjuliani, A.F.; de Abreu Fagundes, V.G.; Francischetti, E.A. Effects of magnesium on blood pressure and intracellular ion levels of Brazilian hypertensive patients. Int. J. Cardiol. 1996, 56, 177–183. [Google Scholar] [CrossRef]
- Costello, R.B.; Moser-Veillon, P.B.; DiBianco, R. Magnesium supplementation in patients with congestive heart failure. J. Am. Coll. Nutr. 1997, 16, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Willett, W.C.; Smith, A.; Brown, L.E.; Rosner, B.; Moore, T.J. Effect on Blood Pressure of Potassium, Calcium, and Magnesium in Women with Low Habitual Intake. Hypertension 1998, 31, 131–138. [Google Scholar] [CrossRef] [PubMed]
- De Valk, H.W.; Verkaaik, R.; van Rijn, H.J.; Geerdink, R.A.; Struyvenberg, A. Oral magnesium supplementation in insulin-requiring Type 2 diabetic patients. Diabet. Med. J. Br. Diabet. Assoc. 1998, 15, 503–507. [Google Scholar] [CrossRef]
- De Lordes Lima, M.; Cruz, T.; Pousada, J.C.; Rodrigues, L.E.; Barbosa, K.; Canguçu, V. The effect of magnesium supplementation in increasing doses on the control of type 2 diabetes. Diabetes Care 1998, 21, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.F.; De Souza, M.C.; Vickers, M.F.; Abeyasekera, S.; Collins, M.L.; Trinca, L.A. Magnesium supplementation alleviates premenstrual symptoms of fluid retention. J. Womens Health 1998, 7, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Weller, E.; Bachert, P.; Meinck, H.; Friedmann, B.; BÄrtsch, P.; MairbÄurl, H. Lack of effect of oral Mg-supplementation on Mg in serum, blood cells, and calf muscle. Med. Sci. Sports Exerc. 1998, 30, 1584–1591. [Google Scholar] [CrossRef] [PubMed]
- Hägg, E.; Carlberg, B.C.; Hillörn, V.S.; Villumsen, J. Magnesium therapy in type 1 diabetes. A double blind study concerning the effects on kidney function and serum lipid levels. Magnes. Res. 1999, 12, 123–130. [Google Scholar] [PubMed]
- Wary, C.; Brillault-Salvat, C.; Bloch, G.; Leroy-Willig, A.; Roumenov, D.; Grognet, J.M.; Leclerc, J.H.; Carlier, P.G. Effect of chronic magnesium supplementation on magnesium distribution in healthy volunteers evaluated by 31P-NMRS and ion selective electrodes. Br. J. Clin. Pharmacol. 1999, 48, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Zorbas, Y.G.; Kakurin, V.J.; Afonin, V.B.; Charapakhin, K.P.; Denogradov, S.D. Magnesium supplements’ effect on magnesium balance in athletes during prolonged restriction of muscular activity. Kidney Blood Press. Res. 1999, 22, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Shechter, M.; Sharir, M.; Labrador, M.J.P.; Forrester, J.; Silver, B.; Merz, C.N.B. Oral Magnesium Therapy Improves Endothelial Function in Patients With Coronary Artery Disease. Circulation 2000, 102, 2353–2358. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.F.; De Souza, M.C.; Marakis, G.; Robinson, P.A.; Morris, A.P.; Bolland, K.M. Unexpected benefit of sorbitol placebo in Mg intervention study of premenstrual symptoms: Implications for choice of placebo in RCTs. Med. Hypotheses 2002, 58, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C.; Golf, S.W.; Völker, K. Effect of magnesium on granulocyte function and on the exercise induced inflammatory response. Magnes. Res. 2003, 16, 49–58. [Google Scholar] [PubMed]
- Rodríguez-Morán, M.; Guerrero-Romero, F. Oral magnesium supplementation improves insulin sensitivity and metabolic control in type 2 diabetic subjects: A randomized double-blind controlled trial. Diabetes Care 2003, 26, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.F.; Marakis, G.; Christie, S.; Byng, M. Mg citrate found more bioavailable than other Mg preparations in a randomised, double-blind study. Magnes. Res. 2003, 16, 183–191. [Google Scholar] [PubMed]
- Závaczki, Z.; Szöllõsi, J.; Kiss, S.A.; Koloszár, S.; Fejes, I.; Kovács, L.; Pál, A. Magnesium-orotate supplementation for idiopathic infertile male patients: A randomized, placebo-controlled clinical pilot study. Magnes. Res. 2003, 16, 131–136. [Google Scholar] [PubMed]
- De Leeuw, I.; Engelen, W.; De Block, C.; Van Gaal, L. Long term magnesium supplementation influences favourably the natural evolution of neuropathy in Mg-depleted type 1 diabetic patients (T1dm). Magnes. Res. 2004, 17, 109–114. [Google Scholar] [PubMed]
- Pokan, R.; Hofmann, P.; von Duvillard, S.P.; Smekal, G.; Wonisch, M.; Lettner, K.; Schmid, P.; Shechter, M.; Silver, B.; Bachl, N. Oral magnesium therapy, exercise heart rate, exercise tolerance, and myocardial function in coronary artery disease patients. Br. J. Sports Med. 2006, 40, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Barragán-Rodríguez, L.; Rodríguez-Morán, M.; Guerrero-Romero, F. Efficacy and safety of oral magnesium supplementation in the treatment of depression in the elderly with type 2 diabetes: A randomized, equivalent trial. Magnes. Res. 2008, 21, 218–223. [Google Scholar] [PubMed]
- Almoznino-Sarafian, D.; Sarafian, G.; Berman, S.; Shteinshnaider, M.; Tzur, I.; Cohen, N.; Gorelik, O. Magnesium administration may improve heart rate variability in patients with heart failure. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Park, H.K.; Son, S.P.; Lee, C.W.; Kim, I.J.; Kim, H.J. Effects of oral magnesium supplementation on insulin sensitivity and blood pressure in normo-magnesemic nondiabetic overweight Korean adults. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. The effect of lowering blood pressure by magnesium supplementation in diabetic hypertensive adults with low serum magnesium levels: A randomized, double-blind, placebo-controlled clinical trial. J. Hum. Hypertens. 2009, 23, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Aydin, H.; Deyneli, O.; Yavuz, D.; Gözü, H.; Mutlu, N.; Kaygusuz, I.; Akalin, S. Short-term oral magnesium supplementation suppresses bone turnover in postmenopausal osteoporotic women. Biol. Trace Elem. Res. 2010, 133, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Kazaks, A.G.; Uriu-Adams, J.Y.; Albertson, T.E.; Shenoy, S.F.; Stern, J.S. Effect of oral magnesium supplementation on measures of airway resistance and subjective assessment of asthma control and quality of life in men and women with mild to moderate asthma: A randomized placebo controlled trial. J. Asthma Off. J. Assoc. Care Asthma 2010, 47, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H.; Johnson, L.K.; Zeng, H. Magnesium supplementation improves indicators of low magnesium status and inflammatory stress in adults older than 51 years with poor quality sleep. Magnes. Res. 2010, 23, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Zorbas, Y.G.; Kakuris, K.K.; Federenko, Y.F.; Deogenov, V.A. Utilization of magnesium during hypokinesia and magnesium supplementation in healthy subjects. Nutrition 2010, 26, 1134–1138. [Google Scholar] [CrossRef] [PubMed]
- Chacko, S.A.; Sul, J.; Song, Y.; Li, X.; LeBlanc, J.; You, Y.; Butch, A.; Liu, S. Magnesium supplementation, metabolic and inflammatory markers, and global genomic and proteomic profiling: A randomized, double-blind, controlled, crossover trial in overweight individuals. Am. J. Clin. Nutr. 2011, 93, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Magnesium improves the beta-cell function to compensate variation of insulin sensitivity: Double-blind, randomized clinical trial. Eur. J. Clin. Investig. 2011, 41, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Tarighat Esfanjani, A.; Mahdavi, R.; Ebrahimi Mameghani, M.; Talebi, M.; Nikniaz, Z.; Safaiyan, A. The effects of magnesium, L-carnitine, and concurrent magnesium-L-carnitine supplementation in migraine prophylaxis. Biol. Trace Elem. Res. 2012, 150, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Laecke, S.V.; Nagler, E.V.; Taes, Y.; Biesen, W.V.; Peeters, P.; Vanholder, R. The effect of magnesium supplements on early post-transplantation glucose metabolism: A randomized controlled trial. Transpl. Int. 2014, 27, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Cosaro, E.; Bonafini, S.; Montagnana, M.; Danese, E.; Trettene, M.S.; Minuz, P.; Delva, P.; Fava, C. Effects of magnesium supplements on blood pressure, endothelial function and metabolic parameters in healthy young men with a family history of metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Moran, M.; Guerrero-Romero, F. Oral Magnesium Supplementation Improves the Metabolic Profile of Metabolically Obese, Normal-weight Individuals: A Randomized Double-blind Placebo-controlled Trial. Arch. Med. Res. 2014, 45, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Setaro, L.; Santos-Silva, P.R.; Nakano, E.Y.; Sales, C.H.; Nunes, N.; Greve, J.M.; Colli, C. Magnesium status and the physical performance of volleyball players: Effects of magnesium supplementation. J. Sports Sci. 2014, 32, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Navarrete-Cortes, A.; Ble-Castillo, J.L.; Guerrero-Romero, F.; Cordova-Uscanga, R.; Juárez-Rojop, I.E.; Aguilar-Mariscal, H.; Tovilla-Zarate, C.A.; del Rocio Lopez-Guevara, M. No effect of magnesium supplementation on metabolic control and insulin sensitivity in type 2 diabetic patients with normomagnesemia. Magnes. Res. 2014, 27, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Romero, F.; Simental-Mendia, L.E.; Hernandez-Ronquillo, G.; Rodriguez-Moran, M. Oral magnesium supplementation improves glycemic status in subjects with prediabetes and hypomagnesaemia: A double-blind placebo-controlled randomized trial. Diabetes Metab. 2015, 41, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Qin, R.; Smith, T.J.; Atherton, P.J.; Barton, D.L.; Sturtz, K.; Dakhil, S.R.; Anderson, D.M.; Flynn, K.; Puttabasavaiah, S.; et al. NCCTG N10C2 (Alliance)—A Double-Blind, Placebo-Controlled Study of Magnesium Supplements to Reduce Menopausal Hot Flashes. Menopause NYN 2015, 22, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Baker, W.L.; Kluger, J.; Coleman, C.I.; White, C.M. Impact of Magnesium L-Lactate on Occurrence of Ventricular Arrhythmias in Patients with Implantable Cardioverter Defibrillators: A Randomized, Placebo-Controlled Trial. Open Cardiovasc. Med. J. 2015, 9, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Joris, P.J.; Plat, J.; Bakker, S.J.; Mensink, R.P. Long-term magnesium supplementation improves arterial stiffness in overweight and obese adults: Results of a randomized, double-blind, placebo-controlled intervention trial. Am. J. Clin. Nutr. 2016, 103, 1260–1266. [Google Scholar] [CrossRef] [PubMed]
- Terink, R.; Balvers, M.G.J.; Hopman, M.T.; Witkamp, R.F.; Mensink, M.; Gunnewiek, J.M.T.K. Decrease in Ionized and Total Magnesium Blood Concentrations in Endurance Athletes Following an Exercise Bout Restores within Hours—Potential Consequences for Monitoring and Supplementation. Int. J. Sport Nutr. Exerc. Metab. 2016, 27, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Moradian, S.T.; Ghiasi, M.S.; Mohamadpour, A.; Siavash, Y. Oral magnesium supplementation reduces the incidence of gastrointestinal complications following cardiac surgery: A randomized clinical trial. Magnes. Res. 2017, 30, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Rajizadeh, A.; Mozaffari-Khosravi, H.; Yassini-Ardakani, M.; Dehghani, A. Effect of magnesium supplementation on depression status in depressed patients with magnesium deficiency: A randomized, double-blind, placebo-controlled trial. Nutrition 2017, 35, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Cunha, A.R.; D’El-Rei, J.; Medeiros, F.; Umbelino, B.; Oigman, W.; Touyz, R.M.; Neves, M.F. Oral magnesium supplementation improves endothelial function and attenuates subclinical atherosclerosis in thiazide-treated hypertensive women. J. Hypertens. 2017, 35, 89. [Google Scholar] [CrossRef] [PubMed]
- Bressendorff, I.; Hansen, D.; Schou, M.; Kragelund, C.; Brandi, L. The effect of magnesium supplementation on vascular calcification in chronic kidney disease—A randomised clinical trial (MAGiCAL-CKD): Essential study design and rationale. BMJ Open 2017, 7, e016795. [Google Scholar] [CrossRef] [PubMed]
- Bressendorff, I.; Hansen, D.; Schou, M.; Silver, B.; Pasch, A.; Bouchelouche, P.; Pedersen, L.; Rasmussen, L.M.; Brandi, L. Oral Magnesium Supplementation in Chronic Kidney Disease Stages 3 and 4: Efficacy, Safety, and Effect on Serum Calcification Propensity—A Prospective Randomized Double-Blinded Placebo-Controlled Clinical Trial. Kidney Int. Rep. 2017, 2, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Toprak, O.; Kurt, H.; Sarı, Y.; Şarkış, C.; Us, H.; Kırık, A. Magnesium Replacement Improves the Metabolic Profile in Obese and Pre-Diabetic Patients with Mild-to-Moderate Chronic Kidney Disease: A 3-Month, Randomised, Double-Blind, Placebo-Controlled Study. Kidney Blood Press. Res. 2017, 42, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Joosten, M.M.; Gansevoort, R.T.; Mukamal, K.J.; van der Harst, P.; Geleijnse, J.M.; Feskens, E.J.; Navis, G.; Bakker, S.J. Urinary and plasma magnesium and risk of ischemic heart disease. Am. J. Clin. Nutr. 2013, 97, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Djurhuus, M.S.; Gram, J.; Petersen, P.H.; Klitgaard, N.A.H.; Bollerslev, J.; Beck-Nielsen, H. Biological variation of serum and urinary magnesium in apparently healthy males. Scand. J. Clin. Lab. Investig. 1995, 55, 549–558. [Google Scholar] [CrossRef]
- Tahiri, M.; Tressol, J.C.; Arnaud, J.; Bornet, F.; Bouteloup-Demange, C.; Feillet-Coudray, C.; Ducros, V.; Pépin, D.; Brouns, F.; Roussel, A.M.; et al. Five-Week Intake of Short-Chain Fructo-Oligosaccharides Increases Intestinal Absorption and Status of Magnesium in Postmenopausal Women. J. Bone Miner. Res. 2001, 16, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Saur, P.M.; Zielmann, S.; Roth, A.T.; Frank, L.; Warneke, G.; Radke, A.; Ensink, F.B.; Kettler, D. Diagnosis of magnesium deficiency in intensive care patients. Anasthesiologie Intensivmedizin Notfallmedizin Schmerztherapie AINS 1996, 31, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Hébert, P.; Mehta, N.; Wang, J.; Hindmarsh, T.; Jones, G.; Cardinal, P. Functional magnesium deficiency in critically ill patients identified using a magnesium-loading test. Crit. Care Med. 1997, 25, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Saur, P.; Niedmann, P.D.; Brunner, E.; Kettler, D. Do intracellular, extracellular or urinary magnesium concentrations predict renal retention of magnesium in critically ill patients? Eur. J. Anaesthesiol. 2005, 22, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Wälti, M.K.; Walczyk, T.; Zimmermann, M.B.; Fortunato, G.; Weber, M.; Spinas, G.A.; Hurrell, R.F. Urinary excretion of an intravenous 26Mg dose as an indicator of marginal magnesium deficiency in adults. Eur. J. Clin. Nutr. 2006, 60, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Martin, B.J. The magnesium load test: Experience in elderly subjects. Aging Clin. Exp. Res. 1990, 2, 291–296. [Google Scholar] [CrossRef]
- Haigney, M.C.P.; Silver, B.; Tanglao, E.; Silverman, H.S.; Hill, J.D.; Shapiro, E.; Gerstenblith, G.; Schulman, S.P. Noninvasive Measurement of Tissue Magnesium and Correlation with Cardiac Levels. Circulation 1995, 92, 2190–2197. [Google Scholar] [CrossRef] [PubMed]
- Coudray, C.; Rambeau, M.; Feillet-Coudray, C.; Tressol, J.C.; Demigne, C.; Gueux, E.; Mazur, A.; Rayssiguier, Y. Dietary inulin intake and age can significantly affect intestinal absorption of calcium and magnesium in rats: A stable isotope approach. Nutr. J. 2005, 4, 29. [Google Scholar] [CrossRef] [PubMed]
- Sojka, J.; Wastney, M.; Abrams, S.; Lewis, S.F.; Martin, B.; Weaver, C.; Peacock, M. Magnesium kinetics in adolescent girls determined using stable isotopes: Effects of high and low calcium intake. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1997, 273, R710–R715. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A. 16—Using stable isotopes to determine mineral bioavailability of functional foods. In Designing Functional Foods; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2009; pp. 415–428. ISBN 978-1-84569-432-6. [Google Scholar]
- Hachey, D.L.; Wong, W.W.; Boutton, T.W.; Klein, P.D. Stable isotopes in the study of human nutrition. Int. J. Radiat. Appl. Instrum. 1988, 39, 503. [Google Scholar] [CrossRef]
- Hansen, K.E.; Nabak, A.C.; Johnson, R.E.; Marvdashti, S.; Keuler, N.S.; Shafer, M.M.; Abrams, S.A. Isotope Concentrations from 24-h Urine and 3-h Serum Samples Can Be Used to Measure Intestinal Magnesium Absorption in Postmenopausal Women. J. Nutr. 2014, 144, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Draxler, J.; Martinelli, E.; Weinberg, A.M.; Zitek, A.; Irrgeher, J.; Meischel, M.; Stanzl-Tschegg, S.E.; Mingler, B.; Prohaska, T. The potential of isotopically enriched magnesium to study bone implant degradation in vivo. Acta Biomater. 2017, 51, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Avioli, L.V.; Berman, M. Mg28 kinetics in man. J. Appl. Physiol. 1966, 21, 1688–1694. [Google Scholar] [CrossRef] [PubMed]
- Danielson, B.G.; Johansson, G.; Ljunghall, S. Magnesium metabolism in healthy subjects. Scand. J. Urol. Nephrol. Suppl. 1979, 51, 49–73. [Google Scholar]
- Watson, W.S.; Hilditch, T.E.; Horton, P.W.; Davies, D.L.; Lindsay, R. Magnesium metabolism in blood and the whole body in man using 28magnesium. Metabolism 1979, 28, 90–95. [Google Scholar] [CrossRef]
- Costello, R.B.; Nielsen, F. Interpreting magnesium status to enhance clinical care: Key indicators. Curr. Opin. Nutr. Metab. Care 2017, 20, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Bilbey, D.L.; Prabhakaran, V.M. Muscle cramps and magnesium deficiency: Case reports. Can. Fam. Physician 1996, 42, 1348–1351. [Google Scholar] [PubMed]
- Abbasi, B.; Kimiagar, M.; Sadeghniiat, K.; Shirazi, M.M.; Hedayati, M.; Rashidkhani, B. The effect of magnesium supplementation on primary insomnia in elderly: A double-blind placebo-controlled clinical trial. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2012, 17, 1161–1169. [Google Scholar]
- Romano, T.J.; Stiller, J.W. Magnesium Deficiency in Fibromyalgia Syndrome. J. Nutr. Med. 1994, 4, 165–167. [Google Scholar] [CrossRef]
- Moorkens, G.; Manuel, B.Y.K.; Vertommen, J.; Meludu, S.; Noe, M.; De, I.L. Magnesium deficit in a sample of the Belgian population presenting with chronic fatigue. Magnes. Res. 1997, 10, 329–337. [Google Scholar] [PubMed]
- Kumeda, Y.; Inaba, M. Metabolic syndrome and magnesium. Clin. Calcium 2005, 15, 97–104. [Google Scholar] [PubMed]
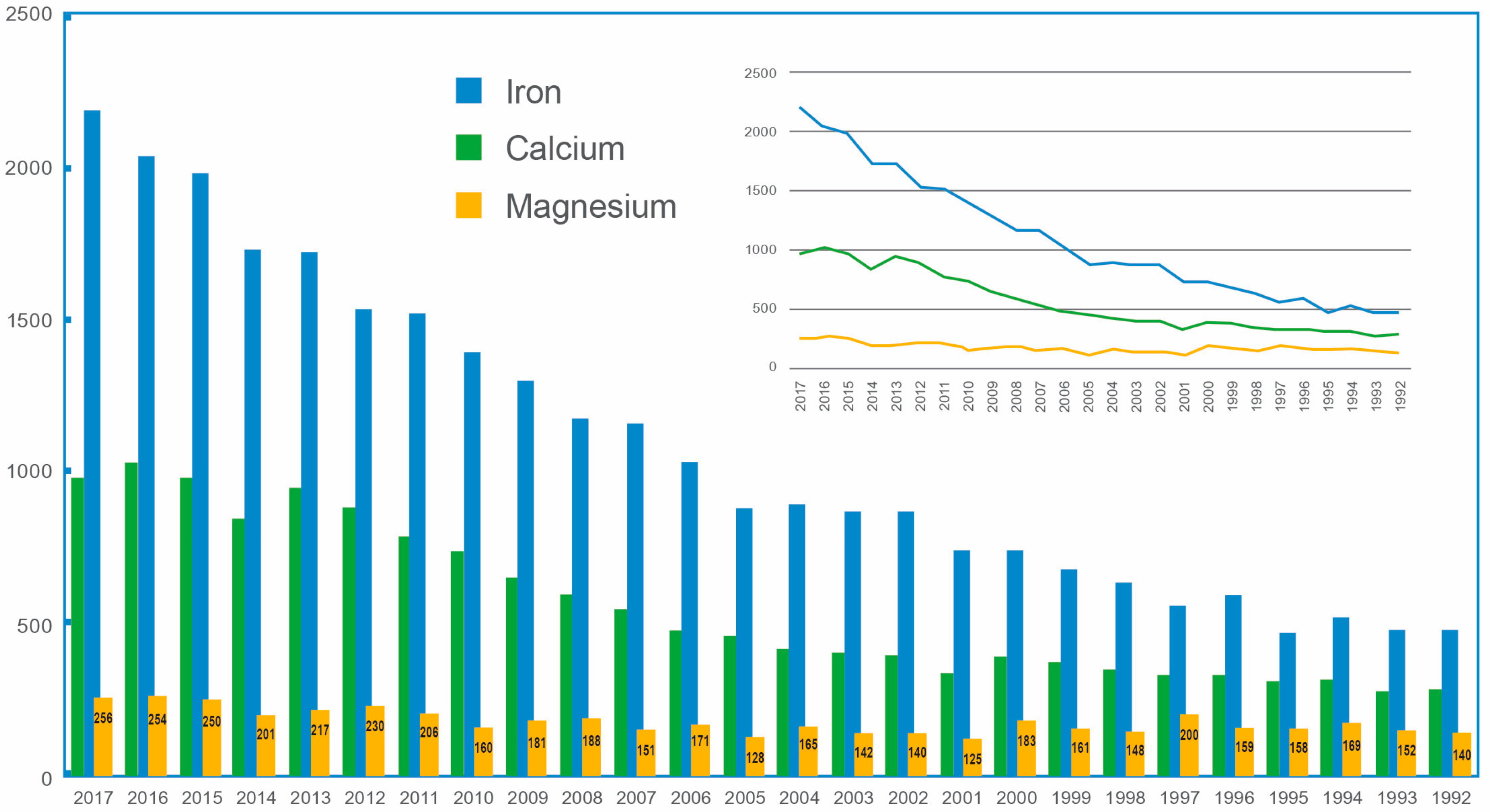
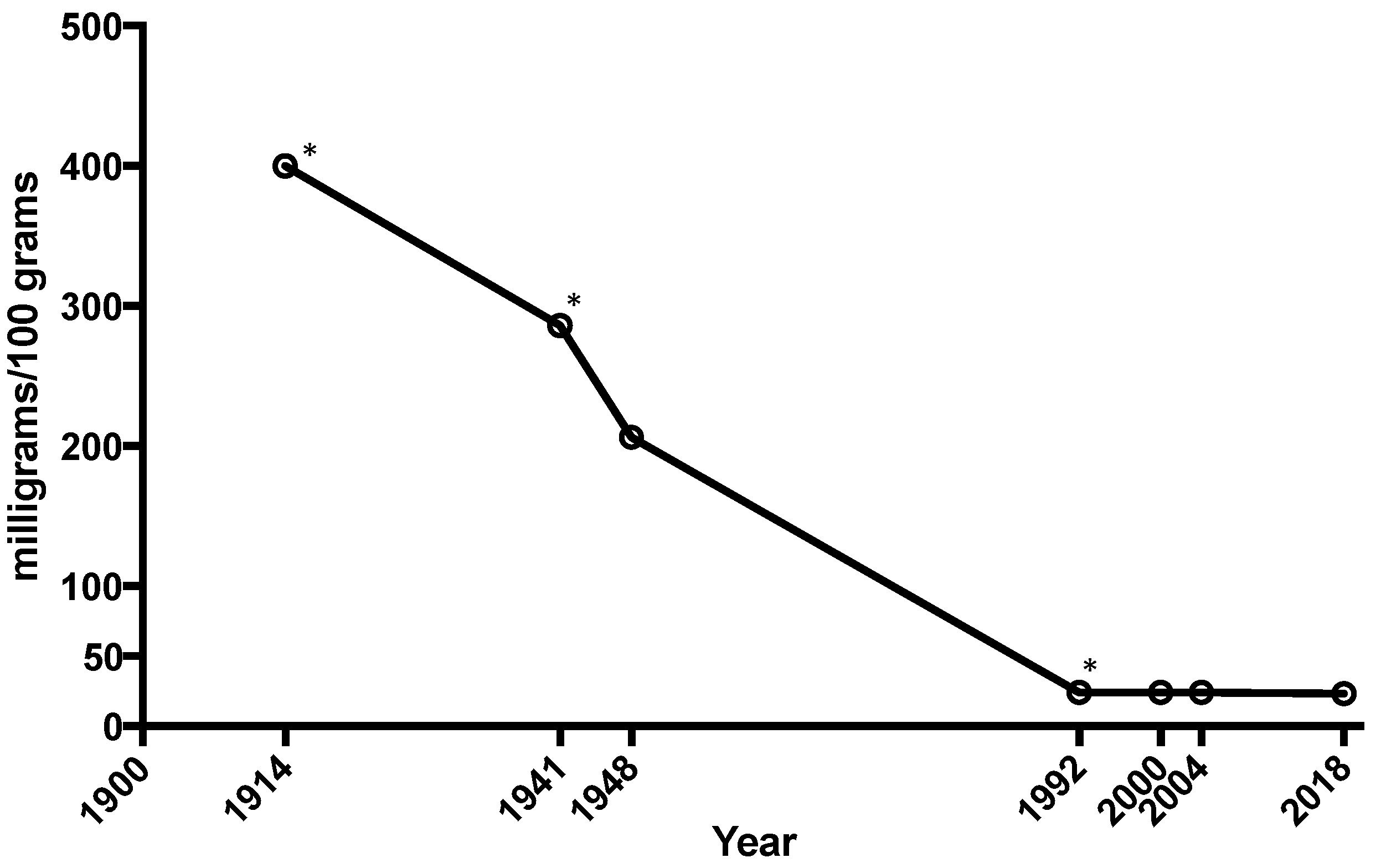
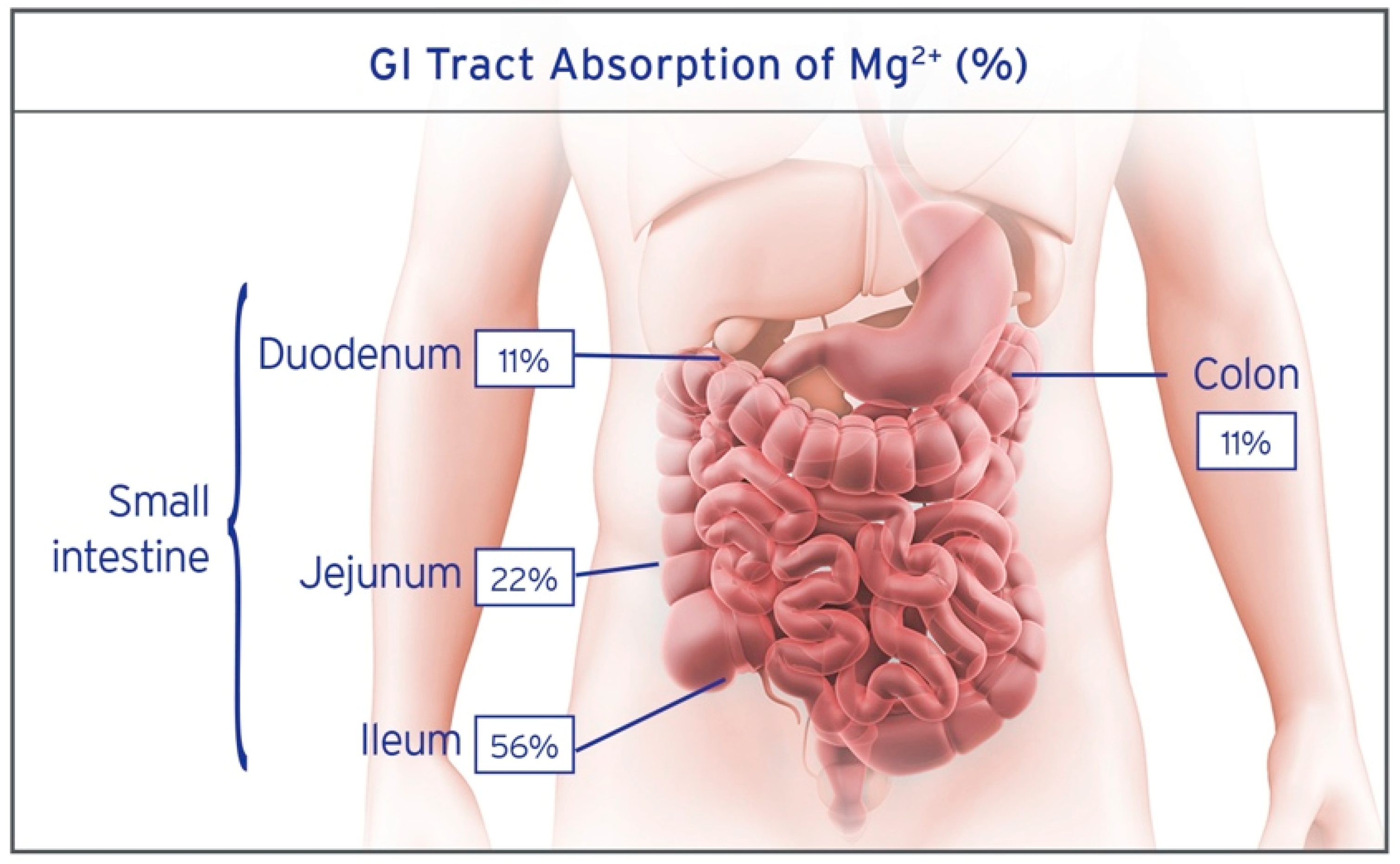
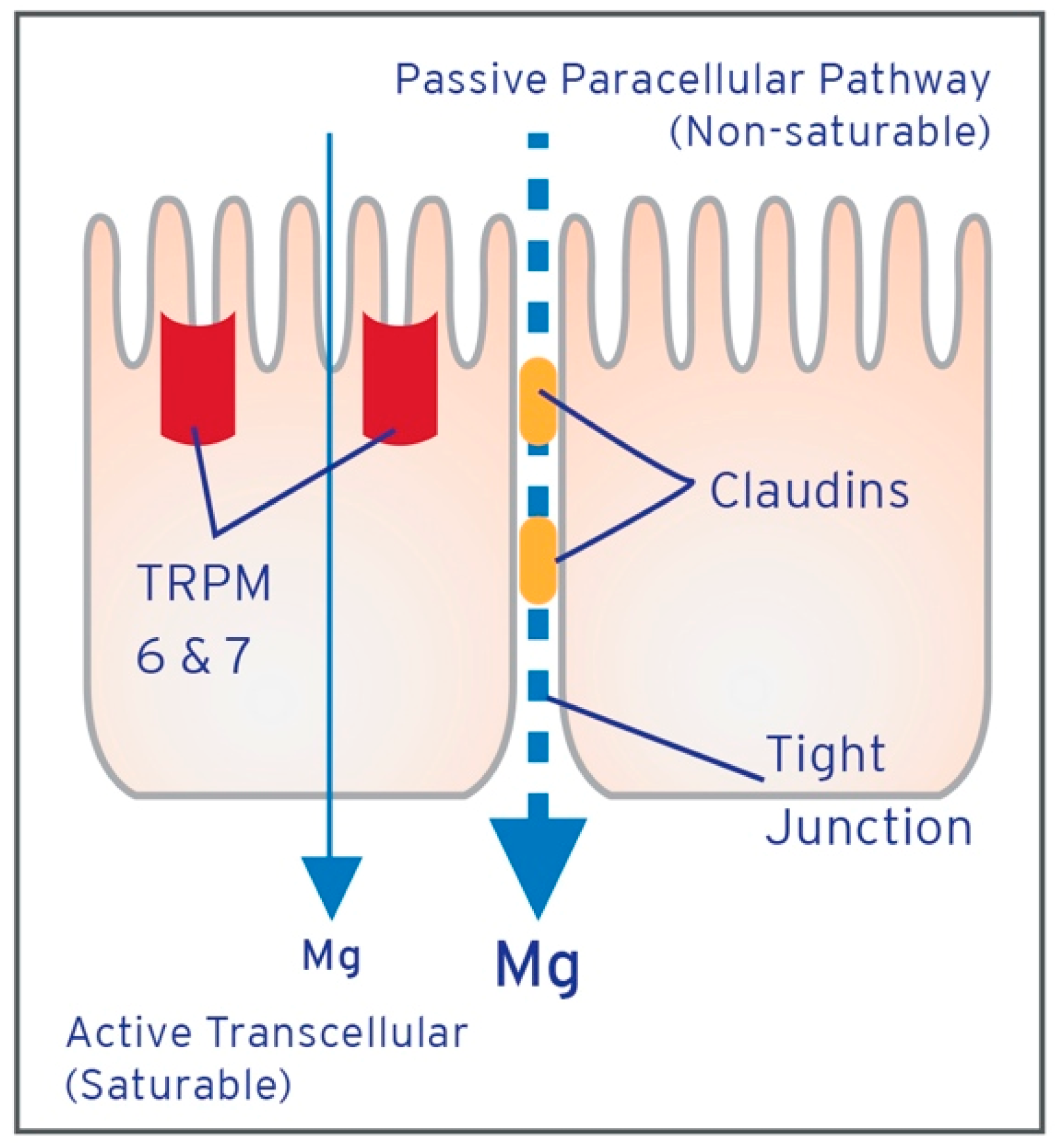
| Study | Blood | Urine | Intracellular | Fecal | Tissue | Challenge Studies | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Serum | Plasma | 24 h | NS | RBC | WBC | SL | Other | Muscle | Other | Balance | Retention | ||
| 1. Zemel, 1990, USA [131] | χ | χ | |||||||||||
| 2. Facchinetti, 1991, Italy [132] | χ | χ | χ | ||||||||||
| 3. Desbiens, 1992, USA [133] | χ | ||||||||||||
| 4. Ferrara, 1992, Italy [134] | χ | χ | |||||||||||
| 5. Bashir, 1993, USA [135] | χ | χ | |||||||||||
| 6. Plum-Wirell, 1994, Sweden [136] | χ | χ | χ | ||||||||||
| 7. Witteman, 1994, Netherlands [137] | χ | χ | |||||||||||
| 8. Eibl, 1995, Austria [138] | χ | χ | |||||||||||
| 9. Eriksson, 1995, Finland [139] | χ | ||||||||||||
| 10. Itoh, 1996, Japan [140] | χ | χ | |||||||||||
| 11. Sanjuliani, 1996, Brazil [141] | χ | ||||||||||||
| 12. Costello, 1997, USA [142] | χ | χ | χ | ||||||||||
| 13. Sacks, 1997, USA [143] | χ | ||||||||||||
| 14. de Valk, 1998, Netherlands [144] | χ | χ | χ | ||||||||||
| 15. Lima, 1998, Brazil [145] | χ | χ | χ | ||||||||||
| 16. Walker, 1998, UK [146] | χ | ||||||||||||
| 17. Weller, 1998, Germany [147] | χ | χ | χ | χ | χ | ||||||||
| 18. Hagg, 1999, Sweden [148] | χ | χ | |||||||||||
| 19. Wary, 1999, French [149] | χ | χ | χ | χ | χ | ||||||||
| 20. Zorbas, 1999, Greece [150] | χ | χ | χ | χ | |||||||||
| 21. Schechter, 2000, USA [151] | χ | χ | |||||||||||
| 22. Walker, 2002, UK [152] | χ | ||||||||||||
| 23. Mooren, 2003, Germany [153] | χ | ||||||||||||
| 24. Rodriguez-Moran, 2003, Mexico [154] | χ | ||||||||||||
| 25. Walker, 2003, UK [155] | χ | χ | χ | ||||||||||
| 26. Závaczki, 2003, Hungary [156] | χ | ||||||||||||
| 27. De Leeuw, 2004, Belgium [157] | χ | χ | |||||||||||
| 28. Pokan, 2006, USA [158] | χ | ||||||||||||
| 29. Rodríguez, 2008, Mexico [159] | χ | ||||||||||||
| 30. Almoznino-Sarafian, 2009, Israel [160] | χ | χ | |||||||||||
| 31. Lee, 2009, South Korea [161] | χ | χ | |||||||||||
| 32. Romero, 2009, Mexico [162] | χ | ||||||||||||
| 33. Aydın, 2010, Turkey [163] | χ | ||||||||||||
| 34. Kazaks, 2010, USA [164] | χ | χ | χ | ||||||||||
| 35. Nielsen, 2010, USA [165] | χ | χ | χ | ||||||||||
| 36. Zorbas, 2010, Greece [166] | χ | χ | χ | χ | |||||||||
| 37. Chacko, 2011, USA [167] | χ | ||||||||||||
| 38. Romero, 2011, Mexico [168] | χ | ||||||||||||
| 39. Esfanjani, 2012, Iran [169] | χ | ||||||||||||
| 40. Laecke, 2014, Belgium [170] | χ | χ | |||||||||||
| 41. Cosaro, 2014, Italy [171] | χ | χ | χ | ||||||||||
| 42. Rodriguez, 2014, Mexico [172] | χ | ||||||||||||
| 43. Setaro, 2014, Brazil [173] | χ | ||||||||||||
| 44. Navarrete-Cortes, 2014, Mexico [174] | χ | χ | |||||||||||
| 45. Guerrero-Romero, 2015, Mexico [175] | χ | ||||||||||||
| 46. Park, 2015, USA [176] | χ | ||||||||||||
| 47. Baker, 2015, USA [177] | χ | ||||||||||||
| 48. Joris, 2016, Netherlands [178] | χ | χ | |||||||||||
| 49. Terink, 2016, Netherlands [179] | χ | ||||||||||||
| 50. Moradian, 2017, Iran [180] | χ | ||||||||||||
| 51. Rajizadeh, 2017, Iran [181] | χ | ||||||||||||
| 52. Cunha, 2017, Brazil [182] | χ | ||||||||||||
| 53. Bressendorff, 2017, Denmark/Norway [183] | χ | χ | |||||||||||
| 54. Bressendorff, 2017, Denmark [184] | χ | χ | χ | ||||||||||
| 55. Toprak, 2017, Turkey [185] | χ | ||||||||||||
| Total | 35 | 11 | 16 | 10 | 12 | 3 | 3 | 3 | 2 | 4 | 2 | 1 | 1 |
| Category | Risk Factor | Criterion |
|---|---|---|
| Disease | Diabetes [4], Heart disease [22] | Major |
| Osteoporosis [26] | Minor | |
| Diet | Soda [41], Processed Foods [39] | Major |
| Coffee [50], Alcohol [49], Protein [42] | Minor | |
| Medication | Diuretics [57], Antacids [51] | Major |
| Oral contraceptives [55], Antibiotics [53] | Minor | |
| Clinical History | Leg Cramps [205] | Major |
| Sleep Disorder [206], Fibromyalgia [207], Chronic fatigue [208] | Minor | |
| Metabolic Status | Metabolic Syndrome [209] | Major |
| BMI > 30 [117] | Minor |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Workinger, J.L.; Doyle, R.P.; Bortz, J. Challenges in the Diagnosis of Magnesium Status. Nutrients 2018, 10, 1202. https://doi.org/10.3390/nu10091202
Workinger JL, Doyle RP, Bortz J. Challenges in the Diagnosis of Magnesium Status. Nutrients. 2018; 10(9):1202. https://doi.org/10.3390/nu10091202
Chicago/Turabian StyleWorkinger, Jayme L., Robert. P. Doyle, and Jonathan Bortz. 2018. "Challenges in the Diagnosis of Magnesium Status" Nutrients 10, no. 9: 1202. https://doi.org/10.3390/nu10091202
APA StyleWorkinger, J. L., Doyle, R. P., & Bortz, J. (2018). Challenges in the Diagnosis of Magnesium Status. Nutrients, 10(9), 1202. https://doi.org/10.3390/nu10091202




