1. Introduction
During endurance exercise, skeletal muscle uses glucose and fatty acids as the main energy sources. Glycogen, the storage form of glucose in muscle and liver, is limited, and its depletion causes fatigue [
1,
2]. Therefore, glycogen sparing is considered an effective and key method to improve endurance capacity and facilitate fatty acid utilization [
3,
4]. We previously reported that the continuous intake of green tea catechins (GTC) increases fatty acid oxidation in skeletal muscle (the major energy metabolism tissue) and improves endurance capacity by suppressing consumption of muscle glycogen [
5,
6].
Conversely, effective supplementation of energy sources before and during exercise is also considered a key strategy to improve endurance capacity. For example, glucose intake as a carbohydrate (CHO) source during exercise significantly increases endurance capacity by continuously suppressing decreases in blood glucose [
7,
8]. Results from several studies have indicated that the single administration of CHO is insufficient to improve endurance performance, while repeated intake of CHO during exercise is very effective to continuously maintain blood glucose levels [
9,
10]. However, in situations like athletic competition there may be restricted access to supplementation. Thus, it would be beneficial to increase the amount of CHO to a level that sustains blood glucose levels with every intake. Exogenous CHO supplementation, endogenous glycogenolysis, and gluconeogenesis contribute to the maintenance of blood glucose levels during exercise. Nogusa et al. [
11] reported that combined ingestion of the gluconeogenic amino acids alanine (Ala) and proline (Pro) with maltodextrin (MD) as a CHO source before exercise effectively maintains blood glucose levels and hepatic glycogen content, and leads to improved endurance performance in mice.
Continuous intake of GTC improves endurance capacity by facilitating metabolic activity to utilize more fatty acids as an energy source, and combined supplementation of Ala and Pro with CHO before exercise immediately contributes to endurance performance by sustainably supplying a CHO energy source during subsequent exercise. These interventions have different mechanisms of action for endurance performance, but they work closely with each other for energy production. Therefore, we hypothesized that the combination of these interventions effectively improves endurance performance. In this study, we investigated this combined effect on endurance performance using treadmill running in mice.
2. Materials and Methods
2.1. Animals
Male 6-week-old BALB/c mice obtained from Charles River (Kanagawa, Japan) were maintained at 23 ± 2 °C under a 12-h light-dark cycle (lights on from 7:00 to 19:00). All animal experiments were approved by the Animal Care Committee of Kao Corporation. All animal experiments followed this committee's Guidelines for the Care and Use of Laboratory Animals. Before any experiment, mice were fed a laboratory diet (CE-2, CLEA Japan, Tokyo, Japan) to stabilize their metabolism. The macronutrient composition of CE-2 was 4.6% fat, 51.4% carbohydrate, and 24.9% protein.
2.2. Materials and Experimental Diets
Polyphenon 70S (Mitsui Norin Co. Ltd., Shizuoka, Japan) used as the GTC mixture contained 80.9% catechins, including epigallocatechin gallate (EGCg, 32.3%), epigallocatechin (EGC, 20%), epicatechin gallate (ECg, 8.8%), epicatechin (EC, 7.9%), gallocatechin gallate (GCg, 3.2%), gallocatechin (GC, 6.1%), catechin (C, 1.8%), and catechin gallate (Cg, 0.8%). Mice were allowed ad libitum access to water and one of two powder diets during experiments. The control diet contained 10% fat (w/w), 20% casein, 55.5% potato starch, 8.1% cellulose, 4% minerals, 2.2% vitamins, and 0.2% methionine. The GTC diet consisting of the control diet supplemented with 0.5% GTC. Dietary intake was measured throughout the experimental period by subtracting the weight of the remaining food from the initial weight of the food given on the previous feeding day. Maltodextrin (TK-16, Matsutani Chemical Industry, Hyogo, Japan), DL-Ala (L-Ala: D-Ala was approximately 1:1), and L-Pro were provided from Ajinomoto Co., Inc. (Tokyo, Japan). MD, DL-Ala, and L-Pro were dissolved in saline as a vehicle immediately before use.
2.3. Experimental Design
We performed two experiments in this study. In Experiment 1, we evaluated the change over time in running endurance capacity with interventions. In Experiment 2, we focused on energy metabolic status during running exercise.
2.3.1. Experiment 1
Male 6-week-old BALB/c mice were allowed ad libitum access to water and the CE-2 diet for acclimation. At 7 weeks of age, mice were assigned to the 3-day treadmill running training (details of treadmill conditions are described in
Section 2.4.). At 8 weeks of age, initial running endurance capacity was measured for 104 of the 120 mice that had adapted to the running exercise without problems. To minimize variations in endurance performance, 56 of 104 mice were selected centering on the overall average running time and then divided into seven experimental groups (
n = 8) with similar average running times and body weight (B.W.) (
Table 1).
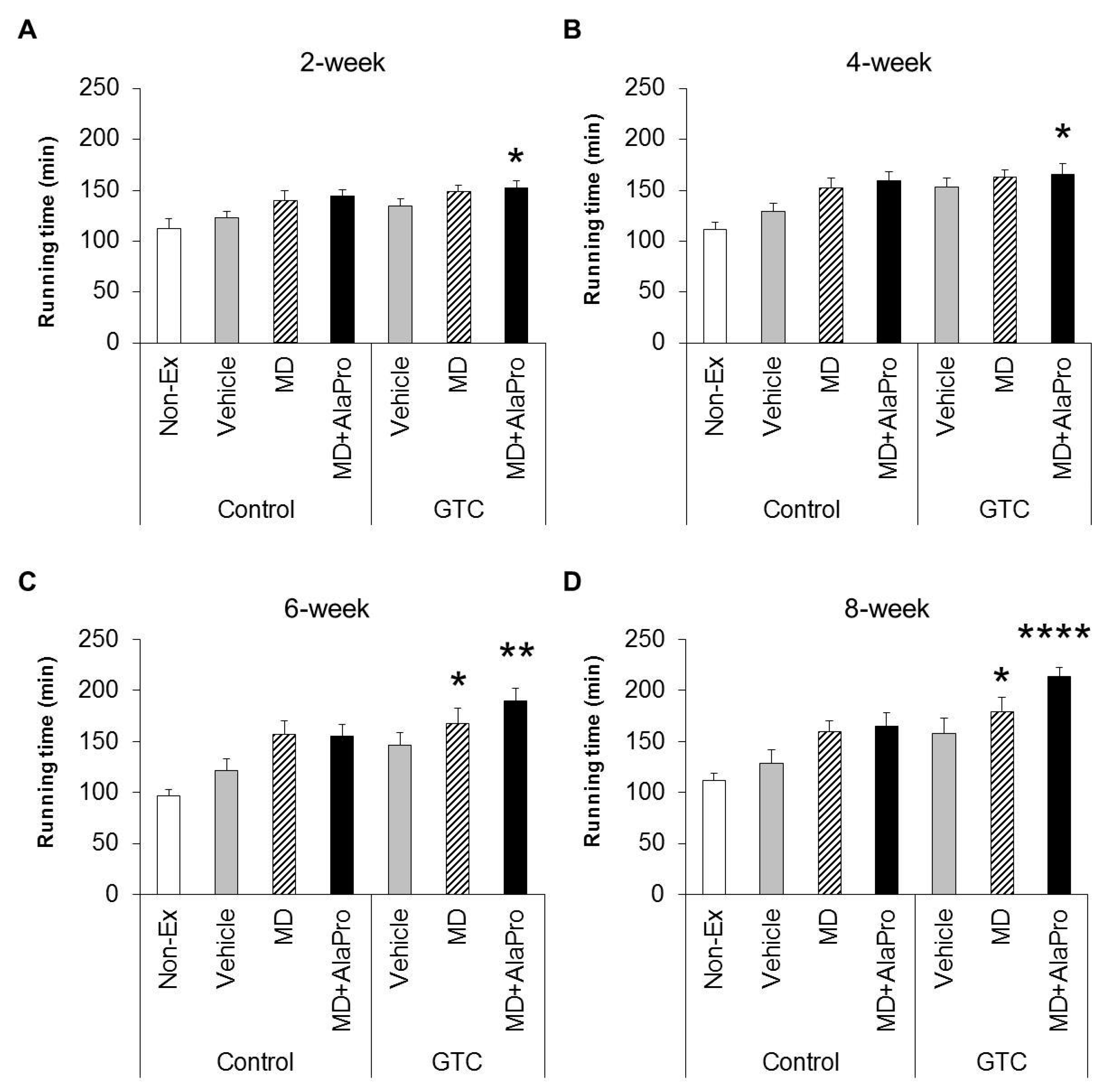
During the experimental period, mice were fed the Control or 0.5% GTC diet and subjected to a 30 mins treadmill running exercise 4 days a week, except for the Non-Ex group. After 2, 4, 6, and 8 weeks from the start of the experiment, endurance capacity was evaluated for each pre-exercise administration using the same methods as the initial measurement. Mice were orally administrated 10 mL/kg B.W. vehicle, MD (2.0 g/kg B.W.), or MD with supplemented AlaPro (1.0 g/kg MD + 0.9 g/kg DL-Ala + 0.1 g/kg L-Pro) solution 60 mins before evaluation of endurance capacity. Two days after final endurance measurements at week 8, mice were anesthetized with isoflurane (Abbott Japan, Tokyo, Japan). Subsequently, blood and tissues, such as liver and skeletal muscles (gastrocnemius, soleus, quadriceps, and tibialis anterior muscles), were collected and weighed. The tissues were immediately frozen in liquid nitrogen and stored at −80 °C.
2.3.2. Experiment 2
According to the same procedure in Experiment 1, 16 of 32 male 8-week-old BALB/c mice were selected and divided into the Control and 0.5% GTC diet group, with similar average running times (145.5 ± 7.6 and 145.2 ± 7.6, respectively) and B.W. Both groups were fed each experimental diet and exercised by treadmill running 4 days a week. Energy metabolism during running was determined by indirect calorimetry analysis (described in
Section 2.5) at week 4. At week 6, both mouse groups were fasted for 2 h and then orally administrated MD supplemented with AlaPro (1.0 g/kg MD + 0.9 g/kg DL-Ala + 0.1 g/kg L-Pro) 60 mins before exercise. Blood and tissue samples were collected from anesthetized mice immediately after 60 mins of running at 20 m/min.
2.4. Running Exercise and Evaluation of Endurance Performance
A 10-lane motorized rodent treadmill (MK-680; Muromachi Kikai, Tokyo, Japan) with an 8° incline was used for the running exercise and to evaluate endurance capacity. The initial 3-day treadmill training was conducted according to the following program: day 1, 10 m/min for 10 mins and 15 m/min for 20 mins; day 2, 10 m/min for 10 mins, 15 m/min for 10 mins, and 20 m/min for 10 mins; and day 3, 15 m/min for 10 mins, 20 m/min for 10 mins, and 25 m/min for 10 min. Evaluation of endurance capacity was according to following program: 10 m/min for 6 mins, 12 m/min for 2 mins, 14 m/min for 2 mins, 16 m/min for 2 mins, 18 m/min for 2 mins, 20 m/min for 2 mins, 22 m/min for 2 mins, 24 m/min for 2 mins, 26 m/min for 2 mins, and 28 m/min until exhaustion. The daily running exercise program during the experimental period was 15 m/min for 10 mins and 20 m/min for 20 mins 4 days a week.
2.5. Indirect Calorimetry Analysis under Exercise Conditions
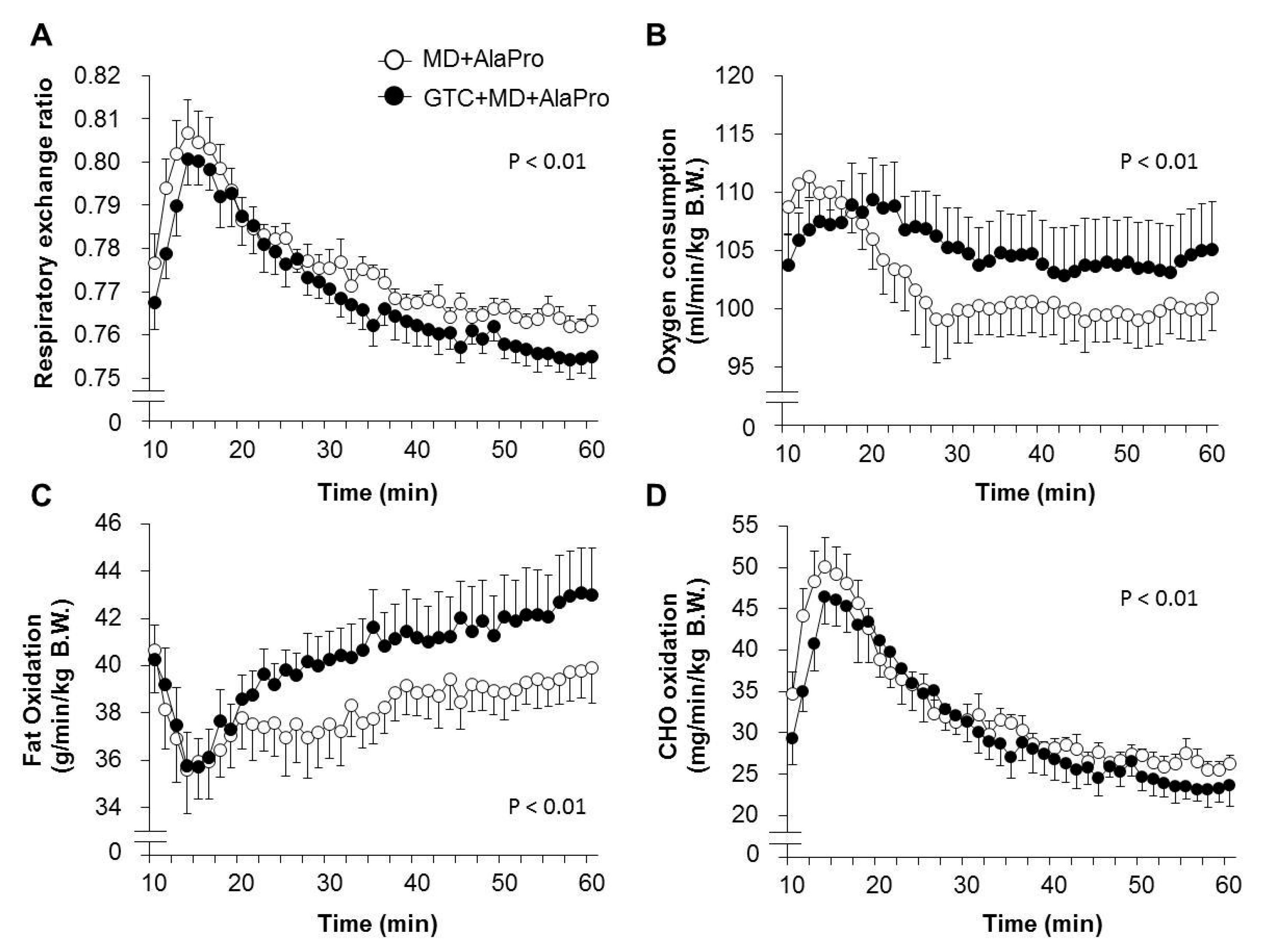
In Experiment 2, energy metabolism analyses under running exercise conditions were performed by using an ARCO-2000 magnetic-type mass spectrometric calorimeter (ARCO System, Chiba, Japan) with a four-lane airtight rodent treadmill with 5° incline setting (Modular Treadmill System; Columbus Instruments, Columbus, OH, USA), with one mouse per lane. Mice were fasted 120 mins before measurements, and MD supplemented with AlaPro (1.0 g/kg MD + 0.9 g/kg DL-Ala + 0.1 g/kg L-Pro) were orally provided to all mice 60 mins before measurements. Mice were placed in a treadmill chamber for 30 mins before measurements to acclimate to the surroundings. Mice ran with an initial speed of 10 m/min for 5 mins, to adapt to running gradually on the treadmill. The running speed was then changed to 15 m/min for 5 mins and subsequently to 20 m/min for 50 mins. Data were collected every 75 secs throughout the 60-min running period. The respiratory exchange ratio (RER) was calculated from the measured values of oxygen consumption (VO
2) and carbon dioxide exhalation (VCO
2), and fat oxidation and carbohydrate oxidation were calculated using the following equations [
12]:
For data comparison between the MD + AlaPro and GTC + MD + AlaPro group, the measurements at the 20 m/min for 50 mins portion of the run were selected for analysis to avoid the effect of a rapid change once running was initiated.
2.6. Liver and Muscle Glycogen Contents
Liver and gastrocnemius muscle glycogen contents after the 60-min running exercise in Experiment 2 were measured as described previously [
6]. In brief, 10 mg of liver and 50 mg of gastrocnemius muscle were digested in 300 L of 30% KOH for 30 mins in a boiling water bath. After 50 L of saturated sodium sulfate was added, the glycogen was precipitated by adding 500 L of 95% ethanol and centrifuged at 1600×
g. The supernatant was decanted and the remaining ethanol was vaporized in an incubator at 80 °C for 15 mins. The pellet was dissolved in 200 L of H
2O and reprecipitated with 250 L of 95% ethanol. The supernatant was decanted after centrifugation at 1600× g, and the remaining alcohol was vaporized. Purified glycogen was hydrolyzed in 600 L of 0.6 N HCl at 100 °C for 3 h. Glucose levels were determined with the Glucose CII test kit (Wako Pure Chemical Industries, Wako, Japan) and converted to the glycogen concentration.
2.7. Statistical Analyses
All values are presented as mean ± standard error of the mean (SE). Statistical analysis was conducted using ANOVA followed by the Dunnett’s test with the Control-Vehicle group in Experiment 1. In Experiment 2, the comparison of the time course changes in the indirect calorimetry analysis between Control and GTC diet group running at 20 m/min was performed using two-way repeated measures ANOVA. This analysis was performed to determine the main effects corresponding to group and time, as well as the interaction between the two. The unpaired t-test was used to compare between two groups. A P-value < 0.05 was considered statistically significant. Data were organized and analyzed using the Microsoft Excel 2010 (Microsoft Corp., Redmond, WA, USA) and GraphPad Prism 6 (GraphPad software Inc., La Jolla, CA, USA).
4. Discussion and Conclusions
Our main findings in this study are as follows: (1) pre-exercise single intake of CHO with Ala and Pro (MD + AlaPro) increased the endurance improving effect of continuous GTC intake more effectively than CHO alone and (2) the increase of sustained fat utilization for energy production during running exercise potentially contributed to this combinational effect.
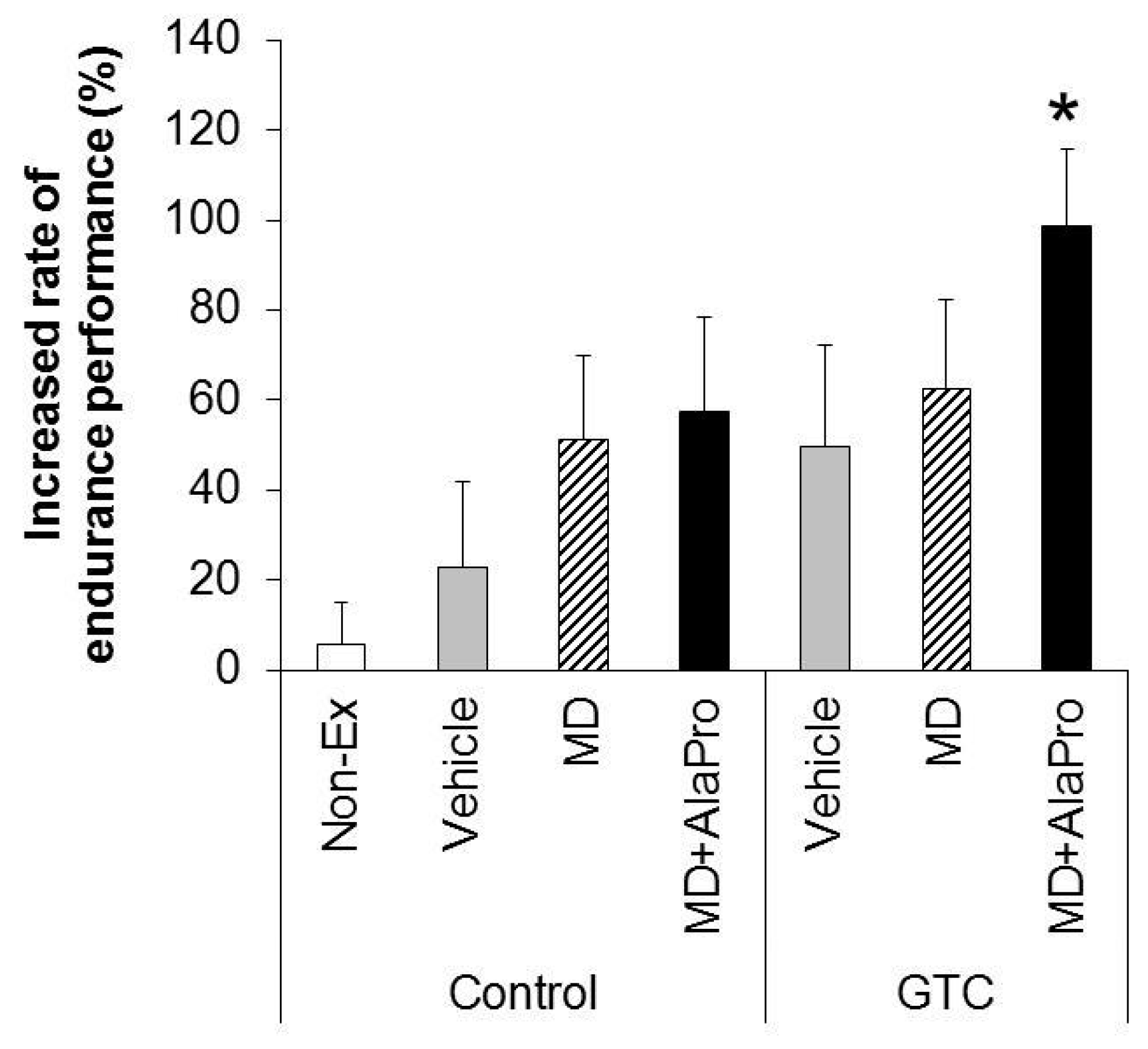
MD + AlaPro increased the effectiveness of improved endurance performance by dietary GTC intake and succeeded in eliciting a significant effect at early 2- and 4-week GTC intervention. The increased rate of endurance performance from the initial time to week 8 of intervention was significantly higher only in the GTC + MD + AlaPro group compared to the Control-Vehicle group. These results suggest that pre-exercise MD + AlaPro intake is quite effective for enhancing the endurance improving effect by continuous GTC intake. In the Control diet group, both MD + AlaPro and MD alone tended to increase endurance performance compared to the Vehicle group. The difference was not significant. In contrast, MD + AlaPro was more effective in endurance improvement than MD alone in a previous study [
11]. We think that the difference in treadmill conditions while evaluating endurance performance could have had a profound effect on this difference between the two studies. The treadmill speed during endurance measurement in the previous study (36 m/min) [
11] was much faster than the speed of 28 m/min used in our study. Since CHO metabolism has a greater influence on energy metabolism at high exercise intensity, the difference between MD and MD + AlaPro was clearly observed in the previous study, whereas our current measurement conditions might be more likely to be affected by lipid metabolism, which is the main mechanism of GTC, than by CHO metabolism. In any case, MD + AlaPro administration was able to consistently increase endurance capacity by only a single intake at any measurement timing. Conversely, GTC showed gradually increased effectiveness in endurance with continuous intake but was less effective in the early stage of intervention. Endurance was synergistically improved by MD + AlaPro in proportion to continuous GTC intake, suggesting that combining these interventions could be beneficial as a comprehensive control method for energy metabolism during exercise.
Fatty acids have higher energy production capacity and larger storage amounts in the body compared to CHO, therefore, enhancing lipid utilization can be an effective strategy for improving endurance [
4]. Fatty acids are metabolized to acetyl-CoA in the mitochondria and converted into energy by entering the tricarboxylic acid (TCA) cycle. Although acetyl-CoA is also supplied from CHO through glycolysis, an increased supply of acetyl-CoA from fatty acid could lead to the suppression of carbohydrate utilization and glycogen sparing. Previous studies revealed that continuous GTC intake significantly decreased RER during exercise, accompanied by high fatty acid-oxidation in skeletal muscle [
5,
6]. In this study, we confirmed higher fat oxidation in the GTC diet group during running exercise than the Control diet group under the same pre-exercise conditions of MD + AlaPro administration. This upregulated fatty acid utilization by continuous GTC intake and the sustained blood glucose maintenance by pre-exercise MD + AlaPro intake might contribute to efficient energy production via acetyl-CoA from fatty acids, resulting in improved endurance capacity.
During prolonged exercise, increases in TCA cycle intermediates are also needed to sustain sufficient aerobic energy production [
13,
14]. To produce ATP through the TCA cycle reaction, oxaloacetate needs to combine with acetyl-CoA and convert to citrate. With increased ATP consumption in the muscle during exercise, increased oxaloacetate induces citrate synthase activation, resulting in ATP production in the TCA cycle and fatty acid oxidation. Endurance exercise training induces fatty acid utilization through the coordinated activation of the mitochondrial fatty acid oxidation enzyme complex and citrate synthase in the skeletal muscle [
15,
16], indicating the importance of the TCA cycle on endurance performance. CHO supplementation before and during exercise increases TCA intermediates in the muscle and contributes to improved endurance performance [
17,
18]. In addition to the role as CHO supplier, the gluconeogenic amino acids, Ala and Pro, are converted to the respective TCA intermediates of oxaloacetate and alpha-ketoglutalate. In the condition that GTC intake increases acetyl-CoA supply through fatty acid oxidation, increased oxaloacetate supply may contribute to the promotion of the TCA cycle reaction. Since pyruvate from glycolysis and Ala are the sources of oxaloacetate, combinations to increase oxaloacetate and acetyl-CoA from fatty acid oxidation through GTC intake may synergistically contribute to TCA cycle facilitation and ATP production. To clarify the precise mechanism in the combinational effect of GTC + MD + AlaPro, further studies including determination of time-course changes in blood components related to energy metabolism, such as glucose, lactate, fatty acid, and amino acids, and TCA intermediates in muscles during exercise, are needed.
We hypothesized that GTC + MD + AlaPro administration would result in glycogen sparing and contribute to increased endurance performance. However, we could not find a difference in liver and muscle glycogen storage after exercise between the MD + AlaPro and GTC + MD + AlaPro groups. To observe the effect on glycogen sparing, we loaded a 60-min running exercise at the 20 m/min speed for glycogen consumption. This is based on the following two reasons. First, it was achievable for all mice according to the endurance measurements in Experiment 1. Second, an increase in fat oxidation was stably observed during the running exercise by indirect calorimetry analysis in Experiment 2. In fact, the minimum running time for mice fed the MD + AlaPro condition was over 130 mins in this study. Therefore, the 60-min running load might not have been enough to observe differences in glycogen consumption. To confirm the precise effect on glycogen sparing, we need to determine appropriate test conditions for glycogen consumption in another study.
In conclusion, the combination of increased fatty acid utilization by continuous GTC intake and effective pre-exercise CHO energy supplementation by MD + AlaPro synergistically improves endurance capacity. These observations indicate that combining nutritional interventions based on different mechanisms of action in energy metabolism may be an effective strategy to improve endurance performance.