Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
2.3. Cell Culture and Differentiation
2.4. Oil Red O Staining
2.5. Biochemical Analysis
2.6. Histological Analysis
2.7. mRNA Quantification by RT-PCR
2.8. Western Blotting
2.9. Statistical Analysis
3. Results
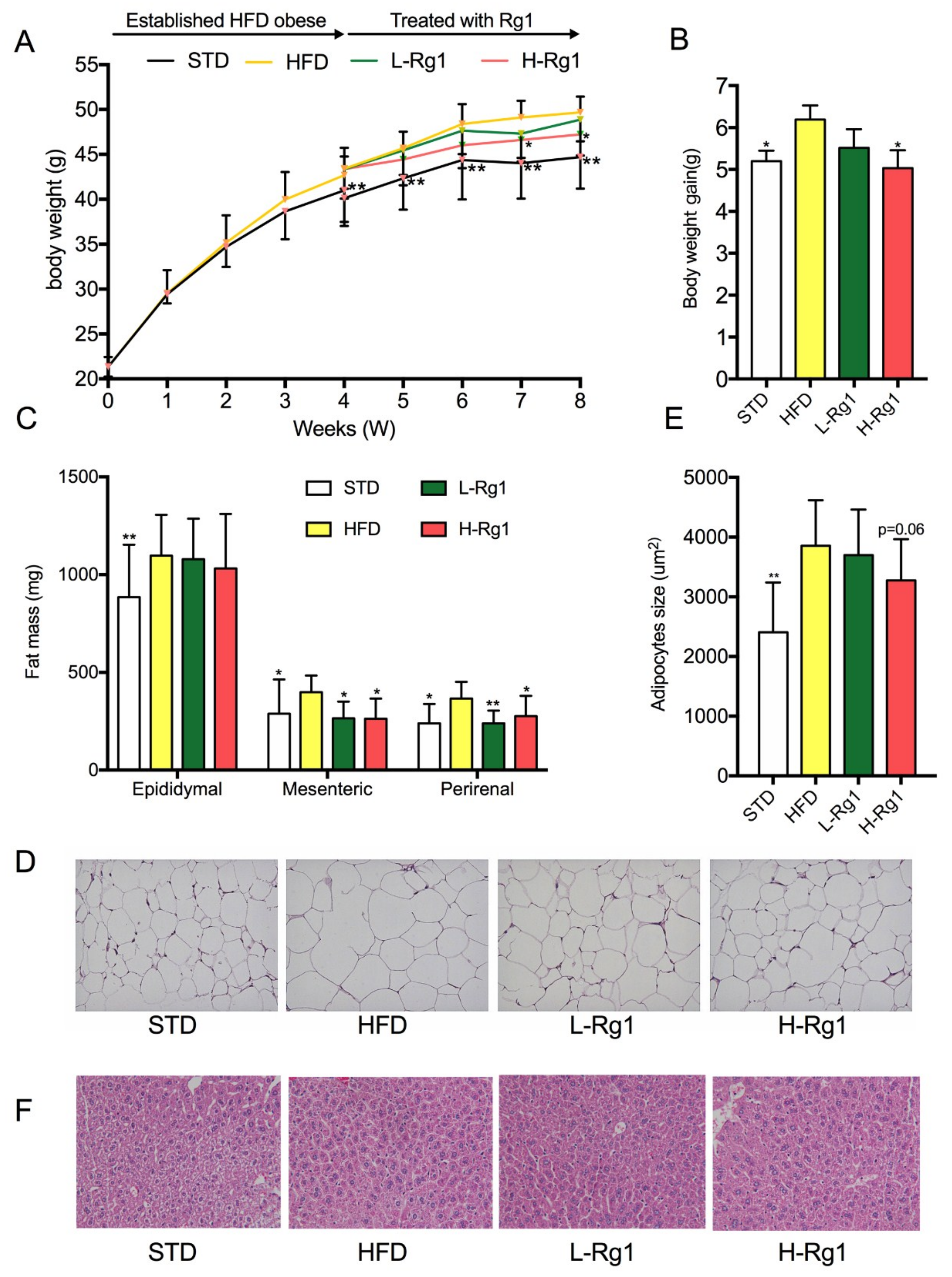
3.1. Effects of Rg1 on Body Weight and Adipose Tissue Mass
3.2. Effect of Rg1 on Serum Biochemical Parameters of Mice
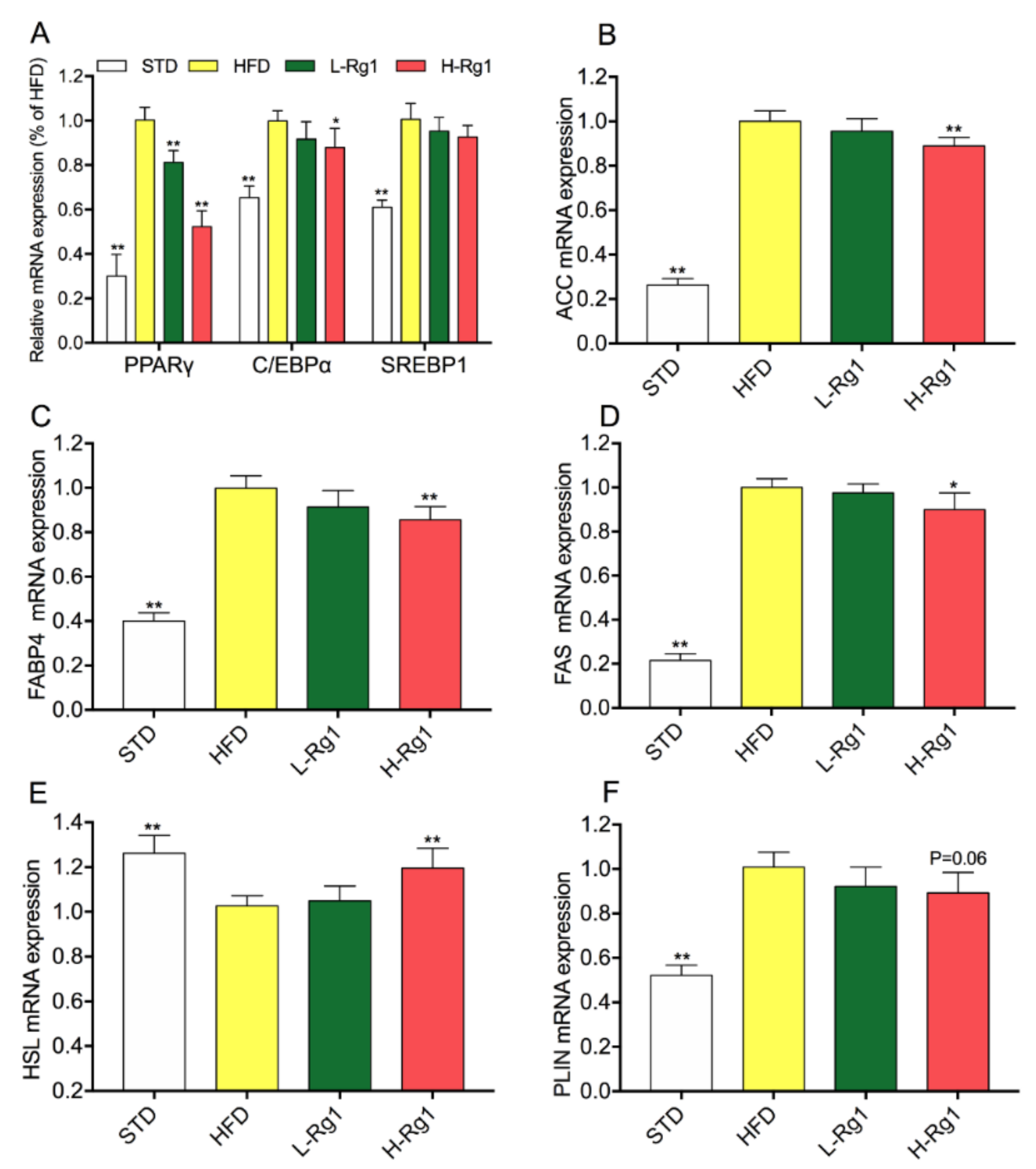
3.3. Effect of Rg1 on Regulated Gene Expression of Lipid Metabolism in Epididymal Adipose Tissue
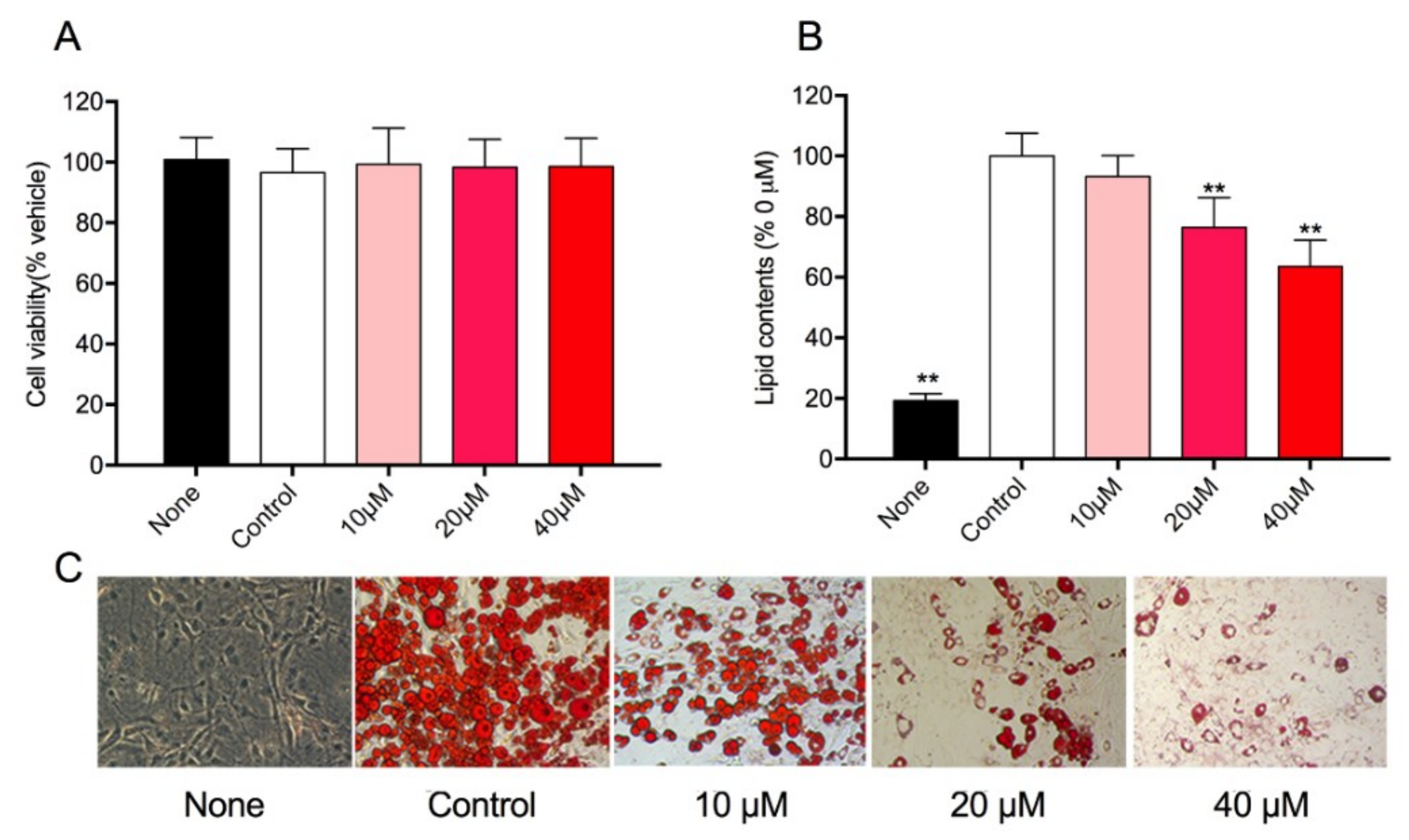
3.4. Effect of Rg1 on Cell Viability and Intracellular Lipid Accumulation in 3T3-L1 Cells
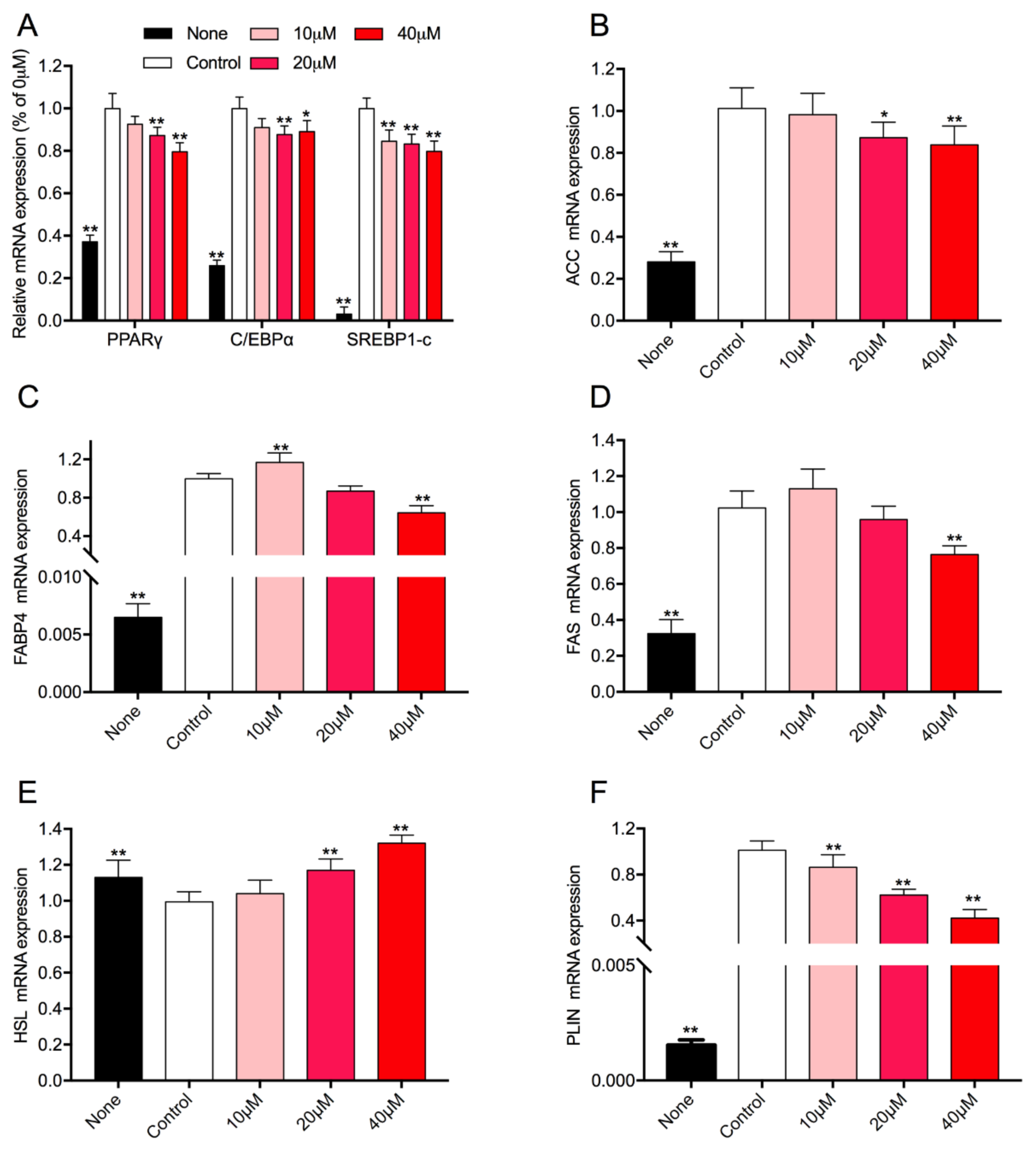
3.5. Effect of Rg1 on the Expression Level of Adipogenic-Specific Genes in 3T3-L1 Cells
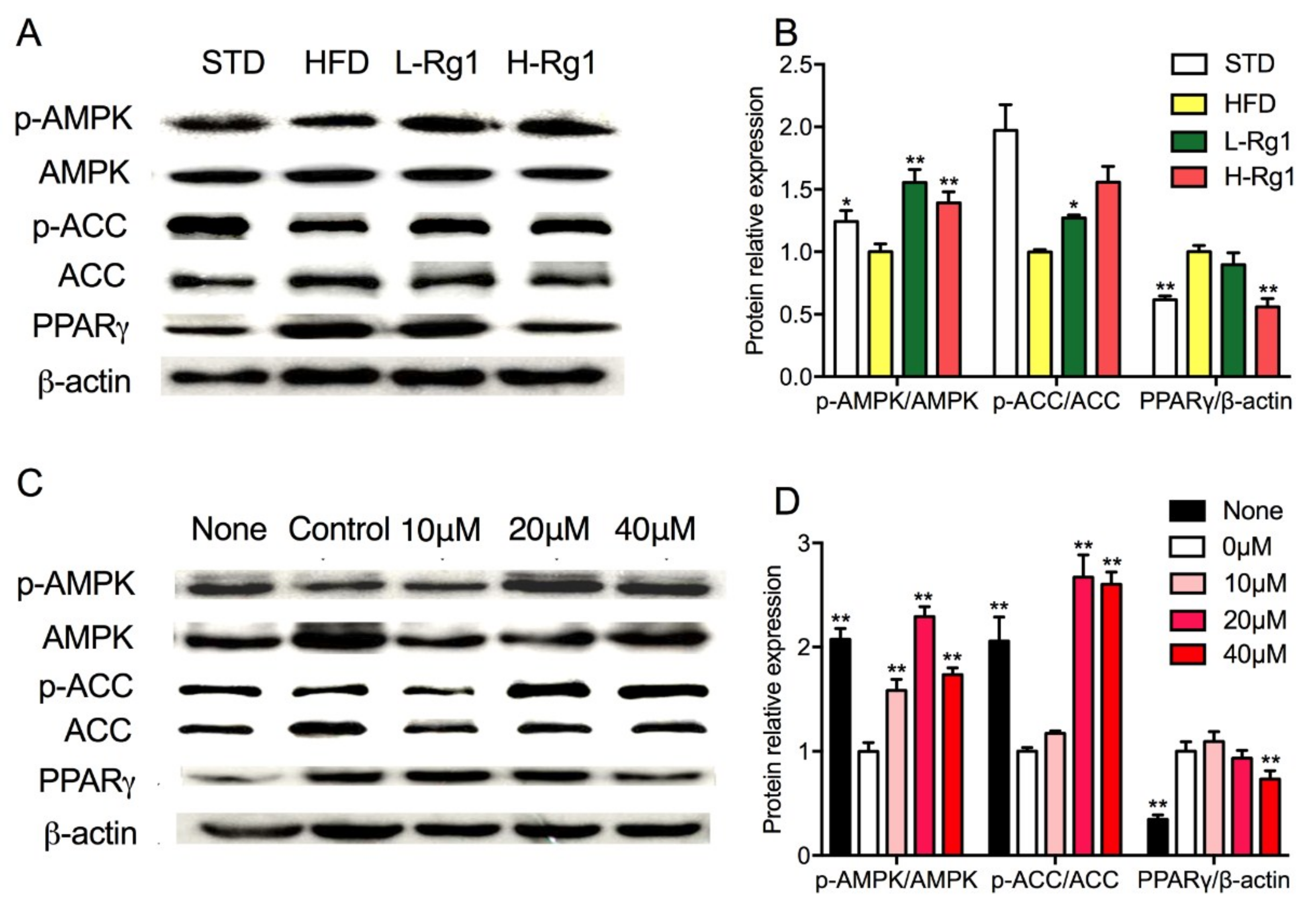
3.6. Effects of Rg1 on AMPK Activation in Adipose Tissue and Adipocytes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Collaborators, G.B.D.O.; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Gadde, K.M.; Martin, C.K.; Berthoud, H.R.; Heymsfield, S.B. Obesity: Pathophysiology and management. J. Am. Coll. Cardiol. 2018, 71, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Saunders, K.H.; Shukla, A.P.; Igel, L.I.; Kumar, R.B.; Aronne, L.J. Pharmacotherapy for obesity. Endocrinol. Metab. Clin. North Am. 2016, 45, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Garach, A.; Cornejo-Pareja, I.; Tinahones, F.J. Does metabolically healthy obesity exist? Nutrients 2016, 8, 320. [Google Scholar] [CrossRef] [PubMed]
- Mota de Sa, P.; Richard, A.J.; Hang, H.; Stephens, J.M. Transcriptional regulation of adipogenesis. Compr. Physiol. 2017, 7, 635–674. [Google Scholar] [PubMed]
- Smith, U.; Kahn, B.B. Adipose tissue regulates insulin sensitivity: Role of adipogenesis, de novo lipogenesis and novel lipids. J. Intern. Med. 2016, 280, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Herzig, S.; Shaw, R.J. Ampk: Guardian of metabolism and mitochondrial homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Day, E.A.; Ford, R.J.; Steinberg, G.R. Ampk as a therapeutic target for treating metabolic diseases. Trends Endocrinol. Metab. 2017, 28, 545–560. [Google Scholar] [CrossRef] [PubMed]
- Ru, W.; Wang, D.; Xu, Y.; He, X.; Sun, Y.E.; Qian, L.; Zhou, X.; Qin, Y. Chemical constituents and bioactivities of panax ginseng (c. A. Mey). Drug Discov. Ther. 2015, 9, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Majeed, F.; Malik, F.Z.; Ahmed, Z.; Afreen, A.; Afzal, M.N.; Khalid, N. Ginseng phytochemicals as therapeutics in oncology: Recent perspectives. Biomed. Pharmacother. 2018, 100, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Shin, B.K.; Kwon, S.W.; Park, J.H. Chemical diversity of ginseng saponins from panax ginseng. J. Ginseng Res. 2015, 39, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ji, G.E. Ginseng and obesity. J. Ginseng Res. 2018, 42, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Kim, S.H.; Lee, M.S.; Kim, S.H.; Yang, H.J.; Kim, M.J.; Kim, H.S.; Ha, J.; Kim, M.S.; Kwon, D.Y. Anti-obesity effects of ginsenoside rh2 are associated with the activation of ampk signaling pathway in 3t3-l1 adipocyte. Biochem. Biophys. Res. Commun. 2007, 364, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Siraj, F.M.; SathishKumar, N.; Kim, Y.J.; Kim, S.Y.; Yang, D.C. Ginsenoside f2 possesses anti-obesity activity via binding with ppargamma and inhibiting adipocyte differentiation in the 3t3-l1 cell line. J. Enzyme Inhib. Med. Chem. 2015, 30, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Shen, L.; Liu, K.J.; Tso, P.; Xiong, Y.; Wang, G.; Woods, S.C.; Liu, M. Antiobesity and antihyperglycemic effects of ginsenoside rb1 in rats. Diabetes 2010, 59, 2505–2512. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.T.; Zhen, J.; Pang, B.; Gu, J.N.; Wu, S.S. Ginsenoside rg1 ameliorates oxidative stress and myocardial apoptosis in streptozotocin-induced diabetic rats. J. Zhejiang Univ. Sci. B 2015, 16, 344–354. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, F.G.; Zhang, W.S.; Pan, A.; Yang, Y.L.; Liu, J.F.; Li, P.; Liu, B.L.; Qi, L.W. Ginsenoside rg1 inhibits glucagon-induced hepatic gluconeogenesis through akt-foxo1 interaction. Theranostics 2017, 7, 4001–4012. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Choi, H.K.; Brinckmann, J.A.; Jiang, X.; Huang, L. Chemical analysis of panax quinquefolius (north american ginseng): A review. J. Chromatogr. A 2015, 1426, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Xiao, W.; Lan, D.; Juan, L.; QU, C.-L.; Chen, L.-G.; Jin, H.-Y.; Zhang, H.-Q. Determination of seven major ginsenosides in different parts of panax quinquefolius l.(american ginseng) with different ages. Chem. Res. Chin. Univ. 2008, 24, 707–711. [Google Scholar]
- Shi, W.; Wang, Y.; Li, J.; Zhang, H.; Ding, L. Investigation of ginsenosides in different parts and ages of panax ginseng. Food Chem. 2007, 102, 664–668. [Google Scholar] [CrossRef]
- Cheng, W.; Jing, J.; Wang, Z.; Wu, D.; Huang, Y. Chondroprotective effects of ginsenoside rg1 in human osteoarthritis chondrocytes and a rat model of anterior cruciate ligament transection. Nutrients 2017, 9, 263. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chu, S.; Zhang, Z.; Chen, N. Hepataprotective effects of ginsenoside rg1—A review. J. Ethnopharmacol. 2017, 206, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Li, J.B.; Zhang, R.; Han, X.; Piao, C.L. Ginsenoside rg1 inhibits dietary-induced obesity and improves obesity-related glucose metabolic disorders. Braz. J. Med. Biol. Res. 2018, 51, e7139. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Liu, H.; Xie, J.; Xu, Q.; Pan, C.; Wang, J.; Wu, X.; Zheng, M.; Liu, J. Anti-obesity effects of zeaxanthin on 3t3-l1 preadipocyte and high fat induced obese mice. Food Funct. 2017, 8, 3327–3338. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yu, Y.; Szabo, A.; Han, M.; Huang, X.F. Central inflammation and leptin resistance are attenuated by ginsenoside rb1 treatment in obese mice fed a high-fat diet. PLoS ONE 2014, 9, e92618. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Xiong, Y.; Wang, D.Q.; Howles, P.; Basford, J.E.; Wang, J.; Xiong, Y.Q.; Hui, D.Y.; Woods, S.C.; Liu, M. Ginsenoside rb1 reduces fatty liver by activating amp-activated protein kinase in obese rats. J. Lipid Res. 2013, 54, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Fang, X.; Li, X.; Zhao, D.; Mo, F.; Jiang, G.; Yu, N.; Zhang, Y.; Guo, Y.; Fu, M.; et al. Ginsenoside rb1 promotes browning through regulation of ppargamma in 3t3-l1 adipocytes. Biochem. Biophys. Res. Commun. 2015, 466, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, L.; Wang, X.; Si, H. Anti-adipogenic effects and mechanisms of ginsenoside rg3 in pre-adipocytes and obese mice. Front. Pharmacol. 2017, 8, 113. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Lee, M.S.; Kim, H.J.; Sung, M.J.; Kim, H.Y.; Kim, M.S.; Kwon, D.Y. Antiobesity effect of ginsenoside rg3 involves the ampk and ppar-gamma signal pathways. Phytother. Res. 2009, 23, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Lee, O.H.; Lee, H.H.; Kim, J.H.; Lee, B.Y. Effect of ginsenosides rg3 and re on glucose transport in mature 3t3-l1 adipocytes. Phytother. Res. 2011, 25, 768–773. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.B.; Yoon, S.J.; Lee, S.H.; Lee, M.S.; Jung, H.; Kim, T.D.; Yoon, S.R.; Choi, I.; Kim, I.S.; Chung, S.W.; et al. Ginsenoside rg3 ameliorated hfd-induced hepatic steatosis through downregulation of stat5-ppargamma. J. Endocrinol. 2017, 235, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, M.S.; Kim, C.T.; Kim, I.H.; Kim, Y. Ginsenoside rg3 reduces lipid accumulation with amp-activated protein kinase (ampk) activation in hepg2 cells. Int. J. Mol. Sci. 2012, 13, 5729–5739. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Kim, K.A.; Kim, D.H. Ginsenoside rh1 ameliorates high fat diet-induced obesity in mice by inhibiting adipocyte differentiation. Biol. Pharm. Bull. 2013, 36, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Yoon, M. Compound k, a novel ginsenoside metabolite, inhibits adipocyte differentiation in 3t3-l1 cells: Involvement of angiogenesis and mmps. Biochem. Biophys. Res. Commun. 2012, 422, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.; Hong, Y.; Xu, J.; Lin, Y.; Si, Q.; Gu, X. Ginsenoside rb2 promotes glucose metabolism and attenuates fat accumulation via akt-dependent mechanisms. Biomed. Pharmacother. 2018, 100, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, J.M.; Stern, J.H.; Scherer, P.E. The cell biology of fat expansion. J. Cell Biol. 2015, 208, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Langin, D. In and out: Adipose tissue lipid turnover in obesity and dyslipidemia. Cell Metab. 2011, 14, 569–570. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ojeda, F.J.; Ruperez, A.I.; Gomez-Llorente, C.; Gil, A.; Aguilera, C.M. Cell models and their application for studying adipogenic differentiation in relation to obesity: A review. Int. J. Mol. Sci. 2016, 17, 1040. [Google Scholar] [CrossRef] [PubMed]
- Rosen, E.D.; Spiegelman, B.M. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 2006, 444, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. Ampk: A nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.J.; Kim, G.W.; Chung, S.H. Amp-activated protein kinase: An emerging target for ginseng. J. Ginseng Res. 2014, 38, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Yuan, H.D.; Chung, S.H. Ginsenoside rg1 suppresses hepatic glucose production via amp-activated protein kinase in hepg2 cells. Biol. Pharm. Bull. 2010, 33, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Park, M.W.; Yuan, H.D.; Lee, H.J.; Kim, S.H.; Chung, S.H. Compound k induces apoptosis via camk-iv/ampk pathways in ht-29 colon cancer cells. J. Agric. Food Chem. 2009, 57, 10573–10578. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Yuan, H.D.; Chung, I.K.; Chung, S.H. Compound k, intestinal metabolite of ginsenoside, attenuates hepatic lipid accumulation via ampk activation in human hepatoma cells. J. Agric. Food Chem. 2009, 57, 1532–1537. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Wang, T.; Yang, L.; Wang, H.Y. Ginsenoside rb2 alleviates hepatic lipid accumulation by restoring autophagy via induction of sirt1 and activation of ampk. Int. J. Mol. Sci. 2017, 18, 1063. [Google Scholar] [CrossRef] [PubMed]
- Quan, H.Y.; Yuan, H.D.; Jung, M.S.; Ko, S.K.; Park, Y.G.; Chung, S.H. Ginsenoside re lowers blood glucose and lipid levels via activation of amp-activated protein kinase in hepg2 cells and high-fat diet fed mice. Int. J. Mol. Med. 2012, 29, 73–80. [Google Scholar] [PubMed]
- Huang, Y.C.; Lin, C.Y.; Huang, S.F.; Lin, H.C.; Chang, W.L.; Chang, T.C. Effect and mechanism of ginsenosides ck and rg1 on stimulation of glucose uptake in 3t3-l1 adipocytes. J. Agric. Food Chem. 2010, 58, 6039–6047. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.R.; Yang, C.; Wong, T.Y.; Popovich, D.G. A quantified ginseng (panax ginseng c.A. Meyer) extract influences lipid acquisition and increases adiponectin expression in 3t3-l1 cells. Molecules 2011, 16, 477–492. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.R.; Lee, S.M.; Popovich, D.G. Ginseng (panax quinquefolius) reduces cell growth, lipid acquisition and increases adiponectin expression in 3t3-l1 cells. Evid.- Based Complement. Altern. Med. 2011, 2011, 610625. [Google Scholar] [CrossRef] [PubMed]
| Formula | g/kg |
|---|---|
| Standard laboratory chow | 333.3 |
| Casein | 200.0 |
| Hydrogenated coconut oil | 220.0 |
| Soybean oil | 16.7 |
| Corn starch | 102.2 |
| Maltodextrin | 102.0 |
| Sodium carbonate | 6.7 |
| Potassium citrate | 2.7 |
| Choline bitartrate | 1.4 |
| Vitamin and mineral mixture | 15.0 |
| Total | 1000.0 |
| Groups | STD | HFD | L-Rg1 | H-Rg1 |
|---|---|---|---|---|
| Lee’s index | 3.49 ± 0.10 | 3.53 ± 0.07 | 3.44 ± 0.10 * | 3.42 ± 0.15 * |
| Relative adipose weight (mg/g bodyweight) | 34.92 ± 3.90 ** | 42.60 ± 3.76 | 41.77 ± 3.00 | 40.32 ± 4.36 |
| Relative liver weight (mg/g bodyweight) | 31.09 ± 5.54 ** | 37.60 ± 4.40 | 33.19 ± 6.2 | 32.51 ± 6.43 * |
| Groups | STD | HFD | L-Rg1 | H-Rg1 |
|---|---|---|---|---|
| TG (mmol/L) | 1.06 ± 0.15 * | 1.27 ± 0.26 | 1.12 ± 0.32 | 0.99 ± 0.23 * |
| TC (mmol/L) | 2.90 ± 0.44 ** | 3.86 ± 0.33 | 3.76 ± 0.55 | 3.10 ± 0.42 * |
| HDL (mmol/L) | 6.20 ± 0.97 ** | 8.16 ± 1.12 | 7.47 ± 1.29 | 8.64 ± 1.15 |
| LDL (mmol/L) | 0.42 ± 0.15 | 0.50 ± 0.22 | 0.51 ± 0.24 | 0.68 ± 0.33 |
| FFA (mol/L) | 730.28 ± 80.05 * | 830.64 ± 125.93 | 796.29 ± 67.08 | 785.22 ± 171.90 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Wang, J.; Liu, M.; Zhao, H.; Yaqoob, S.; Zheng, M.; Cai, D.; Liu, J. Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK. Nutrients 2018, 10, 830. https://doi.org/10.3390/nu10070830
Liu H, Wang J, Liu M, Zhao H, Yaqoob S, Zheng M, Cai D, Liu J. Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK. Nutrients. 2018; 10(7):830. https://doi.org/10.3390/nu10070830
Chicago/Turabian StyleLiu, Huimin, Jing Wang, Meihong Liu, Hongyu Zhao, Sanabil Yaqoob, Mingzhu Zheng, Dan Cai, and Jingsheng Liu. 2018. "Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK" Nutrients 10, no. 7: 830. https://doi.org/10.3390/nu10070830
APA StyleLiu, H., Wang, J., Liu, M., Zhao, H., Yaqoob, S., Zheng, M., Cai, D., & Liu, J. (2018). Antiobesity Effects of Ginsenoside Rg1 on 3T3-L1 Preadipocytes and High Fat Diet-Induced Obese Mice Mediated by AMPK. Nutrients, 10(7), 830. https://doi.org/10.3390/nu10070830






