Regression Analysis to Identify Factors Associated with Urinary Iodine Concentration at the Sub-National Level in India, Ghana, and Senegal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Survey Design
2.2. Survey Administration and Field Procedures
2.3. Indicators/Survey Tools
2.4. Determination of Urinary and Salt Iodine Content
2.5. Data Management
2.6. Data Analysis
2.6.1. Single Variable Regression Analysis
2.6.2. Multiple Variable Regression Analysis
3. Results
3.1. Iodine Status
3.2. Single Variable Regression Analyses
3.3. Multiple Variable Regression Analyses
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- United Nations Children’s Fund; World Health Organization. World Summit for Children-Mid-Decade Goal: Iodine Deficiency Disorders; Special Session; UNICEF-WHO Joint Committee on Health Policy: Geneva, Switzerland, 1994. [Google Scholar]
- Micronutrient Deficiencies. Available online: http://www.who.int/nutrition/topics/idd/en/ (accessed on 15 February 2017).
- World Health Organization (WHO). Forty-Third World Health Assembly A43/36. Available online: http://apps.who.int/iris/bitstream/10665/173415/1/WHA43_36_eng.pdf (accessed on 15 February 2017).
- Division of Communication, UNICEF. The State of the World’s Children 2017; UNICEF: New York, NY, USA, 2017. [Google Scholar]
- UNICEF. Household Consumption of Adequately Iodized Salt. Available online: https://data.unicef.org/wp-content/uploads/2015/12/Iodized-salt-consumption-SOWC-6-November-2014_67.xlsx (accessed on 30 January 2017).
- Global Scorecard of Iodine Nutrition in 2017. Available online: http://www.ign.org/cm_data/IGN_Global_Scorecard_AllPop_and_PW_May2017.pdf (accessed on 4 February 2018).
- Rohner, F.; Wirth, J.; Woodruff, B.; Chiwile, F.; Yankson, H.; Sesay, F.; Koroma, A.; Petry, N.; Pyne-Bailey, S.; Dominguez, E.; et al. Iodine status of women of reproductive age in sierra leone and its association with household coverage with adequately iodized salt. Nutrients 2016, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Laillou, A.; Sophonneary, P.; Kuong, K.; Hong, R.; Un, S.; Chamnan, C.; Poirot, E.; Berger, J.; Wieringa, F. Low urinary iodine concentration among mothers and children in Cambodia. Nutrients 2016, 8, 172. [Google Scholar] [CrossRef] [PubMed]
- Urbanization, Diet Change, and Transformation of Food Supply Chains in Asia. Available online: http://www.perhepi.org/wp-content/uploads/2014/08/3.-Urbanization-diet-change-and-transformations-of-food-supply-change-in-Asia_MSU-GCFSI-Reardon-et-al.pdf (accessed on 27 May 2016).
- Spohrer, R.; Larson, M.; Maurin, C.; Laillou, A.; Capanzana, M.; Garrett, G.S. The growing importance of staple foods and condiments used as ingredients in the food industry and implications for large-scale food fortification programs in Southeast Asia. Food Nutr. Bull. 2013, 34, S50–S61. [Google Scholar] [CrossRef] [PubMed]
- Knowles, J.; van der Haar, F.; Shehata, M.; Gerasimov, G.; Bimo, B.; Cavenagh, B.; Maramag, C.; Otico, E.; Izwardy, D.; Spohrer, R.; et al. Iodine intake through processed food: Case studies from Egypt, Indonesia, the Philippines, the Russian Federation and Ukraine, 2010–2015. Nutrients 2017, 9, 797. [Google Scholar] [CrossRef] [PubMed]
- Office of the Salt Commissioner, Government of India Quality of Salt under the Prevention of Food Adulteration (PFA) ACT. Available online: http://saltcomindia.gov.in/ (accessed on 9 January 2017).
- Ghana. Food and Agricultural Import Regulations and Standards-Narrative. FAIRS Country Report. Available online: https://gain.fas.usda.gov/Recent%20GAIN%20Publications/Food%20and%20Agricultural%20Import%20Regulations%20and%20Standards%20-%20Narrative_Accra_Ghana_12-21-2017.pdf (accessed on 18 April 2018).
- Republic of Senegal Répertoire des Normes Agroalimentaires de l’Association Sénégalaise de Normalisation (ASN). Available online: http://www.sante.gouv.sn/ckfinder/userfiles/files/25.%20normes_ASN.pdf (accessed on 18 April 2018). (In French)
- Measuring Acute Poverty in the Developing World: Robustness and Scope of the Multidimensional Poverty Index. Available online: http://www.ophi.org.uk/wp-content/uploads/ophi-wp-591.pdf (accessed on 27 May 2016).
- What Is the Multidimensional Poverty Index? Human Development Reports. Available online: http://hdr.undp.org/en/content/what-multidimensional-poverty-index (accessed on 26 May 2016).
- Swindale, A.; Bilinsky, P. Household Dietary Diversity Score (HDDS) for Measurement of Household Food Access: Indicator Guide; Food and Nutrition Technical Assistance III Project (FANTA): Washington, DC, USA, 2006. [Google Scholar]
- Kennedy, G.; Ballard, T.; Dop, M.C. Guidelines for Measuring Household and Individual Dietary Diversity; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011; ISBN 978-92-5-106749-9. [Google Scholar]
- Knowles, J.M.; Garrett, G.S.; Gorstein, J.; Kupka, R.; Situma, R.; Yadav, K.; Yusufali, R.; Pandav, C.; Aaron, G.J.; The Universal Salt Iodization Coverage Survey Team. Household coverage with adequately iodized salt varies greatly between countries and by residence type and socioeconomic status within countries: Results from 10 national coverage surveys. J. Nutr. 2017, 147, 1004S–1014S. [Google Scholar] [CrossRef] [PubMed]
- International Council for Control of Iodine Deficiency Disorders; United Nations Children’s Fund (UNICEF); World Health Organization (WHO). Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers; World Health Organization (WHO): Geneva, Switzerland, 2007; ISBN 978-92-4-159582-7. [Google Scholar]
- Pino, S.; Fang, S.-L.; Braverman, L.E. Ammonium persulfate: A safe alternative oxidizing reagent for measuring urinary iodine. Clin. Chem. 1996, 42, 239–243. [Google Scholar] [PubMed]
- Ohashi, T.; Yamaki, M.; Pandav, C.S.; Karmarkar, M.G.; Irie, M. Simple microplate method for determination of urinary iodine. Clin. Chem. 2000, 46, 529–536. [Google Scholar] [PubMed]
- Makhmudov, A.A.; Caldwell, K.L. The Challenge of Iodine Deficiency Disorder. A Decade of CDC’s Ensuring the Quality of Urinary Iodine Procedures Program. Available online: https://www.cdc.gov/labstandards/pdf/equip/EQUIP_Booklet.pdf (accessed on 20 February 2017).
- PT Clarity Research, Indonesia; National Agency of Drugs and Food Control, Indonesia; Ministry of Health, Indonesia. Usage of Iodized Salt in Processed Food in Indonesia; Global Alliance for Improved Nutrition: Jakarta, Indonesia, 2014.
- Lumley, T. Analysis of complex survey samples. J. Stat. Softw. 2004, 9, 1–19. [Google Scholar] [CrossRef]
- AIIMS; GAIN; UNICEF India; IGN South Asia; Nielsen (India) Pvt. Ltd.; NCSOII, India. National Iodine and Sodium Intake Report_India 2015; AIIMS: New Delhi, India, 2015. [Google Scholar]
- Enquete Nationale Sur L’utilisation du Sel Adequatement Iode et des Bouillons Par Les Menages et Sur le Statut en Iode des Femmes Enceintes et en Age de Procreer. Available online: http://www.gainhealth.org/wp-content/uploads/2018/03/Final-Report-on-TDCI-in-Senegal-August-2016-FR.pdf (accessed on 20 March 2018). (In French).
- Pandav, C.; Yadav, K.; Lakshmy, R.; Pandey, R.; Salve, H.; Chakrabarty, A.; Knowles, J.; Sankar, R.; Rah, J.; Ansari, M. Across India, women are iodine sufficient. IDD Newsl. 2015, 43, 1–4. [Google Scholar]
- Zimmermann, M.B.; Andersson, M. Assessment of iodine nutrition in populations: Past, present, and future. Nutr. Rev. 2012, 70, 553–570. [Google Scholar] [CrossRef] [PubMed]
- Shamim, A.A.; Christian, P.; Schulze, K.J.; Ali, H.; Kabir, A.; Rashid, M.; Labrique, A.; Salamatullah, Q.; West, K.P., Jr. Iodine status in pregnancy and household salt iodine content in rural Bangladesh: Iodine status of pregnant women in rural Bangladesh. Matern. Child. Nutr. 2012, 8, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.D.; Hetzel, B.; Fisher, J. Access to iodized salt in 11 low- and lower-middle-income countries: 2000 and 2010. Bull. World Health Organ. 2016, 94, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Globally, 86 Per Cent of the Population Has Access to Iodized Salt. Available online: http://data.unicef.org/nutrition/iodine.html (accessed on 27 May 2016).
- Reardon, T.; Minten, B. The Quiet Revolution in India’s Food Supply Chains. IFPRI Discussion Paper; International Food Policy Research Institute: New Delhi, India, 2011. [Google Scholar]
- Drescher, L.S.; Thiele, S.; Mensink, G.B. A new index to measure healthy food diversity better reflects a healthy diet than traditional measures. J. Nutr. 2007, 137, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P. Water Quality Fact Sheet: Iodine; British Geological Survey: Nottingham, UK, 2000. [Google Scholar]
- Groundwater Quality: Ghana. Available online: https://www.bgs.ac.uk/downloads/directDownload.cfm?id=1281&noexcl=true&t=Country%20Sheets%3A%20Ghana (accessed on 16 August 2017).
- Rah, J.; Anas, A.; Chakrabarty, A.; Sankar, R.; Pandav, C.; Aguayo, V. IDD Control in India: Triumphs and Challenges. 2013. Available online: www.iccidd.org/newsletter/idd_aug13_india.pdf (accessed on 13 March 2018).
- Johnson, C.; Mohan, S.; Rogers, K.; Shivashankar, R.; Thout, S.R.; Gupta, P.; He, F.J.; MacGregor, G.A.; Webster, J.; Krishnan, A.; et al. Mean dietary salt intake in urban and rural areas in India: A population survey of 1395 persons. J. Am. Heart Assoc. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; He, F.J.; MacGregor, G.A. High salt intake: Independent risk factor for obesity? Hypertension 2015, 66, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Abizari, A.-R.; Dold, S.; Kupka, R.; Zimmermann, M.B. More than two-thirds of dietary iodine in children in northern Ghana is obtained from bouillon cubes containing iodized salt. Public Health Nutr. 2017, 20, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Spohrer, R.; Knowles, J.; Jallier, V.; Ndiaye, B.; Indorf, C.; Guinot, P.; Kupka, R. Estimation of population iodine intake from iodized salt consumed through bouillon seasoning in Senegal: Bouillon contribution to iodine intake in Senegal. Ann. N. Y. Acad. Sci. 2015, 1357, 43–52. [Google Scholar] [CrossRef] [PubMed]
- König, F.; Andersson, M.; Hotz, K.; Aeberli, I.; Zimmermann, M.B. Ten repeat collections for urinary iodine from spot samples or 24-hour samples are needed to reliably estimate individual iodine status in women. J. Nutr. 2011, 141, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
| Country | Year | Target Sample Size—HHs (HHs/PSU) | Sample Design | Response Rate (%) | Respondent Characteristics (%) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Target WRA for Urine Samples | Stratification | Sampling Scheme | Completed Interviews | Urinary Iodine Result 1 | Female | WRA (% 15–17 yo) | |||
| India | 2014–2015 | 6048 (12) | All WRA present in every second HH | 12 strata: Urban/Rural by 6 zones: North, North-East, East, West, Central, South | Cross-sectional cluster, PPS within strata | 94.5 | 86.5 | 91.3 | 82.2 (0.7) |
| Ghana | 2015 | 2112 (16) | All WRA present in every HH | 4 strata: North, Mid, South-non-producing, South-salt-producing | Cross-sectional cluster, PPS within strata | 91.3 | 79.9 | 83.8 | 61.4 (0.0) |
| Senegal | 2014 | 1968 (16) | One WRA where present in every HH | 3 strata: Urban, Rural-non-salt producing, Rural-salt-producing | Cross-sectional cluster, PPS within strata | 98.8 | 85.4 | 99.0 | 90.2 (1.4) |
| Variable | Level | Number Samples | Median | IQR | Single Variable Model—UIC µg/L | Multiple Variable Model—UIC µg/L | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | p-Value | Relative to Reference | 95% CI | p-Value (Variable Effect) | |||||
| Strata | South—urban | 201 | 147 | 72, 244 | 183 | 158, 208 | <0.001 | <0.001 | ||
| South—rural | 202 | 162 | 78, 250 | 186 | 163, 209 | 1.1 | 0.9, 1.5 | |||
| West—urban | 188 | 177 | 96, 282 | 208 | 181, 235 | 1.2 | 0.9, 1.5 | |||
| West—rural | 204 | 147 | 81, 265 | 183 | 161, 206 | 1.1 | 0.9, 1.4 | |||
| Central—urban | 189 | 141 | 81, 226 | 181 | 155, 207 | 1.0 | 0.8, 1.2 | |||
| Central—rural | 190 | 115 | 605, 243 | 168 | 139, 197 | 0.9 | 0.7, 1.2 | |||
| North—urban | 157 | 224 a | 162, 309 | 246 | 223, 268 | 1.5 a | 1.2, 1.9 | |||
| North—rural | 176 | 169 | 103, 316 | 207 | 179, 236 | 1.3 | 1.0, 1.6 | |||
| East—urban | 204 | 162 | 90, 304 | 212 | 179, 244 | 1.1 | 0.9, 1.4 | |||
| East—rural | 192 | 155 | 67, 273 | 194 | 162, 225 | 1.1 | 0.8, 1.4 | |||
| North East—urban | 264 | 155 | 84, 246 | 192 | 164, 221 | 1.0 | 0.8, 1.3 | |||
| North East—rural | 253 | 136 | 69, 197 | 162 | 131, 193 | 0.9 | 0.7, 1.1 | |||
| Residence type | Rural | 1217 | 149 | 73, 255 | 182 | 171, 194 | 0.002 | Not included in multiple variable analysis | ||
| Urban | 1203 | 168 | 92, 278 | 202 | 190, 213 | |||||
| MPI education | High (deprived) | 551 | 155 | 82, 238 | 179 | 166, 192 | 0.162 | 0.354 | ||
| Low | 1869 | 160 | 82, 275 | 196 | 186, 205 | 1.1 | 1.0, 1.2 | |||
| MPI health | High (deprived) | 672 | 160 | 79, 253 | 187 | 173, 201 | 0.223 | 0.683 | ||
| Low | 1748 | 158 | 82, 268 | 194 | 184, 203 | 1.0 | 0.9, 1.1 | |||
| MPI living standards | High (deprived) | 1449 | 149 | 73, 246 | 181 | 171, 191 | <0.001 | 0.349 | ||
| Low | 968 | 177 | 98, 289 | 208 | 196, 219 | 1.1 | 1.0, 1.2 | |||
| Heard of iodine deficiency | No | 1046 | 149 | 70, 255 | 180 | 170, 191 | <0.001 | <0.001 | ||
| Yes | 1374 | 165 | 89, 274 | 200 | 189, 211 | 1.2 | 1.1, 1.3 | |||
| Dietary diversity | Not diverse | 1594 | 151 | 76, 256 | 185 | 176, 193 | 0.001 | 0.015 | ||
| Diverse | 826 | 175 | 98, 281 | 206 | 191, 220 | 1.1 | 1.0, 1.2 | |||
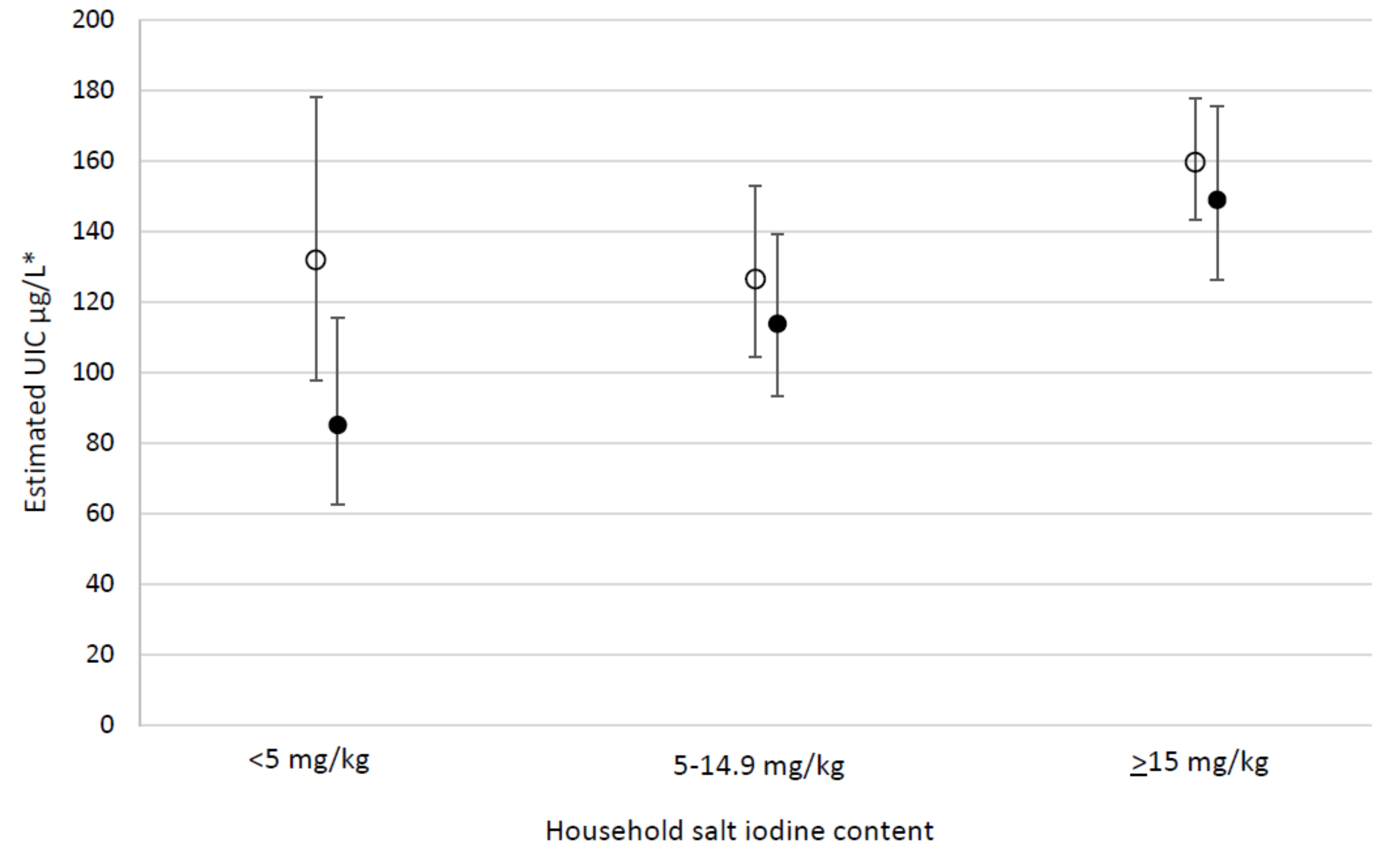
| Household salt iodine content (mg/kg) | <5 | 193 | 112 | 45, 185 | 144 | 123, 166 | <0.001 | <0.001 | ||
| 5–14.9 | 342 | 123 a | 61, 211 | 160 | 144, 176 | 1.3 a | 1.1, 1.6 | |||
| ≥15 | 1876 | 168 a | 91, 279 | 202 | 193, 211 | 1.6 a | 1.3, 2.0 | |||
| Age | <25 | 724 | 165 | 84, 283 | 201 | 186, 216 | 0.662 | 0.239 | ||
| 25–29 | 457 | 152 | 81, 250 | 189 | 174, 204 | 0.9 | 0.8, 1.0 | |||
| 30–34 | 406 | 162 | 88, 248 | 188 | 173, 202 | 1.0 | 0.9, 1.1 | |||
| 35–49 | 833 | 155 | 77, 253 | 187 | 176, 198 | 0.9 | 0.8, 1.0 | |||
| BMI | <18.5 | 443 | 145 | 66, 251 | 180 | 165, 195 | 0.036 | 0.145 | ||
| 18.5–25.0 | 1363 | 161 a | 85, 265 | 193 | 184, 203 | 1.1 a | 1.0, 1.2 | |||
| >25.0 | 584 | 162 a | 89, 277 | 199 | 184, 213 | 1.1 | 1.0, 1.3 | |||
| Variable | Level | Number Samples | Median | IQR | Single Variable Model—UIC µg/L | Multiple Variable Model—UIC µg/L | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | p-Value | Relative to Reference | 95% CI | p-Value (Variable Effect) | |||||
| Strata | South-salt-producing | 338 | 217 | 125, 352 | 265 | 228, 302 | <0.001 | <0.001 | ||
| North | 378 | 168 a | 87, 291 | 228 | 185, 271 | 0.8 | 0.6, 1.0 | |||
| Mid | 298 | 174 | 115, 293 | 20 | 191, 248 | 0.8 | 0.7, 1.0 | |||
| South-non-salt-producing | 258 | 316 a | 187, 467 | 363 | 322, 403 | 1.2 | 1.0, 1.5 | |||
| Residence type | Rural | 492 | 169 | 92, 291 | 224 | 188, 260 | 0.008 | 0.099 | ||
| Urban | 780 | 221 | 138, 361 | 278 | 254, 302 | 1.2 | 1.0, 1.4 | |||
| MPI education | High (deprived) | 572 | 181 | 93, 331 | 239 | 211, 268 | 0.002 | 0.376 | ||
| Low | 700 | 217 | 136, 346 | 270 | 250, 290 | 1.1 | 0.9, 1.2 | |||
| MPI health | High (deprived) | 765 | 185 | 107, 318 | 246 | 219, 272 | 0.008 | 0.222 | ||
| Low | 502 | 216 | 138, 360 | 271 | 251, 291 | 1.1 | 1.0, 1.2 | |||
| MPI living standards | High (deprived) | 1145 | 195 | 115, 333 | 253 | 232, 274 | 0.002 | 0.196 | ||
| Low | 125 | 246 | 151, 386 | 293 | 261, 326 | 1.1 | 1.0, 1.3 | |||
| Heard of iodine deficiency | No | 814 | 191 | 112, 318 | 250 | 225, 275 | 0.054 | 0.779 | ||
| Yes | 458 | 216 | 134, 353 | 269 | 244, 293 | 1.0 | 0.9, 1.1 | |||
| Dietary diversity | Not diverse | 473 | 195 | 126, 343 | 260 | 235, 285 | 0.848 | 0.995 | ||
| Diverse | 799 | 204 | 119, 337 | 256 | 232, 281 | 1.0 | 0. 9, 1.1 | |||
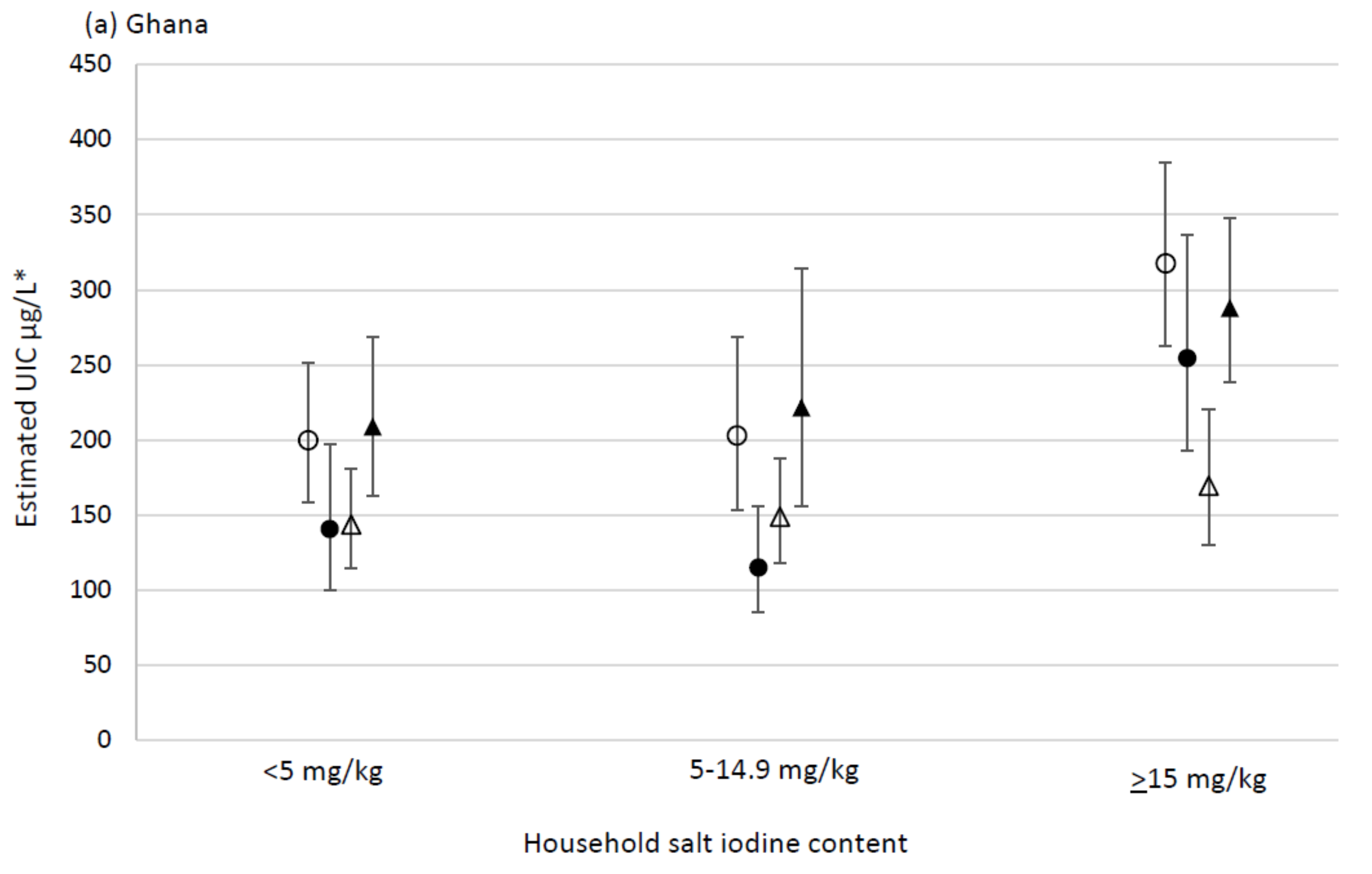
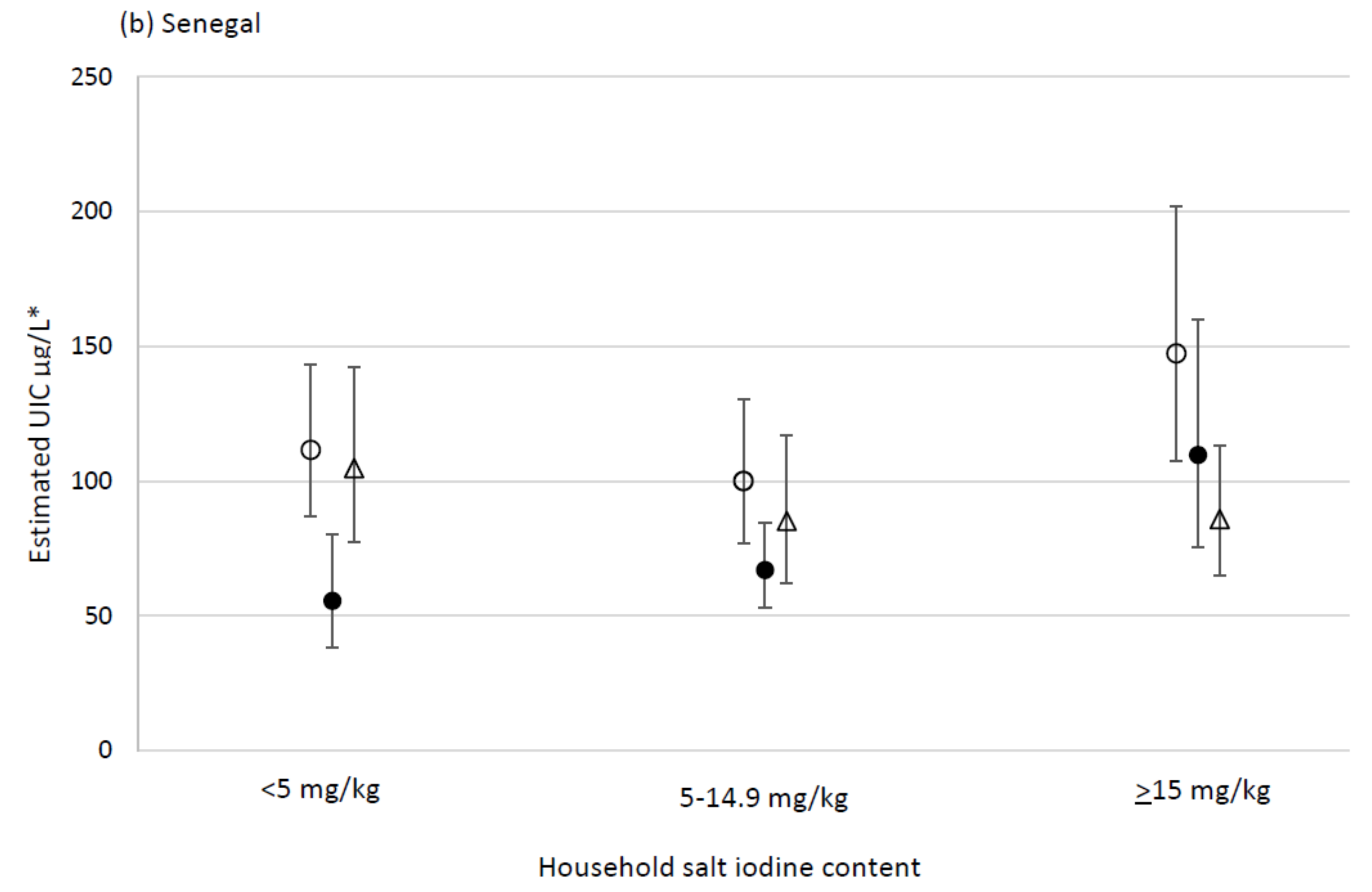
| Household salt iodine content (mg/kg) | <5 | 423 | 180 | 110, 301 | 234 | 208, 260 | <0.001 | <0.001 | ||
| 5–14.9 | 384 | 173 | 105, 312 | 226 | 196, 256 | 1.0 | 0.9, 1.1 | |||
| ≥15 | 301 | 291 a | 173, 447 | 334 | 304, 364 | 1.4 a | 1.2, 1.6 | |||
| Age | <25 | 443 | 202 | 130, 342 | 263 | 234, 292 | 0.705 | 0.073 | ||
| 25–29 | 229 | 186 | 111, 329 | 250 | 222, 279 | 0.9 a | 0.8, 1.0 | |||
| 30–34 | 207 | 188 | 112, 339 | 254 | 220, 289 | 0.8 a | 0.7, 1.0 | |||
| 35–49 | 393 | 216 | 116, 343 | 258 | 231, 285 | 0.9 a | 0. 8, 1.0 | |||
| Tomato paste intake (g in past week) | 0 | 238 | 163 | 84, 261 | 206 | 176, 235 | <0.001 | 0.029 | ||
| <200 | 418 | 211 a | 130, 367 | 281 | 252, 310 | 1.1 | 1.0, 1.3 | |||
| ≥200 | 531 | 197 a | 124, 342 | 258 | 235, 282 | 1.2 a | 1.1, 1.4 | |||
| Bouillon intake (g in past week) | 0 | 179 | 200 | 122, 328 | 244 | 215,273 | 0.003 | 0.023 | ||
| <20 | 691 | 216 | 131, 361 | 277 | 253, 300 | 1.2 a | 1.1, 1.3 | |||
| ≥20 | 366 | 168 | 97, 286 | 221 | 191, 251 | 1.0 | 0.8, 1.2 | |||
| Instant noodle intake | No | 1055 | 194 | 120, 333 | 251 | 232, 271 | 0.064 | 0.436 | ||
| Yes | 217 | 225 | 117, 400 | 289 | 249, 329 | 1.1 | 0.9, 1.2 | |||
| Variable | Level | Number Samples | Median | IQR | Single Variable Model—UIC µg/L | Multiple Variable Model—UIC µg/L | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 95% CI | p-Value | Relative to Reference | 95% CI | p-Value (Variable Effect) | |||||
| Strata | Rural-salt-producing | 462 | 109 | 48, 168 | 129 | 112, 146 | 0.174 | 0.210 | ||
| Rural-non-salt-producing | 437 | 83 | 42, 147 | 115 | 95, 136 | 0.8 | 0.7, 1.1 | |||
| Urban | 409 | 112 | 50, 189 | 140 | 125, 155 | 1.1 | 0.8, 1.4 | |||
| Residence type | Rural | 899 | 83 | 42, 147 | 115 | 95, 136 | 0.074 | Not included in multiple variable analysis | ||
| Urban | 409 | 112 | 50, 189 | 140 | 125, 155 | |||||
| MPI education | High (deprived) | 936 | 94 | 41, 162 | 124 | 110, 138 | 0.133 | 0.465 | ||
| Low | 372 | 111 | 56, 183 | 138 | 122, 155 | 1 | 0.9, 1.4 | |||
| MPI health | High (deprived) | 865 | 90 | 42, 164 | 125 | 110, 141 | 0.626 | 0.701 | ||
| Low | 435 | 113 | 51, 176 | 135 | 119, 150 | 1.0 | 0.8, 1.2 | |||
| MPI living standards | High (deprived) | 975 | 98 | 43, 164 | 127 | 111, 142 | 0.470 | 0.485 | ||
| Low | 323 | 108 | 50, 178 | 133 | 114, 152 | 0.9 | 0.6, 1.2 | |||
| Heard of iodine deficiency | No | 698 | 90 | 42, 162 | 125 | 109, 142 | 0.067 | 0.079 | ||
| Yes | 610 | 113 | 52, 177 | 134 | 122, 147 | 1.2 | 1.0, 1.5 | |||
| Dietary diversity | Not diverse | 440 | 97 | 43, 165 | 126 | 107, 146 | 0.770 | 0.329 | ||
| Diverse | 868 | 103 | 46, 172 | 130 | 117, 143 | 0.9 | 0.8, 1.1 | |||
| Household salt iodine content (mg/kg) | <5 | 363 | 84 | 36, 141 | 107 | 90, 125 | 0.043 | 0.292 | ||
| 5–14.9 | 502 | 85 | 44, 149 | 118 | 102, 134 | 1.1 | 0.8, 1.4 | |||
| ≥15 | 313 | 123 a | 56, 201 | 149 | 131, 168 | 1.3 | 0.9, 1.7 | |||
| Age | <25 | 471 | 107 | 44, 170 | 134 | 117, 151 | 0.542 | 0.360 | ||
| 25–29 | 290 | 91 | 40, 170 | 128 | 108, 149 | 0.9 | 0.7, 1.2 | |||
| 30–34 | 205 | 95 | 44, 162 | 117 | 100, 134 | 0.8 | 0.6, 1.1 | |||
| 35–49 | 342 | 104 | 53, 171 | 129 | 112, 147 | 1.0 | 0.8, 1.3 | |||
| Bouillon intake (g in past week) | 0 | 42 | 85 | 40, 114 | 91 | 70, 112 | 0.454 | 0.761 | ||
| <20 | 205 | 114 | 51, 167 | 124 | 106, 143 | 1.1 | 0.7, 1.8 | |||
| ≥20 | 1059 | 99 | 44, 171 | 131 | 117, 145 | 1.2 | 0.8, 1.7 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knowles, J.; Kupka, R.; Dumble, S.; Garrett, G.S.; Pandav, C.S.; Yadav, K.; Touré, N.K.; Foriwa Amoaful, E.; Gorstein, J. Regression Analysis to Identify Factors Associated with Urinary Iodine Concentration at the Sub-National Level in India, Ghana, and Senegal. Nutrients 2018, 10, 516. https://doi.org/10.3390/nu10040516
Knowles J, Kupka R, Dumble S, Garrett GS, Pandav CS, Yadav K, Touré NK, Foriwa Amoaful E, Gorstein J. Regression Analysis to Identify Factors Associated with Urinary Iodine Concentration at the Sub-National Level in India, Ghana, and Senegal. Nutrients. 2018; 10(4):516. https://doi.org/10.3390/nu10040516
Chicago/Turabian StyleKnowles, Jacky, Roland Kupka, Sam Dumble, Greg S. Garrett, Chandrakant S. Pandav, Kapil Yadav, Ndeye Khady Touré, Esi Foriwa Amoaful, and Jonathan Gorstein. 2018. "Regression Analysis to Identify Factors Associated with Urinary Iodine Concentration at the Sub-National Level in India, Ghana, and Senegal" Nutrients 10, no. 4: 516. https://doi.org/10.3390/nu10040516
APA StyleKnowles, J., Kupka, R., Dumble, S., Garrett, G. S., Pandav, C. S., Yadav, K., Touré, N. K., Foriwa Amoaful, E., & Gorstein, J. (2018). Regression Analysis to Identify Factors Associated with Urinary Iodine Concentration at the Sub-National Level in India, Ghana, and Senegal. Nutrients, 10(4), 516. https://doi.org/10.3390/nu10040516





