The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Dietary Assessment
2.3. The Mediterranean Diet Score
2.4. Principal Component Analysis
2.5. Statistical Analyses
3. Results
3.1. Study Population
3.2. Dietary Assessment
3.3. Dietary Patterns and HrHPV Infection
3.4. Dietary Patterns and Cervical Cancer
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bosch, F.X.; Burchell, A.N.; Schiffman, M.; Giuliano, A.R.; de Sanjose, S.; Bruni, L.; Tortolero-Luna, G.; Kjaer, S.K.; Muñoz, N. Epidemiology and natural history of human papillomavirus infections and type-specific implications in cervical neoplasia. Vaccine 2008, 26 (Suppl. 10), K1–K16. [Google Scholar] [CrossRef] [PubMed]
- Serrano, B.; Brotons, M.; Bosch, F.X.; Bruni, L. Epidemiology and burden of HPV-related disease. Best. Pract. Res. Clin. Obstet. Gynaecol. 2018, 47, 14–26. [Google Scholar] [CrossRef] [PubMed]
- zur Hausen, H. Papillomaviruses and cancer: From basic studies to clinical application. Nat. Rev. Cancer 2002, 2, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Chih, H.J.; Lee, A.H.; Colville, L.; Binns, C.W.; Xu, D. A review of dietary prevention of human papillomavirus-related infection of the cervix and cervical intraepithelial neoplasia. Nutr. Cancer 2013, 65, 317–328. [Google Scholar] [CrossRef] [PubMed]
- González, C.A.; Travier, N.; Luján-Barroso, L.; Castellsagué, X.; Bosch, F.X.; Roura, E.; Bueno-de-Mesquita, H.B.; Palli, D.; Boeing, H.; Pala, V.; et al. Dietary factors and in situ and invasive cervical cancer risk in the European prospective investigation into cancer and nutrition study. Int. J. Cancer 2011, 129, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Dai, B.; Zhang, B.; Wang, Z. Vitamin a and risk of cervical cancer: A meta-analysis. Gynecol. Oncol. 2012, 124, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Meng, Y. Association between serum folate level and cervical cancer: A meta-analysis. Arch. Gynecol. Obstet. 2016, 293, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Shen, K.; Li, Z.; Xu, Y.; Wu, D. Association between vitamin C intake and the risk of cervical neoplasia: A meta-analysis. Nutr. Cancer 2016, 68, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Lee, J.K.; Kim, T.J.; Kim, M.K. The association between fruit and vegetable consumption and HPV viral load in high-risk HPV-positive women with cervical intraepithelial neoplasia. Cancer Causes. Control. 2010, 21, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Siegel, E.M.; Salemi, J.L.; Villa, L.L.; Ferenczy, A.; Franco, E.L.; Giuliano, A.R. Dietary consumption of antioxidant nutrients and risk of incident cervical intraepithelial neoplasia. Gynecol. Oncol. 2010, 118, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Zhu, H.; Lin, C.; Che, J.; Tian, X.; Han, S.; Zhao, H.; Zhu, Y.; Mao, D. Associations between antioxidant vitamins and the risk of invasive cervical cancer in chinese women: A case-control study. Sci. Rep. 2015, 5, 13607. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, M.K.; Lee, J.K.; Kim, J.H.; Son, S.K.; Song, E.S.; Lee, K.B.; Lee, J.P.; Lee, J.M.; Yun, Y.M. Intakes of vitamin A, C, and E, and beta-carotene are associated with risk of cervical cancer: A case-control study in Korea. Nutr. Cancer 2010, 62, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Tomita, L.Y.; Longatto Filho, A.; Costa, M.C.; Andreoli, M.A.; Villa, L.L.; Franco, E.L.; Cardoso, M.A. Diet and serum micronutrients in relation to cervical neoplasia and cancer among low-income Brazilian women. Int. J. Cancer 2010, 126, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Piyathilake, C.J.; Henao, O.L.; Macaluso, M.; Cornwell, P.E.; Meleth, S.; Heimburger, D.C.; Partridge, E.E. Folate is associated with the natural history of high-risk human papillomaviruses. Cancer Res. 2004, 64, 8788–8793. [Google Scholar] [CrossRef] [PubMed]
- García-Closas, R.; Castellsagué, X.; Bosch, X.; González, C.A. The role of diet and nutrition in cervical carcinogenesis: A review of recent evidence. Int. J. Cancer 2005, 117, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Reddy, L.; Odhav, B.; Bhoola, K.D. Natural products for cancer prevention: A global perspective. Pharmacol. Ther. 2003, 99, 1–13. [Google Scholar] [CrossRef]
- Field, C.J.; Johnson, I.R.; Schley, P.D. Nutrients and their role in host resistance to infection. J. Leukoc. Biol. 2002, 71, 16–32. [Google Scholar] [PubMed]
- Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Canto, C.; La Rosa, N.; Cantarella, M.A.; Spampinato, G.; Scalisi, A.; Agodi, A. Line-1 hypermethylation in white blood cell DNA is associated with high-grade cervical intraepithelial neoplasia. BMC Cancer 2017, 17, 601. [Google Scholar] [CrossRef] [PubMed]
- Eveleth, P.B. Physical status: The use and interpretation of anthropometry. Report of a WHO expert committee. World Health Organ. Tech. Rep. Ser. 1995, 854, 1–452. [Google Scholar] [CrossRef]
- Agodi, A.; Barchitta, M.; Valenti, G.; Marzagalli, R.; Frontini, V.; Marchese, A.E. Increase in the prevalence of the MTHFR 677 TT polymorphism in women born since 1959: Potential implications for folate requirements. Eur. J. Clin. Nutr. 2011, 65, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Stampfer, M.J. Total energy intake: Implications for epidemiologic analyses. Am. J. Epidemiol. 1986, 124, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef] [PubMed]
- Couto, E.; Boffetta, P.; Lagiou, P.; Ferrari, P.; Buckland, G.; Overvad, K.; Dahm, C.C.; Tjønneland, A.; Olsen, A.; Clavel-Chapelon, F.; et al. Mediterranean dietary pattern and cancer risk in the epic cohort. Br. J. Cancer 2011, 104, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Quattrocchi, A.; Adornetto, V.; Marchese, A.E.; Agodi, A. Tumor necrosis factor-alpha -308 g>a polymorphism, adherence to mediterranean diet, and risk of overweight/obesity in young women. Biomed. Res. Int. 2014, 2014, 742620. [Google Scholar] [CrossRef] [PubMed]
- Si, C.J.; Shu, L.; Zheng, P.F.; Zhang, X.Y.; Yu, X.L.; Gao, W.; Zhang, L. Dietary patterns and endometrial cancer: A meta-analysis. Eur. J. Cancer Prev. 2017, 26, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.F.; Cantwell, M.M.; Cardwell, C.R.; Velentzis, L.S.; Woodside, J.V. Dietary patterns and breast cancer risk: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 91, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.Y.; Pisu, M.; Oster, R.A.; Urmie, J.M.; Schrag, D.; Huskamp, H.A.; Lee, J.; Kiefe, C.I.; Fouad, M.N. Racial variation in willingness to trade financial resources for life-prolonging cancer treatment. Cancer 2011, 117, 3476–3484. [Google Scholar] [CrossRef] [PubMed]
- Koutoukidis, D.A.; Beeken, R.J.; Manchanda, R.; Burnell, M.; Knobf, M.T.; Lanceley, A. Erratum to: Diet and exercise in uterine cancer survivors (DEUS pilot)—Piloting a healthy eating and physical activity program: Study protocol for a randomized controlled trial. Trials 2017, 18, 28. [Google Scholar] [CrossRef] [PubMed]
- Koutoukidis, D.A.; Beeken, R.J.; Manchanda, R.; Michalopoulou, M.; Burnell, M.; Knobf, M.T.; Lanceley, A. Recruitment, adherence, and retention of endometrial cancer survivors in a behavioural lifestyle programme: The diet and exercise in uterine cancer survivors (DEUS) parallel randomised pilot trial. BMJ Open 2017, 7, e018015. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public. Health. Nutr. 2014, 17, 2769–2782. [Google Scholar] [CrossRef] [PubMed]
- Giacosa, A.; Barale, R.; Bavaresco, L.; Gatenby, P.; Gerbi, V.; Janssens, J.; Johnston, B.; Kas, K.; La Vecchia, C.; Mainguet, P.; et al. Cancer prevention in Europe: The mediterranean diet as a protective choice. Eur. J. Cancer Prev. 2013, 22, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Piyathilake, C.J.; Badiga, S.; Kabagambe, E.K.; Azuero, A.; Alvarez, R.D.; Johanning, G.L.; Partridge, E.E. A dietary pattern associated with line-1 methylation alters the risk of developing cervical intraepithelial neoplasia. Cancer Prev. Res. 2012, 5, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Moga, M.A.; Dimienescu, O.G.; Arvatescu, C.A.; Mironescu, A.; Dracea, L.; Ples, L. The role of natural polyphenols in the prevention and treatment of cervical cancer—An overview. Molecules 2016, 21, 1055. [Google Scholar] [CrossRef] [PubMed]
- Hardy, T.M.; Tollefsbol, T.O. Epigenetic diet: Impact on the epigenome and cancer. Epigenomics 2011, 3, 503–518. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Maugeri, A.; Quattrocchi, A.; Agrifoglio, O.; Agodi, A. The role of miRNAs as biomarkers for pregnancy outcomes: A comprehensive review. Int. J. Genom. 2017, 2017, 8067972. [Google Scholar] [CrossRef] [PubMed]
- Agodi, A.; Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Canto, C.; Marchese, A.E.; Vinciguerra, M. Low fruit consumption and folate deficiency are associated with line-1 hypomethylation in women of a cancer-free population. Genes Nutr. 2015, 10, 480. [Google Scholar] [CrossRef] [PubMed]
- Ong, T.P.; Moreno, F.S.; Ross, S.A. Targeting the epigenome with bioactive food components for cancer prevention. J. Nutrigenet. Nutrigenom. 2011, 4, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Vinciguerra, M.; Agodi, A. Line-1 hypomethylation in blood and tissue samples as an epigenetic marker for cancer risk: A systematic review and meta-analysis. PLoS ONE 2014, 9, e109478. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Human; World Health Organization: Lyon, France, 2007. [Google Scholar]
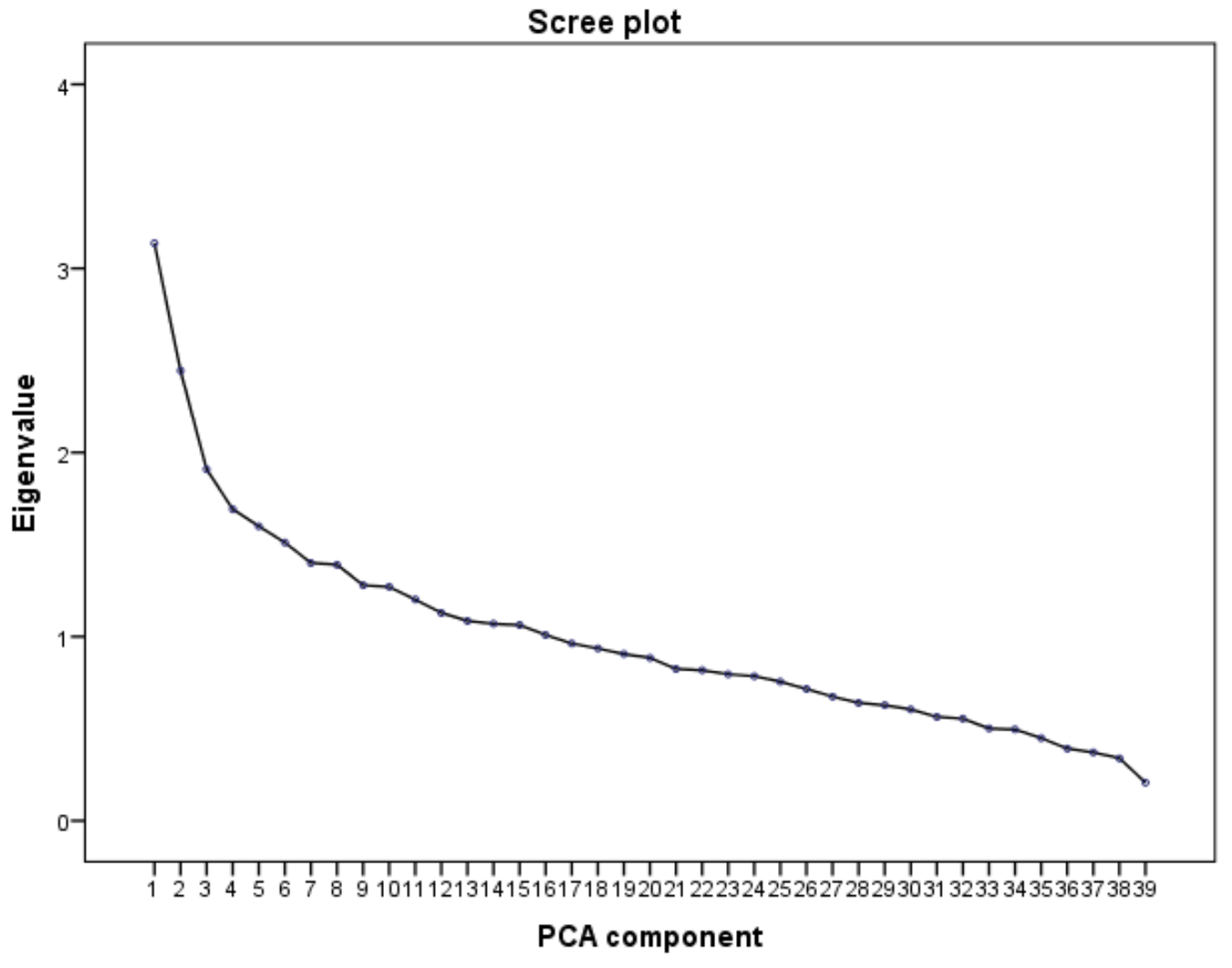
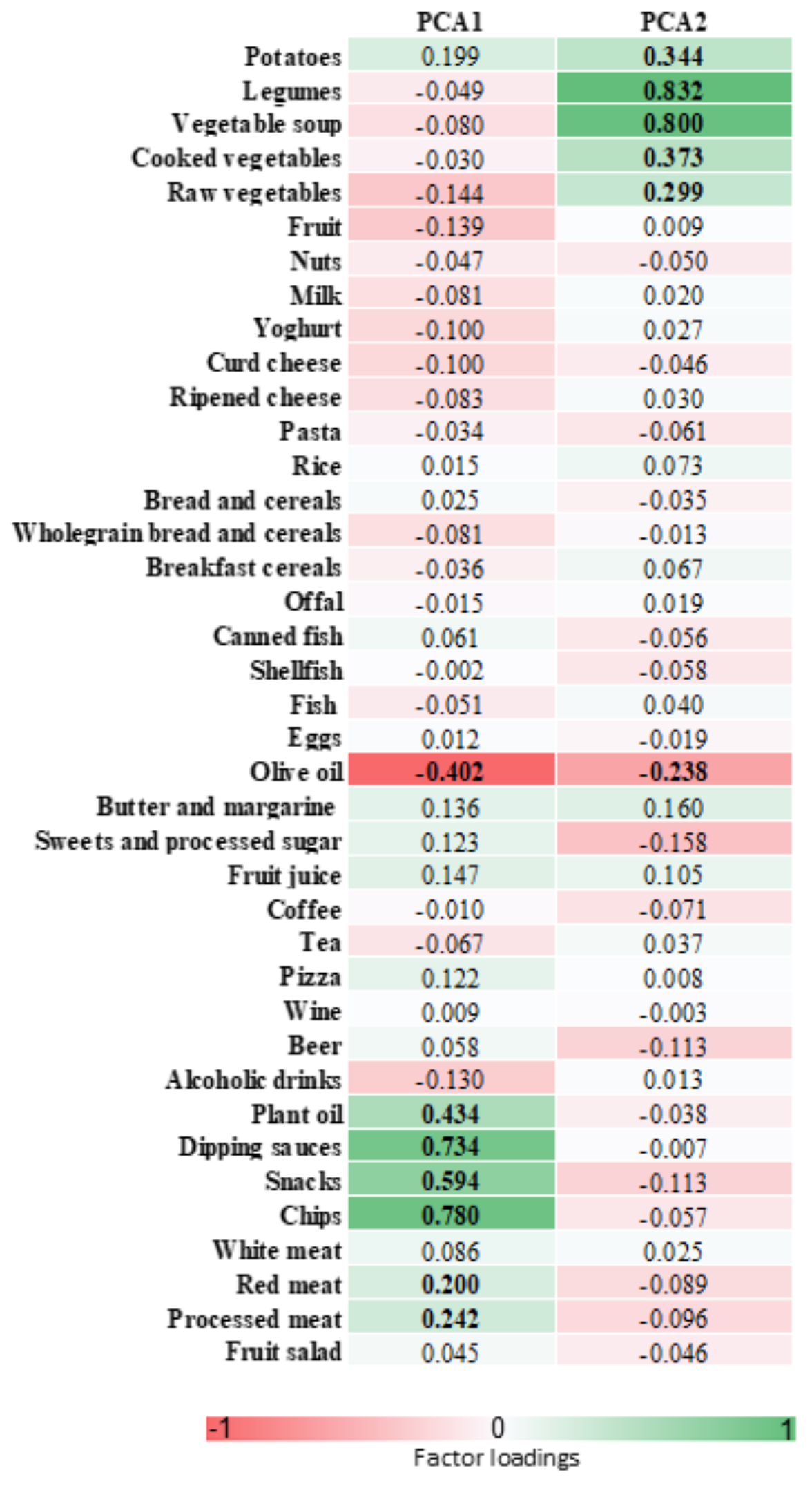
| Characteristics | hrHPV+ (n = 84) | hrHPV– (n = 167) | p-Value a |
|---|---|---|---|
| Age, mean (SD) | 38.63 (10.53) | 43.65 (9.62) | <0.001 |
| Current smokers | 47.0% | 28.1% | 0.003 |
| BMI, mean (SD) | 22.36 (4.01) | 24.34 (4.68) | 0.001 |
| Nutritional status | |||
| Underweight | 8.4% | 5.4% | 0.004 |
| Normal weight | 75.9% | 56.6% | |
| Overweight | 10.8% | 24.1% | |
| Obese | 4.8% | 13.9% | |
| Workers | 51.2% | 41.3% | 0.138 |
| Parity (≥1 live births) | 58.3% | 83.2% | <0.001 |
| Low education level | 31.0% | 41.9% | 0.092 |
| Use of oral contraceptive | 13.1% | 7.8% | 0.177 |
| Characteristics | Cases (n = 127) | Controls (n = 411) | p-Value a |
|---|---|---|---|
| Age, mean (SD) | 36.01 (8.10) | 41.50 (10.21) | <0.001 |
| Current smokers | 53.5% | 36.8% | 0.001 |
| BMI, mean (SD) | 22.47 (3.63) | 23.59 (4.53) | 0.012 |
| Nutritional status | |||
| Underweight | 11.0% | 7.1% | 0.052 |
| Normal weight | 67.5% | 63.1% | |
| Overweight | 18.3% | 19.8% | |
| Obese | 3.2% | 10.0% | |
| Workers | 46.5% | 46.5% | 0.998 |
| Parity (≥1 live births) | 59.8% | 71.8% | 0.011 |
| Low education level | 41.0% | 38.2% | 0.579 |
| Use of oral contraceptive | 14.2% | 8.0% | 0.039 |
| Characteristics | Western Dietary Pattern | Prudent Dietary Pattern | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Q1 (n = 134) | Q2 (n = 135) | Q3 (n = 135) | Q4 (n = 134) | p-value a | Q1 (n = 134) | Q2 (n = 135) | Q3 (n = 135) | Q4 (n = 134) | p-Value a | |
| Age, mean (SD) | 42.37 (10.87) | 43.35 (9.48) | 39.79 (9.25) | 35.28 (8.46) | <0.001 | 40.55 (10.74) | 40.68 (9.90) | 40.52 (9.86) | 39.05 (9.58) | 0.500 |
| Current smokers | 38.1% | 34.1% | 37.3% | 53.7% | 0.005 | 41.0% | 46.3% | 34.8% | 41.0% | 0.300 |
| BMI, mean (SD) | 23.39 (4.23) | 23.69 (4.19) | 23.76 (4.77) | 22.45 (4.13) | 0.053 | 23.18 (4.38) | 23.14 (4.30) | 23.44 (4.30) | 23.53 (4.48) | 0.846 |
| Workers | 36.6% | 50.4% | 47.4% | 54.1% | 0.631 | 44.8% | 45.2% | 51.1% | 44.8% | 0.667 |
| Parity (≥1 live births) | 71.5% | 77.8% | 66.7% | 59.7% | 0.011 | 63.4% | 68.9% | 70.4% | 73.1% | 0.372 |
| Low Education level | 43.3% | 34.8% | 34.1% | 43.3% | 0.218 | 40.3% | 35.6% | 41.5% | 38.1% | 0.762 |
| Use of oral contraceptive | 9.0% | 8.9% | 11.1% | 9.0% | 0.906 | 11.9% | 7.4% | 9.6% | 9.0% | 0.644 |
| Dietary Pattern | Regression Model | adjOR (95%CI) | ||||||
|---|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | p-Trend | Continuous | p-Value | ||
| Western | Model 1 a | 1.00 (ref) | 1.11 (0.48–2.57) | 1.77 (1.04–3.54) | 1.97 (1.14–4.18) | 0.039 | 1.44 (1.03–2.03) | 0.036 |
| Model 2 b | 1.00 (ref) | 1.33 (0.54–3.28) | 1.96 (0.88–4.34) | 2.06 (0.86–4.90) | 0.047 | 1.39 (0.97–1.99) | 0.069 | |
| Prudent | Model 1 a | 1.00 (ref) | 1.18 (0.54–2.60) | 0.86 (0.58–1.28) | 0.85 (0.65–1.11) | 0.842 | 0.83 (0.63–1.11) | 0.215 |
| Model 2 b | 1.00 (ref) | 1.05 (0.45–2.43) | 0.86 (0.57–1.32) | 0.82 (0.62–1.10) | 0.226 | 0.83 (0.62–1.11) | 0.210 | |
| Regression Model | adjOR (95%CI) | |||||
|---|---|---|---|---|---|---|
| Low Adherence | Medium Adherence | High Adherence | p-Trend | MDS (Continuous) | p-Value | |
| Model 1 a | 1.00 (ref) | 0.40 (0.22–0.73) | 0.43 (0.15–1.22) | 0.006 | 0.76 (0.64–0.92) | 0.004 |
| Model 2 b | 1.00 (ref) | 0.40 (0.21–0.75) | 0.50 (0.17–1.50) | 0.015 | 0.79 (0.66–0.96) | 0.018 |
| Dietary Pattern | Regression Model | adjOR (95%CI) | ||||||
|---|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | p-Trend | Continuous | p-Value | ||
| Western | Model 1 a | 1.00 (ref) | 1.35 (0.73–2.51) | 1.05 (0.57–1.94) | 1.35 (0.74–2.45) | 0.560 | 1.11 (0.92–1.35) | 0.281 |
| Model 2 b | 1.00 (ref) | 1.28 (0.63–2.60) | 0.90 (0.45–1.80) | 1.04 (0.53–2.03) | 0.753 | 1.00 (0.81–1.23) | 0.990 | |
| Prudent | Model 1 a | 1.00 (ref) | 0.66 (0.36–1.22) | 0.57 (0.31–1.04) | 0.77 (0.42–1.40) | 0.352 | 0.87 (0.71–1.05) | 0.144 |
| Model 2 b | 1.00 (ref) | 0.62 (0.32–1.22) | 0.50 (0.26–0.98) | 0.58 (0.29–1.14) | 0.076 | 0.83 (0.62–1.11) | 0.210 | |
| Regression Model | adjOR (95%CI) | |||||
|---|---|---|---|---|---|---|
| Low Adherence | Medium Adherence | High Adherence | p-Trend | MDS | p-Value | |
| Model 1 a | 1.00 (ref) | 1.01 (0.65–1.57) | 0.50 (0.18–1..40) | 0.473 | 1.01 (0.88–1.15) | 0.941 |
| Model 2 b | 1.00 (ref) | 1.23 (0.76–2.01) | 0.76 (0.24–2.41) | 0.726 | 1.09 (0.94–1.27) | 0.272 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barchitta, M.; Maugeri, A.; Quattrocchi, A.; Agrifoglio, O.; Scalisi, A.; Agodi, A. The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy. Nutrients 2018, 10, 469. https://doi.org/10.3390/nu10040469
Barchitta M, Maugeri A, Quattrocchi A, Agrifoglio O, Scalisi A, Agodi A. The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy. Nutrients. 2018; 10(4):469. https://doi.org/10.3390/nu10040469
Chicago/Turabian StyleBarchitta, Martina, Andrea Maugeri, Annalisa Quattrocchi, Ottavia Agrifoglio, Aurora Scalisi, and Antonella Agodi. 2018. "The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy" Nutrients 10, no. 4: 469. https://doi.org/10.3390/nu10040469
APA StyleBarchitta, M., Maugeri, A., Quattrocchi, A., Agrifoglio, O., Scalisi, A., & Agodi, A. (2018). The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy. Nutrients, 10(4), 469. https://doi.org/10.3390/nu10040469







