Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Anthropometric Measurements
2.3. Sociodemographic Characteristics
2.4. Dietary Assessment
2.5. Dietary Patterns
2.6. Statistical Analysis
3. Results
3.1. Dietary Salt Intake
3.2. Socio-Demographic Correlates of Dietary Salt Intake
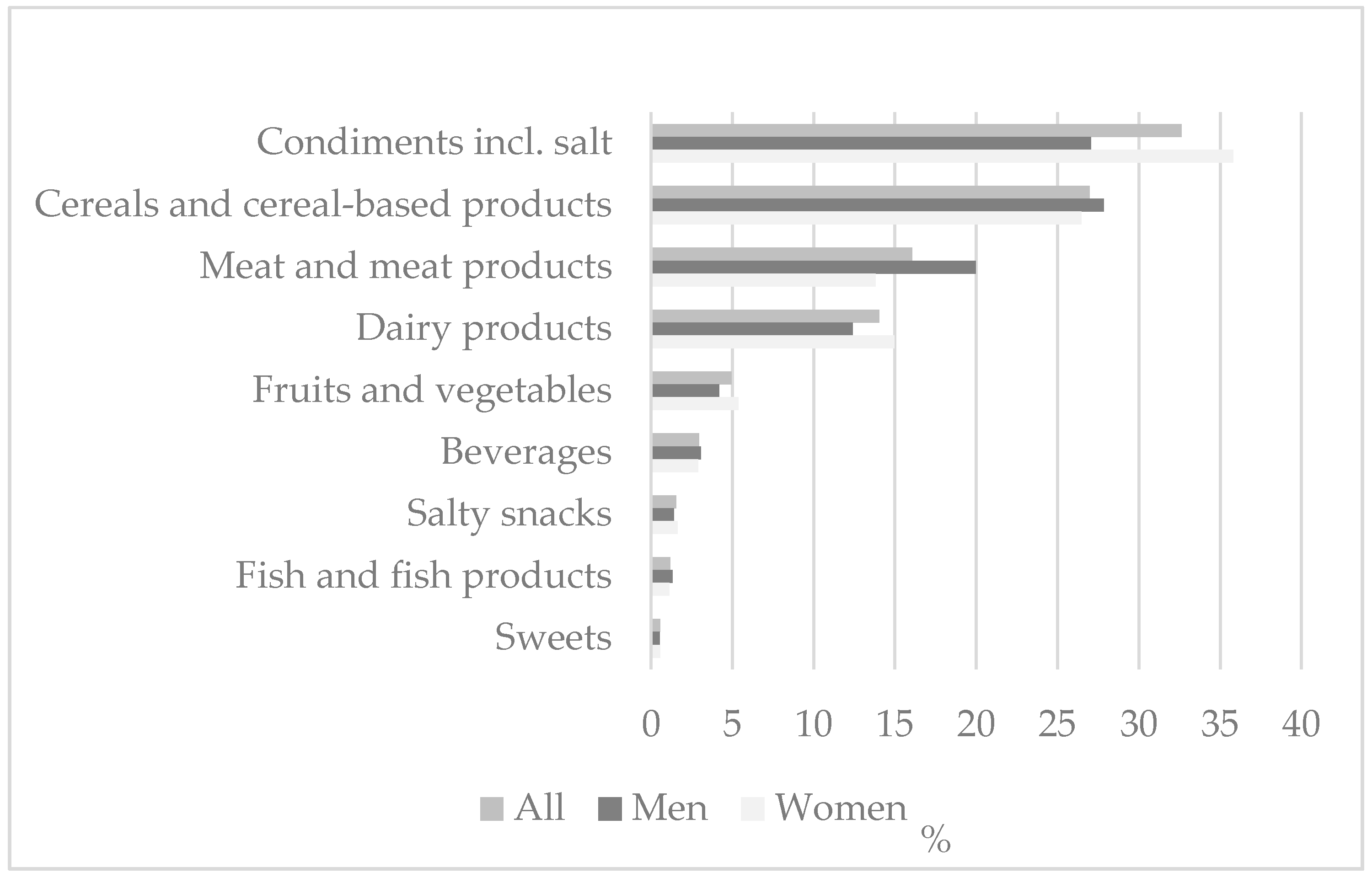
3.3. Contribution of Foods to Dietary Salt Intake
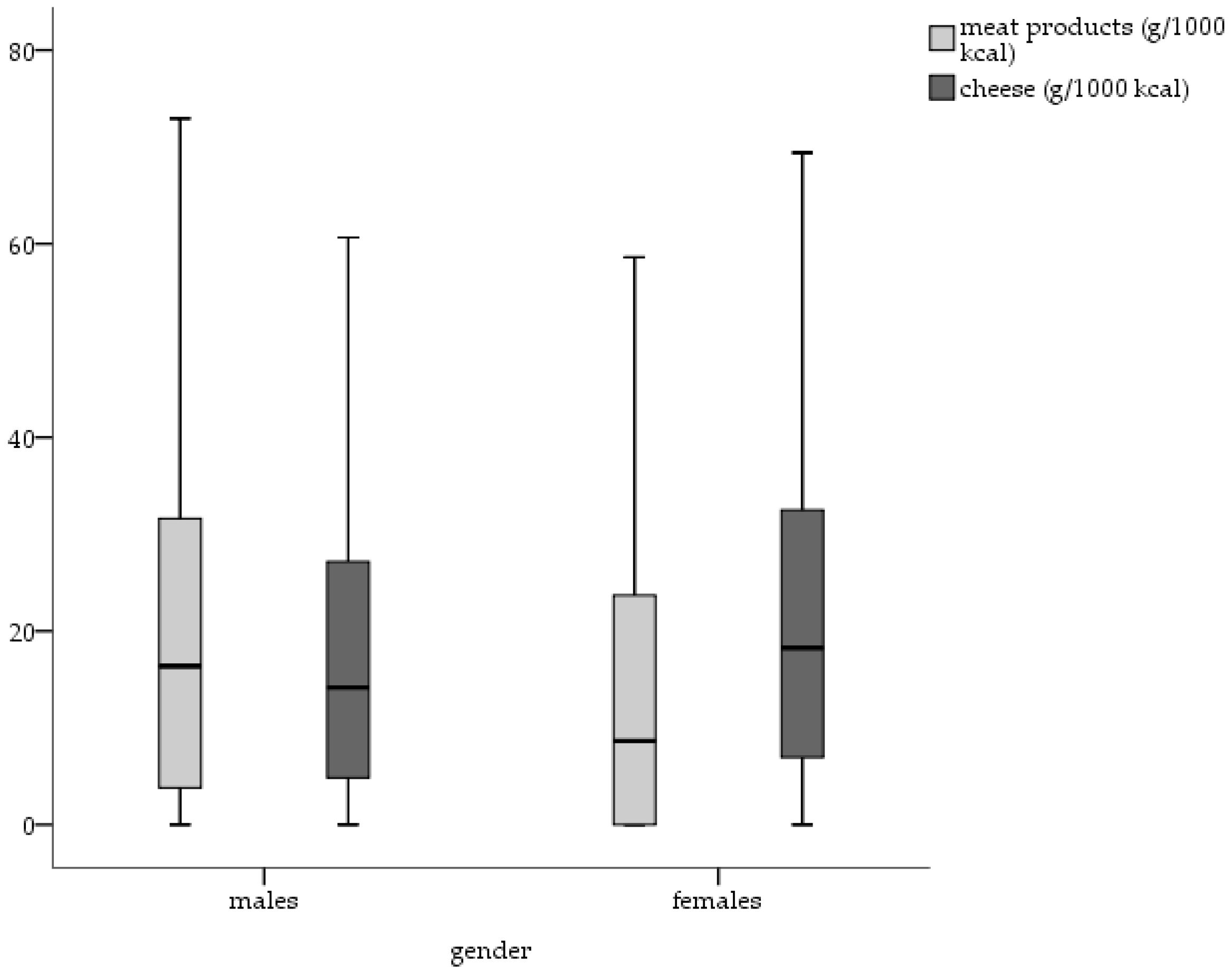
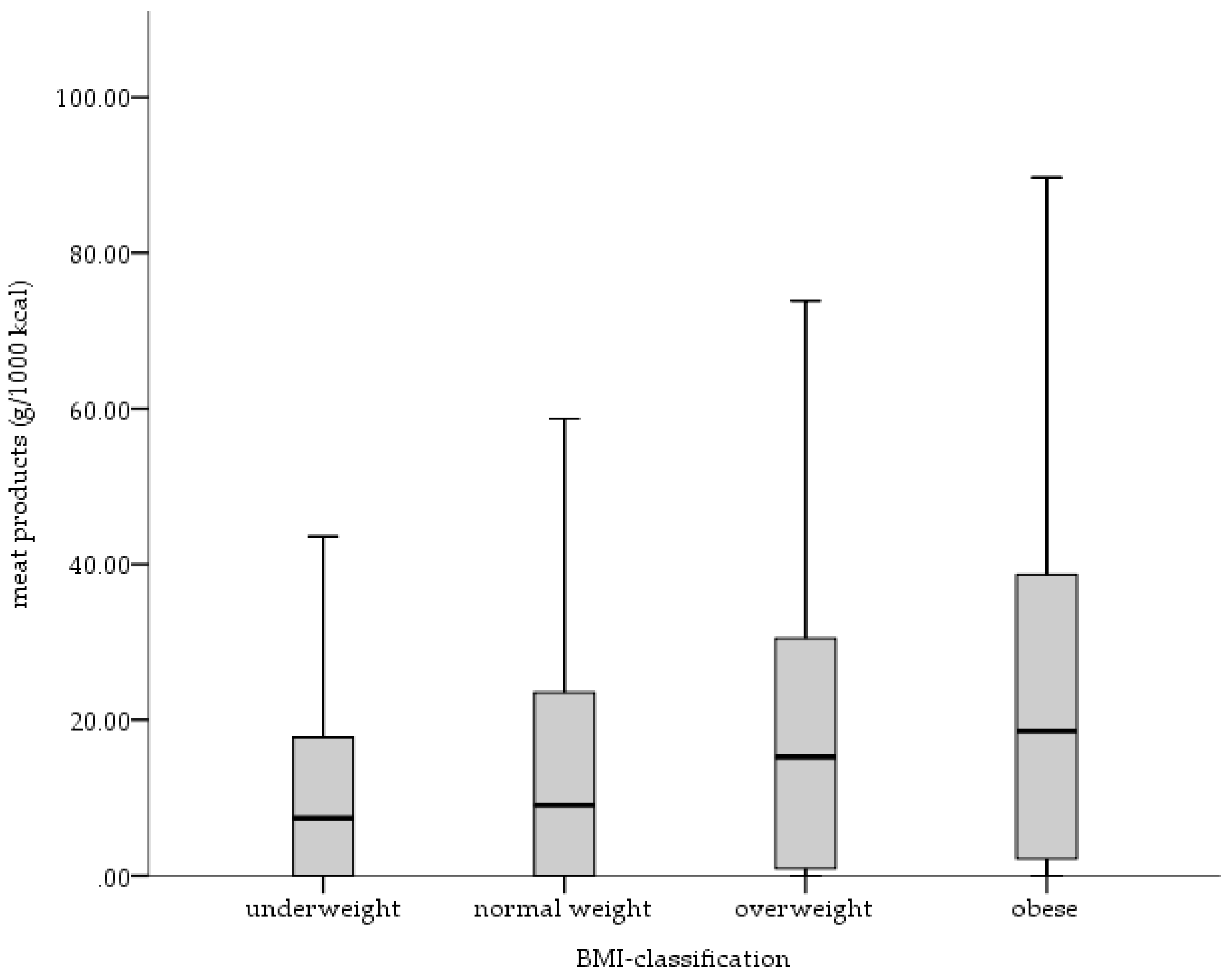
3.4. Contribution of Food Groups to Differences in Dietary Salt Intake
3.5. Predictors of Salt Intake
4. Discussion
4.1. Socio-Demographic Correlates of Dietary Salt Intake
4.2. Contribution of Foods to Dietary Salt Intake
4.3. Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the dietary approaches to stop hypertension (DASH) diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Shi, A.; Tao, Z.; Wei, P.; Zhao, J. Epidemiological aspects of heart diseases (Review). Exp. Ther. Med. 2016, 12, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Polonia, J.; Monteiro, J.; Almeida, J.; Silva, J.A.; Bertoquini, S. High salt intake is associated with a higher risk of cardiovascular events: A 7.2-year evaluation of a cohort of hypertensive patients. Blood Press. Monit. 2016, 21, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Fahirni, S.; Singh, G.M.; Micha, R.; Khatibzadeh, S.; Engell, R.E.; Lim, S.; Danaei, G.; Ezzati, M.; Powles, J. Global Sodium Consumption and Death from Cardiovascular Causes. N. Engl. J. Med. 2014, 371, 624–634. [Google Scholar] [CrossRef] [PubMed]
- Strazzullo, P.; D’Elia, L.; Ngianga-Bakwin, K.; Cappuccio, F.P. Salt intake, stroke, and cardiovascular disease: Meta-analysis of prospective studies. BMJ 2009, 339, b4567. [Google Scholar] [CrossRef] [PubMed]
- Cianciaruso, B.; Bellizzi, V.; Minutolo, R.; Tavera, A.; Capuano, A.; Conte, G.; De Nicola, L. Salt intake and renal outcome in patients with progressive renal disease. Miner. Electrolyte Metab. 1998, 24, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Gibson, S. Salt intake is related to soft drink consumption in children and adolescents: A link to obesity? Hypertension 2008, 51, e55. [Google Scholar] [CrossRef] [PubMed]
- Moosavian, S.P.; Haghighatdoost, F.; Surkan, P.J.; Azadbakht, L. Salt and obesity: A systematic review and meta-analysis of observational studies. Int. J. Food Sci. Nutr. 2017, 68, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M. Cancers attributable to dietary factors in the UK in 2010. IV. Salt. Br. J. Cancer 2011, 105, 31–33. [Google Scholar] [CrossRef] [PubMed][Green Version]
- D’Elia, L.; Rossi, G.; Ippolito, R.; Cappuccio, F.P.; Strazzullo, P. Habitual salt intake and risk of gastric cancer: A meta-analysis of prospective studies. Clin. Nutr. 2012, 31, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Nakandakare, E.R.; Charf, A.M.; Santos, F.C.; Nunes, V.S.; Ortega, K.; Lottenberg, A.M.P.; Mion, D., Jr.; Nakano, T.; Nakajima, K.; D’Amico, E.A.; et al. Dietary salt restriction increases plasma lipoprotein and inflammatory marker concentrations in hypertensive patients. Atherosclerosis 2008, 200, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine of the National Academies. Sodium Intake in Populations-Assessment of Evidence; Institute of Medicine of the National Academies: Washington, DC, USA, 2013. [Google Scholar]
- Deutsche Gesellschaft für Ernährung, Österreichische Gesellschaft für Ernährung, Schweizerische Gesellschaft für Ernährung. Referenzwerte Für Die Nährstoffzufuhr; Österreichische Gesellschaft für Ernährung Deutsche Gesellschaft für Ernährung, Schweizerische Gesellschaft für Ernährung: Bonn, Germany, 2016. [Google Scholar]
- O’Donnell, M.; Mente, A.; Rangarajan, S.; McQueen, M.J.; Wang, X.; Liu, L.; Yan, H.; Lee, S.F.; Mony, P.; Devanath, A.; et al. Urinary Sodium and Potassium Excretion, Mortality, and Cardiovascular Events. N. Engl. J. Med. 2014, 371, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Powles, J.; Fahimi, S.; Micha, R.; Khatibzadeh, S.; Shi, P.; Ezzati, M.; Engell, R.E.; Lim, S.S.; Danaei, G.; Mozaffarian, D. Global, regional and national sodium intakes in 1990 and 2010: A systematic analysis of 24 h urinary sodium excretion and dietary surveys worldwide. BMJ 2013, 3, e003733. [Google Scholar] [CrossRef] [PubMed]
- O’Halloran, S.A.; Lacy, K.E.; Grimes, C.A.; Woods, J.; Campbell, K.J.; Nowson, C.A. A novel processed food classification system applied to Australian food composition databases. J. Hum. Nutr. Diet. 2017, 30, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Brown, I.J.; Tzoulaki, I.; Candeias, V.; Elliott, P. Salt intakes around the world: Implications for public health. Int. J. Epidemiol. 2009, 38, 791–813. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Donnelly, D. Relative contributions of dietary sodium sources. J. Am. Coll. Nutr. 1991, 10, 383–393. [Google Scholar] [CrossRef] [PubMed]
- James, W.P.; Ralph, A.; Sanchez-Castillo, C.P. The dominance of salt in manufactured food in the sodium intake of affluent societies. Lancet 1987, 21, 426–429. [Google Scholar] [CrossRef]
- Anderson, C.A.M.; Appel, L.J.; Okuda, N.; Brown, I.J.; Chan, Q.; Zhao, L.; Ueshima, H.; Kesteloot, H.; Miura, K.; Curb, D.; et al. Dietary Sources of Sodium in China, Japan, the United Kingdom, and the United States, Women and Men Aged 40 to 59 Years: The INTERMAP Study. J. Am. Diet. Assoc. 2010, 110, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Rust, P.; Ekmekcioglu, C. Impact of Salt Intake on the Pathogenesis and Treatment of Hypertension. Adv. Exp. Med. Biol. 2017, 956, 61–84. [Google Scholar] [PubMed]
- Kersting, M.; Rehmer, T.; Hilbig, A. Ermittlung Des Kochsalzkonsums in Verzehrserhebungen Anhand Der Kochsalzausscheidung Im Urin: Eine Sonderauswertung Der DONALD Studie. Available online: http://download.ble.de/05HS048.pdf (accessed on 28 July 2017).
- McLean, R.M. Measuring Population Sodium Intake: A Review of Methods. Nutrients 2014, 6, 4651–4662. [Google Scholar] [CrossRef] [PubMed]
- Pravst, I.; Živa, L.; Kušar, A.; Miklavec, K.; Žmitek, K. Changes in Average Sodium Content of Prepacked Foods in Slovenia during 2011–2015. Nutrients 2017, 9, 952. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Obesity: Preventing and Managing the Global Epidemic; World Health Organization (WHO): Geneva, Switzerland, 2000. [Google Scholar]
- Schindler, K.; Ludvik, B. Methodische und praktische Aspekte der Bestimmung der Körperzusammensetzung [Assessment of body composition-methods and practical aspects, in German]. Wien. Med. Wochenschr. 2004, 154, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Boyce, W.; Torsheim, T.; Currie, C.; Zambon, A. The Familiy Affluence Scale as a measure of National Wealth: Validation of an Adolescent Self-Report Measure. Soc. Indic. Res. 2006, 78, 473–487. [Google Scholar] [CrossRef]
- Armstrong, T.; Bull, F. Development of the World Health Organization Global Physical Activity Questionnaire (GPAQ). J. Public Health 2006, 14, 66–70. [Google Scholar] [CrossRef]
- Slimani, N.; Ferrari, P.; Ocké, M.C.; Welch, A.; Boeing, H.; van Liere, M.; Pala, V.; Amiano, P.; Lagiou, A.; Mattisson, I.; et al. Standardization of the 24-h diet recall calibration method used in the European Prospective Investigation into Cancer and Nutrition (EPIC): General concepts and preliminary results. Eur. J. Clin. Nutr. 2000, 54, 900–917. [Google Scholar] [CrossRef] [PubMed]
- Crispim, S.P.; Vries, J.H.M.D.; Geelen, A.; Souverein, O.W.; Hulshof, P.J.M.; Lafay, L.; Rousseau, A.-S.; Lillegaard, I.T.L.; Andersen, L.F.; Huybrechts, I.; et al. Two non-consecutive 24 h recalls using EPIC-Soft software are sufficiently valid for comparing protein and potassium intake between five European centres-results from the European Food Consumption Validation (EFCOVAL) study. Br. J. Nutr. 2011, 105, 447–458. [Google Scholar] [CrossRef] [PubMed]
- De Boer, E.J.; Slimani, N.; van’t Veer, P.; Boeing, H.; Feinberg, M.; Leclercq, C.; Trolle, E.; Amiano, P.; Andersen, L.F.; Freisling, H.; et al. The European Food Consumption Validation Project: Conclusions and recommendations. Eur. J. Clin. Nutr. 2011, 65, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Slimani, N.; Casagrande, C.; Nicolas, G.; Freisling, H.; Huybrechts, I.; Ocké, M.C.; Niekerk, E.M.; van Rossum, C.; Bellemans, M.; De Maeyer, M.; et al. The standardized computerized 24-h dietary recall method EPIC-Soft adapted for pan-European dietary monitoring. Eur. J. Clin. Nutr. 2011, 65, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, I.; Casagrande, C.; Nicolas, G.; Geelen, A.; Crispim, S.P.; De Keyzer, W.; Freisling, H.; De Henauw, S.; De Maeyer, M.; Krems, C.; et al. Inventory of experiences from national/regional dietary monitoring surveys using EPIC-Soft. Eur. J. Clin. Nutr. 2011, 65, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Max Rubner-Institut-Bundesforschungsinstritut Für Ernährung und Lebensmittel (MRI). Bundeslebensmittelschlüssel (BLS)-Version 3.02. Available online: http://www.blsdb.de (accessed on 25 July 2017).
- Schofield, W.N. Predicting basal metabolic rate, new standards and review of previous work. Hum. Nutr. Clin. Nutr. 1985, 39, 5–41. [Google Scholar] [PubMed]
- Goldberg, G.R.; Black, A.E.; Jebb, S.A.; Cole, T.J.; Murgatroyd, P.R.; Coward, W.A.; Prentice, A.M. Critical evaluation of energy intak data using fundamental principles of energy physiology: 1. Derivation of cut-off limits to identify under-recording. Eur. J. Clin. Nutr. 1991, 45, 569–581. [Google Scholar] [PubMed]
- Black, A.E. Critical evaluation of energy intake using the Goldberg cut-off for energy intake: Basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. 2000, 24, 1119–1130. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Orfanos, P.; Norat, T.; Bueno-de-Mesquita, B.; Ocké, M.C.; Peeters, P.H.; van der Schouw, Y.T.; Boeing, H.; Hoffmann, K.; Boffetta, P.; et al. Modified Mediterranean diet and survival: EPIC-elderly prospective cohort study. BMJ 2005, 330, 991. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Lagiou, P.; Trichopoulos, D. Traditional Greek diet and coronary heart disease. J. Cardiovasc. Risk 1994, 1, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, R.T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef] [PubMed]
- United Nations Educational, Scientific and Cultural Organization (UNESCO). International Standard Classification of Education (ISCED 2011); United Nations Educational Scientific and Cultural Organization (UNESCO): Montreal, QC, Canada, 2012. [Google Scholar]
- World Health Organization (WHO). Global Recommendations on Physical Activity for Health; World Health Organization (WHO): Geneva, Switzerland, 2010. [Google Scholar]
- Elmadfa, I. Österreichischer Ernährungsbericht 2012; Universität Wien: Vienna, Austria, 2012. [Google Scholar]
- Bundesministerium für Ernährung und Landwirtschaft. Ergebnisse der DEGS-Studie; Bundesministerium für Ernährung und Landwirtschaft: Bonn, Germany, 2013. [Google Scholar]
- Cappuccio, F.P.; Ji, C.; Donfrancesco, C.; Palmieri, L.; Ippolito, R.; Vanuzzo, D.; Giampaoli, S.; Strazzullo, P. Geographic and socioeconomic variation of sodium and potassium intake in Italy: Results from the MINISAL-GIRCSI programme. BMJ Open 2015, 5, e007467. [Google Scholar] [CrossRef] [PubMed]
- Vasara, E.; Marakis, G.; Breda, J.; Skepastianos, P.; Hassapidou, M.; Kafatos, A.; Rodopaios, N.; Koulouri, A.A.; Cappuccio, F.P. Sodium and Potassium Intake in Healthy Adults in Thessaloniki Greater Metropolitan Area-The Salt Intake in Northern Greece (SING) Study. Nutrients 2017, 9, 417. [Google Scholar] [CrossRef] [PubMed]
- Kesteloot, H.; Elliott, P.; Lesaffre, E. On the sex ratio of urinary cation excretion obtained from Intersalt and other epidemiological studies. J. Hum. Hypertens. 1990, 4, 603–607. [Google Scholar] [PubMed]
- Santos, J.A.; Webster, J.; Land, M.A.; Flood, V.; Chalmers, J.; Woodward, M.; Neal, B.; Petersen, K.S. Dietary salt intake in the Australian population. Public Health Nutr. 2017, 20, 1887–1894. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Takase, H.; Okado, T.; Sugiura, T.; Yamashita, S.; Kimura, G.; Ohte, N.; Dohi, Y. Significance of adjusting salt intake by body weight in the evaluation of dietary salt and blood pressure. J. Am. Soc. Hypertens. 2016, 10, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food Groups and Risk of Hypertension: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. 2017, 8, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Feng, J.H.; MacGregor, G.A. High salt intake: Independent risk factor for obesity? Hypertension 2015, 66, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Alaniz, M.H.; Brito, L.C.; Borges-Silva, C.N.; Takada, J.; Andreotti, S.; Lima, F.B. High dietary sodium intake increases white adipose tissue mass and plasma leptin in rats. Obesity 2007, 15, 2200–2208. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Alaniz, M.H.; Takada, J.; Andreotti, S.; de Campos, T.B.; Campana, A.B.; Borges-Silva, C.N.; Lima, F.B. High dietary sodium intake enhances insulin-stimulated glucose uptake in rat epididymal adipose tissue. Obesity 2008, 16, 1186–1192. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Pollock, N.K.; Kotak, I.; Gutin, B.; Wang, X.; Bhagatwala, J.; Parikh, S.; Harshfield, G.A.; Dong, Y. Dietary Sodium, Adiposity, and Inflammation in Healthy Adolescents. Pediatrics 2014, 133, e635–e642. [Google Scholar] [CrossRef] [PubMed]
- La Verde, M.; Mulè, S.; Zappalà, G.; Privitera, G.; Maugeri, G.; Pecora, F.; Marranzano, M. Higher adherence to the Mediterranean diet is inversely associated with having hypertension: Is low salt intake a mediating factor? Int. J. Food Sci. Nutr. 2017, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Magriplis, E.; Farajian, P.; Pounis, G.D.; Risvas, G.; Panagiotakos, D.B.; Zampelas, A. High sodium intake of children through ‘hidden’ food sources and its association with the Mediterranean diet: The GRECO study. J. Hypertens. 2011, 29, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Cappuccio, F.P. Socioeconomic inequality in salt intake in Britain 10 years after a national salt reduction programme. BMJ Open 2014, 4, e005683. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, X.; Vaartjes, I.; Neal, B.; Bots, M.L.; Hoes, A.W.; Wu, Y. Does education level affect the efficacy of a community based salt reduction program?—A post-hoc analysis of the China Rural Health Initiative Sodium Reduction Study (CRHI-SRS). BMC Public Health 2016, 16, 759. [Google Scholar] [CrossRef] [PubMed]
- Kloss, L.; Meyer, J.D.; Graeve, L.; Vetter, W. Sodium intake and its reduction by food reformulation in the European Union—A review. NFS J. 2015, 1, 9–19. [Google Scholar] [CrossRef]
- Feng, J.H.; MacGregor, G.A. How Far Should Salt Intake Be Reduced? Hypertension 2003, 42, 1093–1099. [Google Scholar]
- Barr, S.I. Reducing dietary sodium intake: The Canadian context. Appl. Physiol. Nutr. Metab. 2010, 35, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sarmugam, R.; Worsley, A. Current Levels of Salt Knowledge: A Review of the Literature. Nutrients 2014, 6, 5534–5559. [Google Scholar] [CrossRef] [PubMed]
- Grimes, C.; Kelley, S.-J.; Stanley, S.; Bolam, B.; Webster, J.; Khokhar, D.; Nowson, C. Knowledge, attitudes and behaviours related to dietary salt among adults in the state of Victoria, Australia 2015. BMC Public Health 2017, 17, 532. [Google Scholar] [CrossRef] [PubMed]
- Newson, R.S.; Elmadfa, I.; Biro, G.; Cheng, Y.; Prakash, V.; Rust, P.; Barna, M.; Lion, R.; Meijer, G.W.; Neufingerl, N.; et al. Barriers for progress in salt reduction in the general population. An international study. Appetite 2013, 71, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ekmekcioglu, C.; Blasche, G.; Dorner, T.E. Too much salt and how we can get rid of it. Forsch. Komplementmed. 2013, 20, 454–460. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Salt Reduction. Available online: http://www.who.int/mediacentre/factsheets/fs393/en/ (accessed on 17 August 2017).
- Jaenke, R.; Barzi, F.; McMahon, E.; Webster, J.; Brimblecombe, J. Consumer acceptance of reformulated food products: A systematic review and meta-analysis of salt-reduced foods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3357–3372. [Google Scholar] [CrossRef] [PubMed]
- Girgis, S.; Neal, B.; Prescott, J.; Prendergast, J.; Dumbrell, S.; Turner, C.; Woodward, M. A one-quarter reduction in the salt content of bread can be made without detection. Eur. J. Clin. Nutr. 2003, 57, 616–620. [Google Scholar] [CrossRef] [PubMed]
- Dötsch, M.; Busch, J.; Batenburg, M.; Liem, G.; Tareilus, E.; Mueller, R.; Meijer, G. Strategies to reduce sodium consumption: A food industry perspective. Crit. Rev. Food Sci. Nutr. 2009, 49, 841–851. [Google Scholar] [CrossRef] [PubMed]
| Men | Women | |
|---|---|---|
| n | 736 | 1282 |
| Vegetables | 73 | 105 |
| Fruits and nuts | 35 | 72 |
| Legumes | 0 | 0 |
| Cereals including potatoes | 127 | 128 |
| Fish | 0 | 0 |
| Meat | 54 | 36 |
| Dairy products | 96 | 128 |
| Ethanol | 2.8 | 0.3 |
| Lipid ratio 1 | 0.4 | 0.6 |
| Men | Women | |
|---|---|---|
| n (%) | 736 (36.5) | 1282 (63.5) |
| Age (years) 2 | 40 (21) | 37 (21) |
| BMI (kg/m2) 2 | 24.8 (4.5) | 22.8 (4.9) |
| Waist circumference (cm) 2 | 89.7 (12.8) | 77.9 (14.2) |
| Family Affluence Scale (FAS), n (%) 3 | ||
| Low | 4 (0.5) | 5 (0.4) |
| Medium | 186 (25.3) | 378 (29.5) |
| High | 285 (38.7) | 507 (39.5) |
| Not answered | 261 (35.5) | 392 (30.6) |
| Education, n (%) 4 | ||
| Low | 226 (30.7) | 327 (25.5) |
| Medium | 208 (28.3) | 386 (30.1) |
| High | 233 (31.7) | 458 (35.7) |
| Not answered | 69 (9.4) | 111 (8.7) |
| Smoking status, n (%) | ||
| No | 530 (72.0) | 936 (73.0) |
| Yes | 170 (23.1) | 297 (23.2) |
| Not answered | 36 (4.9) | 49 (3.8) |
| Physical activity n (%) 5 | ||
| Recommendation met | 419 (56.9) | 850 (66.3) |
| Recommendation not met | 98 (13.3) | 202 (15.8 |
| Not answered | 219 (29.8) | 230 (17.9) |
| Energy intake (kcal/day) 2 | 2386 (900) | 1792 (729) |
| Characteristics | Proportion by Socio-Demographic Characteristics (%) | Salt Intake (g/day) | |
|---|---|---|---|
| Median | IQ Range | ||
| Overall | 5.0 | 3.1 | |
| Sex | |||
| Men | 36.5 | 6.1 | 3.4 |
| Women | 63.5 | 4.6 | 2.7 |
| BMI-classification | |||
| Underweight | 2.8 | 4.9 | 2.1 |
| Normal weight | 61.7 | 4.9 | 2.9 |
| Overweight | 24.6 | 5.2 | 3.5 |
| Obesity | 10.9 | 5.8 | 3.8 |
| Family Affluence Scale (FAS) | |||
| Low | 0.7 | 6.0 | 3.1 |
| Medium | 41.3 | 4.9 | 2.9 |
| High | 58.0 | 5.0 | 3.2 |
| Education | |||
| Low | 30.1 | 5.1 | 3.3 |
| Medium | 32.3 | 4.9 | 3.0 |
| High | 37.6 | 5.2 | 3.1 |
| Smoking status | |||
| No | 75.8 | 5.0 | 3.1 |
| Yes | 24.2 | 5.1 | 3.2 |
| Physical activity | |||
| Recommendation not met | 19.1 | 5.0 | 3.2 |
| Recommendation met | 80.9 | 5.0 | 3.0 |
| Modified Mediterranean Diet Score (MMDS) | |||
| Low adherence | 44.8 | 5.2 | 3.2 |
| Medium adherence | 42.0 | 4.9 | 3.1 |
| High adherence | 13.2 | 4.8 | 2.9 |
| Parameter | Regression-Coefficient B | Standard Error | 95% CI | p-Value | |
|---|---|---|---|---|---|
| Lower Bound | Upper Bound | ||||
| Sex | |||||
| Men | 1374.908 | 126.416 | 1127.138 | 1622.678 | 0.000 |
| Women | 0 * | ||||
| BMI-classification | |||||
| Underweight | −833.940 | 403.468 | −1624.723 | −43.157 | 0.039 |
| Normal weight | −553.012 | 196.434 | −940.016 | −170.008 | 0.005 |
| Overweight | −350.954 | 215.258 | −772.852 | 70.944 | 0.103 |
| Obesity | 0 * | ||||
| Energy intake | |||||
| Below the recommended energy intake | −1729.149 | 119.809 | −1963.970 | −1494.329 | 0.000 |
| Above the recommended energy intake | 0 * | ||||
| Modified Mediterranean Diet Score (MMDS) | |||||
| Low adherence | 322.207 | 186.669 | −43.657 | 688.071 | 0.084 |
| Medium adherence | 254.975 | 186.549 | −110.654 | 620.603 | 0.172 |
| High adherence | 0 * | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasenegger, V.; Rust, P.; König, J.; Purtscher, A.E.; Erler, J.; Ekmekcioglu, C. Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria. Nutrients 2018, 10, 311. https://doi.org/10.3390/nu10030311
Hasenegger V, Rust P, König J, Purtscher AE, Erler J, Ekmekcioglu C. Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria. Nutrients. 2018; 10(3):311. https://doi.org/10.3390/nu10030311
Chicago/Turabian StyleHasenegger, Verena, Petra Rust, Jürgen König, Anna Elisabeth Purtscher, Judith Erler, and Cem Ekmekcioglu. 2018. "Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria" Nutrients 10, no. 3: 311. https://doi.org/10.3390/nu10030311
APA StyleHasenegger, V., Rust, P., König, J., Purtscher, A. E., Erler, J., & Ekmekcioglu, C. (2018). Main Sources, Socio-Demographic and Anthropometric Correlates of Salt Intake in Austria. Nutrients, 10(3), 311. https://doi.org/10.3390/nu10030311





