Intake of 3 Eggs per Day When Compared to a Choline Bitartrate Supplement, Downregulates Cholesterol Synthesis without Changing the LDL/HDL Ratio
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Experimental Design
2.2. Dietary Records
2.3. Anthropometrics and Plasma Parameters
2.4. Plasma Lipids, Plasma Glucose, and Creatinine
2.5. Apolipoproteins Analysis
2.6. Peripheral Blood Mononuclear Cell Isolation
2.7. Quantitative Real-Time Polymerase Chain Reaction
2.8. Statistical Analysis
3. Results
3.1. Dietary Records
3.2. Anthropometrics and Lipid Profile
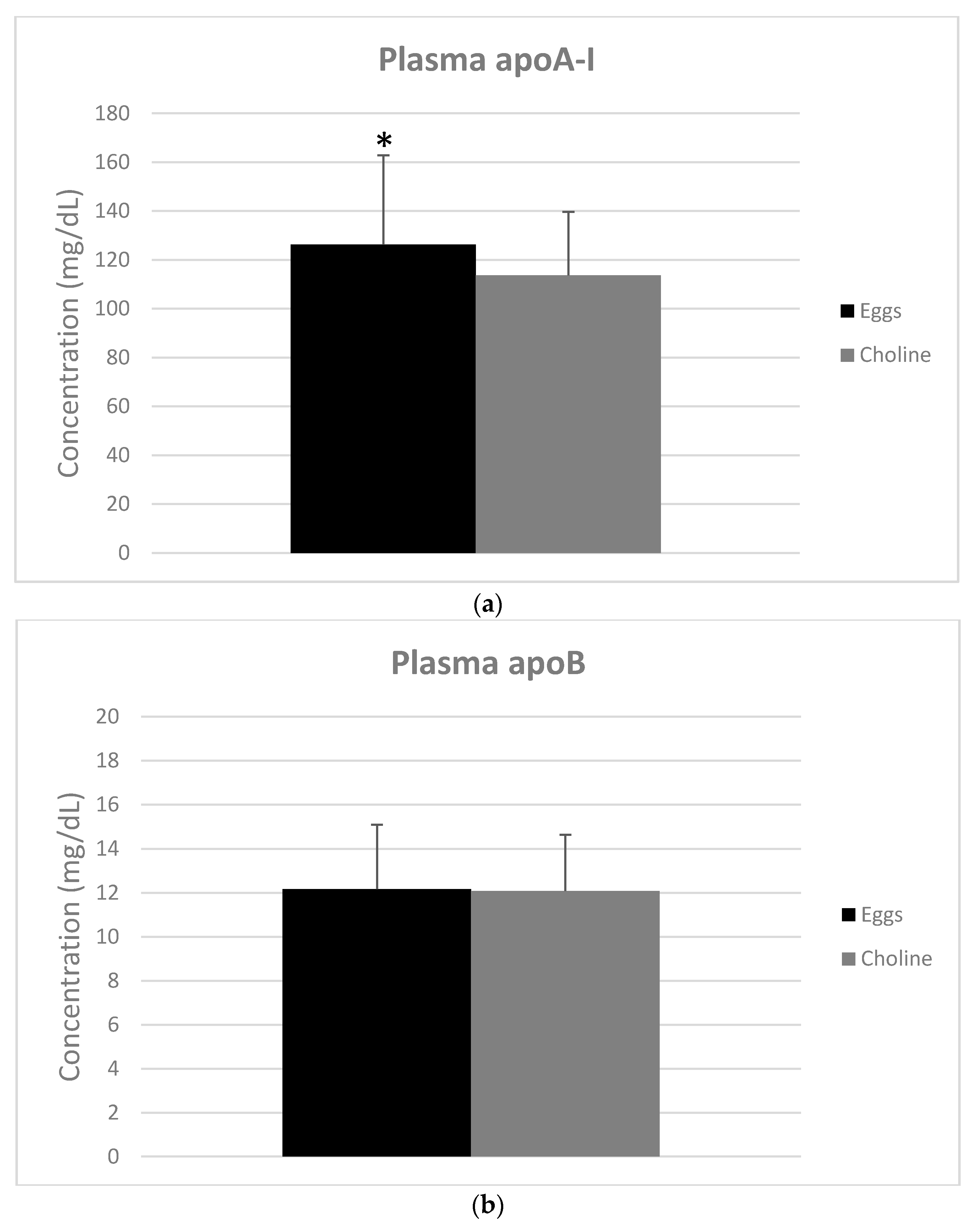
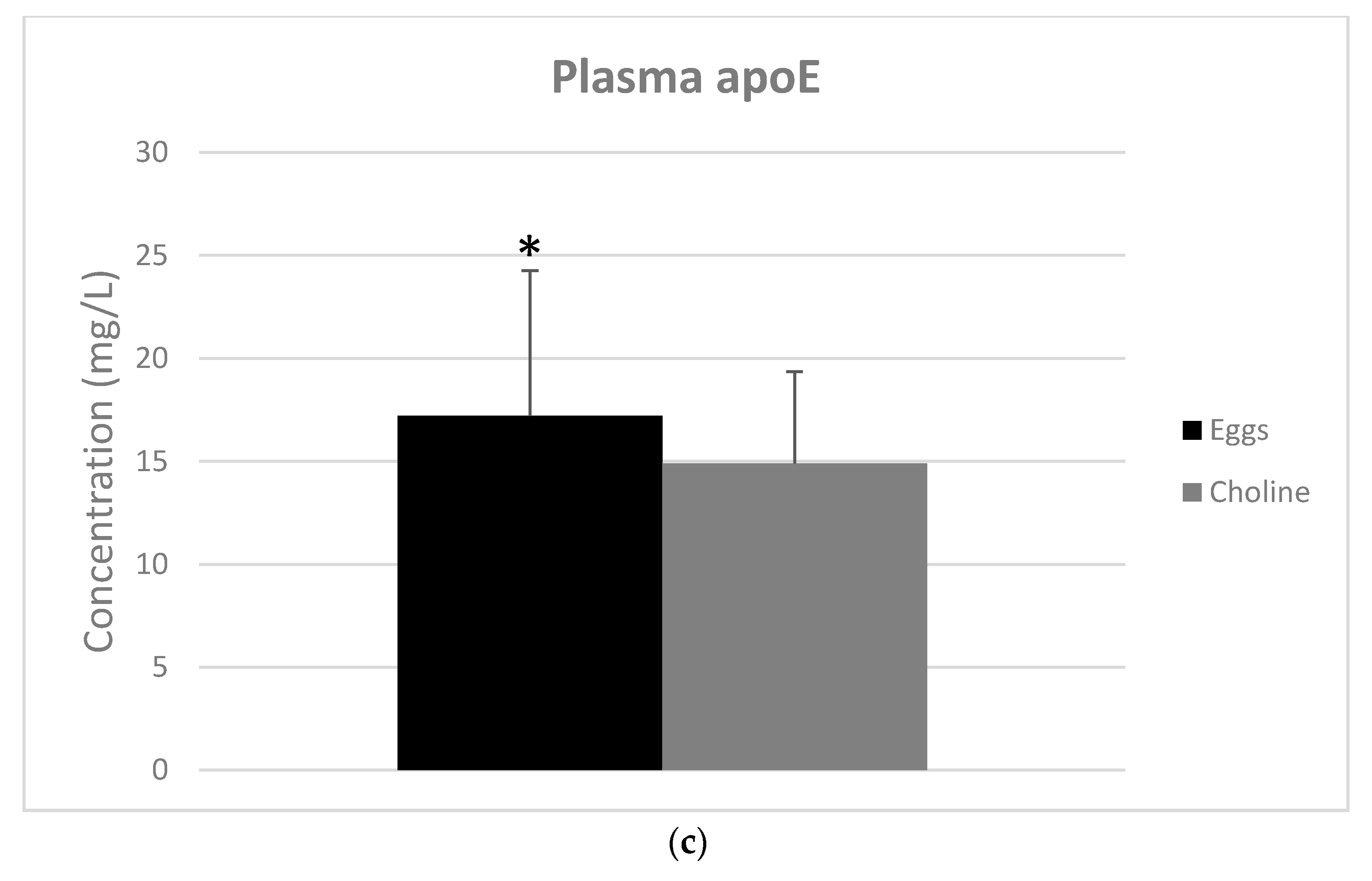
3.3. Apolipoproteins
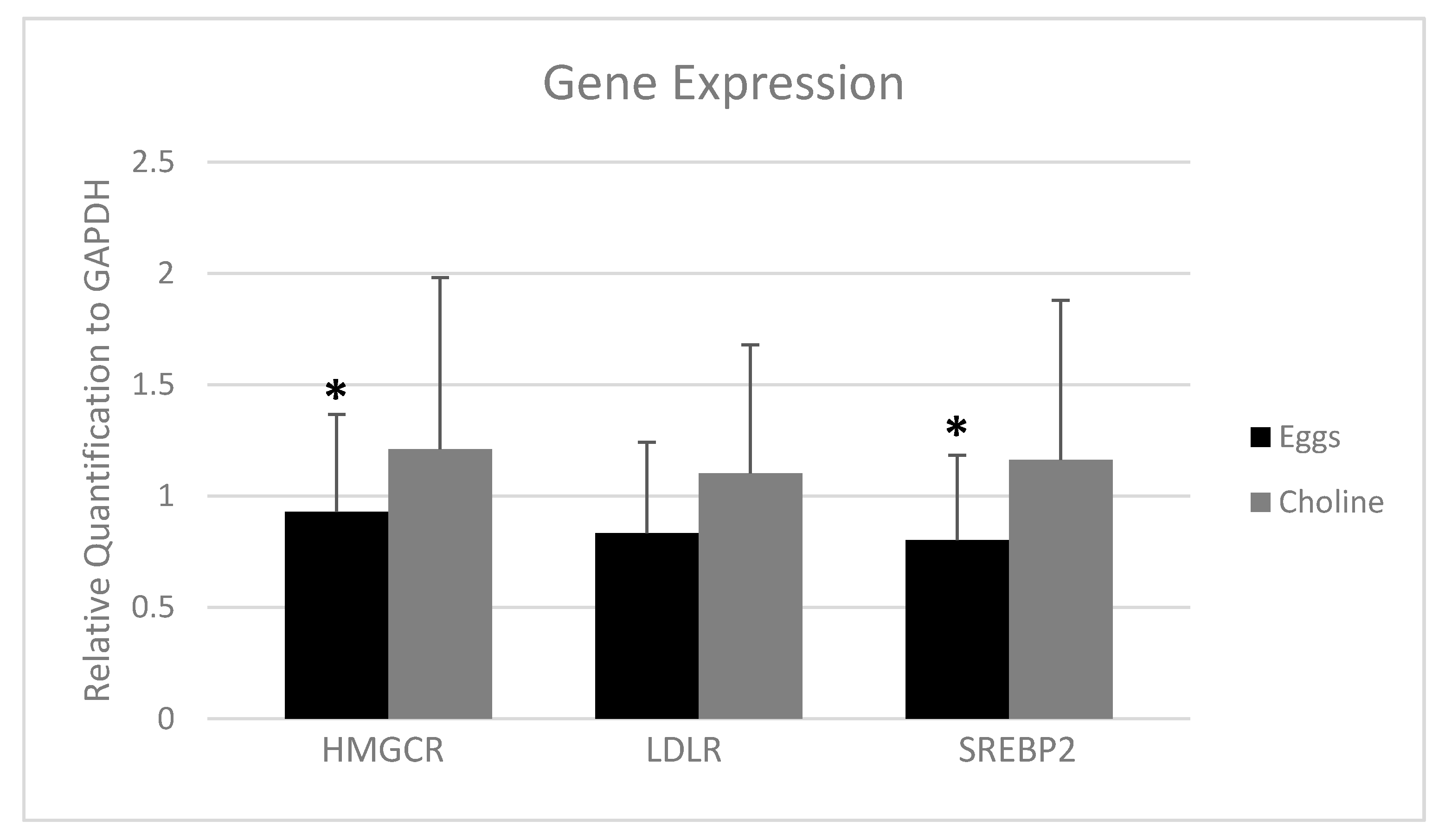
3.4. PBMC Gene Expression
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Imes, C.C.; Austin, M.A. Low-Density Lipoprotein Cholesterol, Apolipoprotein B, and Risk of Coronary Heart Disease: From Familial Hyperlipidemia to Genomics. Changes 2013, 29, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Barona, J.; Fernandez, M.L. Dietary Cholesterol Affects Plasma Lipid Levels, the Intravascular Processing of Lipoproteins and Reverse Cholesterol Transport without Increasing the Risk for Heart Disease. Nutrients 2012, 4, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Chyu, K.Y.; Shah, P.K. HDL/ApoA-1 infusion and ApoA-1 gene therapy in atherosclerosis. Front. Pharmacol. 2015, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nabel, E.G. Cardiovascular disease. N. Engl. J. Med. 2003, 349, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Bertoia, M.L.; Cahill, L.E.; Agarwal, I.; Rimm, E.B.; Mukamal, K.J. Novel metabolic biomarkers of cardiovascular disease. Nat. Rev. Endocrinol. 2014, 10, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Pirillo, A.; Norata, G.D.; Catapano, A.L. LOX-1, OxLDL, and Atherosclerosis. Mediat. Inflamm. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.J.; Freeman, M.W. Scavenger receptors in atherosclerosis: Beyond lipid intake. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1702–1711. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.; Sheedy, F.; Fished, E. Macrophages in atherosclerosis: A dynamic balance. Nat. Rev. Immunol. 2015, 13, 709–721. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ma, K.L.; Ruan, X.Z.; Liu, B.C. Dysregulation of the low-density lipoprotein receptor pathway is involved in lipid disorder-mediated organ injury. Int. J. Biol. Sci. 2016, 12, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J.; Fernandez, M.L. Dietary approaches to improving atheroprotective HDL functions. Food Funct. 2013, 4, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Vilahur, G. High-density lipoprotein benefits beyond the cardiovascular system: A potential key role for modulating acquired immunity through cholesterol efflux. Cardiovasc. Res. 2017, 113, e51–e53. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Andersen, C.J. Bioactive Egg Components and Inflammation. Nutrients 2015, 7, 7889–7913. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C.N. Egg phospholipids and cardiovascular health. Nutrients 2015, 7, 2731–2747. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines Americans, 8th Edition. December 2015. Available online: https://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 15 January 2018).
- Missimer, A.; Dimarco, D.M.; Andersen, C.J.; Murillo, A.G.; Vergara-Jimenez, M.; Fernandez, M.L. Consuming two eggs per day, as compared to an oatmeal breakfast, increases plasma ghrelin while maintaining the LDL/HDL ratio. Nutrients 2017, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Missimer, A.; Fernandez, M.L.; DiMarco, D.M.; Norris, G.H.; Blesso, C.N.; Murillo, A.G.; Vergara-Jimenez, M.; Lemos, B.S.; Medina-Vera, I.; Malysheva, O.V.; et al. Compared to an Oatmeal Breakfast, Two Eggs/Day Increased Plasma Carotenoids and Choline without Increasing Trimethyl Amine N-Oxide Concentrations. J. Am. Coll. Nutr. 2018, 37, 140–148. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Missimer, A.; Murillo, A.G.; Lemos, B.S.; Malysheva, O.V.; Caudill, M.A.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 Eggs/Day Increases HDL Cholesterol and Plasma Choline While Plasma Trimethylamine-N-oxide is Unchanged in a Healthy Population. Lipids 2017, 52, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Blesso, C.N.; Andersen, C.J.; Bolling, B.W.; Fernandez, M.L. Egg intake improves carotenoid status by increasing plasma HDL cholesterol in adults with metabolic syndrome. Food Funct. 2013, 4, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.; Blesso, C.; Lee, J.; Barona, J.; Shah, D.; Thomas, M.; Fernandez, M. Egg Consumption Modulates HDL Lipid Composition and Increases the Cholesterol-Accepting Capacity of Serum in Metabolic Syndrome. Lipids 2013, 29, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Bruce, S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Wu, Y.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Coresh, J.; Greene, T.; Stevens, L.A.; Zhang, Y.L.; Hendriksen, S.; Kusek, J.W.; Lente, F. Van Annals of Internal Medicine Article Using Standardized Serum Creatinine Values in the Modification of Diet in Renal Disease Study Equation for Estimating Glomerular Filtration Rate. Ann. Intern. Med. 2006, 145, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.; Aggarwal, B.B.; Singh, R.B.; Buttar, H.S.; Wilson, D.; De Meester, F. Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention. Diseases 2016, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Salas-salva, J.O.; Bul, M.O.; Babio, N.; Ngel Martnez-gon Alez, M.; Ibarrola-jurado, N.; Basora, J.; Estruch, R.; Isabel Covas, M.; Corella, D.; Os, F.A.; et al. Reduction in the Incidence of Type 2 Diabetes with the Mediterranean Diet: Results of the PREDIMED-Reus nutrition intervention randomized trial. Diabetes Care 2011, 34, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Njike, V.Y.; Ayettey, R.G.; Rajebi, H.; Treu, J.A.; Katz, D.L. Egg ingestion in adults with type 2 diabetes: Effects on glycemic control, anthropometry, and diet quality—A randomized, controlled, crossover trial. BMJ Open Diabetes Res. Care 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Briggs, M.A.; Petersen, K.S.; Kris-Etherton, P.M. Saturated Fatty Acids and Cardiovascular Disease: Replacements for Saturated Fat to Reduce Cardiovascular Risk. Healthcare 2017, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Food Composition Databases Show Foods—Egg, Whole, Raw, Fresh. Available online: https://ndb.nal.usda.gov/ndb/foods/show/112 (accessed on 28 November 2017).
- Siri-Tarino, P.W.; Sun, Q.; Hu, F.B.; Krauss, R.M. Meta-analysis of prospective cohort studies evaluating the association of saturated fat with cardiovascular disease. Am. J. Clin. Nutr. 2010, 91, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of Dietary, Circulating, and Supplement Fatty Acids With Coronary Risk. Ann. Intern. Med. 2014, 160, 398. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, C.E.; Phinney, S.D.; Feinman, R.D.; Brittanie, M.V.; Freidenreich, D.; Quann, E.; Ballard, K.; Puglisi, M.J.; Maresh, C.M.; Kraemer, W.J.; et al. Limited Effect of Dietary Saturated Fat on Plasma Saturated Fat in the Context of a Low Carbohydrate Diet. Lipids 2010, 45, 947–962. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.M.; Anton, X.; Redondo-Valbuena, C.; Roca-Saavedra, P.; Rodriguez, J.A.; Lamas, A.; Franco, C.M.; Cepeda, A. Egg and egg-derived foods: Effects on human health and use as functional foods. Nutrients 2015, 7, 706–729. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.G.; Ford, N.A.; Hu, F.B.; Zelman, K.M.; Mozaffarian, D.; Kris-Etherton, P.M. A healthy approach to dietary fats: Understanding the science and taking action to reduce consumer confusion. Nutr. J. 2017, 16, 53. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hruby, A.; Bernstein, A.M.; Ley, S.H.; Rimm, E.B.; Willett, W.C.; Frank, B. Saturated Fat as Compared With Unsaturated Fats and Sources of Carbohydrates in Relation to Risk of Coronary Heart Disease: A Prospective Cohort Study. J. Am. Coll. Cardiol. 2016, 66, 1538–1548. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L. Effects of eggs on plasma lipoproteins in healthy populations. Food Funct. 2010, 1, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aal, E.S.M.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Leite, J.O.; deOgburn, R.; Smyth, J.A.; Clark, R.M.; Fernandez, M.L. A Lutein-Enriched Diet Prevents Cholesterol Accumulation and Decreases Oxidized LDL and Inflammatory Cytokines in the Aorta of Guinea Pigs. J. Nutr. 2011, 141, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Scicchitano, P.; Cameli, M.; Maiello, M.; Modesti, P.A.; Muiesan, M.L.; Novo, S.; Palmiero, P.; Saba, P.S.; Pedrinelli, R.; Ciccone, M.M. Nutraceuticals and dyslipidaemia: Beyond the common therapeutics. J. Funct. Foods 2014, 6, 11–32. [Google Scholar] [CrossRef]
- DiMarco, D.M.; Norris, G.H.; Millar, C.L.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 Eggs per Day Is Associated with Changes in HDL Function and Increased Plasma Antioxidants in Healthy, Young Adults. J. Nutr. 2017, 147, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L.; Webb, D. The LDL to HDL cholesterol ratio as a valuable tool to evaluate coronary heart disease risk. J. Am. Coll. Nutr. 2008, 27, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Silver, D.L.; Costet, P.; Tall, A.R. Specific binding of ApoA-I, enhanced cholesterol efflux, and altered plasma membrane morphology in cells expressing ABC1. J. Biol. Chem. 2000, 275, 33053–33058. [Google Scholar] [CrossRef] [PubMed]
- Allaire, J.; Vors, C.; Couture, P.; Lamarche, B. LDL particle number and size and cardiovascular risk. Curr. Opin. Lipidol. 2017, 28, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Marcovina, S.; Packard, C.J. Measurement and meaning of apolipoprotein AI and apolipoprotein B plasma levels. J. Intern. Med. 2006, 259, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R.W. Apolipoprotein E: From cardiovascular disease to neurodegenerative disorders. J. Mol. Med. 2016, 94, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.; Vaziri, N.D. HMG-CoA reductase, cholesterol 7α-hydroxylase, LCAT, ACAT, LDL receptor, and SRB-1 in hereditary analbuminemia. Kidney Int. 2003, 64, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Horton, J.D.; Goldstein, J.L.; Brown, M.S. SREBPs: Activators of the complete program of cholesterol and fatty acid synthesis in the liver. J. Clin. Investig. 2002, 109, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.; Quinn, C.M.; Brown, A.J. SREBP-2 positively regulates transcription of the cholesterol efflux gene, ABCA1, by generating oxysterol ligands for LXR. Biochem. J. 2006, 491, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J.; Lee, J.Y.; Blesso, C.N.; Carr, T.P.; Fernandez, M.L. Egg intake during carbohydrate restriction alters peripheral blood mononuclear cell inflammation and cholesterol homeostasis in metabolic syndrome. Nutrients 2014, 6, 2650–2667. [Google Scholar] [CrossRef] [PubMed]
| Target | Forward Primer | Reverse Primer |
|---|---|---|
| HMGCR | 5′-CCCAGTTGTGCGTCTTCCA-3′ | 5′-TTCGAGCCAGGCTTTCACTT-3′ |
| LDLR | 5′-ACTGGGTTGACTCCAAACTTCAC-3′ | 5′-GGTTGCCCCCGTTGACA-3′ |
| SREBP2 | 5′-GGGGATCCCGATGGACGACAGCGGCGGCT-3′ | 5′-GGAATTCTCAGTCTGGCTCATCTTTGACCTT-3′ |
| GAPDH | 5′-TGTGGGCATCAATGGATTTGG-3′ | 5′-ACACCATGTATTCCGGGTCAAT-3′ |
| Parameter | Female | Male | p-Value |
|---|---|---|---|
| Sex | 52% | 48% | 0.910 |
| Age (years) | 25.8 ± 1.95 | 25.2 ± 2.76 | 0.679 |
| BMI (kg/m2) | 23.15 ± 2.46 | 24.39 ± 6.01 | 0.498 |
| Waist Circumference (WC) | 83.5 ± 4.7 | 89.8 ± 7.3 | 0.009 |
| Systolic Blood Pressure (mm Hg) | 103.06 ± 9.34 | 116.00 ± 8.97 | 0.001 |
| Diastolic Blood Pressure (mm Hg) | 68.69 ± 6.17 | 70.33 ± 7.54 | 0.387 |
| Glucose (mg/dL) | 90.8 ± 4.6 | 95.5 ± 3.6 | 0.003 |
| Creatinine (mg/dL) | 0.79 ± 0.10 | 0.98 ± 0.10 | 0.001 |
| eGFR (mL/min) | 94.98 ± 14.65 | 99.39 ± 10.94 | 0.001 |
| Triglycerides (mg/dL) | 64.31 ± 27.09 | 70.31 ± 41.00 | 0.640 |
| Total Cholesterol (mg/dL) | 166.56 ± 30.66 | 159.92 ± 29.62 | 0.561 |
| HDL-C (mg/dL) | 70.81 ± 11.93 | 68.46 ± 7.73 | 0.545 |
| LDL-C (mg/dL) | 82.89 ± 26.51 | 77.40 ± 25.33 | 0.576 |
| LDL-C/HDL-C | 1.20 ± 0.39 | 1.16 ± 0.45 | 0.814 |
| Nutrient | EGGS | CHOLINE | p-Value |
|---|---|---|---|
| Saturated Fat (%) | 13.44 ± 4.46 | 10.91 ± 3.58 | <0.001 |
| Monounsaturated Fat (g) | 29.44 ± 8.84 | 22.42 ± 7.68 | <0.001 |
| Polyunsaturated Fat (g) | 16.09 ± 6.48 | 15.72 ± 5.97 | 0.809 |
| Lutein + Zeaxanthin (μg) | 1474.34 ± 724.72 | 1115.41 ± 746.15 | 0.018 |
| Parameter | Eggs | Choline | p-Value |
|---|---|---|---|
| BMI (kg/m2) | 24.1 ± 2.8 | 24.0 ± 2.60 | 0.347 |
| Systolic Blood Pressure (mm Hg) | 108.1 ± 10.7 | 108.9 ± 10.9 | 0.604 |
| Diastolic Blood Pressure (mm Hg) | 68.8 ± 7.70 | 68.8 ± 6.3 | 0.939 |
| Glucose (mg/dL) | 92.3 ± 6.0 | 90.9 ± 5.7 | 0.226 |
| Creatinine (mg/dL) | 0.85 ± 0.11 | 0.86 ± 0.13 | 0.415 |
| eGFR (mL/min) | 100.6 ± 12.3 | 99.5 ± 12.9 | 0.553 |
| Triglycerides (mg/dL) | 69.6 ± 29.5 | 73.6 ± 36.0 | 0.355 |
| Total Cholesterol (mg/dL) | 172.6 ± 35.8 | 162.7 ± 30.7 | 0.040 |
| HDL-C (mg/dL) | 61.0 ± 16.0 | 57.0 ± 14.3 | 0.030 |
| LDL-C (mg/dL) | 97.7 ± 31.7 | 90.9 ± 26.3 | 0.049 |
| LDL-C/HDL-C | 1.72 ± 0.72 | 1.70 ± 0.67 | 0.775 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemos, B.S.; Medina-Vera, I.; Blesso, C.N.; Fernandez, M.L. Intake of 3 Eggs per Day When Compared to a Choline Bitartrate Supplement, Downregulates Cholesterol Synthesis without Changing the LDL/HDL Ratio. Nutrients 2018, 10, 258. https://doi.org/10.3390/nu10020258
Lemos BS, Medina-Vera I, Blesso CN, Fernandez ML. Intake of 3 Eggs per Day When Compared to a Choline Bitartrate Supplement, Downregulates Cholesterol Synthesis without Changing the LDL/HDL Ratio. Nutrients. 2018; 10(2):258. https://doi.org/10.3390/nu10020258
Chicago/Turabian StyleLemos, Bruno S, Isabel Medina-Vera, Christopher N Blesso, and Maria Luz Fernandez. 2018. "Intake of 3 Eggs per Day When Compared to a Choline Bitartrate Supplement, Downregulates Cholesterol Synthesis without Changing the LDL/HDL Ratio" Nutrients 10, no. 2: 258. https://doi.org/10.3390/nu10020258
APA StyleLemos, B. S., Medina-Vera, I., Blesso, C. N., & Fernandez, M. L. (2018). Intake of 3 Eggs per Day When Compared to a Choline Bitartrate Supplement, Downregulates Cholesterol Synthesis without Changing the LDL/HDL Ratio. Nutrients, 10(2), 258. https://doi.org/10.3390/nu10020258








